Abstract
In this study, field experiments with wheat (Triticum aestivum L.) were conducted consecutively for 2 years. The main objective was to determine (1) effect of seed priming with auxin (IAA), zinc (Zn) or manganese (Mn) on growth and yield parameters of wheat under dry land farming; (2) influence of on-farm priming with Azospirillum under field conditions. In this study, the response of dryland wheat yield to soaking seeds in water, 2 ppm IAA, 0.2% Mn solution and 0.2% aqueous solution of Zn with or without Azospirillum zeae inoculation was investigated under field condition. Total grain yield was significantly enhanced for water primed seeds i.e., 1273 kg ha−1 (6.8%) or seeds primed with IAA, as compared to 1191 kg ha−1 in plants from non-primed seeds. Difference in grain yield between priming with water and 2 ppm IAA was not significant when compared with 1372 kg ha−1 (15%) for seeds primed with 0.2% Mn solution and 1440 kg ha−1 (20%) for seeds primed with 0.2% Zn solution. Grain yield was significantly increased for plants inoculated with Azospirillum, 1411 kg ha−1 (8%) as compared to 1191 t ha−1 for non-Azospirillum-inoculated seeds. Yield parameters, i.e., tiller no. per m−2, grain number per spike, and 1000-grain weight, were all significantly affected by priming treatments and Azospirillum inoculation. Grains with the high nitrogen and phosphorous content were recorded for the treatment where seeds were primed with Zn and inoculated with Azospirillum. In the present study, inoculation of wheat seeds followed by priming is a significant approach to enhance grain yield and grain nutrients (N and P) content.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
On-farm seed priming has potential to improve grain yield. This technique has been adopted by resource-poor farmers for a verity of crops. Plant-growth-promoting bacteria, such as Azospirillum, have also been frequently reported that have potential to increase crop yield both under non-stress and stress conditions.
Various macro- and micronutrients are necessary for the proper plant production. In most agricultural lands, farmers tend to use macronutrients such as N, P and K to gain the higher plant yields, while micronutrients such as Fe, Zn, and Mn have been reported to improve crop growth, grain nutrient enrichment and plant establishment under farm condition [23]; however, applying these micronutrients is not common a practice by farmers in arable land as compared to macronutrients.
Micronutrients play vital role in plants as they are involved in several biological processes. Zn is involved in several biological functions and metabolic reactions [79]. Plants require a low amount of Fe and Zn for their physiological and metabolic processes. Their excess or deficit of these micronutrients induces negative consequences in leaves, root system, plant weight, overall biomass, photosynthesis, can damage the DNA, and directly affect the cell cycle and chromosomes [45], [56], [63], [46], [68].
Mn is involved in photosynthesis and nitrogen metabolism [82], [76]. More than 300 plant enzymes and some vital proteins have Zn in their structure [88], [43]. Plant requirement for micronutrients is low, and excess of these element results in damages to plant such as excessive concentration of Zn in plant causes damage to the DNA, cell cycle and chromosomes [45], [29], [46].
Wheat, as the most important crop, provides nutrition to most of the world population [24] and plays important role in human diet. Deficit of Zn and Mn in human diet is responsible for some diseases [12,14]. Transgenesis and classic breeding methods have been used to improve the micronutrient content of modern wheat cultivars [68]; however, other methods, such as foliar spraying, seed priming and soil application, have been shown to be cost-effective in this regard [26], [9], [23], [77]. Priming seeds among the above-mentioned methods has been reported to be the most effective in improving crop yield as well as the most cost-effective method in increasing micronutrient levels in plants such as wheat [78], [23]. In seed priming, seeds are allowed to be partially hydrated (hydro-priming) to trigger metabolic events without actual germination occurring and then re-dried to attain their near original weight [8]. Primed seeds usually have better germination and establishment, weight and height, root length and grain yield [32], [23], [68].
Improvement in grain yield of wheat followed by priming with Zn has been reported by Padole [61], [75], and Harris et al. [33]. Zn deficiency restricts the growth and yield of wheat [33]. Zn deficiency have been reported to be common in developed and developing countries [92] and, for instant, mentioned as the third most nutrition problem in Pakistan after N and P deficiency [67]. Seeds with higher level of micronutrients have promising results. It had been reported earlier that elevating Zn content in wheat seed improved seedling vigor and enhanced yield [93], [33]. Incidences of micronutrient deficiency in crops such as Mn and Zn seem to have enhanced worldwide in recent years [17]. Garvin et al. [25] reported that Zn contents in wheat grain are inversely correlated with yield. Previous studies showed that use of seeds with elevated micronutrient content resulted in enhanced grain yield and improved seedling vigor [69, 70], [93]. Reis et al. [68] investigated the effect of seed priming with different concentrations of Zn (1–8 mg L−1) on germination and yield of wheat. Result of this experiment showed that, however, Zn dosages higher than 8 mg L−1 had negative effects on germination and grain yield, but seed priming with lower dosages of Zn was non-cytotoxic and improved rate of germination and grain yield.
Among plant hormones, auxins were the first one to be discovered. Indole-3-acetic acid (IAA) is the most prevalent plant hormone of auxin class. IAA is critical for the regulation of the various aspects of plant growth and development [86] as it plays vital role in cell division and differentiation [73]. Lecube et al. [44] reported the role of IAA in protecting plants against oxidative stress in soybean. Environmental factors affect adversely such as drought effect on synthesis of some plant hormones, which causes limitation on physiological processes of the plant [16]. Exogenous application of these hormones at lower concentration regulates differentiation, growth, and development of the plant [55]. The application of these hormones causes the physiological processes to occur at their normal rate [28]. Auxin induces root initiation, and development which can be important under drought stress condition. Improved root system can assist plant in exploring more moisture from depth of soil and enhances absorption of the nutrients as well. Priming seeds with plant growth regulators have been suggested to enhance seed germination, seedling growth, and the yield of plant [22], [21], [91]. Priming seeds of corn [62], wheat [87], rice [91], beet [21], sunflower [36], safflower [3], and perennial grass [47] with plant growth regulators have been reported to result in enhancing yield.
