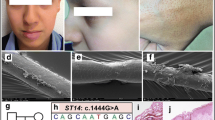
Abstract
Netherton syndrome (OMIM #256500) is a rare but severe autosomal recessive form of ichthyosis that affects the skin, hair, and immune system. The identification of SPINK5, which encodes for the serine protease inhibitor LEKTI, as the gene responsible for Netherton syndrome, enabled the search for causative mutations in Netherton syndrome patients and families. However, information regarding these mutations and their association with the pathological Netherton syndrome phenotype is scarce. Herein, we provide an up-to-date overview of 80 different mutations in exonic as well as intronic regions that have been currently identified in 172 homozygous or compound heterozygous patients from 144 families. Genotypes with mutations located more upstream in LEKTI correlate with more severe phenotypes compared with similar mutations located towards the 3′ region. Furthermore, splicing mutations and post-transcriptional mechanism of nonsense-mediated mRNA decay affect LEKTI expression in variable ways. Genotype–phenotype correlations form the basis of prenatal diagnosis in families with a history of Netherton syndrome and when consanguinity is implied.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
We provide an analysis of the genetic background of Netherton syndrome. |
A correlation of the syndrome’s clinical severity with the position and the nature of the SPINK5 mutations are explored. |
1 Introduction
Netherton syndrome (OMIM #256500) is a rare autosomal recessive disorder that is characterized by congenital ichthyosis, trichorrhexis invaginata, erythroderma, atopic manifestations, high immunoglobulin E levels, hypereosinophilia, and failure to thrive. Although its incidence is reported to be 1 per 200,000 births, it is thought that the incidence might be higher (~1 per 50,000), the underestimation may be due to the difficulty of diagnosis as the symptoms that resemble those of atopic dermatitis, congenital erythodermas, and some forms of ichthyosis [1, 2]. The prognosis of Netherton syndrome is poor for the most severely affected infants, mainly due to dehydration and infections.
The affected gene, serine peptidase inhibitor, Kazal type 5 (SPINK5), which codes for the serine protease inhibitor LEKTI, has been identified through linkage analysis and homozygosity mapping [3, 4]. SPINK5 maps at 5q32 in a cluster of SPINK genes that also includes SPINK 14, 6, 13, 7, and 9, which are located more distally (NC_000005.9) and most of which are not functionally characterized.
SPINK5 is subjected to alternative splicing in human keratinocytes, producing three transcripts (designated SPINK5f-l, SPINK5sh, and SPINK5l) that are translated into different LEKTI precursor isoforms of 145, 125, and 148 kDa, respectively. All three transcripts share the same sequence corresponding to the first 28 exons (domain 13). The splicing of exon 28 to exon 29 generates the SPINK5f-l isoform, which consists of 33 exons (15 domains). The SPINK5sh isoform is composed of 13 domains. The use of an alternative polyadenylation signal within intron 28 produces this shorter isoform of 28 exons. Finally, SPINK5l carries a 30 amino-acid residue insertion encoded by cryptic exon 28a (34 exons), as compared with the full-length transcript [5, 6]. The three alternative transcripts have similar expression patterns, while the expression levels of the main transcript, SPINK5f-l, are about 40-fold higher [5]. At the protein level, the main isoform of LEKTI is localized in the granular and uppermost spinous layers of the epidermis as well as in the most differentiated layers of all stratified epithelia [7].
The precursor main polypeptide is composed of 1064 amino acids and contains 15 putative serine protease inhibitory domains (D1–D15) of the Kazal type (Fig. 1) [8], two of which (domains D2 and D15) contain the highly conserved motif with the canonical 6 cysteine residues, whereas the others have a Kazal-related structure that are devoid of one disulfide bridge. Nevertheless, all of the domains are able to generate the typical Kazal-binding loop hairpin structure [9]. The LEKTI precursor is subjected to proteolytic cleavage, by a furin-driven proteolytic activation cascade, into fragments that contain single or multiple inhibitory domains which are secreted from the cell (Fig. 1) [10].
Proteolytic activation of LEKTI gives five final bioreactive fragments. The first proteolytic cleavage at Arg355 gives two fragments. The first fragment contains D1–D5 and is not cleaved further. The second consists of D6–D15 and is subjected to further proteolysis at Arg625, generating fragments D6–D9 and D10–D15. The fragment containing D10–D15 is not processed further, while D6–D9 is subjected to proteolysis at Arg425, producing fragments consisting of D6 and D7–D9. The latter gives the final fragments containing D7 and D8–D9 by proteolytic cleavage at Arg489 (Fig. 1) [10].
LEKTI major targets for inhibition are the most abundant kallikreins in the stratum corneum—kallikrein (KLK)5, KLK7, and KLK14 [10–13]—as well as trypsin, plasmin, subtilisin A, cathepsin G, and human neutrophil elastase [14]. Each form of LEKTI exhibits its own inhibitory activity with specific as well as overlapping targets [13]. LEKTIf-l has been shown to inhibit trypsin, plasmin, subtilisin A, cathepsin G, and neutrophil elastase but not chymotrypsin [14]. A partial recombinant form of LEKTI consisting of D6–D9 has been shown to exhibit inhibition against trypsin, subtilisin A, chymotrypsin, KLK5, and KLK7 but not cathepsin G, plasmin, or elastase [15, 16]. Multidomain D6–D9 and derived fragments D7–D9 and D8–D9 are strong inhibitors of KLK5 and KLK14 but weak inhibitors of KLK7 [10]. Single domain 6 is a potent inhibitor of both KLK5 and KLK7, and trypsin [11], although single-domain fragments (D6 and D7) show weaker inhibition than corresponding multidomain fragments [10]. The fragment D10–D15 seems to be a weak inhibitor of KLK5 and KLK14, but presents a more efficient inhibition against KLK7 [10]. The balance between serine proteases and their inhibitors ensures the integrity of the stratum corneum, the lipid barrier and the envelope as well as the proper desquamation in the epidermis. In Netherton syndrome, the reduction of LEKTI inhibition on serine proteases can explain the increased desquamation and the clinical manifestations of the skin.
