Abstract
Purpose of Review
Spurred by the successful application of structural vaccinology to other challenging bacterial and viral pathogens, we review the possibility of exploiting 3D structure computational-based recombinant antigen engineering strategies for the development of a protective melioidosis vaccine.
Recent Findings
Structure-based epitope design approaches in the melioidosis field are preliminary and applied essentially by one research network. By combining Burkholderia pseudomallei antigen 3D structures and in silico epitope discovery methods, a panel of synthetic epitope peptides were designed and tested for their B and T cell stimulatory activities. Several peptides were found to be serodiagnostic for B. pseudomallei infection and two elicited bactericidal antibodies.
Summary
A significant amount of B. pseudomallei antigen structures, epitopes, and immunological data is available. Future challenges will be to test all available B. pseudomallei epitopes, focusing on combing multiple B/T cell epitopes onto a single scaffold to generate components, stimulating both arms of the immune system.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Melioidosis is a potentially fatal disease caused by the soil-borne, intracellular Gram-negative bacillus Burkholderia pseudomallei, endemic in the tropical and subtropical regions of the world [1]. Proper diagnostic tests are unavailable, and accordingly, melioidosis is often misdiagnosed and/or underreported, leading to the global distribution of melioidosis being extensively underestimated [2•]. The worst affected areas are North Thailand and North Australia where mortality rates can reach 50 and 19%, respectively [3]. There are a number of risk factors that predispose individuals to B. pseudomallei infection, including diabetes, alcoholism, and the presence of chronic lung and/or liver pathologies. The bacterium enters the body via inhalation, ingestion, and percutaneous inoculation and causes diverse clinical outcomes including pneumonia, multiple abscesses, and fatal septicemia [2•, 4].
B. pseudomallei is resistant to all major antibiotic classes; therefore, empirically administered antibiotics given to patients in the absence of proper diagnosis are ineffective [5, 6]. Such diagnostic and therapeutic challenges, coupled to its classification as a Tier 1 select agent by the Centers for Disease Control and Prevention, have spurred research efforts directed at the discovery and development of a melioidosis vaccine and improved diagnostic tools [7].
In this context, structure-based antigen engineering is evolving as a modern-day strategy to develop improved vaccine components, and it is predicted by experts in the field to deliver future vaccines targeting complicated pathogens such as HIV [8••]. This report reviews the possibility of applying structural vaccinology (SV) approaches for the development of a melioidosis vaccine and outlines some initial SV studies made on specific B. pseudomallei antigens.
Human Immune Responses to B. pseudomallei
Due to its intracellular nature, B. pseudomallei invades and replicates inside both phagocytic and non-phagocytic cells and can do so for prolonged periods. After it is taken up by the cell vacuole, it can escape and replicate in the host cytosol. In fact, a key factor that renders treatment of melioidosis challenging is the ability of B. pseudomallei to persist, eventually leading to chronic disease. For these reasons, a protective melioidosis vaccine must contain antigenic components that induce both cell-mediated and humoral responses of the human immune system [9].
The innate immune system plays a vital and primary role in clearance of Burkholderia from the body following host invasion. A number of key cell types are activated in response to infection, e.g., macrophages and natural killer cells [10]. When replication is not prevented, chronic infection progresses, and the role of CD4+ T cells is critical for long-term infection control [11].
The adaptive immune response to B. pseudomallei infection is less understood. Melioidosis patients with acute phase infections exhibit high antibody titers associated to three IgG isotypes, confirming the importance of the antibody response [12]. A number of vaccine candidates have been tested for their capacity to induce immune protection against challenge with B. pseudomallei in vivo and will be discussed in the following section.
Current Burkholderia Vaccine Research
Tested vaccine candidates range from live-attenuated forms of the bacterium, to recombinant protein antigens and to DNA and polysaccharide subunits; however, a neutralizing vaccine has yet to be formulated [13••].
Mycobacterium tuberculosis and B. pseudomallei have shared characteristics e.g., both are intracellular, they can persist for years and they have similar histological and clinical profiles. Immunization with chronic and acute phase M. tuberculosis antigens expressed on the same polypeptide led to increased immune protection in mice challenged with infection, and human clinical trials are in progress [14,15,16]. This led Champion et al. to conduct a similar study on B. pseudomallei. Three chronic phase B. pseudomallei antigens (BPSL3369, BPSL1897, and BPSL2287) and BPSL2765, due to its link to non-recurrent incidences of melioidosis, were used to immunize mice achieving significant immune protection in comparison with the most protective vaccine candidates tested to date, namely the LolC recombinant protein and capsular polysaccharides [17••, 18, 19].
