Abstract
Purpose of Review
Our aim was to discuss the hygiene hypothesis and current thinking about it.
Recent Findings
Hygiene hypothesis suggests that an underlying reason for the recent prominent rise in allergic diseases may be linked with the diminution of the incidence of early childhood infections, which can be transmitted by means of contact with older siblings. Hygiene hypothesis suggests that contact with microbes in the environment in early life through pets, unpasteurized food, and nonsterilized water may protect from atopic diseases. Exposure to microbial pathogens and animals in infancy prevents atopy by Th1-like cytokine responses or by modifying Th2-like immune responses. Chronic infection and inflammation may protect against atopic disease. Alteration of exposure to microorganisms and access to clean water as well as issues related with food, sanitation, medical and obstetric practices, and urban lifestyle may have significant roles. Decreased exposure to bacteria in the early infancy period may shift the immune system to a predominantly Th2 state which is detected in atopic cases. Environmental exposure may lead to epigenetic changes which causes a shift of immune reactions against microorganisms.
Summary
The hygiene hypothesis implies that modern living conditions can be responsible for the rise in the incidences of allergic disease and asthma. Protective effect of food-borne microbes on allergic diseases is another remarkable issue.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
While infectious disease and hygiene have been key public health issues for centuries [1], allergic diseases have only relatively recently been regarded as a significant health burden. The marked increase in the prevalence of allergic disorders, like eczema [2], allergic rhinitis, and food allergy [3], has been a prominent trend over the past century in all regions of the world, but most characterized in Western countries [4, 5••].
Humans are ecosystems that host microbes, namely the human microbiome, and these microorganisms are necessary for maintenance of our health [6]. The immune system functions as a learning device, and it is similar to a computer that has sufficient hardware and software but has only scarce data. This lack of data must be compensated in the initial few years of life, by means of contact with microorganisms in the environment. In case this contact fails to provide this contiguity, the development of immune regulatory mechanisms may fail. Consequently, the immune system is not only responsible for elimination of harmful organisms that are responsible for infections but also may prevent the reactions triggered by agents responsible for allergic disorders [5••].
The optimal composition of the microbiota is maintained by diet [7], which needs to be diverse, and contains fiber (polysaccharides digested by the microbiota rather than the human host) [8] and polyphenols found in plant products [9,10,11]. A diet deficient in fiber can lead to progressive extinctions of important groups of organisms [12], which are cumulative and increasingly difficult to reverse in subsequent generations [13]. Polyphenols and also fish oils also appear to modulate the composition of the microbiota [5••, 14, 15].
Hygiene hypothesis implies not only an inverse relationship between the size of the family and occurrence of atopic diseases but also in general the contact with microbes in the environment in the early childhood that plays a role of protection such as pets, living in the countryside with livestock, unpasteurized food and nonsterilized water [16], and urban versus rural living [17]. Exposure to microbial pathogens and animals in infancy prevents atopy by Th1-like cytokine responses or by modifying Th2-like immune responses [18]. Yeşil et al. [19] reported that higher prevalence of anti-Helicobacter pylori antibody and higher serum levels of interleukin (IL)-10 in nonatopic subjects suggest that chronic infection and inflammation may protect against atopic disease.
The “hygiene hypothesis” suggests that a reason for the recent increase in allergic diseases can be associated with the lower incidences of infection in the early pediatric period. It is about living with siblings and getting more exposure to infections in early life. Families having pets or living in a farm getting exposure to microbes more have protection to allergies due to need for Th1 response and less of Th2. Some interpretations of the hygiene hypothesis are not upheld by further scientific data. The increase in the prevalence of atopic diseases may not necessarily correlate with the diminution of infection due to pathogenic microorganisms, nor can it be attributed to the alterations of domestic hygiene [20].
What Is Hygiene Hypothesis?
The hygiene hypothesis was initially proposed by Strachan in 1989 [21]. An inverse relationship was identified between the size of the family and occurrence of atopic disorders. Moreover, the cause of the increase in atopic disorders may be related with lower infection incidence in the early childhood period, which avoids the transmission of microorganisms. Further exploration of the concept by allergologists and immunologists yielded that the decrease in microbial exposure is a main cause for the recent increase in the incidence of atopy. Various factors including access to clean water and food as well as sanitation, medical and obstetric practice, and urban lifestyle may be responsible for the alteration of microbial exposure [21].