The generation and analysis of the knockout Spink5 −/− mice [17–19] that reproduce key features of the syndrome have shed light on the pathogenesis of the disease. The SPINK5 loss of function mutations that have been identified in patients with Netherton syndrome cause epidermal protease hyperactivity, resulting in detachment of stratum corneum, skin barrier defect, and over-desquamation. In addition, the upregulated kallikrein KLK5 induces skin inflammation and allergy [19].
An additional consistent feature of Netherton syndrome patients is failure to thrive and/or growth retardation. This could be explained by the dysregulation of human growth hormone (hGH). SPINK5 is expressed in the pituitary gland where it inhibits many kallikreins, which are mainly expressed in tissues that are regulated by steroid hormones [20]. These kallikreins in turn participate in the proteolytic cleavage of hGH. hGH exists as a heterogeneous population of molecules in the human body. The major fraction consists of a 22 kDa single-chain form (single-hGH), which can be altered post-translationally by proteolytic cleavage. The proteolysis of single-hGH creates a two-chain form linked by a disulfide bond. This form shows a slower rate of metabolism in the circulation. Splice variants and fragments of 17 (or 16), 12, and 5 kDa have also been found in the pituitary as well as in the periphery. Patients with Netherton syndrome usually lack domains D6–D15, thus having increased kallikrein activity and hGH proteolysis [21]. Recently, a good response to hGH administration has been reported in three patients with Netherton syndrome who had growth retardation and growth hormone deficiency [22].
Currently, there is no proven cure or consensus on treatments (see, for example, Maatouk et al. [23] and Gallagher and Patel [24]). Different antibacterials are used for skin infections, emollients for the treatment of dryness, and topical corticosteroids for eczema. Furthermore, inhibitors of kallikreins involved in skin diseases have been designed and have tested negative for cytotoxicity toward healthy human keratinocytes [25]. In addition, in a recent case report, administration of the humanized antibody omalizumab resulted in the improvement of allergic skin symptoms [26]. Finally, a phase I trial study protocol has been described [27] for grafting ex vivo gene-corrected keratinocyte stem cells onto patients carrying mutations in SPINK5 (still ongoing, EU Clinical Trials Register, EudraCT number: 2011-003212-22).
In this review we summarize all of the mutations that have been identified to date in SPINK5 and their consequences at the molecular level as well as clinical details of attempting to identify phenotype–genotype correlations.
2 Methodology
Fifty publications were identified that referred to the genetic background of Netherton syndrome and we also consulted the Human Gene Mutation Database (HGDB) [28–31]. Apart from the patients’ genotypes, data regarding their clinical phenotype were also collected. In order to evaluate and compare the clinical phenotype of all patients described, we used a clinical severity score system (see Sect. 2.1). Mutations that affect the expression and/or sequence of LEKTI in different ways were reported. In the case of missense mutations, we performed pathogenicity prediction using Polyphen-2 [32].
2.1 Clinical Severity Score
Severity assessment for the Netherton patients found in the literature was performed independently by two clinical dermatologists (EZ, AP) and was based mainly on clinical manifestations, as described in the published articles, taking into account the severity of each symptom, alone and in combination with the others in order to designate the severity status. A classification of clinical severity score with increasing severity was followed in scale from 1 to 5 where 1 is given to milder phenotypes, such as faintly detectable erythema, as they were designated in the primary studies and 5 to deceased patients. A score of 2 was given to patients with skin manifestations and additional symptoms such as trichorrhexis invaginata, high immunoglobulin E, and allergies, but not infections or developmental derogations. A score of 3 corresponded to intermediate disease severity. Recurring infections was the main extra characteristic of patients in this category. However, patients without an incidence of infections who showed growth retardation or failure to thrive were also included in this class. Finally, the more severe (but not deceased) patients were classified as a 4 on the clinical severity scale. They usually suffered from severe skin manifestations in combination with growth retardation and/or failure to thrive and a burdened clinical state.
3 Results and Discussion
There are registered 84 mutations of SPINK5 that are associated with Netherton syndrome in the HGDB. However, mutations registered using the database codes CM014760, CS001465, CS121225, and CD013874 are not described in the articles cited (for more information visit the HGDB). So far, 80 different mutations in exonic as well as intronic regions have been identified in patients from 144 families (Tables 1, 2, 3). Among them, 18 mutations are nonsense, two are non-synonymous, and 34 are insertion and/or deletion mutations of one or a few nucleotides altering the reading frame and resulting in the generation of premature termination codons (PTCs). Furthermore, 24 mutations disrupting the normal splicing pattern have been found in both splice sites and distant splicing elements. The distribution of the mutation in the genomic sequence of SPINK5 in combination with their effect is shown in Fig. 2c. The absence of interfamilial gene variation in over 100 families leads to the assumption that no other gene(s) in these patients play a noticeable role affecting the clinical phenotypes.