Structural Vaccinology
The 3D architecture of antigens can reveal the structures, the (a)polar and electrostatic surfaces recognized by neutralizing antibodies, and can guide the design of improved immunogens [20, 21]. Introducing structural modifications can address both practical and immunological challenges. For example, as only epitope containing portions of an antigen are required to induce an immune response, 3D structure information can be used to focus on smaller immunogenic substructures of an antigen, thus facilitating its production (e.g., when the production of large multi-domain antigens is problematic). With respect to substructure size, the smallest example is the translation of antigen portions into relatively small peptides, a strategy that potentially brings a considerable potential over traditional vaccination approaches [22]. Also, by identifying antigenic domains that specifically elicit protective immunity, rational antigen engineering based on structural considerations, can be driven. For example, when neutralizing epitope conformations are transient, or hidden by more immunodominant yet non-protective epitopes, attempts may be made to block or improve the presentation of neutralizing conformations. Epitope grafting, peptide cyclization, and stapling techniques are some useful and successful approaches that have been applied in this context [23,24,25,26,27,28].
Computationally Assisted Antigen (re)Design
The prerequisites for structure-based antigen engineering, also termed structural vaccinology, is the 3D antigen structure, obtained via experimental methods (X-ray crystallography or nuclear magnetic resonance [NMR]), or through in silico methods (homology modeling) when suitable structural homologs are available in the Protein Data Bank (PDB; www.rcsb.org), coupled to knowledge of epitope region locations. There have been significant advances in computational biology and the development of depositories hoarding large amounts of epitope data. Consequently, in addition to experimental determination, epitope sequence information may be accurately predicted using more rapid and economical sequence-based and (to a lesser extent) structure-based, in silico epitope prediction methods [29,30,31,32]. When knowledge of both epitope sequence and 3D conformation is known, structure-based antigen engineering may thus proceed to further vaccine design.
SV Approaches Applied to B. pseudomallei Antigens
Examples of SV applied to Burkholderia antigens are limited and focus on the discovery and design of antigenic epitopes as an alternative to the full antigen, using structure-based in silico epitope predictions and design. Target antigens belong to a list of 49 proteins found to be serodiagnostic for B. pseudomallei infection, based on the results of a protein microarray study presenting over 1000 in silico-predicted surface antigens, and on a subsequent study involving convalescent sera that reported 27 proteins that are specifically recognized by recovery IgGs [18, 33••].
B. pseudomallei SV studies involved the use of two 3D structure-based in silico epitope prediction methods called matrix of local coupling energies (MLCE) and electrostatic desolvation profiles (EDP) [34, 35]. These two methods detect different physico-chemical properties that are characteristic of epitopes; therefore, epitope prediction precision is improved by selecting consensus sequences.
MLCE specifically pinpoints antigenic residues that are located in dynamic and conformationally flexible regions of the antigen by identifying solvent-accessible residues that are less energetically coupled with the rest of the protein, e.g., that are not involved in stabilizing interactions within the protein fold. In other words, these regions can adapt to bind a partner, specifically an antibody, with minimal energetic expense and can tolerate well conformational changes determined by antibody recognition [35]. In contrast, EDP identifies generic protein-protein interaction interfaces by looking at surface cavities where desolvation would be energetically favored upon antibody binding [34]. Both methods were successfully tested on a Chlamydia antigen and led to the design of a cross-species immunogenic domain [36].
Peptidoglycan-Associated Lipoprotein
Peptidoglycan-associated lipoprotein (Pal) (BPSL2765) is a seroreactive recovery antigen, recognized by IgGs from patients who have had one episode of melioidosis in comparison with those with recurrent melioidosis, suggesting a role in conferring immune protection [18]. Accordingly, Pal was shown to offer limited protection in a mouse immunization study [17••, 18, 37], and together with two other antigens (FliC and the N-terminal domain of seroreactive antigen BPSS1599) was found to stimulate human memory T and B cells in a humanized melioidosis mouse model [38•].
MLCE and EDP were combined and applied to the Pal crystal structure (3D structure coordinates are available from the PDB under entry code 4B5C) and led to the identification of a highly immunogenic epitope (Pal3) that, when administered to rabbits in peptide form, elicited antibodies that were bactericidal in vitro against B. pseudomallei [39••]. In addition, Pal3 clearly discriminated between melioidosis healthy seronegative, healthy seropositive, and convalescent patient subgroups. The full-length recombinant Pal antigen did not exhibit such properties, underlining the successful outcome of this SV approach and the potential of generating a better immunogen by “extracting” epitope regions from the initial antigen and producing them separately (Fig. 1). Future applications of the Pal3 epitope may be for both vaccine and diagnostic purposes [39••].
