Abstract
Purpose
To provide a systematic review and meta-analysis of published literature characterizing the prognostic value of pre-treatment, volume-based FDG-PET metrics in patients with advanced NSCLC.
Methods
We conducted a systematic PubMed search to identify studies describing the prognostic value of volume-based PET metrics (total metabolic tumor volume [MTV] and/or total lesion glycolysis [TLG]) obtained prior to initiation of first-line systemic therapy for advanced NSCLC. Clinical endpoints examined were progression-free survival (PFS) and overall survival (OS). Hazard ratios for PFS and OS were taken directly from the original reports when available or extracted from survival curves. Inverse variance meta-analyses were performed to assess associations between PET metrics and clinical outcomes.
Results
Thirteen publications including 1,047 patients were included in our analysis. Patients from at least 9 studies received chemotherapy, at least 4 studies utilized targeted therapy, and only 1 study included patients treated with immunotherapy.
Random effects models demonstrated that high MTV is significantly associated with inferior PFS (HR 2.97, 95% CI 2.21–4.00, p < 0.001) and inferior OS (HR 2.73, 95% CI 2.18–3.41, p < 0.001). High TLG is also significantly associated with inferior PFS (HR 2.13, 95% CI 1.56–2.91, p < 0.001) and inferior OS (HR 2.06, 95% CI 1.75–2.44, p < 0.001).
Conclusion
Baseline PET metrics are powerful prognostic factors for advanced NSCLC patients who are treated with chemotherapy or targeted therapy. Further examination of the prognostic value of PET metrics for patients who receive first-line immunotherapy is warranted.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Lung cancer is the leading cause of cancer-related death worldwide [1], despite recent advancements in screening guidelines [2] and treatment options. Non-small cell lung cancer (NSCLC) comprises approximately 80% of all lung cancers, and most patients present with locally advanced to advanced disease. The treatment landscape for patients with advanced disease has changed considerably in recent years, with the incorporation of targeted therapy and immunotherapy into the standard of care [3,4,5,6,7,8]. Outcomes for patients with advanced disease, however, remain poor overall.
Much effort has been placed on identifying prognostic factors in NSCLC, with disease stage and performance status being among the most well-described [9]. Positron emission tomography with 18-fluorodeoxyglucose (FDG PET) is a key procedure in the workup and staging of NSCLC. Metrics extracted from PET have also emerged as potential prognostic tools. The standardized uptake value (SUV) is defined as the ratio of tissue radioactivity concentration and administered dose of radioactivity. The maximum SUV (SUVmax) is often used to quantify a lesion’s metabolic activity, however, it has not been proven to be a valuable prognostic marker. Metabolic tumor volume (MTV) and total lesion glycolysis (TLG), on the other hand, are volumetric parameters that have shown promise in predicting clinical outcomes. MTV is the sum of the volume of all voxels with an SUV above a pre-determined threshold value. TLG is calculated by multiplying the MTV and the average SUV, or SUVmean, combining both volumetric and metabolic information.
In 2015, Im et al. published a meta-analysis demonstrating the prognostic value of these parameters across all stages of non-small cell lung cancer [10]. High MTV and high TLG were associated with increased risk of all-cause mortality, with hazard ratios of 2.31 and 2.43, respectively. Our objective was to perform an updated meta-analysis of the published literature characterizing the prognostic value of pre-treatment, volume-based FDG-PET metrics in NSCLC, specifically focusing on patients with advanced disease and considering recent treatment advancements.
Materials and methods
Data search and study selection
We conducted a systematic PubMed search to identify studies describing the prognostic value of baseline volume-based PET metrics, including total MTV and/or TLG in patients with advanced NSCLC. We searched for English language studies using the search terms “positron emission tomography,” “advanced lung cancer,” “metastatic lung cancer,” “stage IV lung cancer,” and “volume.” We also searched articles related or similar to those yielded in our initial search. We included publications with advanced NSCLC, baseline FDG-PET obtained prior to initiation of first-line systemic therapy, and measurement of total body tumor burden via MTV and/or TLG. We did not include studies where only the primary tumor was measured. Since we wanted to focus on patients with advanced, or metastatic, disease, for whom the standard of care therapy has recently changed, we chose a cutoff of at least 50%, or a majority of study patients having Stage IV disease, for those studies which included a range of early and late stage NSCLC.