The genomic organization and localization of the mutations in the SPINK5 gene found in Netherton syndrome patients. Schematic representation of the exon–intron structure (introns are not in scale). The last exons/introns of the gene are not shown because mutations have not been reported there. Coloring represents the functional category of the mutations, i.e., blue indicates splicing, green indicates indel, red indicates nonsense, orange indicates missense
As shown in Table 1, 86 of the 172 patients in which mutations have been described are homozygous and over 40% of them came from consanguineous families. These patients comprise a valuable source of information for genotype–phenotype correlations as they provide pure genotypes. Patients 1–36 have mutations in the first catalytic fragment of LEKTI (D1–D5), theoretically resulting in the production of a truncated protein. Truncated LEKTI could have a decreased catalytic activity, depending on the domains that are contained in the final product. Sixteen mutations that affect the first fragment of LEKTI have been identified in homozygosity, 12 of which change the codon to PTC while one disrupts the splicing pattern. Clinical phenotype is available for 48 of 49 patients, and the severity score is 4 or 5 in 39 of 48 patients (Table 1). The lower severity score (1 or 2) of five patients (patients 10, 12, 23, 24, and 32) is probably due to the restricted data [34, 36, 38, 44]. In particular, the clinical outcome of over one-quarter of these patients is death. However, this observation is an under-estimation because the information provided corresponds to clinical observations at the time. Poor updating of patients’ clinical data could decrease the real percentage of mortality in this group. Patients 37–41 produce the first catalytic fragment of LEKTI but lack either D6–D15 or D7–D15. The strongest inhibitory activity against KLK5 and KLK14 in vitro is demonstrated from D8–D11 [13]. This is in coordinance with the severity score of patients 1–41 in comparison with that of patients 42–68. Patients 1–43 (lacking D8–D15) display a score of 4 or 5 on the severity scale, while almost 75% of patients 42–68 have a score ≤3. It seems that there is a correlation between genotype and phenotype, indicating that the severity of the phenotype increases as the mutation is nearest to the –NH2 terminal of LEKTI.
Six homozygous splicing mutations have been identified (c.81+2T>A, c.1430+2T>G, c.1431–12G>A, c.1888–1G>A, c.2240+5G>A, and c.2441+3delGAGT). Immunostaining of LEKTI in patient 38 (mutation c.1430+2T>G) showed weak expression of the protein [32]. Similarly, c.1888–1G>Α reduces mRNA levels by more than 75% [4] while immunostaining showed no expression of LEKTI in three homozygous patients [37]. The mutation c.1431–12G>A has been shown using immunoblotting to lead to no protein production [46]. Finally, immunostaining showed no signal of LEKTI in a patient homozygous for c.2441+3delGAGT [59]. There is no information about the functional consequence of c.81+2T>A or c.2240+5G>A on LEKTI. For splicing mutations, nonsense-mediated mRNA decay is probably not complete and occurs in different percentages between individuals. This phenomenon could result in different symptoms between patients carrying the same mutations. For example, patients 39_1 and 39_2 scored 5 on the severity scale while patients 40_1 and 40_2 scored 3 and 4, respectively.
Two missense mutations have been reported in Netherton syndrome patients. p.D106N has been identified in patients 125 and 137 in heterozygous patients: in patient 125, it was found in combination with c.2468delA, while in patient 137 a second mutation could not be identified. p.T808I has been reported once in heterozygosity with p.R790X (patient 120). Based on pathogenicity prediction by Polyphen-2, these mutations are considered to be rather benign substitutions. These patients had relatively low severity scores (2 and 3), which one could say is in agreement with Polyphen-2 prediction.
At this point, it is necessary to consider another important parameter during the evaluation of the clinical phenotype of the patients: the quality of the healthcare system in the patient’s country. Developed countries have a higher quality level of healthcare system than developing or underdeveloped ones. As a result, patients in developing or underdeveloped countries may be at high risk of infections or poor treatment. This is mostly mirrored in the origin of the deceased patients. Nine mutations have been observed with lethal outcomes (c.153delT, c.238insG, c.375_376delAT, c.C649T, c.997C>T, c.1111C>T, c1431–12G>A, c.715insT, and 375delAT) in patients who originated from Turkey, Puerto Rico, Iran, Japan, North Europe, and Bosnia (additionally, two patients were Romanies). These mutations correspond to 23 patients (24 patients were homozygotes and one patient was heterozygous for c.715insT and c.375delAT). Mutations c.238insG and c.1431–12G>A were found to be homozygous in patients from different countries. Concerning c.238insG, two patients who originated from Turkey died in infancy. In contrast, two patients who originated from the USA and Iran presented with severe skin manifestations in combination with failure to thrive but did not die; in fact, at the time of publication of their cases, they were 8 and 3 years old, respectively. Similarly, two Romani patients carrying c.1431–12G>A died while two patients from Bosnia scored 3 (3 years old) and 4 (7 years old) on our severity scale. It is encouraging to note that if patients survive their first year of life, they have good chance of achieving a milder disease severity or even partial recovery. No deaths have been recorded in patients older than 9 months old.
The mutations identified so far are mostly distributed in Europe and Asia. However, three mutations are found with increased frequency in patients who have originated from specific regions. Mutation c.153delT shows evidence of a founder effect in Turkey, based on haplotype analysis [35]. This mutation appears in eight Turkish families and has also been identified in two compound heterozygous patients from Kosovo and Sicily. Similarly, c.392insGTGC was found in two patients from the Dominican Republic (unrelated families) both homozygous and in two African-American patients, both heterozygous. Based on haplotype analysis, it seems that c.392insGTGC is common among West Indies islanders. Finally, haplotype analysis in six families of Finnish origin carrying the c.652C>T (p.R218X) supports a founder effect in these families.