Nitrogen fixing bacteria are frequent colonizer of cereal crops and grasses belonging to the genera Azospirillum, Acetobacter, Azoarcus, Enterobacter, and Herbaspirillum [50]. The genus Azospirillum was first reported by Beijerinck [6] as Spirillum and later by the group of Dr. Johanna Döbereiner in Brazil, in the 1970s [85] reclassified as Azospirillum. It is a diazotrophic bacteria associated with a wide range of plants. A variety of Azospirillum species and other PGPR have been reported to have ability to colonize the aboveground parts and root systems of various plants such as maize [51], [52], wheat [42], rice [41], sugarcane, and other plants [81]. Growth-promoting effect of Azospirillum on yield of cereals such as wheat under field conditions has been elucidated [72], [35], [96], [38]. In cereals, Azospirillum resulted in increased plant biomass, nutrient uptake and root length and volume [72]. Phytohormone production is one of the mechanisms proposed for Azospirillum by which it improves plant growth [57], [5]. Aside from this benefit, Azospirillum directly contributes to make N available to the plants [18].
The current study aimed to assess the growth and yield response of wheat to different seed priming treatments, i.e., Zn, Mn, IAA and distilled water, with or without Azospirillum zeae under dryland farming condition. A preliminary experiment was also conducted to determine the optimum concentration of priming solutions of Mn and Zn, with and without inoculation of Azospirillum. Azospirillum used in this experiment was a native strain, adapted to the region where this experiment was conducted. Numerous reports have been shown that inoculation with Azospirillum resulted in improvement of plant growth and yield of wheat [96]. PGPR can promote plant growth of the host plant through different mechanisms such as biological nitrogen fixation, production of indolic compounds, siderophore production, ACC deaminase activity, phosphate solubilization and plant anatomy modulation [57], [5], [19], [94], [95], [35], [97], [98]. Appreciate agronomical and technical management can affect PGPR performances [96]. In this study, seed priming as an agronomical management was used to investigate whether it can enhance promoting effect of Azospirillum on wheat under dryland farming.
Materials and Methods
Bacterial Strains
Indigenous bacteria used in this study were previously isolated by Dr. MJ. Zarea. It was isolated from the inside of roots of wild wheat, naturally growing in semiarid mountainous areas of west Iran. Preliminary studies conducted in our laboratory indicated that this strain was capable of IAA synthesis and nitrogen fixation. This strain, Azospirillum zeae MK433605, was able to produce indolic compounds at the rates of 37.46 ± 0.84 μg ml−1. The ability of bacterial isolate to solubilize phosphate was 1.89 ± 0.14 mg L−1. Identification of the species of the bacterial isolate based on the molecular approaches was done by Dr S. Mehnaz. Based on the 16S rRNA gene sequence analyses, bacterial strain was identified as Azospirillum zeae, (> 98% similarity with A. zeae). The 16S rRNA gene sequence was deposited in the NCBI databank, getting the accession numbers MK433605. For seed priming with bacterial strain, culture of bacterial strain was prepared in 1 L of NFb [38] using 1 mL of the overnight-grown bacterial culture as initial inoculum.
Preliminary Experiments
The optimization of concentration of Zn and Mn priming solution with and without inoculation of Azospirillum was done in pot culture, with the dosage information available in Johnson et al. 37] and [33] for Zn and [39] for Mn. Wheat seeds (Triticum aestivum L.) cultivar pishgam were first surface sterilized using 98% ethanol for 30 min, 2% sodium hypochlorite for 3 min and, then, washed thoroughly with distilled water. Seeds were primed for 6 h with aqueous solutions of ZnSO4 or MnSO4 at four concentrations, each of Zn and Mn, 0.1%, 0.2%, 0.3% and 0.4%. Non-primed seeds and seeds primed with water served as control treatments.
All seeds from priming treatments and non-primed seeds were divided into two groups and each group of seeds received 1 mL of 2.0 × 107 CFU/g seed of Azospirillum zeae, inoculum or heat-killed Azospirillum inoculum (control), and, then, air-dried in the dark for 2 h at 25 °C.. Fifteen air-dried seeds from each priming treatment inoculated with or without Azospirillum were sown at the depth of 3 cm in plastic pot filled with a field soil and then each pot were thinned to maintain 10 plants per put. The pots were placed in a greenhouse with natural light. At 30 day after sowing, seedlings of each pot were harvested and then top dry mass was measured. The experiment was a 6 × 2 factorial, arranged in a randomized block design with three replications of each treatment. Effects of seed priming and Azospirillum inoculation, and relevant interaction were analyzed, using analysis of variance, by SAS Software version 9.1.
Field Study
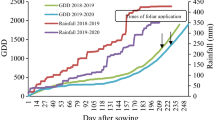
A field experiment was conducted for 2 years (2016–17) at agriculture land in Dehgolan County, Kurdistan Province (35°17′N, 47°22E, West Iran), on clay soil. The preceding crop was wheat in the previous year. Figure 1 shows the sum of rainfalls during the experiments. The experiment was a 5 × 2 factorial, arranged in a randomized complete block design with 3 replicates. Size of each plot was 3 m2, with 6 rows, each one 15 cm apart. Adjacent plots were separated by 1 m. No herbicides were applied before planting. Planting rate was 133 seeds m2. Chemical properties of the soil are presented in Table 1. The soil was slightly alkaline with low amount of organic matter. Seed beds were prepared during autumn. Sowing date was dependent on the time of first sufficient rainfall. A rate of 100 kg ha−1 nitrogen (urea) and phosphorus (50 kg ha−1) in the form of triple superphosphate was applied on a day before sowing. Further 50 kg N ha−1 was applied as top dressing. Because of low density of weeds in plots, no foliar herbicide was applied and weeds were controlled through hand-weeding. Ten treatments included (1) Mn priming + bacterial inoculums (BI), (2) Zn priming + BI, (3) IAA priming + BI, (4) water priming + BI, (5) Mn priming, (6) Zn priming, (7) IAA priming, (8) water priming, (9) no priming, and (10) no inoculum, seeds as control were planted into plots, each with three replicates.
