Abstract
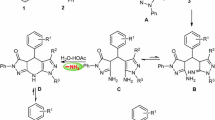
A green and efficient synthesis of 3-pyrazolyl indoles was developed by the cyclocondensation reaction of β-ethylthio-β-indolyl-α, β-unsaturated ketones with semicarbazide hydrochloride as hydrazine equivalent in the presence of 3 equiv. of PEG-400(1a/PEG mole ratio of 1:3) in reflux water. This procedure did not require toxic hydrazine and product purification, eliminating the use of toxic liquid chemicals.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Singh S., Chauhan P., Ravi M., Yadav P. P., New J. Chem., 2018, 42, 6617
Anand D., Yadav P. K., Patel O. P. S., Parmar N., Maurya R. K., Vishwakarma P., Raju K. S. R., Taneja I., Wahajuddin M., Kar S., Yadav P. P., J. Med. Chem., 2017, 60(3), 1041
Gmeiner P., Huebner H., Abu S. K. A., Heterocycles, 2003, 60(6), 1339
Zhang Y. M., Wang R., Shi Z. J., Zhang W., Synthesis, 2011, 11, 1711
Esvan Y. J., Giraud F., Pereira E., Suchaud V., Nauton L., Théry V., Dezhenkova L. G., Kaluzhny D. N., Mazov V. N., Shtil A. A., Anizon F., Moreau P., Bioorgan. Med. Chem., 2016, 24(14), 3116
Sechi M., Innocenti A., Pala N., Rogolino D., Carcelli M., Scozzafava A., Supuran C. T., Bioorgan. Med. Chem. Lett., 2012, 22(18), 5801
Cadoni R., Pala N., Lomelino C., Mahon B. P., McKenna R., Dallocchio R., Dessì A., Carcelli M., Rogolino D., Sanna V., Rassu M., Iaccarino C., Vullo D., Supuran C. T., Sechi M., ACS Med. Chem. Lett., 2017, 8(9), 941
Rao R. M., Reddy G. N., Sreeramulu J., Der. Pharma. Chemica, 2011, 3(5), 301
Somappa S. B., Biradar J. S., Rajesab P., Rahber S., Sundar M., Monatsh. Chem., 2015, 146(12), 2067
El-Mekabaty A., Etman H. A., Mosbah A., J. Heterocyclic Chem., 2016, 53, 894
Ummadi N., Gundala S., Venkatapuram P., Adivireddy P., Med. Chem. Res., 2017, 26, 1574
Gupton J. T., Telang N., Gazzo D. F., Barelli P. J., Lescalleet K. E., Fagan J. W., Mills B. J., Finzel K. L., Kanters R. P. F., Crocker K. R., Dudek S. T., Lariviere C. M., Smith S. Q., Keertikar K. M., Tetrahedron, 2013, 69(29), 5829
El-Mekabaty A., Mesbah A., Fadda A. A., J. Heterocyclic Chem., 2017, 54(2), 916
Conchon E., Aboab B., Golsteyn R. M., Cruzalegui F., Edmonds T., Léonce S., Pfeiffer B., Prudhomme M., Eur. J. Med. Chem., 2006, 41(12), 1470
Wen J. C., Bao Y., Niu Q., Yang J. Y., Fan Y. B., Li J. H., Jing Y. K., Zhao L. X., Liu D., Eur. J. Med. Chem., 2016, 109, 350
Cocconcelli G., Diodato E., Caricasole A., Gaviraghi G., Genesio E., Ghiron C., Magnoni L., Pecchioli E., Plazzi P. V., Terstappen G. C., Bioorgan. Med. Chem., 2008, 16(4), 2043
Krasavin M., Konstantinov I. O., Lett. Org. Chem., 2008, 5(7), 594
Diana P., Carbone A., Barraja P., Martorana A., Gia O., Dalla Via L., Cirrincione, G., Bioorgan. Med. Chem. Lett., 2007, 17(22), 6134
Abo-salem H. M., Ahmed K. M., El-Hallouty S., El-sawy E. R., Mandour A. H., Int. J. Pharm. Pharm. Sci., 2016, 8(12), 113