The original description of hygiene hypothesis [21] is based on the allergic diseases. In this perspective, the hygiene hypothesis explains the increase in prevalences of other immunologic diseases, including Th1-type I diseases and inflammatory bowel disease [22,23,24,25,26].
Strachan’s initial hypothesis for the role of the size of the family and exposure to infections in early childhood for allergic disorders has clearly advanced to merge the probable impacts of hygiene, elimination of parasitic infections, immunizations, improvements in home heating and ventilation, exposure to mites, duration of breastfeeding, diet, parental smoking, air pollution, and exposure to pets and farm animals [21, 27••].
The International Scientific Forum on Home Hygiene (www.ifh-homehygiene.org) carried out a critical review of the hypothesis to determine the implications related with hygiene [20].
The hygiene hypothesis was initially established for explaining the high prevalence of allergic diseases in Western countries. The high prevalence of atopic disorders displays a plateau in industrialized countries [28]. The asthma prevalence rate among Taiwanese children was only 1% when the first recordings were made in the 1980s, but nowadays its rate has raised to about 15% [29]. The asthma prevalence in American children has increased by 38% in the interval between 1980 and 2003 [30, 31]. Asthma prevalence, for example, increased by about 1% a year from around 1980 [32]. Among children 5–18 years of age, the increase has predominantly been among allergic individuals [33] and recent UK studies confirm that atopic, rather than nonatopic, asthma is responsible for much of the rise [34, 35].
A comparison of two British birth cohorts (1958 and 1970) showed increases from 3.1 to 6.4% for eczema and from 12 to 23.3% for hay fever at age 16 years [36].
Another point of view for the hygiene hypothesis implies that an atopic status may result in a higher prevalence of viral respiratory disease and other infections due to the deficit of Th1 response [37] or amelioration of Th1-mediated diseases by parasitic antigens and concurrent Th2 responses [38, 39].
Hygiene Hypothesis and Infectious Agents
The specific impacts of bacteria, viruses, and parasites on the immune response are dependent on the infectious agents as well as the time, severity, and duration of exposure [38]. The hygiene hypothesis that implies the preventive impact of respiratory viruses on atopic diseases is more applicable for food-borne microbes than for air-borne microorganisms [40]. Actually, respiratory viruses were linked with a higher risk of allergy [37, 41, 42]. Furthermore, differences can be emphasized in the same group of infectious microorganisms. Seropositivity for hepatitis A, H. pylori, and Toxoplasma gondii was linked with a decreased atopy prevalence, while seropositivity to Clostridium difficile, Campylobacter jejuni, and Yersinia enterocolitica was related with a definite atopy prevalence [43]. In conjunction, endotoxin was nominated as protective against atopy [44]; whereas, Staphylococcal enterotoxin had no preventive effect but was a potent inducer of IL-5 and served as a cofactor in the pathogenesis of allergy [38, 45].
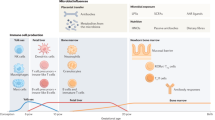
Another mechanism for explanation of the hygiene hypothesis is that exposure of infants to microbial pathogens and pets living in the farm may avoid atopy by amplification of Th1-like cytokine reactions or by modification of Th2-like immune reactions [18, 46,47,48,49]. Atopy in adulthood is characterized with production of Th2 cytokines like IL-4 and IL-5, which trigger production of immunoglobulin E (IgE) and recruitment of eosinophils which is typical for the allergic inflammatory response. On the other hand, nonatopic cases display a predominantly Th1 immune reaction. These reactions are initiated in the intrauterine period; and samples obtained from umbilical cord blood exhibit a prevailing Th2 profile [49]. Restriction of exposure to bacteria in the early infancy period shifts the immune to a predominant state of Th2 response which occurs in atopic patients [49,50,51,52] and viral infections that may cause wheezing can promote the likelihood of developing asthma [18, 53, 54].
Diminution of the variety of the infantile intestinal microbiota may have a noteworthy role in the occurence of allergy and asthma in pediatric population due to the significant impact of flora on mucosal immunity [18, 55,56,57].