Seed Priming and Azospirillum Inoculation
Before starting the experiment, wheat seeds (Triticum aestivum L.) cv. pishgam were surface-sterilized using 98% ethanol for 30 min, 2% sodium hypochlorite for 3 min and then washed thoroughly with distilled water. For the hormo-priming, sterile seeds were soaked for 6 h, in 2 ppm solution of IAA and air-dried under shade, prior sowing. IAA stock was prepared by adding 0.002 g IAA to a 10 mL volumetric flask and dissolved by 95% ethanol, after which sterile distilled water was added to make a total solution of 2 ppm IAA. For nutrient and hydro-priming, surface sterile seeds were soaked in distilled water, Zn (0.2%) or Mn (0.2%) solution for 6 h and then air-dried under shade to original weight to facilitate clump-free sowing. ZnSO4 and MnSO4 were used to prepare the priming solution. Zn and Mn concentrations used in field experiments were based on preliminary experiments.
Primed seeds were then air-dried and inoculated with Azospirillum. Half of each primed seeds were inoculated with bacterial inoculum, and the rest received heat-killed inoculum. Non-primed, non-inoculated seeds were also planted as control treatment. Azospirillum zeae was grown in NFb medium overnight. In inoculated seeds, 1 g of wheat seeds was inoculated with 1 mL suspension of washed overnight bacterial culture at a concentration of 2.0 × 107 CFU mL−1.
Seedling Growth and Seed Emergence
Field emergence of seeds (%) was evaluated 2 weeks after sowing. Plant samples were taken from each plot at 30th and 60th day after planting, for measurements of dry weight. Samples consisted of five randomly selected plants from each plot (fifteen plants per treatment). Plant samples were dried at 75 °C and then weighed.
Yield Component Traits
At maturity, yield components were determined from five randomly selected plants. Yield components included number of tillers, number of spikelet per spike, number of grains per spike and 1000 grain weight (g). Grain yield was recorded from 1 m2 of the plot center. 1000 grain weight was measured from five randomly selected spikes. Total grain yield was presented as kg h−1 using the following formulae (a).
Nitrogen, Phosphorus and Protein Content in Grains
The method described by Van Schouwenberg and Walinge [89] was followed to estimate the nitrogen content in grains. For the estimation of phosphorus contents, ammonium vanadate-molybdate method described by Chapman and Pratt [11] was followed. Flag leaf soluble proteins were determined according to the method described by Bradford [7].
Statistical Analysis
The data recorded on field emergence, seedling fresh weight, yield components, grain yield and P and N content of grain for 2 years were pooled to get a mean value. Effects of seed priming and Azospirillum inoculation, and relevant interaction on all data recorded were analyzed, using the PROC GLM procedure of SAS, by SAS software version 9.1. Differences among treatment means were identified using Fisher’s protected LSD0.05 analysis.
Results
Preliminary Experiment
In preliminary experiment, 30 days after sowing, plants primed with 0.2% or 0.3% Zn showed significantly high dry weight than plants from non-primed seeds, primed with 0.1% Zn or 0.4% Zn (Fig. 2). Priming with 0.4% Zn reduced seedlings dry weight (Fig. 2). Dry weight of seedlings, at 30 days after sowing, in all plants from non-primed seeds and Zn-primed seeds became heavier due to Azospirillum inoculation. Azospirillum inoculation increased dry weight of 30-d old seedlings in non-primed plants and Zn-primed plants by 15.48% and by 26.47%, respectively. Treatment seeds with 0.2% Zn + Azospirillum inoculation significantly increased weight of plants as compared to other treatments (Fig. 2).
The effect of priming wheat seeds for 6 h with various concentration rates (%) of aqueous solutions of Zn and Mn inoculated or not inoculated with Azospirillum on 30-day-old wheat seedlings dry mass. Values with the same letters are not significantly different at P < 0.05. Values are mean ± standard error (SE) (n = 3)
Priming seeds with 0.1%, 0.2%, 0.3% or 0.4% Mn increased dry weight of 30-day-old seedlings compared to non-primed seeds and seeds primed with water (Fig. 2). Using 0.4% Mn decreased dry weight of seedlings compared to 0.2% or 0.1% Mn. Accompanied Mn priming with Azospirillum inoculation resulted in an increase in dry weight of 30 days old seedlings (Fig. 2). Plants from seeds primed with 0.2% or with 0.3% Mn and inoculated with Azospirillum had higher dry weight than those from non-primed seeds, primed with water and primed with 0.1%, or with 0.4% Mn (Fig. 2).
Field Experiments
Seed priming and Azospirillum inoculation significantly influenced (P < 0.05) all parameters recorded, including early fresh seedling weight, grain yield and components, and nutrient (N and P) uptakes (except for field emergence percent). In this study, field emergence percent was not significantly affected by all treatments of seed priming with or without Azospirillum inoculation in comparison with not primed, not inoculated seeds. Results of combined analysis of the 2-year field experiments indicated that priming seeds with water, IAA (2 ppm), MnSO4 (0.2%) or with ZnSO4 (0.2%) did not reduce yield, yield components and N and P content in grain.
Early Seedling Weight
In general, fresh weight of wheat seedlings was improved with priming treatments (Zn, Mn, IAA and water) or inoculated with Azospirillum, but Azospirillum inoculation combined with seed priming significantly elevated the plant biomass (fresh weight) at 30 and 60 days after sowing. Figure 3 shows the effect of seed priming and Azospirillum inoculation on fresh seedling weight at 30 and 60 days after sowing. Plants from seeds primed with Zn, Mn, IAA or water were significantly heavier at 30 and 60 days after sowing than that of from non-primed seeds. Difference in effect of priming and Azospirillum inoculation was more visible at 60 days after sowing (Fig. 3). At 60 days after sowing, plants from seeds primed with 0.2% Zn and inoculated with Azospirillum were significantly heavier than those from non-primed seeds, Zn priming alone and from other seed priming with or without Azospirillum inoculation (Fig. 3).
