Abstract
Acute myeloid leukemia (AML) can affect individuals of all ages, but is more common in older adults. It has been estimated that AML accounted for 1% of all newly diagnosed cancers in the USA in 2022. The diagnostic process varies depending on the presenting symptoms and the healthcare facility that patients attend at diagnosis. The treatment process is long and prone to complications, requiring experienced medical professionals and appropriate infrastructure. Treatment of the disease did not change greatly over the years until 2017 when targeted therapies were licensed. The treatment of AML is associated with significant direct economic costs. A number of obstacles originating both from individual patients and the healthcare system may be encountered during the diagnosis and treatment of the disease, which may negatively impact the optimal management of the disease process. In this article, we focus primarily on the social, operational, and financial obstacles including the corona virus disease 2019 (COVID-19) pandemic experienced during the diagnosis and treatment of AML.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Why carry out this study? |
Social, operational, and financial obstacles are often observed during the diagnosis and management of acute myeloid leukemia (AML). |
The aim of this commentary is to summarize the obstacles encountered during the diagnosis and management of AML. |
What was learned from this study? |
Delays in the diagnosis and financial issues experienced during the diagnostic tests are barriers that can be observed in patients with AML. |
During the management of AML, delays in starting treatment and unavailability of newer treatment options in many countries might have significant outcomes, and, especially, the management of relapsed/refractory disease and allogeneic stem cell transplantation appear to be associated with high economic burden. |
During the COVID-19 era, many challenges were encountered by both the leukemia specialist physicians and the patients and their caregivers. |
Introduction
Acute myeloid leukemia (AML) can affect individuals of all ages, but is more common in older adults. Based on surveillance, epidemiology, and end results (SEER) data, it was estimated that AML accounted for 1% of all newly diagnosed cancers in the USA in 2022 [1, 2]. Unlike acute lymphoblastic leukemia, AML is more prevalent in developed countries in Europe and North America [3]. The onset and intensity of disease-related symptoms vary between individuals. Ideally, patients with urgent conditions should be hospitalized and undergo diagnostic testing in a timely manner. However, the diagnostic process in patients with AML may vary from center to center. Prolongation of the process from diagnosis to treatment may expose patients with AML to the risk of developing disseminated intravascular coagulation, especially in patients with acute promyelocytic leukemia (APL), or to life-threatening complications, such as leukostasis, sepsis, and spontaneous tumor lysis syndrome. Apart from the delay in treatment, the patient's inability to receive appropriate treatment for various reasons is an additional challenge. Barriers to the diagnosis and treatment process negatively affect the optimal management of AML (outlined in Fig. 1). In this article, we attempt to summarize the social, operational, and financial obstacles experienced during the diagnosis and treatment of AML.
This article is based on previously conducted studies and does not contain any new studies with human participants or animals performed by any of the authors.
Time to Diagnosis
The urgency of the situation accelerates the diagnosis process in patients presenting with acute symptoms of AML. Patients may not seek medical attention while waiting for recovery due to nonspecific symptoms in the early period. Additionally, even if cytopenia is detected in patients who are tested for other reasons, blasts that support a diagnosis of AML may not be present in the peripheral blood. It may take some time before a differential diagnosis can be made in these patients based on the results of the requested tests.
In a study conducted in the UK between 2004 and 2011, the median time from symptom onset to seeking medical attention was found to be 13 (range 1–47) days, and the median time to diagnosis of AML was found to be 41 (range 17–85) days [4]. Patients referred to hematology outpatient clinics for differential diagnosis may also experience delays in the diagnosis of their condition if they have to wait for future appointments in a busy outpatient clinic in community-based or academic hospitals. We have encountered this situation mostly as a problem arising from the health service delivery system. For example, to prevent delays in the diagnosis of cancer, the UK government has made the commitment that patients with a suspicion of malignancy who are referred by general practitioners will be examined by a specialist physician within 2 weeks [5]. This might be true for most cancers, even for many AML cases, but the early diagnosis and initiation of all-trans retinoic acid therapy when APL is suspected are extremely crucial for the optimal management of this disease, which is a distinct subtype of AML [6, 7].