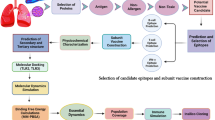
The melioidosis vaccine development puzzle. Pictorial representation of the state-of-the-art for structure-based antigen/epitope discovery and engineering, targeting B. pseudomallei (Bp) seroreactive antigens of known 3D structure. The figure illustrates how diverse advances in diverse scientific areas (structural biology, computational biology, chemistry, and immunology), must come together toward a common aim—melioidosis vaccine development—and summarizes the progress made to date. 1. 3D Bp antigen structures are used for epitope discovery; 2. Epitope sequence and structure data are obtained via in silico and in vitro methods; 3. Chemistry is used to block epitopes in the desired conformation; 4. Epitopes may be combined and presented on vaccine delivery vessels, such as outer membrane vesicles (OMVs) and nanoparticles (e.g., gold); 5. Structure-based epitope grafting may be used as an alternative to combine multiple antigens, generating “super” antigens; 6. B cell epitopes may be presented in microarray format for immunodiagnostic purposes to detect B. pseudomallei infection and infection stage; 7. In vitro immunological data is essential to determine effective B cell and T cell stimulatory activities. The missing piece of the puzzle regards in vivo protection studies with B. pseudomallei epitopes, which are still lagging behind
Oligopeptide Binding Protein
Oligopeptide-binding protein (OppA) (BPSS2141) is a member of the ATP-binding cassette (ABC) transporter family, whose members are known immunogens in Gram-negative bacteria in general. OppA, together with two other ATP-binding cassette system proteins, PotF and LolC, were shown to induce both humoral and cell-mediated immune responses in BALB/c mice challenged with B. pseudomallei [40]. A SV study analogous to that carried out on Pal was applied to the OppA crystal structure (PDB entry 3ZS6) [41]. Given the structural organization of the OppA fold into two lobes, a computational fragmentation into sub-domains prior to predictions using MLCE resulted in improved prediction accuracy. All three OppA epitopes were confirmed to be B cell epitopes, as judged by their immune sera reactivity when synthesized in peptide form [41]. One peptide, in particular, clearly distinguished between healthy seronegative, healthy seropositive, and convalescent melioidosis patient groups, implying a possible role in diagnosis.
Flagellin and the Flagellar Hook-Associated Protein
The flagellar hook-associated protein flagellar hook-associated protein (FlgK) (BPSL0280) and the flagellar subunit flagellin (FliC) (BPSL3319) are two B. pseudomallei antigens that displayed the highest seroreactivity toward immune sera from melioidosis recovery patients [18]. Antibodies raised against FliC proteins from diverse B. pseudomallei species were shown to offer passive protection in vivo [42]. Based on these findings, both antigens were deemed good SV targets. In this case and considering recent improvement in predictions made by MLCE alone, EDP was excluded from epitope predictions applied to the crystal structures of FliC (PDB entry 4CFI) and FlgK (PDB entry 4UT1); for FliC, sequence-based epitope predictions were also carried out [43, 44]. Relevant to melioidosis, two out of three FliC epitope peptides were found to be good joint T and B cell epitopes [44]. With regard to FlgK, epitopes were found to be clustered to discrete domains that may represent good starting points for the design of immunogenic domains [43].
BPSL1050
The NMR structure of seroreactive antigen of unknown function BPSL1050 (PDB entry 2MPE) was used for the application of MLCE and EDP methods, and the two designed epitope peptides successfully induced antibodies with B. pseudomallei agglutination activities that were superior to those induced by antibodies raised against recombinant BPSL1050 [45].
Future Therapeutic Applications for Discovered B. pseudomallei Epitopes
B. pseudomallei epitope peptides may serve as vaccine components (Fig. 1). Although, peptides are poorly immunogenic per se, they can be easily conjugated to other chemically diverse immunogens, e.g., individual proteins or carbohydrates, and their stability in plasma can be improved via diverse engineering/chemical modification strategies [46]. Examples of protein carriers include tetanus and diptheria toxoids, known per se to induce an immune response [47••, 48]. One means of improving their immunogenicity is to present them in vessels that prime the immune response. One such delivery vessel that is gaining attention in the field is outer membrane vesicles (OMVs) derived from the pinching off of portions of the outer membrane (OM) of Gram-negative bacteria. OMVs contain molecules that encounter the key players of the host immune response, e.g., membrane and periplasmic proteins and membrane polysaccharides, which may act as adjuvants of the immune response. OMVs isolated from B. pseudomallei 1026b have been shown to provide good protection in non-human primates, although they did not completely neutralize the bacteria [49]. Decoration of OMVs with B. pseudomallei antigens or epitopes has not yet been exploited; however, the availability of several B. pseudomallei epitopes, with both B and T cell stimulatory activities, and the intrinsic immunogenicity of B. pseudomallei OMVs suggest this to be a vital avenue to pursue in the immediate future.
