Abstract
Biogeography has been difficult to apply as a methodological approach because organismic biology is incomplete at levels where the process of formulating comparisons and analogies is complex. The study of insect biogeography became necessary because insects possess numerous evolutionary traits and play an important role as pollinators. Among insects, the euglossine bees, or orchid bees, attract interest because the study of their biology allows us to explain important steps in the evolution of social behavior and many other adaptive tradeoffs. We analyzed the distribution of morphological characteristics in Colombian orchid bees from an ecological perspective. The aim of this study was to observe the distribution of these attributes on a regional basis. Data corresponding to Colombian euglossine species were ordered with a correspondence analysis and with subsequent hierarchical clustering. Later, and based on community proprieties, we compared the resulting hierarchical model with the collection localities to seek to identify a biogeographic classification pattern. From this analysis, we derived a model that classifies the territory of Colombia into 11 biogeographic units or natural clusters. Ecological assumptions in concordance with the derived classification levels suggest that species characteristics associated with flight performance, nectar uptake, and social behavior are the factors that served to produce the current geographical structure.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
As an empirical, multidisciplinary, and theoretical approach, biogeography aims to explain the conditions and natural phenomena affecting the distribution of organisms (Craw 1983, Wiley 1988, Noonan 1988, Wilson 1991, Crisci & Morrone 1992, Morrone 2001, García-Barros et al 2002, Mackey et al 2008). Different perspectives have been applied to explain such patterns. It is probable that Darwin (1859) made the first contributions to biogeography with his dispersionist theory (Wilson 1991). Due to the lack of an adequate ecological, taxonomic, and systematic consensus, biogeography remains inconsistent (Craw 1983, Seberg 1986, Mackey et al 2008). Biogeographical approaches have a strong ecological background because trends linking organisms with their environment are the principal modulators of such patterns (Pianka 1994, Thompson 2005).
Insects represent 79% of the known global fauna (Noonan 1988). Among insects, bees (Hymenoptera: Apoidea) include the most important pollinator taxa (Danforth et al 2006, Michener 2007). Bees probably originated ca. 125 my ago in extratropical and open areas during the Middle Cretaceous (Roubik 1989), diversifying 125 to 90 my ago in the tropics as they simultaneously tracked the radiation of the angiosperms (Michener 1979). A valid theory explains that bees originated from a wasp-like ancestor (Engel 2000, Poinar & Danforth 2006, Michener 2007), subsequently abandoning their predatory habits to become pollen and nectar feeders as they expanded to temperate regions (Silveira et al 2002, Poinar & Danforth 2006).
The distribution of bees is frequently analyzed according to distinct regional or vegetation terms that are otherwise difficult to define (Roubik & Hanson 2004), and few empirical studies on the geographic distribution of euglossines have been performed (Dick et al 2004, Nemésio 2007b, Ramírez et al 2010).
Of an estimated 20,000–30,000 species worldwide, only 2% to 5% of bees display true social behavior (Michener 2007). Approximately 500 eusocial bee species are reported in the neotropical region, and 240 occur in Colombia (Nates-Parra 2005, Michener 2007). This neotropical diversity includes the orchid bees (Euglossini). These bees are predominant in lowland rain forests (Roubik & Hanson 2004) and are found in habitats from sea level to 2,000 m asl (Ramírez et al 2002, Roubik & Hanson 2004, Nates-Parra et al 2006). It is commonly assumed that orchid bees originated between the Andean foothills and the Amazon region more than 20 my ago with a significant radiation of the group occurred in the Miocene/Pleistocene (Engel 1999, Dick et al 2004, Roubik and Hanson 2004). Recent evidence suggests that euglossines date from the Eocene and that it is not necessarily clear that they originated in South America (Ramírez et al 2010). The current distribution of euglossine bees should reflect geological, environmental, and climatic factors in addition to the occurrence of resource competition, parasites and predators (Nemésio & Silveira 2006, Roubik & Hanson 2004).
Given the relatively recent and widespread distribution of orchid bees (Dick et al 2004), the ancestors of the group should occur at the time of the uplift of the Andean cordillera and at a level of uplift that reached no greater than 40% of the current altitude of the cordillera (Ramírez et al 2010). Bee community structures did not appear in association with the uplift (Gregory-Wodzicki 2000, Dick et al 2004, Michener 2007). In contrast to the cordillera, the Amazon and part of Orinoquia were regions where no important geological activity occurred. In these regions, the most relevant source of variation was the vegetation changes caused by glacial cycles (Colinvaux et al 2000). The Caribbean was influenced by volcanic activity (most likely in the Antilles). In this region, tectonic movements prevailed in the Late Cretaceous. The ocean dynamics should affect the environmental characteristics (Burke 1988).