Characteristics of Healthcare Systems
The process of healthcare services can be influenced by various factors, such as health insurance coverage, socioeconomic status, and the characteristics of the healthcare delivery system itself [8, 9]. Individuals without public or private health insurance may need to pay out-of-pocket health expenses to access proper diagnostic testing and treatment. According to the World Health Statistics published by the World Health Organization in 2022, in 2017 approximately 1 billion people spent > 10% of their household budgets on health services and 290 million people spent > 25% of their budgets [10]. In developing countries, the heterogeneous nature of access to treatment is becoming increasingly more evident. In India, out-of-pocket expenses account for about 62.6% of total healthcare expenditure [11]. Some low- and middle-income countries lack full-coverage healthcare provision, even for cancer patients [12]. Out-of-pocket expenditures may have catastrophic financial consequences for individuals with a disease such as AML, which requires diagnostic invasive interventions and a multidisciplinary approach, and whose treatment decisions are supported by genetic/molecular testing. This situation may also hinder the diagnosis process of the patients.
Limited Infrastructure and Delayed Test Results
In the AML diagnosis and management recommendations of the European Leukemia Net, which were updated in 2022, cytogenetic evaluation, flow cytometry, molecular testing by PCR, and next-generation sequencing are among the recommended standard tests for the diagnosis, risk scoring, treatment decision-making, and patient response monitoring of AML. The same recommendations suggested that the results of cytogenetic tests should be available within 5–7 days and that ideally the results of molecular tests for mutations that can be used as a target in treatment should be available by 3–5 days on average [13]. In our daily clinical practice, obtaining such genetic test results are not that easy, and the duration of time before such results are obtained may vary from center to center, taking relatively longer in non-academic centers. In some cases, the test results cannot be accessed easily due to infrastructure-related obstacles, such as the lack of comprehensive laboratory and experienced personnel (pathology, genetics, etc.) in the center where the patients are examined. In the study by Pollyea and colleagues [14], molecular test results were studied in 67% of AML patients; it has been shown that all recommended molecular test results are studied in < 10% of cases and that the rate of molecular test results being studied is higher in academic centers. Although it is possible to purchase services from centers with comprehensive laboratories, it may not be possible to obtain the results within the short period recommended for such test results. Patients may have to start induction chemotherapy with conventional treatment options before the test results needed for targeted therapies are available.
Treatment Related Barriers
Time to Treatment
There is a risk that delays in the treatment of AML may result in life-threatening complications, such as sepsis, bleeding, leukostasis, and disseminated intravasculary coagulation (DIC) in patients. However, the results obtained from studies comparing the effect of short-term delays (usually < 1 week) from diagnosis to treatment on survival are controversial [15,16,17]. Causes of treatment delays include transfers between hospitals, suspected or documented infections, and management of concomitant acute conditions [15].
Management of the Treatment Process
In the optimal management of acute leukemia, the importance of the contributions of experienced staff and units with the appropriate infrastructure cannot be ignored, as well as the contributions of the physician who organizes the treatment. Blood bank support and quick access to all kinds of blood products are other keys to successful AML management [18]. The results of a retrospective study showed that 1-month mortality is higher in non-academic centers compared to academic centers, while 5-year overall survival is higher in the latter, after adjustment for patient-specific and treatment-related factors [19]. In a comparison of centers based on the annual number of AML patients treated (≤ 75% vs. > 75%), treatment mortality was found to be lower in hospitals with higher volumes of patients with AML [20]. These differences may be due to infrastructure differences between centers and the presence of experienced personnel [21].
Treatment Options and Costs
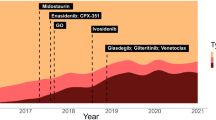
Conventional chemotherapy agents have been available and used for many years to treat AML. More recently, knowledge of the effect of genetic mutations on the pathogenesis of the disease and on its prognosis has supported the development of targeted therapies and led to the reshaping of AML treatment after years during which there were few changes in disease management. Since 2017, agents such as FMS-like tyrosine kinase 3 (FLT3), isocitrate dehydrogenase 1&2 (IDH1&2), and B-cell lymphoma 2 (BCL-2) inhibitors, oral azacytidine, dual-drug liposomal encapsulation of cytarabine and daunorubicin (CPX-351), and glasdegib have been approved for the treatment of various indications in induction and consolidation regimens, both in frontline therapy and in relapsed/refractory AML (r/rAML) patients [13, 22]. The monthly average costs of these current treatments can reach around US$20,000 [23]. The authors of a recent analysis estimated that newly marketed drugs and their preferred use in combination therapeutic regimens will increase drug expenditures in AML; for example, privately insured patients in the USA may face substantial out-of-pocket co-payments for drugs [24].