A meta-analysis was performed to assess the impacts of probiotic supplementation in infants [55]. The results of this meta-analysis indicated that probiotic supplementation significantly diminished the risk of atopic sensitization, but it did not decrease the risk for asthma [55].
Human Helminths and Allergic Disease
Helminth infections are thought to induce the regulatory responses which control the inflammation. Nevertheless, these infections may elicit type-2-associated immune reactions such as helminth-specific IgE which can cross-react with environmental allergens and mediate the IgE-mediated responses. Therefore, there is a balance between the parasites’ allergic and anti-allergic influences which determine the helminth/allergy connection [58].
Multiple studies demonstrated that populations infected with helminths had reduced allergy prevalences [59,60,61]. Further evidence derived from people with hookworm infestations indicated a reduced incidence of asthma [62,63,64]. These findings reminded that experimental infections can be used on asthma patients for research [65]. The conspicuous suppression of Th2 responses by helminths had a neutral impact on concurrent allergic reactions [66]. van den Biggelaar et al. [59] demonstrated that the increase in IL-10 levels induced by schistosome infection was inversely associated with sensitivity for dust mite [67].
Family Size and Structure and Atopy
An inverse relationship was established between family size and atopy in studies which utilized markers like hay fever, skin prick positivity, and specific IgE levels [68,69,70]. Haby et al. [71] noted a protective effect of three or more older siblings for children aged 3 to 5 years. They used an extensive clinical definition for asthma, such as diagnosis of cough or wheeze in the preceding 12 months. Another study yielded that children (aged 3 to 4 years) clinically diagnosed for asthma had a smaller family size compared to healthy controls [72].
The protective effect observed in large families was more apparent than that of older siblings [73] and that of brothers compared with sisters [74, 75]. Interestingly, Bodner et al. [76] has shown that older siblings reduced the likelihood of hay fever and eczema, while the risk of asthma was decreased by the presence of younger siblings. The ALSPAC study analyzed the allergic profiles of 11,042 children [68] and a protective effect of brothers was evident for inhalant allergy. However, there were no remarkable findings for the impacts of family size. Notably, share of a bedroom by children, which is more common in large families, had a preventive role on the consecutive risk for further atopic disorders [74].
Karmaus et al. [77] proposed that the impact of siblings was achieved in the intrauterine period and the inverse relationship between infection and atopic manifestation was not necessarily causal. Another study has shown that the IgE levels in umbilical cord blood decreased with increasing birth order. Further studies [78] demonstrated that maternal IgE was reduced with birth order, which implies that the diminution in cord serum was an indirect impact of the decrease in maternal IgE. This could be an explanation for the reduced incidence of atopy in younger siblings [20].
Ball et al. [79] suggested that asthma was less common in children with ≥1 siblings at home or those who received daycare service in the initial 6 months of life. Nevertheless, children with more exposure to other children at home or in daycare suffered from more frequent wheezing attacks at the age of 2, but this tendency decreased from the age of 6 to 13 years.
Farm and Other Rural Exposure
Attention has been paid for the differences between urban and rural lifestyles. Recent publications supported that the farm, rather than the general rural environment, had a protective role against atopy. For instance, a lower incidence of hay fever has been reported in children of farmers, rather than that in other rural dwellers [46, 48, 80, 81]. Several factors including contact with animals, exposure to stables, and consumption of raw and unpasteurized farm milk were linked with the reduced prevalence of some allergies [20, 46, 48, 82].
Moreover, high levels of serum IgE were found in rural areas in Venezuela [83] and Indonesia [84], corresponding with low prevalences of allergic diseases too [85]. In contrast, the incidence of these disorders in the USA and Europe appears to have doubled in some decades of the last century, especially during the 1960s and 1970s [86].
Hygiene Hypothesis and Vitamin D Deficiency
A high correlation was observed between vitamin D deficiency, malfunction of vitamin D receptors (VDR), composition of gut microbiota, and autoimmune disorders. The biologically active form of vitamin D, 1,25(OH)2 D3, can serve as the primary ligand for VDRs, that plays a fundamental role in reduction of symptoms for autoimmune diseases. Even though the biological roles of VDR, the impacts of its genetic variants, and the influences of epigenetic profiles in its promoter region are not very well known in humans, studies in murine models seem to document that VDRs may have a critical role for suppression of symptomatology for autoimmune diseases. This effect occurs via regulation of autophagy and the production of antimicrobial peptides, such as cathelicidin and β-defensin, which may modify the composition of intestinal microbiota to a more healthy composition [87••].