Grain Yield
The interactive effect of seed priming and A. zeae inoculation on wheat grain yield is presented in Fig. 4. The main effect of priming and Azospirillum inoculation and priming × inoculation was significant for grain yield. In control plants, the grain yield was 1191 kg ha−1 and enhanced to 1411 kg ha−1 (18%) when inoculated with Azospirillum. Priming seed with Zn, Mg, IAA and water alone and in combination with A. zeae inoculation significantly (p < 0.05) enhanced the grain yield of wheat compared to values for non-primed seeds (Fig. 4). Grain yield in plants primed for 6 h in 0.2% Zn or 0.2% Mn solution was significantly higher than that in plants primed with IAA and water (Fig. 4). Grain yield in primed seeds were increased with Azospirillum inoculation. Plants from Zn priming and Azospirillum inoculation produced the highest grain yield (Fig. 4). Azospirillum enhanced grain yield of plants (ha−1) from seeds primed in Zn, Mn, IAA and water by about 49%, 35%, 33% and 24%, respectively.
In this study, the benefits of seeds priming simply with water from those produced by added IAA, Mn and Zn were separated. Figure 5 shows the percent increase in grain yield of plants primed with IAA, Mn and Zn alone in combined or without Azospirillum inoculation over grain yield of plants from seeds primed with water. Grain yield was significantly increased by priming with water alone (1.06%) over the non-primed treatment. Grain yield was significantly increased further by adding small amount of Zn or Mn in water priming (Fig. 5). However, priming with 2 ppm IAA was not significantly superior to water priming (Fig. 5). Percent increase in grain yield was significantly higher when Azospirillum inoculation was accompanied with priming agent, IAA, Mn and Zn (Fig. 5). Supplying 0.2% Zn through water priming and then inoculation with Azospirillum gained an additional increase in grain yield of 35% as compared to water priming alone (Fig. 5).
Yield Components
Although the grain number per spike was not significantly affected by the seed priming treatments, it was significantly enhanced by Azospirillum inoculation (Table 2). Plants from seeds inoculated with Azospirillum produced, on average, 5.29% higher grain number per spike as compared to non-inoculation treatment. Azospirillum inoculation resulted in improving effect on plants in terms of grain no. per spike. When seed priming combined with Azospirillum inoculation, enhancing in ration of grain no. per spike was more pronounced (Table 2). Azospirillum increased grain number per spike of plants from Zn, Mn, IAA and water priming treatments from 20.28 to 21.89 (7.93%), 20.11 to 22.64 (12.59%), 20.61 to 24.58 (19.27%) and 21.3 to 26.43 (24.09%), respectively. The highest grain number per spike was recorded for plants primed with 0.2% Zn solution for 6 h and inoculated with Azospirillum (Table 2).
Although 1000-grain weight was not significantly affected by the seed priming treatments, it was significantly enhanced by Azospirillum inoculation (Table 2). Plants from Azospirillum-inoculated seeds produced, on average, heavier 1000 grain weight (9%) than non-inoculated plants. There was a significant interaction between seed priming and Azospirillum inoculation on grain weight (Table 2). The greatest 1000 grain weight was obtained for plants, primed with Zn solution and inoculated with Azospirillum (Table 2).
Another yield component, spike number per m2, was significantly affected by Azospirillum inoculation, but not by priming treatment. In control plants (non-priming) and in plants primed with water (hydro-priming), increase in number of spike per m2 was the same and increased, on average, by 4.5% due to Azospirillum inoculation. The highest number of spike number per m2 was recorded in plants inoculated with Azospirillum and primed with Zn.
Nitrogen and Phosphorus Content
Figure 6 shows a comparison of the grain N content of wheat among the priming treatments with and without Azospirillum inoculation. Grain N content in non-priming plants was 0.76% and in response to Azospirillum inoculation increased to 0.91%. Priming seeds with Zn and Mn had a positive effect on grain N content. Grain N content in plants from seeds primed in Zn and Mn solution increased by 20% and 15% compared with those of plants from non-priming seeds, IAA priming and hydro-priming. There was no significant difference between seed priming with water and IAA with control (not primed seeds) on N content of grain. Priming seeds in 2% Zn solution increased grain N content compared with all other priming treatments (Fig. 6). There was a significant interaction between seed priming and Azospirillum on grain N content. Grains with the highest N content obtained from plants underwent Zn priming and Azospirillum inoculation (Fig. 6).
The effect of priming seeds for 6 h in water, IAA (2 ppm), or with aqueous solutions of Zn (0.2%) or Mn (0.2%) in combination or not with Azospirillum inoculation on grain N and P contents. Values with the same letters are not significantly different at P < 0.05. Values are mean ± standard error (SE) (n = 3)
Grain P content followed the same trend as observed for grain N content (Fig. 6). P content of grain was increased in non-primed treatment by Azospirillum inoculation, 5.93%. In this study, Zn priming among priming treatments was the most important influence on grain P content. Plants from seeds primed in 0.2% Zn solution for 6 h produced the grains with higher P content compared with those of plants from all the other priming treatment (Fig. 6). Plants raised from Zn-primed seeds and inoculated with Azospirillum had the higher P in grain (Fig. 6). When plants from Zn-primed seeds inoculated with Azospirillum grain P content was increased up to 6.8%.
Discussion
Soaking seeds in water prior to sowing followed by surface-drying (on-farm seed priming) can be simultaneously combined with micro-dosing of fertilizer, small amount of micronutrients, plant growth regulators and water restricting agents (polyethylene glycol). In this study, simultaneously to priming, wheat seeds were treated with Zn, Mn or IAA added to the conditioning solution. Seeds primed with water, auxin, Mn and, especially Zn solution resulted in increased grain weight, spike no per plant and seed no per spike. Plants of the Zn-primed seeds among different seed priming agents had the highest grain yield and yield components. Harris et al. [30], [53, 54], [23] and [10] reported that on-farm, soaking seeds in water prior to sowing resulted in faster emergence, higher vigor and density, accelerated flowering and maturity and higher yields as compared with non-prime crops. Considerable studies have shown that priming seeds with micronutrients can improve yield of crops [23]. Working with wheat and barley crops under field conditions, [33] demonstrated that grain yield increased by on-farm seed priming with zinc. Likewise, Ajouri et al. [1] found that priming barley seeds with Zn enhanced seed germination and seedling development. Harris et al. [31] reported that seed priming with ZnSO4 increased grain yield of wheat. At research and Extension Center, Arkansas, USA, [80] treated rice (Oryza sativa L.) seeds with Zn and found that growth and grain yield were increased. Micronutrients such as zinc were evaluated as cost-effective in several crops and suggested to be a promising alternative to soil- or foliar-applied Zn. Johnson et al. [37] found that grain yield of chickpea, lentil, rice and wheat decreased or was not affected, when seeds were primed with Zn.
