Abstract
Samples of corbicular and stored pollen gathered by Tetragonisca angustula (Latreille, 1811) in an Amazon-Chiquitano transitional forest during the dry season were analyzed. The pollen spectrum was established as well as the dynamics of the relationship between the stingless bee and the surrounding flora. Pollen samples obtained from three rational hives were subjected to acetolysis and 55 pollen types were identified, the most frequent being from Anadenanthera (Fabaceae), Chenopodiaceae, and Dydimopanax (Araliaceae). Significant differences in pollen families used between hives along the months of collection were found in stored pollen and non-significant differences in corbicular pollen. Mean values of alpha diversity (H′) showed T. angustula as a generalist while beta diversity qualitatively showed that pollen composition was similar between two hives both of which differed from the third hive. Pollen types in corbicular and stored pollen were in general related with richness of flowering plants in the foraging area; the pollen offer was less diverse than the pollen collected, and a trend to prefer the collection of pollen from the most abundant flowering species could be discerned. T. angustula showed polylectic feeding habits and a capacity to adapt to food availability.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The well-functioning of many ecosystems depends partly on plant-animal interactions such as pollination, which indirectly ensure their productive sustainability and maintenance of their biodiversity (Klein et al 2007). These mutualistic interactions form complex networks that help to maintain the architecture of biodiversity and even of the food network of terrestrial communities (Bascompte & Jordano 2008, Aizen & Chacoff 2009). It is estimated that one third of the food consumed on a worldwide basis is available through the service of pollinators (Michener 2007). Around 78% of plants with flowers are pollinated by animals—mainly insects—in temperate zone communities and the figure rises to 94% in tropical communities (Ollerton et al 2011, Ollerton 2017); furthermore, over 40% of the animals that pollinate tropical plants are bees (Ollerton 2017). Nowadays, these plant-animal networks suffer from the reduction of populations of native pollinators due mainly to loss of their natural habitats (Freitas et al 2009), thus negatively affecting natural ecosystems and crop productivity (Klein et al 2007). In Bolivia and other countries in the Neotropics, there are threats such as fast deforestation for farming, livestock, and the introduction of exotic bee species causing the loss of native species of bees (Freitas et al 2009).
In the Amazon rainforest, native bees play a key role in the maintenance of regional ecosystems through pollination (Absy et al 2018). For example, the stingless bee, Scaptotrigona hellwegeri (Friese, 1900), uses a large proportion of native tree pollen types as resources, thus contributing to the preservation of vegetation (Quiroz et al 2011). Another stingless bee, Tetragonisca angustula (Latreille, 1811), is a constant floral visitor of certain native species of plants (Obregon et al 2013) and is used principally in meliponiculture (Almeida et al 2012) despite the fact that it is attracted by plants with different amounts of the resources pollen or nectar. In this context, the identification of the native flora used by native stingless bees is essential for interpreting their role in native ecosystems and for using them as food supply plants in programs aimed at their conservation or their use in natural, agricultural, and urban ecosystems (Carvalho & Marchini 1999). In Bolivia, basic research on the ecology of stingless bees has been mainly limited to the description of their nesting patterns (De la Riva 2004), and results have been applied to the improvement of bee breeding and honey production (Copa 2004, Ferrufino & Vit 2013). The present study was aimed at characterizing the preference patterns on the pollinic resources used during the dry season by the stingless bee, T. angustula, in a transitional Chiquitano-Amazonian forest in the locality of Santa Cruz, Bolivia. In particular, we sought to (1) evaluate qualitatively and quantitatively the pollen resources available to T. angustula, (2) identify pollen loaded and stored by the bees, (3) compare the alpha and beta diversities of loaded and stored pollen used by T. angustula in rational hives, and (4) compare pollen use of T. angustula with pollen availability through time.