New therapeutic agents have positive aspects, such as efficacy, safety, ease of administration, and shortening of hospital stay [24]. However, molecules may also have characteristic negative effects. In this context, IDH inhibitors can cause specific side effects, including differentiation syndrome; patients receiving FLT3 inhibitors may experience gastrointestinal toxicities; and venetoclax requires dynamic dose changes and close monitoring during the ramp-up period, and drug-drug interactions can be problematic in patients receiving this drug. It is important for the correct management of the process that the patients and the healthcare institutions where the patients are being treated, when necessary, have information about the treatment plan, side effects of the drugs, and possible drug interactions.
These new targeted therapies come at a high cost and, consequently, these agents may not be accessible to patients who are uninsured and/or of low socioeconomic status, or to patients in developing countries. Due to the relatively long duration of the treatment of AML and the presence of a treatment process open to complications, it can be predicted that conventional treatments are also associated with high costs, with long-term hospitalizations and treatment-related complications accounting for most of these costs. In a study conducted in the USA covering the years 1999–2006, i.e., before the licensing of new drugs, treatment costs were found to be around US$123,000 and US$130,000 for patients who had private insurance or public insurance, respectively, while they were approximately US$100,000 for uninsured patients. This difference was attributed to shorter hospital stays and discontinuation of treatment after induction [25]. In the study by Pandya et al. [26], the cost of AML treatment services was analyzed between 2008 and 2016. Treatment costs were found to have increased over time, amounting to US$198,657 and US$53,081 for the high- and low-intensity induction chemotherapies, respectively, and US$73,428 for high-intensity consolidation chemotherapy. Allogeneic stem cell transplantation (allo-SCT) costs were around US$329,621, and the treatment costs were considerably higher, namely, US$439,104 in r/rAML [26].
Management of the r/rAML Patient
Treatment of r/rAML is a therapeutic challenge. The effect of previous treatment processes on the performance capacity of the patients and the permanent comorbid conditions that develop during the treatment process make the management of the cases difficult and may limit the treatment options. The curative treatment of AML is allo-SCT, which may be preferred in the first remission or relapse according to the risk group assessment. Considering the advances in supportive care, risk stratification, donor/graft selection, and peri-transplant management, the application of allo-SCT has increased over time and today is applied in licensed centers that meet appropriate conditions [26]. In the treatment process, it is important to be in contact with experienced and comprehensive centers in the evaluation of patients suitable for allo-SCT, planning the transplantation process, and directing selected cases that are not suitable for other treatments to clinical studies. The participation rate of cancer patients in clinical studies was found below 5% and the most important obstacles were determined as protocol, physician triage, and patient decision [27]. Because of this dynamic and complex process, it is difficult to follow up r/rAML patients in non-academic centers.
AML management during COVID-19 pandemic
The severe acute respiratory syndrome-coronavirus 2 (SARS-Cov-2) pandemic, which began in 2019 (COVID-19), paralyzed healthcare systems on global scale [28]. Patients with AML were at risk for SARS-CoV-2 infection not only due to malignancy-related immunodeficiency but also related to the treatment they received [29]. Hospitalization and repeated outpatient visits have been shown to increase the risk of infection [30]. Travel restrictions during the pandemic period and patients’ avoidance of infection risk may have reduced outpatient attendance [29]. During the pandemic, bed occupancy rates of hospitals increased, inpatient leukemia treatment and allogeneic transplantation processes became more difficult, blood donations decreased, and blood bank reserves decreased, all of which led to problems with supportive care [28]. Under these conditions, alternative treatment options were introduced according to various guideline recommendations, such as induction treatment options that do not require hospitalization in suitable patients, blood product use and transfusion thresholds, treatment of COVID-19 infection, vaccination policies, and management of the transplantation process [30,31,32,33,34].
Conclusion
A better understanding of the barriers in AML management will guide the review of healthcare policies and the development of interventions aimed at eliminating inequalities [35]. There are various solutions from the simple to the complex, such as improving the infrastructure of healthcare centers, optimizing the time required for diagnostic test results, facilitating the communication of centers in terms of rapid access to specific treatment options, such as allo-SCT and clinical trials, expanding the treatment coverage of health insurance systems, and expanding access to up-to-date treatment options in developing countries. If we compare the AML diagnosis and treatment process to a road, clearing the stones on the road will improve the driving experience.