The molecular consequences of 11 different mutations on protein production, mRNA levels, and splicing pattern have been studied so far through Western blot, Northern blot, and reverse transcription–polymerase chain reaction (RT-PCR), respectively. Mutation c.715insT results in the absence of detectable LEKTI, as has been shown through Western blotting (patient 33, homozygous) [46]. Along the same lines, the c.2471_2475delAAGAGinsT mutation also inhibits LEKTI production, as it cannot be detected by Western blot in the patient 128 [61]. However, this patient is referred to as homozygous [61] and also as compound heterozygous (together with the c.2049delAGAGGAAAGAA mutation) [77]. Thus, a safe conclusion can be made only for the c.2471_2475delAAGAGinsT mutation. Mutation c.316delAG results in reduced SPINK5 expression, based on RT-PCR results in one heterozygous patient [65]. However, since no second mutation was identified in this patient, these results should be considered with caution. Splicing mutations c.283–12T/A and c.1820+53G/A cause the maintenance of the last 10 nt of intron 4 and of the first 54 nt of intron 19, respectively, in a proportion of the mature transcripts, as has been shown by RT-PCR [61]. However, the normal transcripts are possibly translated in wild-type LEKTI. Concerning the mutations found only in a heterozygous state, we can draw conclusions only for those that are found in compound heterozygosity in combination with mutations that have been studied in homozygosity and their consequences at the molecular level are known. It has been shown by Northern analysis in homozygous patients that mutation c.153delT reduces mRNA transcript levels by at least 27% [4, 34]. However, this mutation produces PTC in domain I of LEKTI, so no functional protein can be produced. Immunostaining in a homozygous patient showed no LEKTI signal [37]. Based on this, we can conclude the result of 891C>T mutation through a compound heterozygote, patient 90 (c.153delT/c.891C>T). c.891C>T has been shown by RT-PCR to cause skipping of exon 11 and PTC after 4 aa of exon 12, producing a truncated protein that contains domains 1–4, which can be identified by Western blot analysis [62]. However, the exon skipping is not complete (studied by RT-PCR), and a reduced amount of wild-type LEKTI is produced [62]. Mutation c.474G>A (p.Gln158Gln) (patient 83) also causes exon skipping (exon 6) as well as the production of other transcripts with parts of intron 6. However, in this case the exon skipping was complete (showed by RT-PCR) [66]. Interestingly, one of the mutant transcripts (in frame insertion of 108 bp) produced a protein that was expressed at a similar level to LEKTI (immunostaining results) [66]. Patient 83 also carried c.1732C>T (p.R578X), producing a truncated protein. However, no reduction of the transcript expression was observed (RT-PCR results) [66]. The mutation c.2468insA abolishes LEKTI expression as has been shown by immunostaining in the epidermis of a homozygous patient [36] and two heterozygous patients [37] and by Western blot in a compound heterozygous patient (c.2468insA combined with c.1432–13G/A) [46]. From the latter patient we can deduce that the c.1432–13G>A mutation also abolishes LEKTI expression, which is also confirmed by Western blotting [46] and immunostaining [51] in two homozygous patients (40_1, 40_2 and 39_1, respectively). Patient 80 is heterozygous for c.283−2A>T (combined with c.2468insA); due to the absence of LEKTI signal in his epidermis we can speculate that c.283−2A>T also abolishes LEKTI expression [37]. Several mutations seem to cause loss of LEKTI expression, although this is based only on immunostaining of LEKTI in the epidermis of six patients [37]. Two patients are homozygous for c.268_269insT and c.2313G>A, three are genotyped as c.238insG/p.Arg217X and c.2041delAG/p.Arg371X, and in three patients only one mutation has been identified in heterozygosity (p.Arg217X/nd and c.238insG/nd) [37]. Although immunostaining can give us a hint of the molecular consequences of the abovementioned mutations, further studies using more sensitive techniques are necessary in order to elucidate whether expression of LEKTI is significantly decreased or completely abolished.
4 Conclusions
Mutations that are located more upstream in LEKTI will produce a more severe phenotype than similar mutations located towards the 3′ region. The fact that mutations have not been identified in the 3′ region of the SPINK5 gene (downstream of exon 27) could imply that these mutations remain undetected because they don’t have a dramatic effect on LEKTI function. Mutations affecting the splicing pattern could be associated with a severe or a mild phenotype, which possibly depends on the proportion of normally spliced transcripts that are produced. The presence of normally spliced transcripts could conceivably be correlated with a milder phenotype. The post-transcriptional mechanism of nonsense-mediated mRNA decay (NMD) results in a decreased abundance of mRNAs that contain PTCs. This observation suggests that different PTC mutations in SPINK5 have variable effects on mRNA stability. Patients 133–143 are heterozygotes with different severity scores; no second mutation has been identified (Table 3). However, this could be due to hidden mutations, deeper in introns, in regions that have not been screened or, although there are no indications of this, an indication for mutations that result in the gain of function and are sufficient to cause the pathological phenotype. Additionally, several patients have a more severe phenotype than expected based on their genotype. This paradox could be explained if aberrant translation products are created that escape NMD and act in a dominant negative fashion, thus leading to the appearance of Netherton syndrome. NMD can act in either a beneficial or detrimental way; the latter if it prevents the production of proteins with some residual function and the former if it prevents the synthesis of toxic truncated proteins.