Environmental Exposures and Epigenetic Changes
Environmental exposures may lead to epigenetic alterations via DNA methylation, histone acetylation, and micro-RNA changes, and these reactions modify the immune response against the microbes [88, 89]. As the multiple genome-wide association studies (GWAS) are completed worldwide, various genes associated with asthma and its related phenotypes, as well as other genes including those in the GSDMB/ORMDL3 region (17q12-q21 region) [84], NFKBI, IL1R2, LBP, IL18RAP, and TLR1 interacted with viral infections for modification of the risk for asthma and allergic diseases in the first year of life [18, 53, 90, 91].
Conclusion
The hygiene hypothesis implies that modern living conditions can be responsible for the rise in the incidences of allergic disease and asthma. A country’s gross domestic products or an individual’s affluence or exposure to infection risk factors may be important for hygiene hypothesis. It means less exposure to microbes that induce Th1 response. The hygiene hypothesis may not be applicable to all populations worldwide, but it is more relevant in populations with increasing affluence or subsequent to migration from less to more affluent countries [92].
References
Papers of particular interest, published recently, have been highlighted as: •• Of major importance
Smith V. Chapter 3. Greek hygiene. In: Smith V, editor. Clean: a history of personal hygiene and purity. Oxford: Oxford University Press; 2007. p. 74–83.
Deckers IA, McLean S, Linssen S, Mommers M, van Schayck CP, Sheikh A. Investigating international time trends in the incidence and prevalence of atopic eczema 1990–2010: a systematic review of epidemiological studies. PLoS One. 2012;7(7):e39803.
Asher MI, Montefort S, Björkstén B, Lai CK, Strachan DP, Weiland SK et al.; ISAAC Phase Three Study Group. Worldwide time trends in the prevalence of symptoms of asthma, allergic rhinoconjunctivitis, and eczema in childhood: ISAAC Phases One and Three repeat multicountry cross-sectional surveys. Lancet 2006; 368(9537): 733–743.
Prescott SL, Pawankar R, Allen KJ, Campbell DE, Sinn JK, Fiocchi A, et al. A global survey of changing patterns of food allergy burden in children. World Allergy Organ Journal. 2013;6:21.
•• Bloomfield SF, Rook GA, Scott EA, Shanahan F, Stanwell-Smith R, Turner P. Time to abandon the hygiene hypothesis: new perspectives on allergic disease, the human microbiome, infectious disease prevention and the role of targeted hygiene. Perspect Public Health. 2016;136(4):213–24. doi:10.1177/1757913916650225. Hygiene hypothesis and allergic diseases and the importance of human microbiome.
McFall-Ngai M, Hadfield MG, Bosch TC, Carey HV, Domazet-Lošo T, Douglas AE, et al. Animals in a bacterial world, a new imperative for the life sciences. Proceedings of the National Academy of Sciences U S A. 2013;110:3229–36.
Thorburn AN, Macia L, Mackay CR. Diet, metabolites, and ‘Western-lifestyle’ inflammatory diseases. Immunity. 2014;40:833–42.
Zeng H, Chi H. Metabolic control of regulatory T cell development and function. Trends Immunol. 2015;36:3–12.
Martín-Peláez S, Mosele JI, Pizarro N, Farràs M, de la Torre R, Subirana I et al. Effect of virgin olive oil and thyme phenolic compounds on blood lipid profile: implications of human gut microbiota. Eur J Nutr. 2015; doı: 10.1007/s00394-015-1063-2.
Cuervo A, Hevia A, López P, Suárez A, Diaz C, Sánchez B, et al. Phenolic compounds from red wine and coffee are associated with specific intestinal microorganisms in allergic subjects. Food Funct. 2016;7:104–9.
Vanamala JK, Knight R, Spector TD. Can your microbiome tell you what to eat? Cell Metab. 2015;22:960–1.
Sonnenburg ED, Smits SA, Tikhonov M, Higginbottom SK, Wingreen NS, Sonnenburg JL. Diet-induced extinctions in the gut microbiota compound over generations. Nature. 2016;529:212–5.
