Abstract
This study investigates symptom relief in patients whose hemifacial spasms (HFS) were completely relieved following microvascular decompression (MVD). Patients with HFS who were operated on from January 2007 to May 2011, at our department, were studied. The medical records and operation videos of each patient were reviewed. Follow-up was performed via telephone communication. A total of 410 patients with HFS were divided into two groups: Group A (277 cases): HFS caused by small vascular compression, and Group B (133 cases): HFS caused by vertebral artery compression. Magnetic resonance imaging (MRI) was performed on all the patients to rule out those with secondary HFS. Patients who had undergone previous MVD were also excluded at that time. In all, 462 patients underwent MVD for HFS in our center during the study period. Follow-up was available for 410 patients. In the small vascular compression cohort, 72 (72/277, 26.0 %) HFS patients achieved relief with delayed healing. While in the vertebral artery compression cohort, 52 (52/133, 39.1 %) HFS patients experienced relief with delayed healing. There was significant difference between these two cohorts (P < 0.05). 93.75% of delayed healing patients were cured and experienced spasm relief within 6 months. Surgeons should be aware that delayed symptom relief after MVD for HFS is more common than has been reported, and time course of symptom relief is various as well as unpredictable; however, most HFS patients are cured, and experience spasm relief within 6 months.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Most neurosurgery experts have accepted the efficiency of microvascular decompression (MVD) for treatment of hemifacial spasm (HFS), which has a high cure rate [1]. However, we discovered delayed spasm relief amongst a number of patients in our normal clinical practices. Over a 53-month period from January 1, 2007 to May 31, 2011, we prospectively and retrospectively collected information on 462 patients with HFS who were evaluated in our center. 144 cases showed delayed spasm relief. We analyzed the postoperative cure-delay of distinct offending vessels in patients with HFS following MVD, and investigated pathogeneses and treatment principles.
Materials and methods
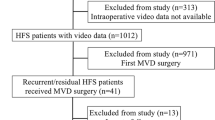
We studied patients with HFS who were operated on from January 2007 to May 2011 at Drum Tower Hospital’s Department of Neurosurgery at the School of Medicine in Nanjing University in Nanjing, China. Each patient’s medical records and operation videos were reviewed. Follow-up was performed via telephone communication. We were unable to follow-up with 52 patients, due to outdated contact numbers and deaths caused by severe diseases.
The diameters of the anterior inferior cerebellar artery (AICA) and posterior inferior cerebellar artery (PICA) were smaller than the vertebral artery (VA). Moreover, AICA and PICA vessels were separated from the facial nerve root easily, and there was no statistical difference between the cure rate and the delayed healing rate of the two vessels. We assigned AICA and PICA to the small vascular compression group.
A total of 410 patients with HFS were divided into two groups: Group A (277 cases): HFS caused by small vascular compression (149 cases of AICA; 128 cases of PICA) and Group B (133 cases): HFS caused by vertebral artery compression. Magnetic resonance imaging (MRI) was performed for all the patients to rule out those with secondary HFS. Patients who had undergone previous MVD were also excluded at this time.
Facial nerve microvascular decompression
After suboccipital retrosigmoidal craniotomy, the cerebellopontine angle is exposed. Subsequently, an inspection of the root exit zone (REZ) of the facial nerve from the brainstem takes place. After microsurgical dissection of the vessel from the facial nerve, a Teflon sponge is placed between the brainstem and vessel, to permanently prevent vascular compression. During the operation, brainstem auditory evoked potentials (BAEPs) for hearing loss and abnormal muscle response (AMR) wave, which is regarded as an exclusive electrophysiological phenomenon of HFS, were closely monitored, which facilitated a complete decompression.
Statistical analysis
Statistical analysis for outcomes was performed using the Fisher exact test and the FREQ procedure from Statistical Analysis Systems. Statistical significance was inferred at P < 0.05.