Material and Methods
Study Site
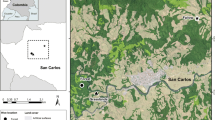
The study was performed in the private Reserve Potrerillos del Güendá (17°39′S, 63°27.5′W) in the southeast of Amboró in the department of Santa Cruz, Bolivia. This Reserve has an area of 350 ha, is surrounded by the rivers Güendá and Las Conchas, and lies in the lowlands within a biogeographical zone where the Amazonic and Chiquitano ecoregions converge (Navarro et al 1996). Lowlands in Bolivia are characterized by a dry season (April–September) and a wet season with heavy rains (October–March) (Navarro 2011, Toledo et al 2011). In the study site, higher areas are occupied by a humid rainforest with some elements of semi-deciduous Chiquitano forest. It is characterized by a forest canopy 13 to 30 m high depending on the zone (Navarro & Maldonado 2002) and contains species such as Aspidosperma cylindricarpon Müll. Arg, Astrocaryum murumuru Mart., Bactris major Jacq., Calycophyllum spruceanum (Benth.) Hook. f. ex K. Schum., and Dypteryx odorata (Aubl.) Willd. In the lower areas, there are fragments of Chiquitano vegetation with Allagoptera leucocalyx (Drude) Kuntze, Astronium fraxinifolium Schott, Curatella americana L., and Zamia boliviana (Brongn.) A. DC. (Navarro 2001). The largest part of the Reserve has elements of both Chiquitano and Amazonic forests, with characteristic species such as Aspidosperma, Anadenanthera colubrina (Vell.) Brenan, Batocarpus amazonicus (Ducke) Fosberg, Myracrodruon urundeuva M. Allemão, Tipuana tipu (Benth.) Kuntze, 1898, Aspidosperma rigidum Rusby, and Vitex cymosa Bertero ex Spreng. (Navarro 2001). During the dry season, the deciduous and semi-deciduous vegetation loses most of its foliage and there is even a reduction in the flowering and growth of herbaceous plants (Navarro 2011, Devisscher et al 2016), in contrast to the wet season where the constant rains allow the flowering of the different strata that make up the forest including lianas and different epiphytes (Navarro 2001, Ibich & Merida 2003).
Rational Beehives and Pollen Samples
Three vertical rational hives were formed in February 2015 by transplanting individuals of T. angustula from natural hives in local trees. Two days after forming them, they were transferred and installed at the final destination at distances of 10 m from each other (Fig 1). Sampling of stored and corbicular pollen was conducted monthly from April to August 2015; it was carried out during 3 days of the first week of each month during the hours of highest activity of the bees, from 11:00 h until 14:00 h. Three-centimeters-long pieces of disposable plastic straws (de Novais et al 2014) were used to collect stored pollen from the pots most recently sealed by the bees (usually lighter in color than older pots), i.e., pots containing the pollen stored by bees mostly during the month previous to the sampling period. Corbicular pollen was collected by covering the main entrance of the hive, catching returning bees (3–15 individuals) and scraping their corbicula. Pollen samples were stored in Eppendorf tubes and frozen until used in laboratory procedures. Due to an attack to the hives by the invasive bee Lestrimelitta limao Friese, 1903 (Meliponini), no sampling of stored or corbicular pollen was possible in July, and only two hives (hives 2 and 3) could be sampled for stored and corbicular pollen in August. Furthermore, no corbicular pollen was found in bees collected at the entrance of hive 1 in June.
Thus, stored pollen samples collected in April, May, June, and August corresponded to pollen collected by bees during the months of March, April, May, and July, respectively, whereas corbicular pollen samples collected in April, May, June, and August corresponded to pollen transported by the bees during those same months.
Botanical Samples and Reference Pollen Collection
Four orthogonal transects (4 × 200 m) were established around the hives (Fig 1), following the flight range reported for the species (Nogueira-Neto 1997, Gamero 2006). Flowers and flower buds were collected and pollen was extracted from them to elaborate a reference pollen collection. Herbarium samples were made from all plant species whose flowers and flower buds were collected. Identifications were achieved by comparison with the collections of the Herbarium of Eastern Bolivia (USZ) and by using taxonomical keys (Gentry 1996). Plants were identified mostly to the species level (26 cases); in some cases, only the genus could be discerned (17 cases). Identifications were confirmed by Dr. Michael H. Nee (New York Botanical Garden).