In this review of the mutations in SPINK5 found in patients with Netherton syndrome, and their consequences at the molecular level we hoped to explain the wide variety of clinical phenotypes and to shed light on the gaps in our knowledge regarding the majority of the syndrome’s mutations. Study of the functional impact of each mutation on LEKTI, and by extension on patients’ phenotype, is crucial for prenatal diagnosis in cases of a family history of Netherton syndrome and when consanguinity is implied.
References
Sprecher E, Amin S, Nielsen K, Prendiville JS, Uitto J, Richard G, et al. The spectrum of pathogenic mutations in SPINK5 in 19 families with Netherton syndrome: implications for mutation detection and first case of prenatal diagnosis. J Invest Dermatol. 2001;117(2):179–87.
Pruszkowski A, Bodemer C, Fraitag S, Teillac-Hamel D, Amoric JC, de Prost Y. Neonatal and infantile erythrodermas: a retrospective study of 51 patients. Arch Dermatol. 2000;136(7):875–80.
Chavanas S, Garner C, Bodemer C, Ali M, Teillac DH, Wilkinson J, et al. Localization of the Netherton syndrome gene to chromosome 5q32, by linkage analysis and homozygosity mapping. Am J Hum Genet. 2000;66(3):914–21.
Chavanas S, Bodemer C, Rochat A, Hamel-Teillac D, Ali M, Irvine AD, et al. Mutations in SPINK5, encoding a serine protease inhibitor, cause Netherton syndrome. Nat Genet. 2000;25(2):141–2.
Tartaglia-Polcini A, Bonnart C, Micheloni A, Cianfarani F, Andrè A, Zambruno G, et al. SPINK5, the defective gene in netherton syndrome, encodes multiple LEKTI isoforms derived from alternative pre-mRNA processing. J Invest Dermatol. 2006;126(2):315–24.
Deraison C, Bonnart C, Lopez F, Besson C, Robinson R, Jayakumar A, et al. LEKTI fragments specifically inhibit KLK5, KLK7, and KLK14 and control desquamation through a pH-dependent interaction. Mol Biol Cell. 2007;18(9):3607–19.
Bitoun E, Micheloni A, Lamant L, Bonnart C, Tartaglia-Polcini A, Cobbold C, et al. LEKTI proteolytic processing in human primary keratinocytes, tissue distribution and defective expression in Netherton syndrome. Hum Mol Genet. 2003;12(19):2417–30.
Mägert HJ, Ständker L, Kreutzmann P, Zucht HD, Reinecke M, Sommerhoff CP, et al. LEKTI, a novel 15-domain type of human serine proteinase inhibitor. J Biol Chem. 1999;274(31):21499–502.
Lauber T, Schulz A, Schweimer K, Adermann K, Marx UC. Homologous proteins with different folds: the three-dimensional structures of domains 1 and 6 of the multiple Kazal-type inhibitor LEKTI. J Mol Biol. 2003;328(1):205–19.
Fortugno P, Bresciani A, Paolini C, Pazzagli C, El Hachem M, D’Alessio M, et al. Proteolytic activation cascade of the Netherton syndrome-defective protein, LEKTI, in the epidermis: implications for skin homeostasis. J Invest Dermatol. 2011;131(11):2223–32.
Egelrud T, Brattsand M, Kreutzmann P, Walden M, Vitzithum K, Marx UC, et al. hK5 and hK7, two serine proteinases abundant in human skin, are inhibited by LEKTI domain 6. Br J Dermatol. 2005;153(6):1200–3.
Borgoño CA, Michael IP, Komatsu N, Jayakumar A, Kapadia R, Clayman GL, et al. A potential role for multiple tissue kallikrein serine proteases in epidermal desquamation. J Biol Chem. 2007;282(6):3640–52.
Deraison C, Bonnart C, Lopez F, Besson C, Robinson R, Jayakumar A, et al. LEKTI fragments specifically inhibit KLK5, KLK7, and KLK14 and control desquamation through a pH-dependent interaction. Mol Biol Cell. 2007;18(9):3607–19.
Mitsudo K, Jayakumar A, Henderson Y, Frederick MJ, Kang Y, Wang M, et al. Inhibition of serine proteinases plasmin, trypsin, subtilisin A, cathepsin G, and elastase by LEKTI: a kinetic analysis. Biochemistry. 2003;42(13):3874–81.
Jayakumar A, Kang Y, Mitsudo K, Henderson Y, Frederick MJ, Wang M, et al. Expression of LEKTI domains 6-9’ in the baculovirus expression system: recombinant LEKTI domains 6–9′ inhibit trypsin and subtilisin A. Protein Expr Purif. 2004;35(1):93–101.
Schechter NM, Choi EJ, Wang ZM, Hanakawa Y, Stanley JR, Kang Y, et al. Inhibition of human kallikreins 5 and 7 by the serine protease inhibitor lympho-epithelial Kazal-type inhibitor (LEKTI). Biol Chem. 2005;386(11):1173–84.
Descargues P, Deraison C, Bonnart C, Kreft M, Kishibe M, Ishida-Yamamoto A, et al. Spink5-deficient mice mimic Netherton syndrome through degradation of desmoglein 1 by epidermal protease hyperactivity. Nat Genet. 2005;37(1):56–65.