Data extraction and statistical analysis
We recorded study characteristics, including first author, location and year of publication, number of patients, percentage of patients having Stage IV disease, forms of treatment administered, PET techniques, and cutoffs used for high MTV and TLG. The clinical endpoints examined were progression-free survival (PFS) and overall survival (OS). Hazard ratios for PFS and OS were extracted directly from the original reports when available, or estimated indirectly from survival curves, as previously suggested by Parmar et al. [11], using customized scripts in Matlab (The Mathworks, Natick, MA). Briefly, our customized scripts function by digitizing published survival curves and inferring the timing of each patient’s clinical event or censoring. Cox proportional hazards models are then fit to the derived patient-level data. Inverse variance meta-analyses using random effects models were performed to assess associations between PET metrics and clinical outcomes. The Q Test was used to identify potential outliers within the data set.
Results
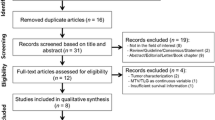
Our electronic search results yielded 416 records. We eliminated 358 entries upon initial screening and identified 58 abstracts to assess further for eligibility. Fourteen full-text articles met our eligibility criteria. We also reviewed articles that were listed as similar to these fourteen entries and upon further investigation, we identified an additional 7 publications to include for analysis. One article was subsequently found to have been retracted and was excluded. Another 7 articles had either duplicate or insufficient data. Thirteen publications were included in our final analysis (Fig. 1), incorporating 1047 patients [12,13,14,15,16,17,18,19,20,21,22,23,24].
The percentage of patients with Stage IV disease ranged from 67 to 100%. There were 3 studies we included [21, 23, 24] which did not provide a breakdown of stage, but these articles specifically stated patients with advanced disease were studied. Two of these articles specified that patients with at least Stage IIIB were included [23, 24]. Patients from at least nine studies received chemotherapy and patients from at least 4 studies were treated with targeted therapy. Patients in one study with PD-L1 scores of at least 50% were treated with first-line immunotherapy (Table 1). The majority of the included publications described methods of MTV and TLG delineation, and PET/CT techniques including patient fasting time prior to imaging, injected activity, uptake time, scan time, and reconstruction and attenuation correction details (Table 2).
The median cutoff used to define high MTV across studies was 93 cm3. Random effects models demonstrated that high MTV is significantly associated with inferior PFS (Fig. 2), with a hazard ratio (HR) of 2.97 (95% CI 2.21–4.00, p < 0.001), as well as inferior OS (Fig. 3), with a HR of 2.73 (95% CI 2.18–3.41, p < 0.001). Similar findings were seen with regards to TLG. The median cutoff used to define high TLG across studies was 444 cm3. Random effects models demonstrated that high TLG is significantly associated with inferior PFS (Fig. 4), with a HR of 2.13 (95% CI 1.56–2.91, p < 0.001) as well as inferior OS (Fig. 5), with a HR of 2.06 (95% CI 1.75–2.44, p < 0.001).
Discussion
In our analysis, high MTV and high TLG were significantly associated with worse PFS and OS outcomes in patients with advanced NSCLC. Several studies have been published in recent years examining the prognostic value of pre-treatment PET/CT in advanced NSCLC across multiple lines of therapy [25,26,27,28,29]. This meta-analysis establishes its value prior to first line therapy, thus providing a useful tool to help guide expectations at the time of initial diagnosis.
Our findings largely corroborate a previous meta-analysis published by Im, et al. Unlike the previous study, we focused on patients with advanced NSCLC, mitigating concerns that prior findings could be mediated by associations between disease stage and volumetric measures of disease burden. Additionally, we considered primary tumors and regional and distant metastases as components of patients’ disease burden, while the previous study focused only on primary lung tumors. Of note, future studies may implement machine learning technique to identify more robust PET-based prognostic variables (e.g., textural features [30]).
Of note, only one study meeting criteria for this analysis utilized immunotherapy as first-line treatment for NSCLC. Our group has begun to examine the value of volume-based PET metrics in our own cohort of patients treated with first-line immunotherapy, and has shown that MTV may be an important prognostic factor for these patients with advanced disease [31]. Additional studies that reflect this current standard of care treatment paradigm for NSCLC are sorely needed to confirm the benefit of PET/CT in the modern treatment era.
Stratification factors employed in recent randomized trials for advanced NSCLC include race, ECOG performance status, tumor histological type, region of enrollment, PD-L1 tumor proportion score (TPS), and choice of chemotherapy [3,4,5,6,7,8]. The hazard ratios for PFS and/or OS associated with these factors range from 1.23 to 2.94. MTV and TLG, as evidenced by the HR’s detailed in our study, are arguably more powerful than many of these prognostic tools. These findings support the use of volume-based measures of disease burden as stratification factors to improve clinical trial efficiency.