Results
Operative findings
The responsible blood vessel is divided into small vascular compression and vertebral artery compression types, based on the preoperative cranial MRI combined with intraoperative findings. The small vascular compression type is characterized in that: REZ of the facial nerve from the brainstem with small vessel cyclic-like, point-like or loop-like compressions. The small vessel type can be easily separated from the facial nerve root. This type is the most common cause of HFS. We released the responsible vessel first, and then shifted the vessel after the tension reduction. Teflon sponges were placed out the facial nerve compression point to decompress the facial nerve root.
The vertebral artery compression type includes: (1) the vertebral artery combined with small vascular compressions, often present as a thickened, shifted vertebral artery which compresses the facial nerve. During the operation, we found that the accompanying small vessel, beneath the vertebral artery, was the actual responsible vessel after shifting the vertebral artery. In this case, we released and removed the vertebral artery in the lower cranial nerve level first, and then placed a Teflon sponge to expose the facial nerve root and acquire sufficient operative space. After the facial nerve root was revealed, we discovered that the small accompanying vascular was the responsible vascular. Afterwards, we isolated the small vascular with a Teflon sponge to decompress the facial nerve root. In a few cases, the facial nerve root was only exposed with difficulty. We built a bridge for the vertebral artery, and then placed one Teflon sponge in the lower cranial nerve level and another in the distal vertebral artery. Eventually, we removed the responsible vascular to prevent vascular compression. (2) Vertebral basilar artery compression alone, which is rare and difficult to operate on.
Outcomes
A total of 462 patients underwent MVD for HFS at our center during the study period. Follow-up was available for 410 patients. In the small vascular compression cohort, 74 (74/277, 26.7 %) HFS patients experienced relief with delayed healing. While in the vertebral artery compression cohort, 70 (70/133, 52.6 %) HFS patients experienced relief with delayed healing. There was significant difference between these two cohorts (P < 0.05).
93.75% of delayed healing patients were cured and experience spasm relief within 6 months. The course of delayed symptom relief over time is shown in Table 1. The AMR wave vanished in all of the patients postoperatively. 120 patients in 144 cases without symptom relief underwent electrophysiological monitoring again the next day, and AMR was recorded in 118 individuals. Among the 118 who were AMR positive, 112 patients obtained relief within 6 months.
Discussion
HFS, a syndrome of unilateral facial nerve hyperactive dysfunction, is a common cranial nerve disease, which is characterized by slight intermittent twitching of a single facial muscle, and gradually becomes more intense and frequent, eventually spreading to involve other facial muscles. HFS is a severe functional disease that causes impairment in patients’ quality of life. Clinical surveys suggest that the prevalence rate of HFS is ten out of 100,000 persons in the United States’ and Norway’s populations [2, 3]. Moreover, it is most common within Asian populations [4].
HFS is generally caused by pulsatile vascular compression on the 7th nerve REZ [5–7]. MVD has been established for the treatment of HFS, and is efficacious and durable [8]. With the development of microsurgical techniques, the cure rate of MVD has increased significantly [9]. However, the cure rate of MVD has been reported to range from approximately 70 to 99.3 % [10, 11]. Our department performed approximately 2000 MVD surgeries in the past ten years, for which the cure rate is 91.7 %. Upon review of our cases, we found that our attainment of a satisfactory cure rate is greatly impacted by our ability to make accurate, strategic procedural choices that are in accordance with the different type of offending vascular facial nerves.
For small vascular compressed facial nerves, we released the offending vessel first. A wide arachnoid dissection was then performed to reduce the offending vessel tension and make the vessel more movable. Teflon was interposed at the facial nerve compression point to decompress the facial nerve root. We avoided placing Teflon sponges between the vessel and the facial nerve root, in case of postoperative invalidation or recurrence.