Multiple-Epitope Presentation
A subunit melioidosis vaccine represents a safer alternative to a live-attenuated vaccine and efforts should focus on formulating a multivalent vaccine containing several immunogenic epitopes that lead to improved immune responses [50•]. In this context, once the sequence and 3D structure of epitopes are known, SV methods can be used to engineer multiple epitopes for presentation on a single protein scaffold. Structure-based antigen engineering, such as epitope grafting, can be used to transplant neutralizing epitopes from one antigen onto a structurally homologous region of a diverse protein scaffold [24,25,26,27]. When the protein scaffold itself is a full-length protein antigen, this can result in multiple-epitope presentation that can lead to accentuated immune responses. With regard to B. pseudomallei, in light of promising subunit protection studies with three chronic phase antigens and BPSL2765, combining T and B cell B. pseudomallei epitopes on the same scaffold could prove an ideal strategy to contemporarily induce both cell and antibody immune responses; however, no studies of this type have been reported to date.
Future Diagnostic Applications for Discovered B. pseudomallei Epitopes
Current B. pseudomallei diagnostic tests are based on lengthy bacterial culture procedures that exhibit poor sensitivity and specificity, leading to many melioidosis cases being unreported and to disease progression. Potential biomarkers include O-polysaccharide (OPS) and hemolysin co-regulated protein 1 (Hcp1) [51]. A recent report cites preliminary data on the use of a monoclonal antibody-based immunofluorescent assay (IFA) that recognizes a B. pseudomallei exopolysaccharide [52].
A more rapid method is the indirect hemagglutination assay (IHA) used to diagnose infections in Australia by measuring antibody titers to three lipopolysaccharide (LPS) types; however, its sensitivity is poor and is as low as 25% in North Eastern Thai populations, where complications are encountered due to high antibody titers in the local population, resulting from natural exposure to the non-pathogenic B. thailandensis species that co-habits with B. pseudomallei [53, 54].
The key to developing a serological-based test is the identification of specific biomarkers that do not lead to ambiguous diagnosis. In this context, the antigens identified in the protein microarray studies carried out by Felgner et al. and Suwannasaen et al. represent serodiagnostic antigens for further evaluation as biomarkers [18].
There are over 40 highly conserved species pertaining to the Burkholderia genus. Other members that are pathogenic include B. mallei, responsible for glanders in horses and other solipeds, and B. cenocepacia, which causes opportunistic infections in cystic fibrosis (CF) patients. Peptide-based immunodiagnostic tests are advantageous, as peptides are easy to produce and chemical modifications may be easily introduced to constrain peptide conformation, thus presenting peptides in microarrays in conformations that are optimally recognized by serum IgGs (Fig. 1).
The possibility of using B. pseudomallei synthetic peptide epitopes to diagnose B. cenocepacia infections in CF patients was confirmed by a recent study [55••]. Silica chips were used to present the synthetic epitope peptides from Pal, FliC, OppA, and BPSL1050 [55••]. All B. pseudomallei peptides were seroreactive against immune sera form CF patients harboring B. cenocepacia infections. Moreover, the sensitivity of this microarray was found to be excellent, and the same peptides were not recognized by IgGs from healthy controls or CF patients with different bacterial infections, such as Pseudomonas aeruginosa [55••]. For peptide-based immunodiagnostics, the extensive natural variability of Burkholderia species must be taken into consideration when selecting cross-reactive antigens. Genome sequence information for diverse Burkholderia species should be screened, and selected candidates should belong to the conserved core genome.
In a separate study, the effect of peptide conformational flexibility was evaluated relative to immune sera recognition and the elicitation of bactericidal antibodies. The more rigid α-helical conformation of Pal3, constrained by introducing a 1,4-disubstituted-1,2,3-triazole chemical staple could discriminate better between diverse melioidosis patient serotypes than the linear peptide [56•]. In contrast, the constrained peptide elicited a more limited repertoire of antibodies with reduced bactericidal properties in comparison to the linear epitope peptide, suggesting that peptide epitope design strategies should evaluate peptide conformation and dynamics, relative to its desired application [56•].
Conclusion
Based on the results, data, and recent developments that we review in this paper, we conclude that the full integration of computer-based approaches with structural biology is coming of age, and that the next few years will witness a huge increase in the use of designed biomolecular agents for immunological applications. The increasing availability of 3D B. pseudomallei antigen structures solved by X-ray crystallography or NMR, and the increasing accuracy of in silico homology models, together with computational-based epitope prediction tools raises the possibility of rapidly generating large libraries of immunoreactive peptides for immunological testing and hence, diagnostic and therapeutic potential. There are 11 structures of known seroreactive antigens (identified by Felgner et al. [33••]) deposited in the PDB; structural homologs with higher than 30% sequence identity to 18 B. pseudomallei antigens are available, together with two additional structure homologs for chronic phase antigens BPSL2287 and BPSL3369 tested in the most successful protection study carried out to date [17••]. We can also consider 3D structures pertaining to other Burkholderia species, such as B. mallei and B. cenocepacia. Such wealth of structural data can be valuable for designing improved immunogens when complemented with in vitro and in vivo immunological studies.