The tribe Euglossini is represented by five genera with approximately 200 species, of which 110 occur in Colombia (Ramírez et al 2002, Nates-Parra 2005). Although the genera are clearly defined, the occurrence of many differences within the tribe has produced discussions about the relationships among the species (Cameron 2004, Michel-Salzat et al 2004, Michener 2007). Although the Euglossini are accurately characterized as corbiculate (i.e., the hypothesis of monophyly is supported), the biology of the tribe differs from that of its relatives in aspects such as the lack of true social behavior (Noll 2002, Cameron 2004, Nates-Parra 2005) and substantial morphological variability (Michener 2007). Such dissimilarity should contribute to the evolutionary success of the Euglossini (Roubik & Hanson 2004) as well as to their habitat use (Otero & Sandino 2003, Sandino 2004, Uehara-Prado & Garófalo 2006, Parra-H & Nates-Parra 2007).
Adaptation can be linked to certain morphological and behavioral features. In groups that use traplining foraging strategies, for example, tongue length is related to the preference for the nectar of certain species (Ackerman et al 1982, Ackerman 1985, Kato et al 1992, Roubik & Hanson 2004). In addition, the atmospheric pressure influences the process of nectar intake when the tongue is extruded making feeding efficiency (in relation to viscosity) environmentally dependent (Borrell 2004, 2007a, b, Borrell & Krenn 2006). Because nectar resources are equally exploited by males and females (Ackerman 1985), intraspecific variation for tongue length should not be expected between the sexes (Roubik personal communication).
Body size is involved in thermoregulatory capacity and in the optimization of the strength of vertical flight. By increasing the flux of hemolymph from the thorax to the abdomen, large bees can release heat and improve their flight capacity. This ability can be used, for example, for flight across open areas exposed to solar radiation (Inouye 1975, Armbruster & Berg 1994, Dudley 1995, Borrell & Medeiros 2004). Dwarf forms have also been reported in the Euglossini (Roubik 2004). The frequency of these forms is unclear, and their effect on interspecific variability cannot be assessed.
Social behavior significantly contributes to defense against parasites and to resource optimization (Roubik & Hanson 2004), and it occurs in each genus of euglossines. Eufriesea Cockerell is predominantly solitary, although cooperative forms are reported (Kimsey 1982, Eberhard 1989, Garófalo 1994, Soucy et al 2003). More complex levels of cooperation exist in Eulaema Lepeletier and Euglossa Latreille (Bennett 1965, Santos & Garófalo 1994, Augusto & Garófalo 2004, 2009, 2010). Exaerete Hoffmannsegg and Aglae Lepeletier & Serville are obligate parasites of other euglossines (Roubik & Hanson 2004). An example of the biogeographic reliance on social behavior is the comparative study of Nemésio & Silveira (2006), which demonstrated a potential geographical correlation between parasites and hosts.
Despite these results, the knowledge of euglossine nesting biology remains incomplete (information on the nests is only available for approximately 20% of the species) (Roubik & Hanson 2004, Garófalo 2006, Augusto & Garófalo 2010). The available evidence strongly suggests that the euglossine bees are by no means strictly solitary (Soucy et al 2003, Augusto & Garófalo 2004, 2009, 2010, Garófalo 2006, Otero et al 2008, Cocom-Pech et al 2008).
Species adapted to other species and environments show differences in their geographic distributions, and such relations are defined by evolutionary interactions (Thomson 2005). As indicated above, the sensitivity of the Euglossini to habitat conditions is evident and clearly responds to selective pressure along gradients, such as those imposed by relief. Hence, we seek to reveal biogeographical patterns by describing the interaction between the morphological characteristics of the tribe and its species distribution. In addition, a reviewed and updated checklist showing the richness of the orchid bee fauna is presented for the natural regions of Colombia, a country known for its ecosystem diversity and for including part of the region in which the tribe could have originated.
Material and Methods
By reviewing approximately 7,000 specimens of Euglossini from the specialized Mellitology collections at the Museum of the “Laboratorio de Investigaciones en Abejas, Departamento de Biología, Universidad Nacional de Colombia, Sede Bogotá” (LABUN), we assessed the geographical and altitudinal distribution of the group in Colombia. We supplemented the resulting information on species distribution by reviewing approximately 3,000 specimens from the D.W. Roubik bee collection at Smithsonian Tropical Research Institute, Panama. The expected occurrence of species was also assessed based on selected references on Euglossini (see Moure et al 2008) and on a previous proposal by Ramírez et al (2002).