Yang T, Liang D, Koch PJ, Hohl D, Kheradmand F, Overbeek PA. Epidermal detachment, desmosomal dissociation, and destabilization of corneodesmosin in Spink5−/− mice. Genes Dev. 2004;18(19):2354–8.
Briot A, Deraison C, Lacroix M, Bonnart C, Robin A, Besson C, et al. Kallikrein 5 induces atopic dermatitis-like lesions through PAR2-mediated thymic stromal lymphopoietin expression in Netherton syndrome. J Exp Med. 2009;206(5):1135–47.
Clements JA, Mukhtar A, Verity K, Pullar M, McNeill P, Cummins J, et al. Kallikrein gene expression in human pituitary tissues. Clin Endocrinol. 1996;44:223–31.
Komatsu N, Saijoh K, Otsuki N, Kishi T, Micheal IP, Obiezu CV, et al. Proteolytic processing of human growth hormone by multiple tissue kallikreins and regulation by the serine protease inhibitor Kazal-Type5 (SPINK5) protein. Clin Chim Acta. 2007;377(1–2):228–36.
Aydın BK, Baş F, Tamay Z, Kılıç G, Süleyman A, Bundak R, et al. Netherton syndrome associated with growth hormone deficiency. Pediatr Dermatol. 2014;31(1):90–4.
Maatouk I, Moutran R, Tomb R. Narrowband ultraviolet B phototherapy associated with improvement in Netherton syndrome. Clin Exp Dermatol. 2012;37(4):364–6.
Gallagher JL, Patel NC. Subcutaneous immunoglobulin replacement therapy with Hizentra® is safe and effective in two infants. J Clin Immunol. 2012;32(3):474–6.
Tan X, Soualmia F, Furio L, Renard JF, Kempen I, Qin L, et al. Toward the first class of suicide inhibitors of kallikreins involved in skin diseases. J Med Chem. 2015;58(2):598–612.
Yalcin AD. A case of netherton syndrome: successful treatment with omalizumab and pulse prednisolone and its effects on cytokines and immunoglobulin levels. Immunopharmacol Immunotoxicol. 2016;38(2):162–6.
Di WL, Mellerio JE, Bernadis C, Harper J, Abdul-Wahab A, Ghani S, et al. Phase I study protocol for ex-vivo lentiviral gene therapy for the inherited skin disease, Netherton syndrome. Hum Gene Ther Clin Dev. 2013;24(4):182–90.
Stenson PD, Ball EV, Mort M, Phillips AD, Shiel JA, Thomas NS, et al. Human Gene Mutation Database (HGMD): 2003 update. Hum Mutat. 2003;21(6):577–81.
Krawczak M, Cooper DN. The human gene mutation database. Trends Genet. 1997;13(3):121–2.
Cooper DN, Ball EV, Krawczak M. The human gene mutation database. Nucleic Acids Res. 1998;26(1):285–7.
Krawczak M, Ball EV, Fenton I, Stenson PD, Abeysinghe S, Thomas N, et al. Human gene mutation database-a biomedical information and research resource. Hum Mutat. 2000;15(1):45–51.
Adzhubei IA, Schmidt S, Peshkin L, Ramensky VE, Gerasimova A, Bork P, et al. A method and server for predicting damaging missense mutations. Nat Methods. 2010;7(4):248–9.
Müller FB, Hausser I, Berg D, Casper C, Maiwald R, Jung A, et al. Genetic analysis of a severe case of Netherton syndrome and application for prenatal testing. Br J Dermatol. 2002;146(3):495–9.
Bitoun E, Chavanas S, Irvine AD, Lonie L, Bodemer C, Paradisi M, et al. Netherton syndrome: disease expression and spectrum of SPINK5 mutations in 21 families. J Invest Dermatol. 2002;118(2):352–61.
Bitoun E, Bodemer C, Amiel J, de Prost Y, Stoll C, Calvas P, et al. Prenatal diagnosis of a lethal form of Netherton syndrome by SPINK5 mutation analysis. Prenat Diagn. 2002;22(2):121–6.
Ong C, O’Toole EA, Ghali L, Malone M, Smith VV, Callard R, et al. LEKTI demonstrable by immunohistochemistry of the skin: a potential diagnostic skin test for Netherton syndrome. Br J Dermatol. 2004;151(6):1253–7.
Descargues P, Deraison C, Prost C, Fraitag S, Mazereeuw-Hautier J, D’Alessio M, et al. Corneodesmosomal cadherins are preferential targets of stratum corneum trypsin- and chymotrypsin-like hyperactivity in Netherton syndrome. J Invest Dermatol. 2006;126(7):1622–32.
Tiryakioğlu NO, Önal Z, Saygili SK, Önal H, Ersoy Tunali N. Treatment of ichthyosis and hypernatremia in a patient with Netherton syndrome with a SPINK5 c.153delT mutation using kallikrein inhibiting ointment. Int J Dermatol. doi:10.1111/ijd.13248 [Epub 2016 Feb 17].
Bingol B, Tasdemir S, Gunenc Z, Abike F, Esenkaya S, Tavukcuoglu S, et al. Prenatal diagnosis of Comel-Netherton syndrome with PGD, case report and review article. J Assist Reprod Genet. 2011;28(7):615–20.
Itoh K, Kako T, Suzuki N, Sakurai N, Sugiyama K, Yamanishi K. Severe lethal phenotype of a Japanese case of Netherton syndrome with homozygous founder mutations of SPINK5 c.375_376delAT. J Dermatol. 2015;42(12):1212–4.