In the case of a vertebral artery compressed facial nerve, we cut the arachnoid from the lower cranial nerve level to the facial nerve level, fully freed the vertebral artery, and then built a bridge for it. We then placed Teflon in the lower cranial nerve level, and then placed a Teflon sponge in the distal vertebral artery to expose the facial nerve root and acquire sufficient operative space. After the facial nerve root was revealed, we would be able to find that the small accompanying vascular was the vascular that was actually responsible. Afterwards, we isolated the small vascular with a Teflon sponge to decompress the facial nerve root. In such cases, operations needed to be gentle and careful, and it was important to probe accompanying small blood vessels to avoid omitting an offending vessel.
In our study, 144 patients showed delayed disappearance of symptom after operation. The rate of delayed symptom disappearance was 35.1 %. It is well known that patients have various rates of symptom disappearance after MVD. The highest reported percentage of the delayed disappearance was 61.8 % [12]. After MVD for HFS, there are various reports of symptom improvement over the course of time [13, 14]. The average postoperative symptom duration ranges from days to years. Sindou et al reported postoperative symptom disappearance over more than 3.5 years [15]. In our research, the vertebral artery compression group contained one case in which a patient’s healing was delayed for 2 years. In terms of the postoperative outcome and complications, there are no significant differences between these two different compression groups; however, there was a significant difference in cure rate between these two groups.
Delayed symptom disappearance after MVD for HFS is more common than has been reported. For the pathogeneses of HFS, four hypotheses of delayed healing are most recognized. One of the recognized pathogeneses of HFS is the compression of the facial nerve root by the vascular, which causes the demyelination of the nerve fibers to occur. The restoration of the facial nerve’s physiological function does not take place immediately following MVD’s relief of vascular compression. Symptoms are relieved over different courses of time, according to the extent of various compressions.
The second hypothesis is that hyperexcitability in the facial motor nucleus is generated as the facial nerve stimulated by the offending vascular. For patients with severe facial motor nucleus compression injury, especially when thick, tortuous, arteriosclerotic vertebral arteries with strong pulsatile impact force are responsible for the compression, the reduction of facial motor nucleus excitability will occur over a certain amount of time after MVD.
The third hypothesis is that MVD may cause edema of the facial nerve. Facial nerve injury induced by vascular compression, combined with nerve edema, may cause postoperative symptoms to worsen or persist for a period of time. Spasms disappear when the facial nerve’s function returns to normal after the edema subsides. Another hypothesis is that, despite sufficient decompression by MVD, the facial nerve root is still likely to be offended by Teflon cotton and cerebrospinal fluid’s transferred pulsatile assault. This slight pressure can lead to prolonged duration of postoperative symptoms.
In our study, 144 patients showed delayed disappearance of HSF after MVD, and the delay healing rate was 35.1 % (144/410). In the small vascular compression group, 74 (74/277, 26.7 %) HFS patients experienced symptom relief with delayed healing. While in the vertebral artery compression group, 70 (70/133, 52.6 %) HFS patients were cured with delayed healing. There was significant difference between these two groups (P < 0.05). 93.75 % delayed healing patients were cured with spasm relief within 6 months.
The reason why the delayed healing rate of vertebral artery compression groups is higher than that of small vascular compression groups may be due to these two hypotheses: The first hypothesis is that the vertebral artery compresses the facial nerve severely, which causes the demyelination of the nerve fibers to occur and results in hyperexcitability in the facial motor nucleus. In these cases, the surgery has fully decompressed the facial nerve. The restoration of physiological function to the facial nerve, and the reduction of facial motor nucleus excitability, requires a certain amount of time following MVD. The second hypothesis is that the operative space in the vertebral artery compression group is relatively narrow. The pulsatile impact of the blood vessel may be transferred to the facial nerve root via Teflon cotton and cerebrospinal fluid. However, the exact reason for the delay in healing still needs further study. AMR is considered to be a useful indicator, which aids in the intraoperative complete decompression of the facial nerve. Among all of the patients without immediate symptom relief, 94.9 % patients obtained relief within 6 months.