In the present study, Mn priming treatment had also potential to improve grain yield of wheat, but at the rate less than that achieved by Zn-seed priming. In Australia, [49] soaked wheat seeds in aqueous solution of MnSO4 and reported that grain yield and grain Mn content were increased. Priming seeds with MnSO4 has been proposed to be the effective in improving wheat yield [23].
Since in this study seeds were primed with IAA, therefore improved yield components and grain yield can be related to the effect of auxin in the early growth of seedlings. In the present study, dry weights of plants of auxin-primed seeds were increased over non-primed seedling plants. It seems that auxin absorbed by seeds through priming could modulate plant processes. Auxin induces the root formation and coleoptile elongation (Taiz and Zeiger [84]). Diverse plant processes such as cell elongation and root initiation and development [60], [71] are regulated by auxin. Even regeneration of xylem and development of vascular development are modulated by auxin (Mattsson [48]). Partitioning of photo-assimilate from vegetative organs into reproductive organ is affected by the auxin (Cole and Patrick [15]), an important factor causing grain yield to increase.
In the present study, neither field emergence nor emergence rate was significantly affected by seed priming treatments for both years of the study (data not shown) but seedlings fresh weights from primed seeds with different priming agents (Zn, Mn, IAA and water) were significantly higher than non-primed seeds after 30 and 60 days of emergence. In this experiment, treatments that improved seedling growth during early plant growth also increased yield components (spike no, 1000-grains weight and harvest index) and total grain yield, suggesting that increment in yield due to seed priming can be attributed to the improved seedling growth. Ashraf and Foolad [2] and Chen and Arora [13] indicated that changes in important metabolic pathways, exerting during early plant growth by seed priming, are able to continue until later stages of plant development. Semiarid area is characterized by irregular rainfall pattern, low and unpredictable rain conditions. This study was performed in a semiarid area, with irregular and insufficient rainfall during both years of the experiments. Virk et al. [90] and [59] attributed greater yield increments to the rainfall profiles, rain seasons with restricted precipitation resulted in greater yield increments. Additionally, early growth and development of seedlings from primed seeds allow plant to access and take up more fertilizer [30]. Any delay in development of seedling leads the fertilizer to be leached or volatized from soil surface.
In this study, Azospirillum-inoculated plants produced more grain yield than non-inoculated seeds. Plants inoculated with Azospirillum produced grains with higher amount of N and P content. When inoculation was coupled with priming, additional increase in grain yield and N and P content in grains was exhibited. The benefits of inoculation on enhanced growth and yield could be attributed to the phytohormone production, N2 fixation, and P solubilization. Several literature regarding the promoting effects of Azospirillum on yield and other agronomic traits in wheat has been published previously [74], [65, 66], [4], [35], [38]. Increased phosphorus contents in grains due to Azospirillum inoculation could not merely refer to inoculation since the used bacterial strain in this study exhibited low potential activity of solubilizing inorganic phosphate in the modified Pikovskaya medium (data not shown). Higher N content in grains followed by inoculation could be attributed to some extent to the biological nitrogen fixation activity of this strain. In this study, inoculation resulted in attaining the early growth promotion in wheat seedlings. Auxin produced by Azospirillum has been assumed to have a promoting effect on attaining the early growth promotion in wheat [40]. Auxin induces rooting, root initiation, root development [60], [71] and increases root surface area [34]. Developed root system assists the plant in enhancing nutrient absorption [20], Gray and Smith [27], [83], [5] which can be important under dryland farming [38], Zarea [97]. Developed root system assists plant in uptaking soil minerals and water in a better way [58], [64]. In the present study, additional increase in growth and yield occurred when seed priming was combined with Azospirillum. Relationships of environment, Azospirillum and environment are complex. Various agronomic management methods such as applying N fertilizers and priming seed with chemical agent like 2,4-D and hydrogen peroxide have been reported to improve promoting effect of Azospirillum on plants [96].
Conclusions
It should be noted that there are a few reports dealing with PGPR inoculation coupled with priming in the field study. PGPR can be applied simultaneously to priming or following by priming. In the present study, Azospirillum inoculation following by seed priming is a significant additional approach in terms of grain yield and grain nutrient content.