With regard to melioidosis diagnostics, initial findings render the notion of using a single peptide-based immunodiagnostic test to diagnose diverse Burkholderia infections a reality. Incorporating multiple epitopes from diverse bacteria raises the possibility of screening multiple bacterial infections in one shot.
SV applications to vaccine design and production have been so far limited, despite initial promising results reported for protein antigens from diverse pathogens such as the respiratory syncytial virus, Neisseria meningitidis serotype B, and Haemophilus influenzae [57]. There is no reason to suggest that a melioidosis vaccine may not be achieved using analogous approaches; however, we believe that a large multidisciplinary research effort is required; in vivo studies currently represent one of the missing pieces of the puzzle and are essential to understand whether any of the B. pseudomallei epitopes identified to date display effective protection in a vaccine formulation (Fig. 1).
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Wiersinga WJ, Currie BJ, Peacock SJ. Melioidosis. N Engl J Med. 2012;367(11):1035–44. doi:10.1056/NEJMra1204699.
• Limmathurotsakul D, Golding N, Dance DA, Messina JP, Pigott DM, Moyes CL, et al. Predicted global distribution of Burkholderia pseudomallei and burden of melioidosis. Nat Microbiol. 2016;1:15008. doi:10.1038/nmicrobiol.2015.8. Importantly, this paper highlights the immense global incidence of melioidosis and underlines why the development of a melioidosis vaccine is a must.
Limmathurotsakul D, Wongratanacheewin S, Teerawattanasook N, Wongsuvan G, Chaisuksant S, Chetchotisakd P, et al. Increasing incidence of human melioidosis in Northeast Thailand. Am J Trop Med hyg. 2010;82(6):1113–7. doi:10.4269/ajtmh.2010.10-0038.
Currie BJ, Ward L, Cheng AC. The epidemiology and clinical spectrum of melioidosis: 540 cases from the 20 year Darwin prospective study. PLoS Negl Trop Dis. 2010;4(11):e900. doi:10.1371/journal.pntd.0000900.
Harris P, Engler C, Norton R. Comparative in vitro susceptibility of Burkholderia pseudomallei to doripenem, ertapenem, tigecycline and moxifloxacin. Int J Antimicrob Agents. 2011;37(6):547–9. doi:10.1016/j.ijantimicag.2011.02.001.
Peacock SJ, Schweizer HP, Dance DA, Smith TL, Gee JE, Wuthiekanun V, et al. Management of accidental laboratory exposure to Burkholderia pseudomallei and B. mallei. Emerg Infect Dis. 2008;14(7):e2. doi:10.3201/eid1407.071501.
Moran GJ. Threats in bioterrorism. II: CDC category B and C agents. Emerg Med Clin North Am. 2002;20(2):311–30.
•• De Gregorio E, Rappuoli R. From empiricism to rational design: a personal perspective of the evolution of vaccine development. Nat Rev Immunol. 2014;14(7):505–14. doi:10.1038/nri3694. A comprehensive review written by the expert in the field about structural vaccinology, its application to date and future perspectives.
Healey GD, Elvin SJ, Morton M, Williamson ED. Humoral and cell-mediated adaptive immune responses are required for protection against Burkholderia pseudomallei challenge and bacterial clearance postinfection. Infect Immun. 2005;73(9):5945–51. doi:10.1128/IAI.73.9.5945-5951.2005.
Silva EB, Dow SW. Development of Burkholderia mallei and pseudomallei vaccines. Front Cell Infect Microbiol. 2013;3:10. doi:10.3389/fcimb.2013.00010.
Haque A, Easton A, Smith D, O'Garra A, Van Rooijen N, Lertmemongkolchai G, et al. Role of T cells in innate and adaptive immunity against murine Burkholderia pseudomallei infection. J Infect Dis. 2006;193(3):370–9. doi:10.1086/498983.
Ho M, Schollaardt T, Smith MD, Perry MB, Brett PJ, Chaowagul W, et al. Specificity and functional activity of anti-Burkholderia pseudomallei polysaccharide antibodies. Infect Immun. 1997;65(9):3648–53.
•• Titball RW, Burtnick MN, Bancroft GJ, Brett P. Burkholderia pseudomallei and Burkholderia mallei vaccines: are we close to clinical trials? Vaccine. 2017; doi:10.1016/j.vaccine.2017.03.022. Fundamental reading for a current update on where melioidosis vaccine research presently stands.