Benn CS, Melbye M, Wohlfahrt J, Bjorksten B, Aaby P. Cohort study of sibling effect, infectious diseases, and risk of atopic dermatitis during first 18 months of life. Br Med J. 2004;328:1223–8.
Caesar R, Tremaroli V, Kovatcheva-Datchary P, Cani PD, Backhed F. Crosstalk between gut microbiota and dietary lipids aggravates WAT inflammation through TLR signaling. Cell Metab. 2015;22:658–68.
Kaliannan K, Wang B, Li XY, Kim KJ, Kang JX. A host-microbiome interaction mediates the opposing effects of omega-6 and omega-3 fatty acids on metabolic endotoxemia. Sci Rep. 2015;5:11276.
von Hertzen L, Haahtela T. Disconnection of man and the soil: reason for the asthma and atopy epidemic? J Allergy Clin Immunol. 2006;117:334–44.
Kilpeläinen M, Terho EO, Helenius H, Koskenvuo M. Farm environment in childhood prevents the development of allergies. Clin Exp Allergy. 2000;30:201–8.
Daley D. The evolution of the hygiene hypothesis: the role of early-life exposures to viruses and microbes and their relationship to asthma and allergic diseases. Curr Opin Allergy Clin Immunol. 2014;14:390–6.
Yeşil Ş, Kan A, Abdulmajed O, Bakirtaş A, Sultan N, Demirsoy MS. Role of hygienic factors in the etiology of allergic disorders in children. Turk J Med Sci. 2017;47(2):627–32. doi:10.3906/sag-1603-22.
Bloomfield SF, Stanwell-Smith R, Crevel RW, Pickup J. Too clean, or not too clean: the hygiene hypothesis and home hygiene. Clin Exp Allergy. 2006;36(4):402–25.
Strachan DP. Hay fever, hygiene and household size. Br Med J. 1989;299:1259–60.
Marshall AL, Chetwynd A, Morris JA, et al. Type 1 diabetes mellitus in childhood: a matched case control study in Lancashire and Cumbria, UK. Diabet Med. 2004;21:1035–40.
Sheikh A, Smeeth L, Hubbard R. There is no evidence of an inverse relationship between TH2-mediated atopy and TH1-mediated autoimmune disorders: lack of support for the hygiene hypothesis. J Allergy Clin Immunol. 2003;111:131–5.
Sotgiu S, Pugliatti M, Sotgiu A, et al. Does the ‘hygiene hypothesis’ provide an explanation for the high prevalence of multiple sclerosis in Sardinia? Autoimmunity. 2003;36:257–60.
Danese S, Sans M, Fiocchi C. Inflammatory bowel disease: the role of environmental factors. Autoimmun Rev. 2004;3:394–400.
Johnson RJ, Hurtado A, Merszei J, et al. Hypothesis: dysregulation of immunologic balance resulting from hygiene and socioeconomic factors may influence the epidemiology and cause of glomerulonephritis worldwide. Am J Kidney Dis. 2003;42:575–81.
•• Patel S, Gruchalla R. Can we be too clean for our own good? The hygiene hypothesis reviewed. Tex Med. 2017;113(2):54–9. Update for hygiene hypothesis, a review paper.
Nowak D, Suppli Ulrik C, von Mutius E. Asthma and atopy: has peak prevalence been reached? Eur Resp I. 2004;23:359–60.
Chang TW, Wu PC, Hsu CL, Hung AF. Anti-IgE antibodies for the treatment of IgE-mediated allergic diseases. Adv Immunol. 2007;93:63–119.
Versini M, Jeandel P-Y, Bashi T, Bizarro G, Blank M, Shoenfeld Y. Unraveling the hygiene hypothesis of helminthes and autoimmunity: origins, pathophysiology and clinical applications. BMC Med. 2015;13:81–96.
Allaerts W, Chang TW. Skewed exposure to environmental antigens complements hygiene hypothesis in explaining the rise of allergy. Acta Biotheor. 2017;65(2):117–34. doi:10.1007/s10441-017-9306-7. Epub 2017 Mar 24
Warner JO. Worldwide variations in the prevalence of atopic symptoms: what does it all mean? Thorax. 1999;54(Suppl 2):546–51.