Pollen Treatment, Identification, and Counting Protocol
Pollen was acetolyzed according to Erdtman (1986) and preserved in 0.1–0.5 ml of glycerin contained in Eppendorf tubes. Three slides per sample were prepared for identification and quantification of pollen under the microscope. Identification of pollen types collected and stored by bees was based on comparisons with the reference pollen collection and with pollen catalogs (Colinvaux et al 1990, Roubik & Moreno 1991). The level of identification of pollen types which were collected by bees but were absent from the reference pollen collection varied according to the taxonomic group; in some cases, only pollen morphotypes could be discerned. Stock suspensions of pollen grains from different pots or bees were introduced into a Neubauer chamber and pollen types determined from the contents of the corner cells (Celeromics 2009).
Pollen counts of different morphotypes were determined by counting 300 pollen grains (Vossler 2012) of both corbicular or stored pollen for each hive and each sampling period; counting was carried out in triplicates. Frequency classes were determined based on the percentage of each pollen type in a given sample, following Louveaux et al (1978): predominant pollen (> 45%), secondary pollen (16–45%), important minor pollen (3–15%), and minor pollen (< 3%).
Statistical Analysis
Abundance of pollen according to families was analyzed along time in corbicular and stored pollen using similarity analysis of permutations (ANOSIM) with 10,000 random permutations and the Bonferroni correction factor. The results were analyzed graphically using the nonmetric multidimensional scaling (NMDS) method based on the Bray-Curtis matrix. The pollen families in the hives responsible for the variability found between months were analyzed with the similarity of percentages (SIMPER) method. The Shannon-Weaver (H′) diversity index was used to compare alpha diversity between corbicular and stored pollen, pollen used in different hives, and pollen used in different months. Bray-Curtis index was used to determine beta diversity between hives. All analyses were performed with the PAST software (Hammer et al 2001).
Results
Pollen Types
Forty-three plant species and their corresponding pollen types were registered in the transects (Table 1), while 55 pollen morphotypes belonging to 22 botanical families were found in the hives (Table 2); 18 of those pollen types were also found in the transects (Table 3). The highest number of pollen types was found among the Fabaceae (eight types), Sapindaceae and Araliaceae (five), Euphorbiaceae, Malpighiaceae and Rubiaceae (four), Asteraceae, Anacardiaceae, Bignonaceae and Boraginaceae (three), and Urticaceae (two); the other eleven families were represented by a single pollen type (Table 2). The pollen types with highest occurrence were from Didymopanax (88% of samples), Chenopodiaceae-msp11 (88%), Anadenanthera (63%), Astronium (50%), Senna (38%), Banisteriopsis (38%), Heteropteris (37.5%), and Zanthoxylum (38%).
Differences in Consumption of Pollen Families in the Different Months
ANOSIM revealed that abundance of pollen belonging to different families collected during different months showed significant differences between hives (one-way ANOSIM: R = 0.39, P = 0.007); the graph resulting from NMDS (Fig 2) showed the closer similitude between pollen families in hives 2 and 3. Furthermore, SIMPER analysis showed that the families Asteraceae, Bignonaceae, Chenopodiaceae, Euphorbiaceae, Malpigiaceae, Rutaceae, and Sapindaceae were mainly responsible for the differences between the hives. Abundance of corbicular pollen belonging to different families collected during different months showed non-significant differences between hives (one-way ANOSIM: R = 0.15, P = 0.97).
Non-metric multidimensional scaling (NMDS) of stored pollen collected by Tetragonisca angustula during different months (indicated in the graph) based on Bray-Curtis similarity matrices of pollen families (2D stress value, 0.123). Three rational hives were analyzed: hives 1 (square), 2 (circle), and 3 (diamond).
Alpha and Beta Diversity of Pollen Types
The mean Shannon Weaver index (H′) for stored pollen (0.73) was higher than for corbicular pollen (0.57). Hive 1 had the highest diversity of both stored and corbicular pollen (H′ = 1 and 0.85, respectively) compared with hives 2 (H′ = 0.76 and 0.59, respectively) and 3 (H′ = 0.62 and 0.68, respectively). Diversity and abundance of blooming plants (with flowers or flower buds) was high and showed a decrease at the beginning of the study period and an increase towards the end of the study period (Fig 3). However, this measure of diversity hides the fact that the composition of species showed high variability; in fact, all but five species occurred in only one observation period and presence of a species was highly scattered (Table 1). Diversity of stored pollen was higher than that of corbicular pollen; however, both followed similar trends through time, and similar to that of blooming plants (with flowers or flower buds) in the transects (Fig 3).