Aside from volume-based measures, an additional method of assessing tumor burden in advanced disease is to count the number of sites of metastases. The presence of only a few metastatic sites is termed oligometastatic disease and has been associated with a favorable prognosis. Multiple studies are exploring the role of radical local therapy for oligometastatic cancer patients [32,33,34]. Future studies with patient-level data are warranted to examine the relationship between volumetric and count-based measures of disease burden, and the merits of each of these metrics as prognostic factors should be compared.
Limitations to this study include the lack of patient-level PET data and the use of variable definitions of high/low MTV and TLG. Of note, sensitivity analyses performed after removing trials with extreme MTV and TLG cutoffs did not yield meaningful changes to the results of our meta-analyses (data not shown). Future analyses with patient-level PET data would allow more refined characterization of the relationship between disease burden measured on PET and NSCLC prognosis.
Conclusion
Baseline PET metrics are powerful prognostic factors for advanced NSCLC patients who are treated with first line therapy. Future studies are needed to examine the prognostic value of PET metrics for patients who receive first-line immunotherapy.
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Sung H et al (2021) Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 71(3):209–249
Jonas DE et al (2021) Screening for lung cancer with low-dose computed tomography: an evidence review for the us preventive services task force. JAMA 325:971
Soria JC et al (2018) Osimertinib in untreated EGFR-mutated advanced non-small-cell lung cancer. N Engl J Med 378(2):113–125
Reck M et al (2016) Pembrolizumab versus chemotherapy for PD-L1-positive non-small-cell lung cancer. N Engl J Med 375(19):1823–1833
Langer CJ et al (2016) Carboplatin and pemetrexed with or without pembrolizumab for advanced, non-squamous non-small-cell lung cancer: a randomised, phase 2 cohort of the open-label KEYNOTE-021 study. Lancet Oncol 17(11):1497–1508
Mok TSK et al (2019) Pembrolizumab versus chemotherapy for previously untreated, PD-L1-expressing, locally advanced or metastatic non-small-cell lung cancer (KEYNOTE-042): a randomised, open-label, controlled, phase 3 trial. Lancet 393(10183):1819–1830
Gandhi L et al (2018) Pembrolizumab plus chemotherapy in metastatic non-small-cell lung cancer. N Engl J Med 378(22):2078–2092
Paz-Ares L et al (2018) Pembrolizumab plus chemotherapy for squamous non-small-cell lung cancer. N Engl J Med 379(21):2040–2051
Brundage MD, Davies D, Mackillop WJ (2002) Prognostic factors in non-small cell lung cancer: a decade of progress. Chest 122(3):1037–1057
Im HJ et al (2015) Prognostic value of volumetric parameters of (18)F-FDG PET in non-small-cell lung cancer: a meta-analysis. Eur J Nucl Med Mol Imaging 42(2):241–251
Parmar MK, Torri V, Stewart L (1998) Extracting summary statistics to perform meta-analyses of the published literature for survival endpoints. Stat Med 17(24):2815–2834
Yoo SW et al (2012) Metabolic tumor volume measured by F-18 FDG PET/CT can further stratify the prognosis of patients with stage IV non-small cell lung cancer. Nucl Med Mol Imaging 46(4):286–293
Zaizen Y et al (2012) Prognostic significance of total lesion glycolysis in patients with advanced non-small cell lung cancer receiving chemotherapy. Eur J Radiol 81(12):4179–4184
Chung HW et al (2014) FDG PET/CT metabolic tumor volume and total lesion glycolysis predict prognosis in patients with advanced lung adenocarcinoma. J Cancer Res Clin Oncol 140(1):89–98
Hyun MH et al (2019) Clinical implications of circulating cell-free DNA quantification and metabolic tumor burden in advanced non-small cell lung cancer. Lung Cancer 134:158–166
Nygaard AD et al (2014) The correlation between cell-free DNA and tumour burden was estimated by PET/CT in patients with advanced NSCLC. Br J Cancer 110(2):363–368
Sharma A et al (2018) Role of various metabolic parameters derived from baseline 18F-FDG PET/CT as prognostic markers in non-small cell lung cancer patients undergoing platinum-based chemotherapy. Clin Nucl Med 43(1):e8–e17