Platts-Mills TAE, Carter MC, Heymann PW. Specific and nonspecific obstructive lung disease in childhood: causes of changes in the prevalence of asthma. Environ Health Persp. 2000;108(Suppl 4):725–31.
Russell G, Helms PJ. Trends in occurrence of asthma among children and young adults. BMJ. 1997;315:1014–5.
Upton MN, McConnachie A, McSharry C, et al. Intergenerational 20 year trends in the prevalence of asthma and hay fever in adults: the Midspan family study surveys of parents and offspring. BMJ. 2000;321:88–92.
Bloomfield SF, Stanwell-Smithw R, Crevelz RWR, Pickup J. Too clean, or not too clean: the hygiene hypothesis and home hygiene. Clin Exp Allergy. 36:402–25.
Mallia P, Johnston SL. Respiratory viruses: do they protect from or induce asthma? Allergy. 2002;57:1118–29.
Bresciani M, Parisi C, Menghi G, Bonini S. The hygiene hypothesis: does it function worldwide? Curr Opin Allergy Clin Immunol. 2005;5(2):147–51.
Whary MT, Fox JG. Th1-mediated pathology in mouse models of human disease is ameliorated by concurrent Th2 responses to parasite antigens. Curr Op Med Chem. 2004;4:531–8.
Sheikh A, Strachan DP. The hygiene theory: fact or fiction? Curr Opin Otolaryngol Head Neck Surg. 2004;12:232–6.
Umetsu DT. Flu strikes the hygiene hypothesis. Nat Med. 2004;10:232–4.
Dahl ME, Dabbagh K, Liggitt D, et al. Viral-induced T helper type 1 responses enhance allergic disease by effects on lung dendritic cells. Nat Immunol. 2004;5:337–43.
Linneberg A, Ostergaard C, Tvede M, et al. IgG antibodies against microorganisms and atopic disease in Danish adults: the Copenhagen Allergy Study. J Allergy Clin Immunol. 2003;111:847–53.
Liu AH. Something old, something new: indoor endotoxin, allergens and asthma. Paediatr Respir Rev. 2004;5(Suppl 1A):S65–71.
Heaton T, Mallon D, Venaille T, Holt P. Staphylococcal enterotoxin induced IL-5 stimulation as a cofactor in the pathogenesis of atopic disease: the hygiene hypothesis in reverse? Allergy. 2003;58:252–6.
Riedler J, Braun-Fahrlander C, Eder W, et al. Exposure to farming in early life and development of asthma and allergy: a cross-sectional survey. Lancet. 2001;358:1129–33.
Rook GA, Stanford JL. Skin-test responses to mycobacteria in atopy and asthma. Allergy. 1999;54:285–6.
Von Ehrenstein OS, Von Mutius E, Illi S, et al. Reduced risk of hay fever and asthma among children of farmers. Clin Exp Allergy. 2000;30:187–93.
Prescott SL, Macaubas C, Holt BJ, et al. Transplacental priming of the human immune system to environmental allergens: universal skewing of initial T cell responses toward the Th2 cytokine profile. J Immunol. 1998;160:4730–7.
Prescott SL, Macaubas C, Smallacombe T, et al. Development of allergenspecific T-cell memory in atopic and normal children. Lancet. 1999;353:196–200.
Baldini M, Lohman IC, Halonen M, et al. A polymorphism_ in the 5′ flanking region of the CD14 gene is associated with circulating soluble CD14 levels and with total serum immunoglobulin E. Am J Respir Cell Mol Biol. 1999;20:976–83.
Baldini M, Vercelli D, Martinez FD. CD14: an example of gene by environment interaction in allergic disease. Allergy. 2002;57:188–92.
Caliskan M, Bochkov YA, Kreiner-Moller E, et al. Rhinovirus wheezing illness and genetic risk of childhood-onset asthma. N Engl J Med. 2013;368:1398–407.
Cox DW, Bizzintino J, Ferrari G, et al. Human rhinovirus species C infection in young children with acute wheeze is associated with increased acute respiratory hospital admissions. Am J Respir Crit Care Med. 2013;188:1358–64.
Elazab N, Mendy A, Gasana J, et al. Probiotic administration in early life, atopy, and asthma: a meta-analysis of clinical trials. Pediatrics. 2013;132:e666–76.