Richness of pollen types varied between hives (32, 21, and 27 pollen types for hives 1, 2, and 3, respectively). Beta diversity based on the Bray-Curtis index showed that pollen abundance was more similar between hives 2 and 3 (index = 0.60) than between hives 1 and 2 (index = 0.15) and between hives 1 and 3 (index = 0.28).
Pollen Collected and Flower Occurrence
The 18 pollen types from the transects which were collected and stored by the bees had mellitophilous features. Additionally, Chenopodiaceae-msp11 is a non-mellitophilic pollen type which was absent from the transects but present as stored pollen in all hives at all collection times.
The time course for diversity and abundance of blooming plants (with flowers or flower buds) and for corbicular and stored pollen types followed similar trends, i.e., a decrease during the first 2 months followed by an increase towards the end of the study period (Fig 4).
Pollen types used by bees were seldom associated to flower taxa present in the transects. In fact, in only 9 of the 108 possible (pollen type-blooming taxon) associations, pollen was used which occurred as a resource in the transects. In the remaining associations, pollen was used but was not found in the transects (10 associations) or pollen was neither found as resource nor as used pollen (43 associations) (Table 3).
Discussion and Conclusions
T. angustula collected 55 pollen types belonging to 22 plant families, thus showing a polylectic feeding habit (Martínez et al 1994). The Fabaceae was the most represented family at the species level. This family is considered an important resource for stingless bees such as Scaptotrigona fulvicutis (Moure 1964) (Marquez-Souza et al 2007), since it generally offers large amounts of pollen over long periods of time thus providing resources to perennial hives (Michener 2007).
The ten most frequent pollen types accounted for almost 80% of all pollen collected. It should be noted that none of the pollen types had an occurrence index > 90%, corresponding to a temporary specialization sensu Rech & Absy (2011a). This type of variation of the use of the resources available may correspond to a strategy common in the Meliponinae that involves maintaining fidelity towards certain abundant pollinic food sources while in parallel seeking to diversify the collection of pollen (Rech & Absy 2011a, b, de Novais et al 2015). This leads to the finding of infrequent or rare pollen types (Marques-Souza 2010), which are considered alternative resources. Fidelity towards certain pollinic sources involves the quantity and/or quality of the reward, cost/benefit considerations, and seasonal changes that could affect the pollen spectra available (Ramalho et al 1990, de Novais et al 2015). The broad pollen spectrum utilized by T. angustula involves perennial and deciduous plant species with a variety of life forms (e.g., trees, shrubs, herbs, climbing plants). Moreover, the fact that the richness of pollen types characterized in transects only represents around 30% of the pollen types found in corbicular and stored samples suggests the existence of a wide niche in relation with pollen resources used by T. angustula, a factor that may increase food availability during the dry season thus favoring the development of the hive.
A morphotype of Chenopodiaceae was one of the most frequently occurring pollen types in spite of lacking mellitophilic features, thus supporting the idea that pollination syndrome is less relevant when it comes to food consumption. It is probably used as a resource only during the dry season when other flowering plants become scarce, thus corresponding to a temporary specialization in which benefits to the plant and to its floral visitor are not symmetrical (Rech & Absy 2011a). This may be interpreted as a case of bee plasticity in its collection habits, in which a resource of relatively poorer quality is used because of its high abundance relative to other more rewarding species (Marquez-Souza 2010); alternatively, the flowers of this Chenopodiaceae morphotype may possess some hitherto unknown features that make them attractive to the bee.
Corbicular and stored pollen showed a certain degree of parallelism along time (Fig 3), in spite of the facts that corbicular pollen was assessed during a very short period of time (3 days per month, 3 h per day) in comparison with stored pollen (all hours of bee activity during the whole month) and that blooming individuals may have varied along the month. Moreover, a certain degree of parallelism was observed between families of blooming plants in the transects and pollen collected along time (Fig 3) and between availability of blooming species and of blooming individuals and pollen collected and stored through time (Fig 4). These observations suggest that pollen collection responds to the blooming dynamics in the transect rather than specificity in collection behavior (de Novais et al 2014). This is supported by the high variation of species composition in the transects through time.