References
Ajouri A, Asgedom H, Becker M (2004) Seed priming enhances germination and seedling growth of barley under conditions of P and Zn deficiency. J Plant Nutr Soil Sci 167:630–636
Ashraf M, Foolad MR (2005) Pre-sowing seed treatment-a shotgun approach to improve germination, plant growth, and crop yield under saline and non-saline conditions. Adv Agron 88:223–271
Aymen EM, Kaouther Z, Fredj MB, Cherif H (2012) Seed priming for better growth and yield of safflower (Carthamus tinctorius) under saline condition. J Stress Physiol Biochem 8:135–143
Ayyaz K, Zaheer A, Rasul G, Mirza MS (2016) Isolation and identification by 16S rRNA sequence analysis of plant growth-promoting Azospirilla from the rhizosphere of wheat. Braz J Microbiol 47:542–550
Bashan Y, Holguin G, de Bashan LE (2004) Azospirillum–plant relationship: physiological, molecular, agriculture, and environmental advances (1997–2003). Can J Microbiol 50:521–577
Beijerinck MW (1925) Über ein Spirillum, welches freien Stickstoff binden kann. Zentralbl Bakteriol Parasitenkd Infekt Abt. 63:353
Bradford M (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Bradford KJ (1986) Manipulation of seed water relations via osmotic priming to improve germination under stress conditions. HortSci. 21:1105–1112
Cakmak I (2008) Enrichment of cereal grains with zinc: agronomic or genetic biofortification? Plant Soil 302:1–17
Carrillo-Reche J, Vallejo-Marín M, Quilliam RS (2018) Quantifying the potential of ‘on-farm’ seed priming to increase crop performance in developing countries. A meta-analysis. Agron Sustain Dev 38:64
Chapman HD, Pratt PF (1961) Ammonium vanadate–molybdate method for determination of phosphorus. In: Chapman HD, Pratt PF (eds) Methods of analysis for soils. Plants and Water University of California, Berkeley, pp 184–203
Chasapis CT, Loutsidou AC, Spiliopoulou CA, Stefanidou ME (2012) Zinc and human health: an update. Arch Toxicol 86:521–553
Chen K, Arora R (2013) Priming memory invokes seed stress-tolerance. Environ Exp Bot 94:33–45
Chen P, Bornhorst J, Aschner M (2018) Manganese metabolism in humans. Front Biosci (Landmark Ed). 23:1655–1679
Cole DMA, Patrick JW (1998) Auxin control of photoassimilate transport to and within developing grains of wheat. Aust J Plant Physiol 25:69–77
Copur O, Demirel U, Karakus M (2010) Effects of several plant growth regulators on the yield and fiber quality of cotton (Gossypium hirusutum L.). Notulae Botanicae HortiAgrobotanici Cluj 38:104–110
Curtin D, Martin RJ, Scott CL (2008) Wheat (Triticum aestivum) response to micronutrients (Mn, Cu, Zn, B) in Canterbury, New Zealand. New Zealand J Crop Hortic Sci 36:169–181
de Bashan LE, Mayali X, Bebout BM, Weber PK, Detweiler AM, Hernandez JP, Prufert-Bebout L, Bashan Y (2016) Establishment of stable synthetic mutualism without co-evolution between microalgae and bacteria demonstrated by mutual transfer of metabolites (NanoSIMS isotopic imaging) and persistent physical association (fluorescent in situ hybridization). Algal Res 15:179–186
de Souza R, Ambrosini A, Passaglia LM (2015) Plant growth-promoting bacteria as inoculants in agricultural soils. Genet Mol Biol. 38:401–419
Dey R, Pal KK, Bhatt DM, Chauhan SM (2004) Growth promotion and yield enhancement of peanut (Arachis hypogaea L.) by application of plant growth promoting rhizobacteria. Microbiol Res 159:371–394
Dotto L, Silva VN (2017) Beet seed priming with growth regulators. Semina: CienciasAgrarias 38:1785–1798
Farahmandfar E, Shirvan MB, Sooran SA, Hoseinzadeh D (2013) Effect of seed priming on morphological and physiological parameters of fenugreek seedlings under salt stress. Intl J Agri Crop Sci 5:811–815
Farooq M, Basra SMA, Wahid A (2006) Priming of field-sown rice seed enhances germination, seedling establishment, allometry and yield. Plant Growth Regul 49:285–294
Gao Y, Wu P, Zhao X, Wang Z (2014) Growth, yield, and nitrogen use in the wheat/maize intercropping system. Field Crops Res 167:19–30
Garvin DF, Welch RM, Finley JW (2006) Historical shifts in the seed mineral micronutrient concentration of US hard red winter wheat germplasm. J Sci Food Agric 86:2213–2220
Gibson RS (2006) Zinc: the missing link in combating micronutrient malnutrition in developing countries. Proc Nutr Soc 65:51–60
Gray EJ, Smith DL (2005) Intracellular and extracellular PGPR: Commonalities and distinctions in the plant-bacterium signaling process. Soil Biol Biochem 37:395–412
Gulluoglu L (2004) Determination of usage of plant growth regulators in soybean (Glycine max Merr) farming under Harran plain conditions. J Fac. Agri 8:17–23
Hafeez B, Khanif YM, Saleem M (2013) Role of zinc in plant nutrition—a review. Am J Exp Agric 3:374–391
Harris D, Raghuwanshi BS, Gangwar JS, Singh SC, Joshi KD, Rashid A, Hollington PA (2001) Participatory evaluation by farmers of “on-farm” seed priming in wheat in India. Exp Agric 37:403–415
Harris D, Rashid A, Arif M, Yunas M (2005) Alleviating micronutrient deficiencies in alkaline soils of the North-West Frontier Province of Pakistan: Onfarm seed priming with zinc in wheat and chickpea. In: Andersen P, Tuladhar JK, Karki KB, Maskey SL (eds) Micronutrients in South and South East Asia, pp 143–151
Harris D, Rashid A, Miraj G, Arif M, Shah H (2007) On-farm seed priming with zinc sulphate solution–a cost-effective way to increase the maize yields of resource-poor farmers. Field Crops Res. 10:119–127
Harris D, Rashid A, Miraj G, Arif M, Yunas M (2008) ‘On-farm’ seed priming with zinc in chickpea and wheat in Pakistan. Plant Soil 306:3–10
Ivanchenko MG, Napsucialy-Mendivil S, Dubrovsky JG (2010) Auxin-induced inhibition of lateral root initiation contributes to root system shaping in Arabidopsis thaliana. Plant J 64:740–752