Hachem JP, Wagberg F, Schmuth M, Crumrine D, Lissens W, Jayakumar A, et al. Serine protease activity and residual LEKTI expression determine phenotype in Netherton syndrome. J Invest Dermatol. 2006;126(7):1609–21.
Moskowitz DG, Fowler AJ, Heyman MB, Cohen SP, Crumrine D, Elias PM, et al. Pathophysiologic basis for growth failure in children with ichthyosis: an evaluation of cutaneous ultrastructure, epidermal permeability barrier function, and energy expenditure. J Pediatr. 2004;145(1):82–92.
Kilic G, Guler N, Ones U, Tamay Z, Guzel P. Netherton syndrome: report of identical twins presenting with severe atopic dermatitis. Eur J Pediatr. 2006;165(9):594–7.
Israeli S, Sarig O, Garty BZ, Indelman M, Bergman R, Sprecher E, et al. Molecular analysis of a series of Israeli families with Comèl-Netherton syndrome. Dermatology. 2014;228(2):183–8.
Hannula-Jouppi K, Laasanen SL, Ilander M, Furio L, Tuomiranta M, et al. Intrafamily and interfamilial phenotype variation and immature immunity in patients with Netherton syndrome and Finnish SPINK5 founder mutation. JAMA Dermatol. 2016;152(4):435–42.
Raghunath M, Tontsidou L, Oji V, Aufenvenne K, Schürmeyer-Horst F, Jayakumar A, et al. SPINK5 and Netherton syndrome: novel mutations, demonstration of missing LEKTI, and differential expression of transglutaminases. J Invest Dermatol. 2004;123(3):474–83.
Fong K, Akdeniz S, Isi H, Taskesen M, McGrath JA, Lai-Cheong JE. New homozygous SPINK5 mutation, p.Gln333X, in a Turkish pedigree with Netherton syndrome. Clin Exp Dermatol. 2011;36(4):412–5.
Macknet CA, Morkos A, Job L, Garberoglio MC, Clark RD, Macknet KD Jr, et al. An infant with Netherton syndrome and persistent pulmonary hypertension requiring extracorporeal membrane oxygenation. Pediatr Dermatol. 2008;25(3):368–72.
Diociaiuti A, Castiglia D, Fortugno P, Bartuli A, Pascucci M, Zambruno G, et al. Lethal Netherton syndrome due to homozygous p.Arg371X mutation in SPINK5. Pediatr Dermatol. 2013;30(4):e65–7.
Zhao Y, Ma ZH, Yang Y, Yang SX, Wu LS, Ding BL, et al. SPINK5 gene mutation and decreased LEKTI activity in three CN patients with Netherton’s syndrome. Clin Exp Dermatol. 2007;32(5):564–7.
Capri Y, Vanlieferinghen P, Boeuf B, Dechelotte P, Hovnanian A, Lecomte B. A lethal variant of Netherton syndrome in a large inbred family Arch Pediatr. 2011;18(3):294–8.
Akagi A, Kitoh A, Moniaga CS, Fujimoto A, Fujikawa H, Shimomura Y, et al. Case of Netherton syndrome with an elevated serum thymus and activation-regulated chemokine level. J Dermatol. 2013;40(9):752–3.
Numata S, Hamada T, Teye K, Matsuda M, Ishii N, Karashima T, et al. Complete maternal isodisomy of chromosome 5 in a Japanese patient with Netherton syndrome. J Invest Dermatol. 2014;134(3):849–52.
Renner ED, Hartl D, Rylaarsdam S, Young ML, Monaco-Shawver L, Kleiner G, et al. Comèl-Netherton syndrome defined as primary immunodeficiency. J Allergy Clin Immunol. 2009;124(3):536–43.
Chao SC, Richard G, Lee JY. Netherton syndrome: report of two TW siblings with staphylococcal scalded skin syndrome and mutation of SPINK5. Br J Dermatol. 2005;152(1):159–65.
Hosomi N, Fukai K, Nakanishi T, Funaki S, Ishii M. Caspase-1 activity of stratum corneum and serum interleukin-18 level are increased in patients with Netherton syndrome. Br J Dermatol. 2008;159(3):744–6.
Komatsu N, Takata M, Otsuki N, Ohka R, Amano O, Takehara K, et al. Elevated stratum corneum hydrolytic activity in Netherton syndrome suggests an inhibitory regulation of desquamation by SPINK5-derived peptides. J Invest Dermatol. 2002;118(3):436–43.
Komatsu N, Saijoh K, Jayakumar A, Clayman GL, Tohyama M, Suga Y, et al. Correlation between SPINK5 gene mutations and clinical manifestations in Netherton syndrome patients. J Invest Dermatol. 2008;128(5):1148–59.
Tüysüz B, Ojalvo D, Mat C, Zambruno G, Covaciu C, Castiglia D, et al. A new SPINK5 donor splice site mutation in siblings with Netherton syndrome. Acta Derm Venereol. 2010;90(1):95–6.
Di WL, Hennekam RC, Callard RE, Harper JI. A heterozygous null mutation combined with the G1258A polymorphism of SPINK5 causes impaired LEKTI function and abnormal expression of skin barrier proteins. Br J Dermatol. 2009;161(2):404–12.
Lacroix M, Lacaze-Buzy L, Furio L, Tron E, Valari M, Van der Wier G, et al. Clinical expression and new SPINK5 splicing defects in Netherton syndrome: unmasking a frequent founder synonymous mutation and unconventional intronic mutations. J Invest Dermatol. 2012;132(3 Pt 1):575–82.