The large number of pollen types collected by the bees but not found in the transects could reflect a foraging pattern different from that established by Nogueira-Neto (1997) and by Gamero (2006). For example, Didymopanax and Anadenanthera sp. were among the most frequently occurring pollen types but were absent from the transects. Alternative explanations may be offered: (i) the flight range of the bee under the prevailing environmental conditions could be greater than the 200 m reported for the species (Nogueira-Neto 1997, Gamero 2006), thus having access to flowering species not represented in the transects; (ii) transects may not be representative of all flowering plants within a radius of 200 m from the central hive; and (iii) observations in the transect were performed at floor level whereas the presence of flowers and flower buds at the canopy level could be different (Ramalho 2004), since many species in the transect were tall trees (10 of the 43 species) or epiphytes (4 of the 43 species) growing high up in the canopy.
Some pollinic resources from plants in the transects with mellitophilic flowers were not collected and/or stored by the bees (in fact, only 18 of the 29 mellithophilic species in the transects were used as pollen resources). This is probably related to the bee not foraging on species that are scarcely represented (Vossler 2012); nevertheless, this can also be related to a behavioral preference by this bee towards some specific pollen types. Thus, pollen collection would occur mostly from the most represented species, independent of their quality (Ramalho et al 1990, Vossler 2012). In addition, an initial pollen collection based on resource availability rather than quality may lead to learning and memorization of certain plant features and produce fidelity towards a poorer resource (Nicolson 2007, de Novais et al 2014, Benitez-Vieyra et al 2014). Additional studies are required to evaluate bee preference for different resources available.
References
Absy ML, Rech AR, Ferreira MG (2018) Pollen collected by stingless bees: a contribution to understanding Amazonian diversity. In: Vit P, Pedro SRM, Roubik DW (eds) Pot-pollen in stingless bee melittology. Springer international publishing AG, pp 29–46
Aizen M, Chacoff N (2009) Las interacciones planta-animal como servicio ecosistemico: el caso del mutualismo de la polinización. In: Medel R, Aizen M, Zamora R (eds) Ecología y evolución de interacciones planta-animal. Editorial Universitaria, Santiago de Chile, pp 316–329
Almeida JL, Oliveira E, Soares J, Menezes M, Monica O, Lorezon MC (2012) Floral sources to Tetragonisca angustula (Hymenoptera: Apidae) and their pollen morphology in a Southeastern Brazilian Atlantic Forest. Rev Biol Trop 60:1491–1501
Bascompte J, Jordano P (2008) Redes mutualistas de especies. Investigación y Ciencia 1:50–59
Benitez-Vieyra S, Fornoni J, Pérez-Alquicira J, Boege K, Domínguez CA (2014) The evolution of final—reward correlations in bee- and hummingbird pollinated species of Salvia. Proc Royal Soc B 281:1–6
Carvalho C, Marchini LC (1999) Tipos polinicos colectados por Nannotrigona testaceicornis e Tetragonisca angustula (Hymenoptera, Apidae, Meliponinae). Sci Agric 56:1–6
Celeromics (2009) Cell counting error for manual cell counting. http://www.celeromics.com/ Accessed 5 Oct 2016
Colinvaux P, De Oliveira P, Moreno E (1990) Amazon Pollen Manual and Atlas/ Manual e Atlas Palinológico da Amazonia. Harwood Academic Publishers, Amsterdam 413 pp
Copa AM (2004) Patrones de nidificación de Trigona (Tetragonisca) angustula y Melipona rufiventris (Hymenoptera: Meliponini) en el norte de La Paz, Bolivia. Ecol Apl 3:82–86