Abu Safieh K. A., El-Abadelah M. M., Sabri S. S., Abu Zarga M. H., Voelter W., Mössmer C. M., J. Heterocyclic Chem., 2001, 38(3), 623
Gmeiner P., Hübner H., Abu Safieh K. A., Fasfous I. I., El-Abadelah M. M., Sabri S. S., Voelter W., Heterocycles, 2003, 60(6), 1339
Reddy C. R., Vijaykumar J. R., Synthesis, 2013, 45, 830
Usachev B. I., Obydennov D. I., Kodess M. I., Sosnovskikh V. Y., Tetrahedron Lett., 2009, 50, 4446
Kumari S., Shekharb A., Pathak D. D., New J. Chem., 2016, 40, 5053
Usachev B. I., Obydennov D. L., Sosnovskikh V. Y., J. Fluorine Chem., 2012, 135, 278
Kumari S., Shekhar A., Pathak D. D., New J. Chem., 2016, 40, 5053
Hazra S., Deb M., Elias A. J., Green Chem., 2017, 19, 5548
Wu C., Lu L. H., Peng A. Z., Jia G. K., Peng C., Cao Z., Tang Z., He W. M., Xu X., Green Chem., 2018, 20, 3683
Pakamorė I., Rousseau J., Rousseau C., Monflier E., Szilágyi P. Á., Green Chem., 2018, 20, 5292
Xie L. Y., Peng S., Liu F., Liu Y. F., Sun M., Tang Z. L., Jiang S., Cao Z., He W. M., ACS Sustainable Chem. Eng., 2019, 77, 7193
Zhang Q. Q., Ge Y. C., Yang C. J., Zhang B. G., Deng K. J., Green Chem., 2019, 21, 5019
Dong D. W., Ouyang Y., Yu H. F., Liu Q., Liu J., Wang M., Zhu J., J. Org. Chem., 2005, 70, 4535
Yu H. F., Liao P. Q., Chemistry, 2013, 5, 435
Yu H. F., Liao P. Q., Tetrahedron Lett., 2016, 57, 2868
Qi F., Yu H. F., Wang Y. N., Lv Y., Li Y. X., Han L., Wang R., Feng X. N., Synth. Commun., 2017, 47(23), 2220
Yu H. F., Yu Z. K., Angew. Chem. Int. Ed., 2009, 48, 2929
Yu H. F., Li T. C., Liao P. Q., Synthesis, 2012, 44, 3743
Wang W. J., Yu, H. F., Synth. Commun., 2019, 49(3), 377
Marković V., Joksović M. D., Green Chem., 2015, 17, 842
Dickerson T. J., Reed N. N., Janda K. D., Chem. Rev., 2002, 102, 3325
Burley G. A., Davies D. L., Griffith G. A., Lee M., Singh K., J. Org. Chem., 2010, 75, 980
Kidwai M., Bhatnagar D., Tetrahedron Lett., 2010, 51, 2700
Jawale D. V., Pratap U. R., Bhosale M. R., Mane R. A., J. Heterocyclic Chem., 2016, 53(5), 1626
Brückner R.; Ed. Hamata M., Organic Mechanisms: Reactions, Stereochemistry and Synthesis, 3rd Edn., Springer, Berlin, 2010, 344
Author information
Authors and Affiliations
Corresponding authors
Additional information
Supported by the National Natural Science Foundation of China(No.20902010) and the Foundation of Science and Technology Research Projects of the 13th Five-year Plan of Jilin Provincial Department of Education, China(No.2016037).
Supporting information
Rights and permissions
About this article
Cite this article
Zhao, Y., Yu, H., Liao, P. et al. Green and Efficient Synthesis of 3-Pyrazolyl Indoles in Water. Chem. Res. Chin. Univ. 36, 847–852 (2020). https://doi.org/10.1007/s40242-019-0011-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40242-019-0011-8