Bisgaard H, Li N, Bonnelykke K, et al. Reduced diversity of the intestinal microbiota during infancy is associated with increased risk of allergic disease at school age. J Allergy Clin Immunol. 2011;128:646–52. (e1-5)
Abrahamsson TR, Jakobsson HE, Andersson AF, et al. Low gut microbiota diversity in early infancy precedes asthma at school age. Clin Exp Allergy. 2014;44:842–50.
Santiago HC, Nutman TB. Human helminths and allergic disease: the hygiene hypothesis and beyond. Am J Trop Med Hyg. 2016;95(4):746–53. Epub 2016 Aug 29
vanden Biggelaar AH, van Ree R, Rodrigues LC, Lell B, Deelder AM, Kremsner PG, et al. Decreased atopy in children infected with Schistosoma haematobium: a role for parasite-induced interleukin-10. Lancet. 2000;356:1723–7. doi:10.1016/S0140-6736 (00)03206-2.
Cooper PJ, Chico ME, Rodrigues LC, Ordonez M, Strachan D, Griffin GE, et al. Reduced risk of atopy among school-age children infected with geohelminth parasites in a rural area of the tropics. J Allergy Clin Immunol. 2003;111:995–1000. doi:10.1067/mai.2003.1348.
Rodrigues LC, Newcombe PJ, Cunha SS, Alcantara-Neves NM, Genser B, Cruz AA, et al. Early infection with Trichuristrichiura and allergen skin test reactivity in later childhood. Clin Exp Allergy. 2008;38:1769–77. doi:10.1111/j.1365-2222.2008.03027.x.
Leonardi-Bee J, Pritchard D, Britton J. Asthma and current intestinal parasite infection: systematic review and meta-analysis. Am J Respir Crit Care Med. 2006;174:514–23. doi:10.1164/rccm.200603-331OC.
Scrivener S, Yemaneberhan H, Zebenigus M, Tilahun D, Girma S, Ali S, et al. Independent effects of intestinal parasite infection and domestic allergen exposure on risk of wheeze in Ethiopia: a nested case-control study. Lancet. 2001;358:1493–9. doi:10.1016/S0140-6736(01)06579-5.
Flohr C, Tuyen LN, Quinnell RJ, Lewis S, Minh TT, Campbell J, et al. Reduced helminth burden increases allergen skin sensitization but not clinical allergy: a randomized, double-blind, placebo-controlled trial in Vietnam. Clin Exp Allergy. 2010;40:131–42. doi:10.1111/j.1365-2222.2009.03346.x.
Feary JR, Venn AJ, Mortimer K, Brown AP, Hooi D, Falcone FH, et al. Experimental hookworm infection: a randomized placebo-controlled trial in asthma. Clin Exp Allergy. 2010;40:299–306. doi:10.1111/j.1365-2222.2009.03433.x.
Mc Sorley HJ, Maizels RM. Helminth infections and host immune regulation. Clin Microbiol Rev. 2012;25(4):585–608. doi:10.1128/CMR.05040-11.
Fitzsimmons CM, Falcone FH, Dunne DW. Helminth allergens, parasite-specific IgE, and its protective role in human immunity. Front Immunol. 2014 Feb 14;5:61. doi:10.3389/fimmu.2014.00061.eCollection 2014.
Strachan DP, Harkins LS, Johnston IDA, Anderson HR. Clinical aspects of allergic disease. Childhood antecedents of allergic sensitisation in young British adults. J Allergy Clin Immunol. 1997;99:6–12.
von Mutius E, Martinez FD, Fritzsch C, Nicolai T, Roell G, Thiemann HH. Prevalence of asthma and atopy in two areas of West and East Germany. Am J Respir Crit Care Med. 1994;49:358–64.
Jarvis D, Chinn S, Luczynska C, Burney P. The association of family size with atopy and atopic disease. Clin Exp Allergy. 1997;27:240–5.
Haby MM, Peat JK, Marks GB, Woolcock AJ, Leeder SR. Asthma in preschool children: prevalence and risk factors. Thorax. 2001;56:589–95.
Infante-Rivard C, Amre D, Gautrin D, Malo JL. Family size, daycare attendance and breastfeeding in relation to the risk of childhood asthma. Am J Epidemiol. 2001;153:653–8.
Strachan D. Socioeconomic factors and the development of allergy. Toxicol Lett. 1996;86:199–203.