De la Riva PA (2004) Evaluación de la densidad de colmenas de abejas sin aguijón (Meliponinae) con relación a la oferta y uso de especies de plantas con flor en diferentes épocas en Santa Fe – Ixiamas (La Paz, Bolivia). Tesis de licenciatura, Carrera de Biología, UMSA. La Paz, Bolivia. 60 pp
de Novais JS, Absy ML, Riveiro F (2014) Pollen types collected by Tetragonisca angustula (hymenoptera: Apidae) in dry vegetation in Northeastern Brazil. Eur J Entomol 111:25–34
de Novais JS, Garcez AC, Absy ML, dos Santos FAR (2015) Comparative pollen spectra of Tetragonisca angustula (Apidae, Meliponini) from the Lower Amazon (N Brazil) and caatinga (NE Brasil). Apidologie 46:417–431
Devisscher T, Malhi Y, Landívar VDR, Oliveras I (2016) Understanding ecological transition under recurrent wildfire: a case study in the seasonally dry tropical forest of the Chiquitania, Bolivia. Forest Ecol Manag 360:273–286
Erdtman G (1986) Pollen morphology and plant taxonomy (an introduction to pollen analysis). EJ Brill, Leiden 426 pp
Ferrufino U, Vit P (2013) Pot-honey of six Meliponines from Amboró National Park, Bolivia. In: Vit P, Silva P, Roubik D (eds) Pot honey a legacy of stingless bees. Springer, New York – EEUU, pp 409–416
Freitas BM, Imperatriz- Fonseca VL, Medina LM, Kleinert AMP, Galetto L, Nates-Parra G, Quezada-Euán JJG (2009) Diversity, threats and conservation of native bees in the Neotropics. Apidologie 40:332–346
Gamero R (2006) Evaluación de la adaptación de la abeja Tetragonica angustula Latreille (Hymenoptera: Apidae: Meliponinae) en cámaras artificiales. Tesis de licenciatura, Carrera de Agronomía, Universidad Earth. Guácimo, Costa Rica, 55 pp
Gentry A (1996) A field guide to the families and genera of woody plants of northwest South America (Colombia, Ecuador, Peru), with supplementary notes on herbaceous taxa. University of Chicago, Chicago 920 pp
Hammer Q, Harper D, Ryan P (2001) PAST: paleontological statistics software package for education and data analysis. Paleontol Electron 4:4
Ibich PL, Merida G (2003) Biodiversidad: la riqueza de Bolivia. Estado de conocimiento y conservación. FAN. Santa Cruz, Bolivia 737 pp
Klein AM, Vaissiere BE, Cane JH, Steffan-Dewenter I, Cunningham SA, Kreme C, Tscharntke T (2007) Importance of pollinator in changing landscapes for world crops. Proc Royal Soc B 274:303–313
Louveaux J, Maurizio A, Vorwohl G (1978) Methods of melissopalynology. Bee World 59:139–157
Marquez-Souza AC, Absy ML, Kerr WE (2007) Pollen harvest features of the Central Amazonian bee Scaptotrigona fuvicutis Moure 1964 (Apidae: Meliponinae), in Brasil. Acta Bot Bras 21:11–20
Marquez-Souza AC (2010) Ocorrencia do pólen de Podocarpus sp. (Podocarpaceae) nas coletas de Frieseomelitta varia Lepeletier 1836 (Apidae: Meliponinae) em uma área de Manaus, AM, Brasil. Acta Bot Bras 24:558–566
Martínez E, Cuadriello J, Ramírez E, Medina M, Sosa M, Melchor J (1994) Foraging of Nannotrigona testaceicornis, Trigona (T. angustula), Scaptotrigona Mexicana and Plebeia sp. in the Tacaná region, Chiapas, Mexico. Grana 33:205–217
Michener CD (2007) The bees of the world. The Johns Hopkins University Press, Baltimore 913 pp
Navarro GI, Vargas G, Jardim A, Toledo M, Barra N, Nee M (1996) Clasificación y diagnóstico de la vegetación para la conservación de la región del Parque Nacional Amboró (Santa Cruz, Bolivia). Informe inedito a FAN/TNC y la DNCB
Navarro GI (2001) Descripción de la vegetación de Potrerillos. Unpublished report, 2 pp
Navarro GI, Maldonado M (2002) Geografía ecológica de Bolivia: vegetación y ambientes acuáticos. Fundación Simón I. Patiño, Cochabamba, Bolivia 719 pp