Conclusion
Surgeons should be aware that delayed spasm disappearance after MVD for HFS is common, and most cases are cured with spasm relief within 6 months. For patients who experience no significant improvement in symptoms after MVD, we suggest avoiding reoperation in a short term. The effect of MVD takes at least 2 years to be fully evaluated.
References
Sekula RF Jr, Frederickson AM, Arnone GD, Quigley MR, Hallett M (2013) Microvascular decompression for hemifacial spasm in patients >65 years of age: an analysis of outcomes and complications. Muscle Nerve 48(5):770–776
Auger RG, Whisnant JP (1990) Hemifacial spasm in Rochester and Olmsted County, Minnesota, 1960 to 1984. Arch Neurol 47:1233–1234
Nilsen B, Le KD, Dietrichs E (2004) Prevalence of hemifacial spasm in Oslo, Norway. Neurology 63:1532–1533
Wu Y, Davidson AL, Pan T, Jankovic J (2010) Asian over-representation among patients with hemifacial spasm compared to patients with cranial-cervical dystonia. J Neurol Sci 298:61–63
Campos-Benitez M, Kaufmann AM (2008) Neurovascular compression findings in hemifacial spasm. J Neurosurg 109:416–420
Chung SS, Chang JH, Choi JY, Chang JW, Park YG (2001) Microvascular decompression for hemifacial spasm: a long-term follow-up of 1,169 consecutive cases. Stereotact Funct Neurosurg 77:190–193
Dannenbaum M, Lega BC, Suki D, Harper RL, Yoshor D (2008) Microvascular decompression for hemifacial spasm: Long-term results from 114 operations performed without neurophysiological monitoring. J Neurosurg 109:410–415
Moller AR, Jannetta PJ (1985) Hemifacial spasm: results of electrophysio-logic recording during microvascular decompression operations. Neurology 35:969–974
Huh R, Han IB, Moon JY et al (2008) Microvascular decompression for hemifacial spasm:analyses of operative complications in 1582 consecutive patients. Surg Neurol 69:153–157
Hyun SJ, Kong DS, Park K (2010) Microvascular decompression for treating hemifacial spasm:lesons learned from a prospective study of 1174 operations. Neurosurg Rev 33:325–334
Chung SS, Chang JH, Choi JY et al (2001) Microvascular decompression for hemifacial spasm:a long-term follow-up of 1169 consecutive cases. Stereotact Funct Neurosurg 77:190–193
Oh ET, Kim E, Hyun DK, Yoon SH, Park H, Park HC (2008) Time course of symptom disappearance after microvascular decompression for hemifacial spasm. J Korean Neurosurg Soc 44:245–248
Li CS (2005) Varied patterns of postoperative course of disappearance of hemifacial spasm after microvascular decompression. Acta Neurochir (Wien) 147:617–620
Shin JC, Chung UH, Kim YC (1990) Park CI : prospective study of microvascular decompression in hemifacial spasm. Neurosurgery. 40:730–734
Sindou MP (2005) Microvascular decompression for primary hemifacial spasm. Importance of intraoperative neurophysiological monitoring. Acta Neurochir 147:1019–1026
Conflict of interest
The authors do not have any possible conflicts of interest.
Ethical Standard
Approval was obtained from the local ethical standards committee on human experimentation and written informed consent from all subjects before enrollment. The study has been performed in accordance with the ethical standards laid down in the 1964 declaration of Helsinki and its later amendments.
Author information
Authors and Affiliations
Corresponding author
Additional information
Yuxiang Dai and Hongbin Ni have contributed equally to this work.
Rights and permissions
About this article
Cite this article
Dai, Y., Ni, H., Xu, W. et al. Clinical analysis of hemifacial spasm patients with delay symptom relief after microvascular decompression of distinct offending vessels. Acta Neurol Belg 116, 53–56 (2016). https://doi.org/10.1007/s13760-015-0471-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13760-015-0471-7