Ho TY et al (2015) Total lesion glycolysis determined per RECIST 1.1 criteria predicts survival in EGFR mutation-negative patients with advanced lung adenocarcinoma. Clin Nucl Med 40(6):e295–e299
Han EJ et al (2015) Prognostic value of early response assessment using 18F-FDG PET/CT in chemotherapy-treated patients with non-small-cell lung cancer. Nucl Med Commun 36(12):1187–1194
Liao S et al (2012) Prognostic value of the quantitative metabolic volumetric measurement on 18F-FDG PET/CT in Stage IV nonsurgical small-cell lung cancer. Acad Radiol 19(1):69–77
Ooi H et al (2016) Fluorodeoxyglucose uptake in advanced non-small cell lung cancer with and without pulmonary lymphangitic carcinomatosis. Anticancer Res 36(8):4313–4320
Pellegrino S et al (2019) Total metabolic tumor volume by 18F-FDG PET/CT for the prediction of outcome in patients with non-small cell lung cancer. Ann Nucl Med 33(12):937–944
Wang D et al (2016) Prognostic value of baseline 18F-FDG PET/CT functional parameters in patients with advanced lung adenocarcinoma stratified by EGFR mutation status. PLoS ONE 11(6):e0158307
Seban RD et al (2020) FDG-PET biomarkers associated with long-term benefit from first-line immunotherapy in patients with advanced non-small cell lung cancer. Ann Nucl Med 34(12):968–974
Chin AL et al (2018) Prognostic value of pretreatment FDG-PET parameters in high-dose image-guided radiotherapy for oligometastatic non-small-cell lung cancer. Clin Lung Cancer 19(5):e581–e588
Olivier A et al (2014) Higher predictive value of tumour and node [18F]-FDG PET metabolic volume and TLG in advanced lung cancer under chemotherapy. Nucl Med Commun 35(9):908–915
Seban RD et al (2020) Baseline metabolic tumor burden on FDG PET/CT scans predicts outcome in advanced NSCLC patients treated with immune checkpoint inhibitors. Eur J Nucl Med Mol Imaging 47(5):1147–1157
Winther-Larsen A et al (2016) Metabolic tumor burden as marker of outcome in advanced EGFR wild-type NSCLC patients treated with erlotinib. Lung Cancer 94:81–87
Minamimoto R et al (2019) 18 F-FDG and 11 C-4DST PET/CT for evaluating response to platinum-based doublet chemotherapy in advanced non-small cell lung cancer: a prospective study. EJNMMI Res 9(1):4
Brodin NP et al (2021) Validating the SumMean. Pract Radiat Oncol 11(1):e46–e51
Andraos T et al (2020) Prognostic value of FDG-PET metrics for advanced NSCLC patients treated with first-line immunotherapy. IJROBP 108:e116–e117
Palma DA et al (2019) Stereotactic ablative radiotherapy versus standard of care palliative treatment in patients with oligometastatic cancers (SABR-COMET): a randomised, phase 2, open-label trial. Lancet 393(10185):2051–2058
Gomez DR et al (2016) Local consolidative therapy versus maintenance therapy or observation for patients with oligometastatic non-small-cell lung cancer without progression after first-line systemic therapy: a multicentre, randomised, controlled, phase 2 study. Lancet Oncol 17(12):1672–1682
Iyengar P et al (2018) Consolidative radiotherapy for limited metastatic non-small-cell lung cancer: a phase 2 randomized clinical Trial. JAMA Oncol 4(1):e173501
Funding
This work was supported by RefleXion Medical, Inc. (Hayward, CA, USA).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Dr. Berkowitz has no relevant financial or non-financial interests to disclose. Dr. Halmos is a consultant at AstraZeneca, Boehringer Ingelheim, Genentech/Roche, Pfizer, Lilly, Foundation Medicine, Guardant Health, Takeda, Novartis, Merck, Bristol-Myers Squibb, Spectrum Pharmaceuticals and TPT Therapeutics. Dr. Cheng is a consultant at AstraZeneca and Bayer, and she received research grants from Roche/Genentech, Spectrum Pharmaceuticals, and Vaccinex. Mr. Huntzinger is an employee at RefleXion Medical, where he also has stock and stock options. Dr. Ohri is a consultant at Merck and AstraZeneca.
Ethical approval
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Berkowitz, A.C., Halmos, B., Cheng, H. et al. FDG-PET metrics in advanced non-small cell lung cancer (NSCLC): a review and meta-analysis. Clin Transl Imaging 11, 381–387 (2023). https://doi.org/10.1007/s40336-023-00542-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40336-023-00542-y