Svanes C, Jarvis D, Chinn S, Burney P. Childhood environment and adult atopy: results from the European Community Respiratory Health Survey. J Allergy Clin Immunol. 1999;103:415–20.
Strachan DP, Harkins LS, Golding J. Sibship size and self reported inhalant allergy among adult women. Clin Exp Allergy. 1997;27:151–5.
Bodner C, Godden D, Seaton A. Family size, childhood infections and atopic disease. Thorax. 1998;53:28–32.
Karmaus W, Arshad H, Mattes J. Does the sibling effect have its origin in utero? Investigating birth order, cord serum immunoglobulin E concentration and allergic sensitisation at age 4 years. Am J Epidemiol. 2001;154:909–15.
Karmaus W, Arshad H, Sadeghnejad A, Twistleton R. Does maternal immunoglobulin E decrease with increasing order of live offspring? Investigation into maternal immune tolerance. Clin Exp Allergy. 2004;34:853–9.
Ball TM, Castro-Rodriguez JA, Griffith KA, Holberg CJ, Martinez FD, Wright AL. Siblings, day-care attendance and the risk of asthma and wheezing during childhood. N Engl J Med. 2000;343:538–43.
Braun-Fahrlander C, Gassner M, Grize L, et al. Prevalence of hay fever and allergic sensitisation in farmers’ children and their peers living in the same rural community. Clin Exp Allergy. 1999;29:28–34.
Gassner-Bachman M, Wuthrich B. Farmers’ children suffer less from hay fever and asthma. Dtsch Med Wochenschr. 2000;125:924–31.
Maziak W. Asthma and farming. Lancet. 2002;359(9306):623–4.
Lynch NR, Hagel IA, Palenque ME, Di Prisco MC, Escudero JE, Corao LA, et al. Relationship between helminthic infection and IgE response in atopic and nonatopic children in a tropical environment. J Allergy Clin Immunol. 1998;101:217–21.
Hamid F, Wiria AE, Wammes LJ, Kaisar MM, Djuardi Y, Versteeg SA, et al. Risk factors associated with the development of atopic sensitization in Indonesia. PLoS One. 2013;8(6):e67064. doi:10.1371/journal.pone.Print 2013.
Chang TW. Changing patterns of antigen exposure and their impact on the prevalence of allergy. Pediatr Allergy Immunol. 2014;25:733–9.
Rook GAW, Brunet LR. Microbes, immunoregulation, and the gut. Gut. 2005;54:317–20.
•• Clark A, Mach N. Role of vitamin D in the hygiene hypothesis: the interplay between vitamin D, vitamin D receptors, gut microbiota, and immune response. Front Immunol. 2016;7:627. doi:10.3389/fimmu.2016.00627.eCollection 2016. Vitamin D and hygiene hypothesis.
Abrahamsson TR, Jakobsson HE, Andersson AF, et al. Low diversity of the gut microbiota in infants with atopic eczema. J Allergy Clin Immunol. 2012;129:434–40. (40 e1-2)
Durham AL, Wiegman C, Adcock IM. Epigenetics of asthma. Biochim Biophys Acta. 1810;2011:1103–9.
Daley D, Park JE, He JQ, et al. Associations and interactions of genetic polymorphisms in innate immunity genes with early viral infections and susceptibility to asthma and asthma-related phenotypes. J Allergy Clin Immunol. 2012;130:1284–93.
Ali S, Hirschfeld AF, Mayer ML, et al. Functional genetic variation in NFKBIA and susceptibility to childhood asthma, bronchiolitis, and bronchopulmonary dysplasia. J Immunol. 2013;190:3949–58.
Leong RW, Mitrev N, Ko Y. Hygiene hypothesis: is the evidence the same all over the world? Dig Dis. 2016;34:35–42. doi:10.1159/000442922. Epub 2016 Mar 16
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
This article is part of the Topical Collection on Special Topics In Otorhinolaryngology: Otolaryngic Allergy
Rights and permissions
About this article
Cite this article
Cingi, C., Muluk, N.B. Hygiene Hypothesis: What Is the Current Thinking?. Curr Otorhinolaryngol Rep 5, 175–180 (2017). https://doi.org/10.1007/s40136-017-0158-0
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40136-017-0158-0