Bertholet S, Ireton GC, Ordway DJ, Windish HP, Pine SO, Kahn M, et al. A defined tuberculosis vaccine candidate boosts BCG and protects against multidrug-resistant Mycobacterium tuberculosis. Sci Transl Med. 2010;2(53):53ra74. doi:10.1126/scitranslmed.3001094.
Lin PL, Dietrich J, Tan E, Abalos RM, Burgos J, Bigbee C, et al. The multistage vaccine H56 boosts the effects of BCG to protect cynomolgus macaques against active tuberculosis and reactivation of latent Mycobacterium tuberculosis infection. J Clin Invest. 2012;122(1):303–14. doi:10.1172/JCI46252.
Montagnani C, Chiappini E, Galli L, de Martino M. Vaccine against tuberculosis: what’s new? BMC Infect Dis. 2014;14(Suppl 1):S2. doi:10.1186/1471-2334-14-S1-S2.
•• Champion OL, Gourlay LJ, Scott AE, Lassaux P, Conejero L, Perletti L, et al. Immunisation with proteins expressed during chronic murine melioidosis provides enhanced protection against disease. Vaccine. 2016;34(14):1665–71. doi:10.1016/j.vaccine.2016.02.038. Here, the first in vivo protection trial combining B. pseudomallei antigens from both chronic and acute infection phases is reported. This immunization trial has given the best results to date, with regards to protection in challenged mice.
Suwannasaen D, Mahawantung J, Chaowagul W, Limmathurotsakul D, Felgner PL, Davies H, et al. Human immune responses to Burkholderia pseudomallei characterized by protein microarray analysis. J Infect Dis. 2011;203(7):1002–11. doi:10.1093/infdis/jiq142.
Nelson M, Prior JL, Lever MS, Jones HE, Atkins TP, Titball RW. Evaluation of lipopolysaccharide and capsular polysaccharide as subunit vaccines against experimental melioidosis. J Med Microbiol. 2004;53(Pt 12):1177–82. doi:10.1099/jmm.0.45766-0.
Liljeroos L, Malito E, Ferlenghi I, Bottomley MJ. Structural and computational biology in the design of immunogenic vaccine antigens. J Immunol Res. 2015;2015:156241. doi:10.1155/2015/156241.
Malito E, Carfi A, Bottomley MJ. Protein crystallography in vaccine research and development. Int J Mol Sci. 2015;16(6):13106–40. doi:10.3390/ijms160613106.
Gori A, Longhi R, Peri C, Colombo G. Peptides for immunological purposes: design, strategies and applications. Amino Acids. 2013;45(2):257–68. doi:10.1007/s00726-013-1526-9.
Bird GH, Irimia A, Ofek G, Kwong PD, Wilson IA, Walensky LD. Stapled HIV-1 peptides recapitulate antigenic structures and engage broadly neutralizing antibodies. Nat Struct Mol Biol. 2014;21(12):1058–67. doi:10.1038/nsmb.2922.
Capelli R, Marchetti F, Tiana G, Colombo G. SAGE: a fast computational tool for linear epitope grafting onto a foreign protein scaffold. J Chem Inf Model. 2017;57(1):6–10. doi:10.1021/acs.jcim.6b00584.
McLellan JS, Correia BE, Chen M, Yang Y, Graham BS, Schief WR, et al. Design and characterization of epitope-scaffold immunogens that present the motavizumab epitope from respiratory syncytial virus. J Mol Biol. 2011;409(5):853–66. doi:10.1016/j.jmb.2011.04.044.
Mulder GE, Quarles van Ufford HL, van Ameijde J, Brouwer AJ, Kruijtzer JA, Liskamp RM. Scaffold optimization in discontinuous epitope containing protein mimics of gp120 using smart libraries. Org Biomol Chem. 2013;11(16):2676–84. doi:10.1039/c3ob27470e.
Ofek G, Guenaga FJ, Schief WR, Skinner J, Baker D, Wyatt R, et al. Elicitation of structure-specific antibodies by epitope scaffolds. Proc Natl Acad Sci U S A. 2010;107(42):17880–7. doi:10.1073/pnas.1004728107.
Timmerman P, Puijk WC, Boshuizen RS, van Dijken P, Slootstra JW, Beurskens FJ, et al. Functional reconstruction of structurally complex epitopes using CLIPSTM technology. Open Vaccine J. 2009;2:56–67.
Fleri W, Paul S, Dhanda SK, Mahajan S, Xu X, Peters B, et al. The immune epitope database and analysis resource in epitope discovery and synthetic vaccine design. Front Immunol. 2017;8:278. doi:10.3389/fimmu.2017.00278.