Navarro GI (2011) Clasificación de la vegetación de Bolivia. Fundación Simón I. Patiño, Santa Cruz 711 pp
Nicolson SW (2007) Nectar consumer. In: Nicolson SW, Nepi M, Pacini E (eds) Nectaries and nectar. Springer, Dordrecht, pp 289–342
Nogueira-Neto P (1997) Vida e criaҫão de abelhas indígenas sem ferrão. Urna Edição Nogueirapis, São Paulo 445 pp
Obregon D, Rodríguez CA, Chamorro FJ, Nates Parra G (2013) Botanical origin of pot- honey from Tetragonisca angustula Latreille in Colombia. In: Vit P, Pedro SR, Roubik D (eds) Pot—honey: a legacy of stingless bees. Springer, New York, pp 337–346
Ollerton J (2017) Pollinator diversity: distribution, ecological function, and conservation. Annu Rev Ecol Evol Syst 48:353–376
Ollerton J, Winfree R, Tarrant S (2011) How many flowering plants are pollinated by animals? Oikos 120:321–326
Quiroz DL, Arreguín ML, Fernández R (2011) Patrones estacionales de utilización de recursos florales por Scaptotrigona hellwegeri en la estación de biología Chamela, Jalisco, México. Palibotánica 31:89–119
Ramalho M (2004) Stingless bees and mass flowering trees in the canopy of Atlantic Forest: a tight relationship. Acta Bot Bras 18:37–47
Ramalho M, Kleinert A, Fonseca I (1990) Important bee plants for stingless bees (Melipona and Trigona) and Africanized honeybees (Apis mellifera) in Neotropical habitats: a review. Apidologie 21:469–488
Rech AR, Absy ML (2011a) Pollen storages in nests of bees of the genera Partamona, Scaura and Trigona (Hymenoptera, Apidae). Rev Bras Entomol 55:361–372
Rech AR, Absy ML (2011b) Pollen sources used by species of Meliponini (Hymenoptera: Apidae) along the Rio Negro channel in Amazonas, Brasil. Grana 50:150–161
Roubik DW, Moreno JE (1991) Pollen and spores of Barro Colorado Island. Missouri Botanical Garden, St. Louis 268 pp
Toledo M, Poorter L, Peña- Claros M, Alarcón A, Balcázar J, Leaño C, Licona JC, Llanque O, Vroomans V, Zuimeda P, Bongers F (2011) Climate is a stronger driver of tree and forest growth rates than soil and disturbance. J Ecol 99:254–264
Vossler F (2012) Estudio palinológico de las reservas alimentarias (miel y masas de polen) de “abejas nativas sin aguijón” (Hymenoptera, Apidae, Meliponini): un aporte al conocimiento de la interacción abejas planta en el Chaco Seco de Argentina. Tesis doctoral, Carrera de Ciencias Naturales, Universidad Nacional de la Plata. La Plata, Argentina, 166 pp
Acknowledgments
We thank UAGRM Botanical Laboratory for their support in the analysis of pollen samples, Herbario del Oriente Boliviano (USZ) for their collaboration with botanical identifications, Museo de Historia Natural Noel Kempff Mercado for logistical support in field trips, and Herbario Nacional de Bolivia for their collaboration in reconfirming the identification of pollen grains. Finally, we thank Tonny Bonaso and the private Natural Reserve “Potrerillos del Güenda” for authorizing and supporting this research.
Funding Information This work received financial support from LANBIO (Latin American Network for Research on Bioactive Natural Compounds) and BOL-01.
Author information
Authors and Affiliations
Corresponding author
Additional information
Edited by Fernando B Noll – UNESP
Rights and permissions
About this article
Cite this article
Saravia-Nava, A., Niemeyer, H.M. & Pinto, C.F. Pollen Types Used by the Native Stingless Bee, Tetragonisca angustula (Latreille), in an Amazon-Chiquitano Transitional Forest of Bolivia. Neotrop Entomol 47, 798–807 (2018). https://doi.org/10.1007/s13744-018-0612-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13744-018-0612-9