Jafarian T, Zarea MJ (2016) Hydrogen peroxide affects plant growth promoting effects of Azospirillum. Crop Sci Biotech 19:167–175
Jafri N, Mazid M, Mohammad F (2015) Responses of seed priming with gibberellic acid on yield and oil quality of sunflower (Helianthus annus L.). Indian J. Agric. Res. 49:235
Johnson SE, Lauren JG, Welch RM, Duxbury JM (2005) A comparison of the effects of micronutrient seed priming and soil fertilization on the mineral nutrition of chickpea (Cicer arietinum), lentil (Lens culinaris), rice (Oryza sativa) and wheat (Triticum aestivum) in Nepal. Exp Agric 41:427–448
Karimi N, Zarea MJ, Mehnaz S (2018) Endophytic Azospirillum for enhancement of growth and yield of wheat. Environ Sustain 1:149–158
Khalid BY, Malik NSA (1982) Pre-sowing soaking of wheat seeds in copper and manganese solutions. Commun Soil Sci Plant Analy 13:981–986
Khalid A, Arshad M, Zahir ZA (2004) Screening plant growth-promoting rhizobacteria for improving growth and yield of wheat. J Appl Microbiol 96:473–480
Khammas KM, Ageron E, Grimont PAD, Kaiser P (1989) Azospirillum irakense sp. nov., a nitrogen-fixing bacterium associated with rice roots and rhizosphere soil. Res Microbiol 140:679–693
Kopylov IP, Spyrydonov VH, Patyka VP (2009) Identification of Azospirillum genus bacteria isolated from the spring wheat root zone. Mikrobiol Z 71:13–19
Laity JH, Lee BM, Wright PE (2001) Zinc finger proteins: new insights into structural and functional diversity. Curr Opin Struct Biol 11:39–46
Lecube ML, Noriega GO, Santa Cruz DM, Tomaro ML, Batlle A, Balestrasse KB (2014) Indole acetic acid is responsible for protection against oxidative stress caused by drought in soybean plants: the role of heme oxygenase induction. Redox Rep 19:242–250
Liu DH, Jiang WS, Wuang C (1996) Effects of Zn2+ on root growth, cell division, and nucleoli of Allium cepa L. J Environ Sci 8:21–27
Ma T, Duan XH, Yang YY, Yao J, Gao TP (2017) Zinc-alleviating effects on iron-induced phytotoxicity in roots of Triticum aestivum. Biol Plant 61:733–740
Ma HY, Zhao DD, Ning QR, Wei JP, Li Y, Wang MM, Liu XL, Jiang Ch-J (2018) A multi-year beneficial effect of seed priming with gibberellic acid-3 (GA3) on plant growth and production in a perennial grass. Leymus Chinensis Sci Rep 8:13214
Mattsson J, Ckurshumova W, Berleth T (2003) Auxin signaling in Arabidopsis leaf vascular development. Plant Physiol 131:1327–1339
Marcar NE, Graham RD (1986) Effect of seed manganese content on the growth of wheat (Triticum aestivum) under manganese deficiency. Plant Soil 96:165–173
Mehnaz S, Mirza MS, Haurat J, Bally R, Normand P, Bano A, Malik KA (2001) Isolation and 16S rRNA sequence analysis of the beneficial bacteria from the rhizosphere of rice. Can J Microbiol 47:110–117
Mehnaz S, Weselowski B, Lazarovits G (2007) Azospirillum zeae sp. nov., diazotrophic bacteria isolated from rhizosphere soil of Zea mays. Int J Syst Evol Microbiol 57:2805–2809
Mehnaz S, Weselowski B, Lazarovits G (2007) Azospirillum canadense nov., a nitrogen fixing bacterium isolated from corn rhizosphere. Int J Syst Evol Microbiol 57:620–624
Murungu FS, Nyamugafata P, Chiduza C et al (2003) Effects of seed priming, aggregate size and soil matric potential on emergence of cotton (Gossypium hirsutum L.) and maize (Zea mays L.). Soil Tillage Res 74:161–168
Murungu FS, Chiduza C, Nyamugafata P, Clark LJ, Whalley WR (2004) Effect of on-farm seed priming on emergence, growth and yield of cotton and maize in a semi-arid area of Zimbabwe. Exp Agric 40:23
Naeem M, Bhatti I, Ahmad RH, Ashraf MY (2004) Effect of some growth hormones (GA3, IAA and Kinetin) on the morphology and early or delayed initiation of bud of lentil (Lens culinaris Medik). Pak J Bot 36:801–809
Naidoo G, Chirkoot HD (2004) The effects of coal dust on photosynthetic performance of the mangrove, Avicennia marina in Richards Bay, South Africa. Environ Pollut 127:359–366
Okon Y, Kapulnik Y (1986) Development and function of Azospirillum-inoculated roots. Plant Soil 90:3–16
Okon Y, Bloemberg GV, Lugtenberg BJJ (1998) Biotechnology of biofertilization and phytostimulation. In: Altman A (ed) Agricultural biotechnology. Marcel Dekker, New York, pp 327–349
Ousman A, Aune JB (2011) Effect of seed priming and micro-dosing of fertilizer on groundnut, sesame and cowpea in Western Sudan. Exp Agric 47(3):431–443
Overvoorde P, Fukaki H, Beeckman T (2010) Auxin control of root development. Cold Spring Harb Perspect Biol 2(6):a001537
Padole VR (1979) Effect of presoaking seed treatment of wheat seed with chemicals and hormones on yield and uptake of NPK. J Maharashtra AgricUniv 4(1):85–88
Pallaoro DS et al (2016) Priming corn seeds with plant growth regulator. J Seed Sci 38:227–232
Palmer CM, Guerinot ML (2009) Facing the challenges of Cu, Fe and Zn homeostasis in plants. Nat Chem Biol 5:333–340
Pérez-Montaño F, Alías-Villegas C, Bellogín RA, del Cerro P, Espuny MR, Jiménez-Guerrero I, López-Baena FJ, Ollero FJ, Cubo T (2014) Plant growth promotion in cereal and leguminous agricultural important plants: from microorganism capacities to crop production. Microbiol Res 169:325–336
Piccinin GG, Dan LGM, Braccini AL, Mariano DC, Okumura RS, Bazo G, Ricci TT (2011) Agronomic efficiency of Azospirillum brasilense in physiological parameters and yield components in wheat crop. J Agron 10:132–135
Piccinin GG, Braccini AL, Dan LGM, Scapim CA, Ricci TT, Bazo GL (2013) Efficiency of seed inoculation with Azospirillum brasilense on agronomic characteristics and yield of wheat. Ind Crop Prod 43:393–397
Rashid A (1996) Secondary and micronutrients. In: Bashir E, Bantel R (eds) Soil science. National Book Foundation, Islamabad, Pakistan, p 374