Lundegaard C, Lund O, Buus S, Nielsen M. Major histocompatibility complex class I binding predictions as a tool in epitope discovery. Immunology. 2010;130(3):309–18. doi:10.1111/j.1365-2567.2010.03300.x.
Malito E, Rappuoli R. Finding epitopes with computers. Chem Biol. 2013;20(10):1205–6. doi:10.1016/j.chembiol.2013.10.002.
Nielsen M, Lund O, Buus S, Lundegaard C. MHC class II epitope predictive algorithms. Immunology. 2010;130(3):319–28. doi:10.1111/j.1365-2567.2010.03268.x.
•• Felgner PL, Kayala MA, Vigil A, Burk C, Nakajima-Sasaki R, Pablo J, et al. A Burkholderia pseudomallei protein microarray reveals serodiagnostic and cross-reactive antigens. Proc Natl Acad Sci U S A. 2009;106(32):13499–504. doi:10.1073/pnas.0812080106. A list of 49 seroreactive melioidosis antigens on which all current B. pseudomallei structural vaccinology studies have been based is reported.
Fiorucci S, Zacharias M. Prediction of protein-protein interaction sites using electrostatic desolvation profiles. Biophys J. 2010;98(9):1921–30. doi:10.1016/j.bpj.2009.12.4332.
Scarabelli G, Morra G, Colombo G. Predicting interaction sites from the energetics of isolated proteins: a new approach to epitope mapping. Biophys J. 2010;98(9):1966–75. doi:10.1016/j.bpj.2010.01.014.
Soriani M, Petit P, Grifantini R, Petracca R, Gancitano G, Frigimelica E, et al. Exploiting antigenic diversity for vaccine design: the chlamydia ArtJ paradigm. J Biol Chem. 2010;285(39):30126–38. doi:10.1074/jbc.M110.118513.
Hara Y, Mohamed R, Nathan S. Immunogenic Burkholderia pseudomallei outer membrane proteins as potential candidate vaccine targets. PLoS ONE. 2009;4(8):e6496. doi:10.1371/journal.pone.0006496.
• Nithichanon A, Gourlay LJ, Bancroft GJ, Ato M, Takahashi Y, Lertmemongkolchai G. Boosting of post-exposure human T-cell and B-cell recall responses in vivo by Burkholderia pseudomallei-related proteins. Immunology. 2017;151(1):98–109. doi:10.1111/imm.12709. The identification of two FliC epitope peptides with joint T-cell and B-cell activities using computational biology is reported. This is relevant for a melioidosis vaccine where both cell-mediated and humoral responses should be triggered.
•• Gourlay LJ, Peri C, Ferrer-Navarro M, Conchillo-Sole O, Gori A, Rinchai D, et al. Exploiting the Burkholderia pseudomallei acute phase antigen BPSL2765 for structure-based epitope discovery/design in structural vaccinology. Chem Biol. 2013;20(9):1147–56. doi:10.1016/j.chembiol.2013.07.010. This is the first example of how SV was used to extract and generate a B. pseudomallei epitope with improved immunological properties in comparison to its parental antigen. This report underlines the possibility of improving the immunogenicity of any B. pseudomallei antigen with limited protective properties.
Harland DN, Chu K, Haque A, Nelson M, Walker NJ, Sarkar-Tyson M, et al. Identification of a LolC homologue in Burkholderia pseudomallei, a novel protective antigen for melioidosis. Infect Immun. 2007;75(8):4173–80. doi:10.1128/IAI.00404-07.
Lassaux P, Peri C, Ferrer-Navarro M, Gourlay LJ, Gori A, Conchillo-Sole O, et al. A structure-based strategy for epitope discovery in Burkholderia pseudomallei OppA antigen. Structure. 2013;21(1):167–75. doi:10.1016/j.str.2012.10.005.
Brett PJ, Mah DC, Woods DE. Isolation and characterization of Pseudomonas pseudomallei flagellin proteins. Infect Immun. 1994;62(5):1914–9.
Gourlay LJ, Thomas RJ, Peri C, Conchillo-Sole O, Ferrer-Navarro M, Nithichanon A, et al. From crystal structure to in silico epitope discovery in the Burkholderia pseudomallei flagellar hook-associated protein FlgK. FEBS J. 2015;282(7):1319–33. doi:10.1111/febs.13223.
Nithichanon A, Rinchai D, Gori A, Lassaux P, Peri C, Conchillio-Sole O, et al. Sequence- and structure-based immunoreactive epitope discovery for Burkholderia pseudomallei Flagellin. PLoS Negl Trop Dis. 2015;9(7):e0003917. doi:10.1371/journal.pntd.0003917.