References
Juliusson G, Antunovic P, Derolf A, et al. Age and acute myeloid leukemia: real world data on decision to treat and outcomes from the Swedish Acute Leukemia Registry. Blood. 2009;113(18):4179–87.
National Cancer Institute (NCI) Surveillance, Epidemiology, and End Results (SEER). Cancer stat facts: leukemia—acute myeloid leukemia. 2023. https://seer.cancer.gov/statfacts/html/amyl.html. Accessed 3 Jan 2023.
Dong Y, Shi O, Zeng Q, et al. Leukemia incidence trends at the global, regional, and national level between 1990 and 2017. Exp Hematol Oncol. 2020;9:14.
Howell DA, Smith AG, Jack A, et al. Time-to-diagnosis and symptoms of myeloma, lymphomas and leukaemias: a report from the Haematological Malignancy Research Network. BMC Hematol. 2013;13(1):9.
Jones R, Rubin G, Hungin P. Is the two week rule for cancer referrals working? BMJ. 2001;322(7302):1555–6.
Agency for Healthcare Research and Quality (AHRQ). 2012 National healthcare disparities report. AHRQ publication no. 13-0003. Rockville: AHRQ; 2013.
Agency for Healthcare Research and Quality (AHRQ). 2012 National healthcare quality report. AHRQ publication no. 13-0002. Rockville: AHRQ; 2013.
Sanz MA, Fenaux P, Tallman MS, et al. Management of acute promyelocytic leukemia: updated recommendations from an expert panel of the European LeukemiaNet. Blood. 2019;133(15):1630–43.
Yilmaz M, Kantarjian H, Ravandi F. Acute promyelocytic leukemia current treatment algorithms. Blood Cancer J. 2021;11(6):123.
World health statistics 2022: monitoring health for the SDGs, sustainable development goals. Geneva: World Health Organization; 2022. Licence: CC BY-NC-SA 3.0 IGO. ISBN 978-92-4005114-0 https://www.who.int/publications/i/item/9789240051157. Accessed 26 Dec 2022.
Sriram S, Khan MM. Effect of health insurance program for the poor on out-of-pocket inpatient care cost in India: evidence from a nationally representative cross-sectional survey. BMC Health Serv Res. 2020;20(1):839.
Haier J, Sleeman J, Schäfers J. Editorial series: cancer care in low- and middle-income countries. Clin Exp Metastasis. 2019;36(6):477–80.
Döhner H, Wei AH, Appelbaum FR, et al. Diagnosis and management of AML in adults: 2022 recommendations from an international expert panel on behalf of the ELN. Blood. 2022;140(12):1345–77.
Pollyea DA, George TI, Abedi M, et al. Diagnostic and molecular testing patterns in patients with newly diagnosed acute myeloid leukemia in the Connect® MDS/AML Disease Registry. EJHaem. 2020;1(1):58–68.
Ibrahimi S, Mukherjee S, Machiorlatti MG, et al. Effect of time of admission to treatment initiation on outcomes of patients with acute myeloid leukemia: a tertiary care referral center experience. JCSO. 2018;16(5):e188–93.
Bertoli S, Bérard E, Huguet F, et al. Time from diagnosis to intensive chemotherapy initiation does not adversely impact the outcome of patients with acute myeloid leukemia. Blood. 2013;121(14):2618–26.
Sekeres MA, Elson P, Kalaycio ME, et al. Time from diagnosis to treatment initiation predicts survival in younger, but not older, acute myeloid leukemia patients. Blood. 2009;113(1):28–36.
Jillella AP, Cortes JE, Kota VK. Optimizing management of acute leukemia in community centers and when to refer. Hematol Am Soc Hematol Educ Progr. 2020;2020(1):123–8.
Bhatt VR, Shostrom V, Giri S, et al. Early mortality and overall survival of acute myeloid leukemia based on facility type. Am J Hematol. 2017;92(8):764–71.
Giri S, Pathak R, Aryal MR, Karmacharya P, Bhatt VR, Martin MG. Impact of hospital volume on outcomes of patients undergoing chemotherapy for acute myeloid leukemia: a matched cohort study. Blood. 2015;125(21):3359–60.