Reis S, Pavia I, Carvalho A, Moutinho-Pereira J, Correia C, Lima-Brito J (2018) Seed priming with iron and zinc in bread wheat: effects in germination, mitosis and grain yield. Protoplasma 255:1179–1194
Rengel Z, Graham RD (1995) Importance of seed Zn content for wheat grown on Zn-deficient soil I vegetative growth. Plant Soil 173:259–266
Rengel Z, Graham RD (1995) Importance of seed Zn content for wheat grown on Zn-deficient soil II grain yield. Plant Soil 173:267–274
Saini S, Sharma I, Kaur N, Pati PK (2013) Auxin: a master regulator in plant root development. Plant Cell Rep 32:741–757
Salantur A, Ozturk R, Akten S (2006) Growth and yield response of spring wheat (Triticum aestivum L.) to inoculation with rhizobacteria. Plant Soil Environ. 52:111–118
Salisbury FB (1994) The role of plant hormones. In: Wilkinson RE (ed) Plant environment interactions. Dekker, New York
Santa ORD, Hernadez RF, Alvarez GLM, Ronzelli P, Soccol CR (2004) Azospirillum sp. inoculation in wheat, barley and oats seeds greenhouse experiments. Braz Arch Biol Technol 47:843–850
Saraswat VK, Bansal KN (1991) Methods of zinc application and its effect on yield and zinc content of rice (Oryza sativa) and wheat (Triticum vulgare). Madras Agric J 78(5–8):174–177
Schmidt SB, Jensen PE, Husted S (2016) Manganese deficiency in plants: the impact on photosystem II. Trends Plant Sci 21:622–632
Sheteiwy M, Shen H, Xu J, Guan Y, Song W, Hu J (2017) Seed polyamines metabolism induced by seed priming with spermidine and 5-amino levulinic acid for chilling tolerance improvement in rice (Oryza sativa L.) seedlings. Environ Exp Bot 137:58–72
Singh B, Natesan SKA, Singh BK, Usha K (2003) Improving zinc efficiency of cereals under zinc deficiency. Curr Sci 88:36–44
Singh SP, Keller B, Gruissem W, Bhullar NK (2017) Rice nicotianamine synthase 2 expression improves dietary iron and zinc levels in wheat. Theor Appl Genet 130:283–292
Slaton NA, Wilson-Jr CE, Ntamatungiro S, Norman RJ, Boothe DL (2001) Evaluation of zinc seed treatments for rice. Agron J 93:152–157
Steenhoudt O, Vanderleyden J (2000) Azospirillum, a free-living nitrogen-fixing bacterium closely associated with grasses: genetic, biochemical and ecological aspects. FEMS Microbiol 24:487–506
Stout PR, Arnon DI (1939) Experimental methods for the study of the role of copper, manganese, and zinc in the nutrition of higher plants. Am J Bot 26:144–149
Sukumar P, Legué V, Vayssières A, Martin F, Tuskan GA, Kalluri UC (2013) Involvement of auxin pathways in modulating root architecture during beneficial pant–microorganism interactions. Plant, Cell Environ 36:909–919
Taiz L, Zeiger E (2010) Plant Physiology, 5th edn. Sinauer Associates, Inc., Publishers, Sunderland, MA, USA
Tarrand JJ, Krieg NR, Döbereiner J (1978) A taxonomic study of the Spirillum lipoferum group, with the descriptions of a new genus, Azospirillum gen. nov. and two species Azospirillum lipoferum (Beijerinck) comb. nov. and Azospirillum brasilense sp. nov. Can J Microbiol 24:967–980
Teale WD, Paponov IA, Palme K (2006) Auxin in action: signalling, transport and the control of plant growth and development. Nat Rev Mol Cell Biol 7:847–859
Ulfat A, Majid SA, Hameed A (2017) Hormonal seed priming improves wheat (Triticum aestivum L.) field performance under drought and non-stress conditions. Pak J Bot 49:1239–1253
Vallee BL, Falchuk KH (1993) The biochemical basis of zinc physiology. Physiol Rev 73:79–118
Van Schouwenberg J, Walinge I (1973) Methods of analysis for plant material. Agricultural University, Wageningen
Virk DS, Chakraborty M, Ghosh J, Harris D (2006) Participatory evaluation of horsegram (Macrotyloma uniflorum) varieties and their on-station responses to on-farm seed priming in eastern India. Exp Agric 42:411
Wei LX et al (2017) Priming of rice (Oryza sativa L.) seedlings with abscisic acid enhances seedling survival, plant growth, and grain yield in saline-alkaline paddy fields. Field Crops Res. 203:86–93
White JG, Zasoski RJ (1999) Mapping soil micronutrients. Field Crops Res 60:11–26
Yilmaz A, Ekiz H, Gultekin I, Torun B, Barut H, Karanlik S, Cakmak I (1998) Effect of seed zinc content on grain yield and zinc concentration of wheat grown in zinc-deficient calcareous soils. J Plant Nutr 21:2257–2264
Zahid M, Abbasi MK, Hameed S, Rahim N (2015) Isolation and identification of indigenous plant growth promoting rhizobacteria from Himalayan region of Kashmir and their effect on improving growth and nutrient contents of maize (Zea mays L.). Front Microbiol 6:207
Zahid M, Abbasi MK, Hameed S, Rahim N (2015) Isolation and identification of indigenous plant growth promoting rhizobacteria from Himalayan region of Kashmir and their effect on improving growth and nutrient contents of maize. Front Microbiol 6:207
Zarea MJ (2017) Azospirillum and wheat production. In: Kumar V, Kumar M, Sharma SH (eds) Probiotics in agroecosystem. Springer, Singapore, pp 329–348
Zarea MJ (2019) Applications of Beneficial Microbe in Arid and Semiarid Agroecosystem: IAA-Producing Bacteria. In: Kumar V, Prasad R, Kumar M, Choudhary D (eds) Microbiome in Plant Health and Disease. Springer, Singapore, pp 105–118
Zarea MJ (2019) Salt-tolerant microbes: isolation and adaptation. In: Varma A, Giri B (eds) Microorganisms in saline environments strategies and functions. Springer, Switzerland, pp 285–301
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Karimi, N., Goltapeh, E.M., Amini, J. et al. Effect of Azospirillum zeae and Seed Priming with Zinc, Manganese and Auxin on Growth and Yield Parameters of Wheat, under Dryland Farming. Agric Res 10, 44–55 (2021). https://doi.org/10.1007/s40003-020-00480-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40003-020-00480-5