Gaudesi D, Peri C, Quilici G, Gori A, Ferrer-Navarro M, Conchillo-Sole O, et al. Structure-based design of a B cell antigen from B. pseudomallei. ACS Chem Biol. 2015;10(3):803–12. doi:10.1021/cb500831y.
Purcell AW, McCluskey J, Rossjohn J. More than one reason to rethink the use of peptides in vaccine design. Nat Rev Drug Discov. 2007;6(5):404–14. doi:10.1038/nrd2224.
•• Black M, Trent A, Tirrell M, Olive C. Advances in the design and delivery of peptide subunit vaccines with a focus on toll-like receptor agonists. Expert Rev Vaccines. 2010;9(2):157–73. doi:10.1586/erv.09.160. A comprehensive summary of the types of delivery vessels available for peptide-based subunit vaccine components.
Putz MM, Ammerlaan W, Schneider F, Jung G, Muller CP. Humoral immune responses to a protective peptide-conjugate against measles after different prime-boost regimens. Vaccine. 2004;22(31–32):4173–82. doi:10.1016/j.vaccine.2004.06.013.
Nieves W, Petersen H, Judy BM, Blumentritt CA, Russell-Lodrigue K, Roy CJ, et al. A Burkholderia pseudomallei outer membrane vesicle vaccine provides protection against lethal sepsis. Clin Vaccine Immunol: CVI. 2014;21(5):747–54. doi:10.1128/CVI.00119-14.
• Muruato LA, Torres AG. Melioidosis: where do we stand in the development of an effective vaccine? Future Microb. 2016;11(4):477–80. doi:10.2217/fmb-2015-0018. An important paper reviewing the future outlook towards melioidosis vaccine development.
Pumpuang A, Dunachie SJ, Phokrai P, Jenjaroen K, Sintiprungrat K, Boonsilp S, et al. Comparison of O-polysaccharide and hemolysin co-regulated protein as target antigens for serodiagnosis of melioidosis. PLoS Negl Trop Dis. 2017;11(3):e0005499. doi:10.1371/journal.pntd.0005499.
Dulsuk A, Paksanont S, Sangchankoom A, Ekchariyawat P, Phunpang R, Jutrakul Y, et al. Validation of a monoclonal antibody-based immunofluorescent assay to detect Burkholderia pseudomallei in blood cultures. Trans R Soc Trop Med Hyg. 2017; doi:10.1093/trstmh/trw079.
Sorenson AE, Williams NL, Morris JL, Ketheesan N, Norton RE, Schaeffer PM. Improved diagnosis of melioidosis using a 2-dimensional immunoarray. Diagn Microbiol Infect Dis. 2013;77(3):209–15. doi:10.1016/j.diagmicrobio.2013.07.009.
Suttisunhakul V, Wuthiekanun V, Brett PJ, Khusmith S, Day NP, Burtnick MN, et al. Development of rapid enzyme-linked Immunosorbent assays for detection of antibodies to Burkholderia pseudomallei. J Clin Microbiol. 2016;54(5):1259–68. doi:10.1128/JCM.02856-15.
•• Peri C, Gori A, Gagni P, Sola L, Girelli D, Sottotetti S, et al. Evolving serodiagnostics by rationally designed peptide arrays: the Burkholderia paradigm in Cystic Fibrosis. Sci Rep. 2016;6:32873. doi:10.1038/srep32873. The use of B. pseudomallei epitope peptides for the diagnosis of both B. pseudomallei and B. cenocepacia infections in melioidosis and Cystic Fibrosis patients, is reported. This illustrates the possibility of designing cross-reactive diagnostic molecules and cross-protective vaccine components.
• Gori A, Peri C, Quilici G, Nithichanon A, Gaudesi D, Longhi R, et al. Flexible vs rigid epitope conformations for diagnostic- and vaccine-oriented applications: novel insights from the Burkholderia pseudomallei BPSL2765 Pal3 epitope. ACS Infect Dis. 2016;2(3):221–30. doi:10.1021/acsinfecdis.5b00118. The use of click-chemistry approaches to restrict peptide 3D conformation and improve their recognition in serological based immunodiagnostics, is described.
Dormitzer PR, Grandi G, Rappuoli R. Structural vaccinology starts to deliver. Nat Rev Microbiol. 2012;10(12):807–13. doi:10.1038/nrmicro2893.
Acknowledgements
We gratefully acknowledge PRIN-2015 contract number 2015JTL4HL for funding.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
This article is part of the Topical Collection on Melioidosis and Tropical Bacteriology
Rights and permissions
About this article
Cite this article
Gori, A., Bolognesi, M., Colombo, G. et al. Structural Vaccinology for Melioidosis Vaccine Design and Immunodiagnostics. Curr Trop Med Rep 4, 103–110 (2017). https://doi.org/10.1007/s40475-017-0117-3
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40475-017-0117-3