Fortugno P, Grosso F, Zambruno G, Pastore S, Faletra F, Castiglia D. A synonymous mutation in SPINK5 exon 11 causes Netherton syndrome by altering exonic splicing regulatory elements. J Hum Genet. 2012;57(5):311–5.
Chatziioannidis I, Babatseva E, Patsatsi A, Galli-Tsinopoulou A, Sarri C, Lithoxopoulou M, et al. Netherton syndrome in a neonate with possible growth hormone deficiency and transient hyperaldosteronism. Case Rep Pediatr. 2015;2015:818961.
Kogut M, Salz M, Hadaschik EN, Kohlhase J, Hartmann M. New mutation leading to the full variety of typical features of the Netherton syndrome. J Dtsch Dermatol Ges. 2015;13(7):691–3.
Roedl D, Oji V, Buters JT, Behrendt H, Braun-Falco M. rAAV2-mediated restoration of LEKTI in LEKTI-deficient cells from Netherton patients. J Dermatol Sci. 2011;61(3):194–8.
Numata S, Teye K, Krol RP, Okamatsu Y, Hashikawa K, Matsuda M, et al. A compound synonymous mutation c.474G>A with p.Arg578X mutation in SPINK5 causes splicing disorder and mild phenotype in Netherton syndrome. Exp Dermatol. 2016;25(7):568–70.
Geyer AS, Ratajczak P, Pol-Rodriguez M, Millar WS, Garzon M, Richard G. Netherton syndrome with extensive skin peeling and failure to thrive due to a homozygous frameshift mutation in SPINK5. Dermatology. 2005;210(4):308–14.
Sprecher E, Tesfaye-Kedjela A, Ratajczak P, Bergman R, Richard G. Deleterious mutations in SPINK5 in a patient with congenital ichthyosiform erythroderma: molecular testing as a helpful diagnostic tool for Netherton syndrome. Clin Exp Dermatol. 2004;29(5):513–7.
Alpigiani MG, Salvati P, Schiaffino MC, Occella C, Castiglia D, Covaciu C, et al. A new SPINK5 mutation in a patient with Netherton syndrome: a case report. Pediatr Dermatol. 2012;29(4):521–2.
Mizuno Y, Suga Y, Haruna K, Muramatsu S, Hasegawa T, Kohroh K, et al. A case of a Japanese neonate with congenital ichthyosiform erythroderma diagnosed as Netherton syndrome. Clin Exp Dermatol. 2006;31(5):677–80.
Guerra L, Fortugno P, Pedicelli C, Mazzanti C, Proto V, Zambruno G, et al. Ichthyosis linearis circumflexa as the only clinical manifestation of Netherton syndrome. Acta Derm Venereol. 2015;95(6):720–4.
Mizuno Y, Suga Y, Muramatsu S, Hasegawa T, Shimizu T, Ogawa H. A Japanese infant with localized ichthyosis linearis circumflexa on the palms and soles harbouring a compound heterozygous mutation in the SPINK5 gene. Br J Dermatol. 2005;153(3):661–3.
Shimomura Y, Sato N, Kariya N, Takatsuka S, Ito M. Netherton syndrome in two Japanese siblings with a novel mutation in the SPINK5 gene: immunohistochemical studies of LEKTI and other epidermal molecules. Br J Dermatol. 2005;153(5):1026–30.
Lin SP, Huang SY, Tu ME, Wu YH, Lin CY, Lin HY, et al. Netherton syndrome: mutation analysis of two TW families. Arch Dermatol Res. 2007;299(3):145–50.
Konishi T, Tsuda T, Sakaguchi Y, Imai Y, Ito T, Hirota S, et al. Upregulation of interleukin-33 in the epidermis of two Japanese patients with Netherton syndrome. J Dermatol. 2014;41(3):258–61.
Chao SC, Tsai YM, Lee JY. A compound heterozygous mutation of the SPINK5 gene in a TW boy with Netherton syndrome. J Formos Med Assoc. 2003;102(6):418–23.
Goujon E, Beer F, Fraitag S, Hovnanian A, Vabres P. ‘Matchstick’ eyebrow hairs: a dermoscopic clue to the diagnosis of Netherton syndrome. J Eur Acad Dermatol Venereol. 2010;24(6):740–1.
Xi-Bao Z, San-Quan Z, Yu-Qing H, Yu-Wu L, Quan L, Chang-Xing L. Netherton syndrome in one CN adult with a novel mutation in the SPINK5 gene and immunohistochemical studies of LEKTI. Indian J Dermatol. 2012;57(4):265–8.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Authors C. A. Sarri, A. Roussaki-Schulze, Y. Vasilopoulos, E. Zafiriou, A. Patsatsi, C. Stamatis, P. Gidarokosta, D. Sotiriadis, T. Sarafidou, and Z. Mamuris declare that they have no conflicts of interest.
Funding
Funding was received from Postgraduate Courses “Biotechnology—Quality Assessment in Nutrition and the Environment” and “Applications of Molecular Biology—Genetics—Diagnostic Biomarkers” of the Department of Biochemistry and Biotechnology, University of Thessaly, Larissa, Greece.
Rights and permissions
About this article
Cite this article
Sarri, C.A., Roussaki-Schulze, A., Vasilopoulos, Y. et al. Netherton Syndrome: A Genotype-Phenotype Review. Mol Diagn Ther 21, 137–152 (2017). https://doi.org/10.1007/s40291-016-0243-y
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40291-016-0243-y