Shallis RM, Wang R, Davidoff A, Ma X, Zeidan AM. Epidemiology of acute myeloid leukemia: Recent progress and enduring challenges. Blood Rev. 2019;36:70–87.
Kucukyurt S, Eskazan AE. New drugs approved for acute myeloid leukaemia in 2018. Br J Clin Pharmacol. 2019;85(12):2689–93.
Kantarjian HM, Short NJ, Fathi AT, et al. Acute myeloid leukemia: historical perspective and progress in research and therapy over 5 decades. Clin Lymphoma Myeloma Leuk. 2021;21(9):580–97.
Forsythe A, Sandman K. What does the economic burden of acute myeloid leukemia treatment look like for the next decade? An analysis of key findings, challenges and recommendations. J Blood Med. 2021;12:245–55.
Bradley CJ, Dahman B, Jin Y, Shickle LM, Ginder GD. Acute myeloid leukemia: how the uninsured fare. Cancer. 2011;117(20):4772–8.
Pandya BJ, Chen CC, Medeiros BC, et al. Economic and clinical burden of acute myeloid leukemia episodes of care in the United States: a retrospective analysis of a commercial payer database. J Manag Care Spec Pharm. 2020;26(7):849–59.
Go RS, Frisby KA, Lee JA, et al. Clinical trial accrual among new cancer patients at a community-based cancer center. Cancer. 2006;106(2):426–33.
Koo J, Auletta JJ, Hartley DM, et al. Secondary impact of the coronavirus disease 19 pandemic on patients and the cellular therapy healthcare ecosystem. Transpl Cell Ther. 2022;28(11):737–46.
Philip CC, Selvarajan S, Nayak L, et al. Acute myeloid leukemia during the COVID pandemic: impact and the indian experience. Blood. 2022;140:8127–8.
Brissot E, Labopin M, Baron F, et al. Management of patients with acute leukemia during the COVID-19 outbreak: practical guidelines from the acute leukemia working party of the European Society for Blood and Marrow Transplantation. Bone Marrow Transpl. 2021;56(3):532–5.
Wilde L, Isidori A, Keiffer G, Palmisiano N, Kasner M. Caring for AML patients during the COVID-19 crisis: an American and Italian experience. Front Oncol. 2020;10:1689.
Algwaiz G, Aljurf M, Koh M, et al. Real-world issues and potential solutions in hematopoietic cell transplantation during the COVID-19 pandemic: perspectives from the Worldwide Network for Blood and Marrow Transplantation and Center for International Blood and Marrow Transplant Research Health Services and International Studies Committee. Biol Blood Marrow Transpl. 2020;26:2181–9.
Waghmare A, Abidi MZ, Boeckh M, et al. Guidelines for COVID-19 management in hematopoietic cell transplantation and cellular therapy recipients. Biol Blood Marrow Transpl. 2020;26(11):1983–94.
Modemann F, Ghandili S, Schmiedel S, Weisel K, Bokemeyer C, Fiedler W. COVID-19 and adult acute leukemia: our knowledge in progress. Cancers (Basel). 2022;14(15):3711.
Majhail NS, Omondi NA, Denzen E, Murphy EA, Rizzo JD. Access to hematopoietic cell transplantation in the United States. Biol Blood Marrow Transpl. 2010;16(8):1070–5.
Acknowledgements
Funding
No funding or sponsorship was received for this work or publication of this article.
Authorship
All named authors meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this article, take responsibility for the integrity of the work as a whole, and have given their approval for this version to be published.
Author Contributions
All named authors (Emine Eylem Genç, İrem Sena Saraç, Hayrunnisa Arslan and Ahmet Emre Eşkazan) have contributed to conceptualization, critical review, and final approval of the manuscript.
Disclosures
Emine Eylem Genç, İrem Sena Saraç, Hayrunnisa Arslan and Ahmet Emre Eşkazan have nothing to disclose.
Compliance with Ethics Guidelines
This article is based on previously conducted studies and does not contain any new studies with human participants or animals performed by any of the authors.
Data Availability
Data sharing is not applicable to this article as no new datasets were created or analyzed in this study.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Genç, E.E., Saraç, İ.S., Arslan, H. et al. Diagnostic and Treatment Obstacles in Acute Myeloid Leukemia: Social, Operational, and Financial. Oncol Ther 11, 145–152 (2023). https://doi.org/10.1007/s40487-023-00229-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40487-023-00229-4