We adopted the natural regions used by Ramírez et al (2002) as our provisional classification of the collection localities. Gorgona Island and the Sierra Nevada de Santa Marta were treated as isolated and independent units belonging to the Pacific and Caribbean regions, respectively. The validity of the taxonomic nomenclature was assessed according to the Moure et al (2008) catalog of the bee fauna of the Neotropics.
We defined the following categories for body size: small, up to 11 mm in length; middle-sized, 12–22 mm in length; and large, greater than 23 mm in length. For tongue length, we used the following four categories proposed by Sandino (1992): very long, exceeding 21 mm; long, 14–20 mm; short, including mouth parts and up to 8 mm; and medium, 9–13 mm. The specific body size dimensions and tongue length given in the original descriptions were used.
For the purpose of categorization, social behavior was determined as follows: Aglae and Exaerete were classified as parasitic (Cameron 2004, Garófalo 2006, Nemésio and Silveira 2006), Eufriesea as solitary (Kimsey 1982, Cameron 2004, Garófalo 2006), Euglossa as communal–solitary (Eberhard 1989, Soucy et al 2003, Augusto & Garófalo 2004, 2009, 2010), and Eulaema as communal (Bennett 1965, Santos & Garófalo 1994, Cameron 2004).
We performed a simple correspondence analysis (CA) followed by hierarchical clustering with Spad® 4.5 software (CEISA-CERESTA, Montreuil, France 2000) to identify patterns of grouping. For diversity, estimators of richness (Chao 2, Jackknife, beta diversity and Species abundance,Shannon diversity, and species evenness (SHE) analysis) and assemblages of similar species we used Past® 2.04 (University of Oslo, Norway 2010). The classes resulting from this analysis provided the data associations used later for regional classifications by comparing them with the species similarity by localities.
Results
We determined 134 orchid bee species for Colombia: Aglae (monotypic), Eufriesea (34 species), Euglossa (74 species), Eulaema (20 species), and Exaerete (5 species). Few euglossine bee species reach 3,000 m asl, and most species are found below 2,000 m asl (Online Supplementary Material Table S1).
According to the CA, the value of inertia was influenced most strongly by body size (32.4%), social structure (32.7%), and tongue length (25.9%). We found significant differences in all cases at α = 0.05. The null model was always supported (H a , λ 1 ≠ λ 2). The hypotheses of equality and homogeneity of the inertia values were rejected (H o , λ 1 = λ 2) (Online Supplementary Material Table S2). We obtained the following richness estimators: Chao 2, 150.687 (variance, 1.375); Jackknife 1, 163.067; Jackknife 2, 175.448; and beta diversity (Whittaker), 4.8294.
By grouping the data into seven levels (classes) with ordinate clustering, we defined a classification model. In the model, the first class comprises large parasitic species with very long tongues. The second class comprises all species that reach 3,000 m asl, typically distributed through Andean municipalities and showing very long tongues, communal behavior, and large body size. The third class includes large communal species with long tongues. The fourth class is represented by solitary species of medium size, primarily from Amazonian departments up to 500 m asl. The fifth class includes species found exclusively in the Andean localities and the Sierra Nevada de Santa Marta. The species belonging to this class were found frequently between 1,500 and 2,000 m asl. This class excludes large and/or solitary–communal species as well as species from the savanna region, Pacific littoral, and Amazon basin. The sixth class is characterized by small solitary–communal species with long tongues found throughout the Pacific municipalities between 1,000 and 1,500 m asl. The seventh class includes small solitary–communal species of medium tongue length from the savanna. This class also includes species from the piedmont of the eastern llanos below 1,000 m asl (Online Supplementary Material Table S2).
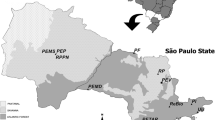
Based on the classification suggested above and species similarities (Online Supplementary Material Fig S3), we suggest 11 biogeographic provinces for Colombia (Fig 1). The high Andean Province includes environments occupied primarily by species with high and proficient flight. The Amazon Provinces comprise the Southern and the Northeastern Amazon Provinces. The Inter-Andean Provinces (below 2,000 m asl) correspond to the Cauca Province and Magdalena Province as well as the Sierra Nevada de Santa Marta, which is evidently related more closely to the Andes than to the Caribbean. The valleys corresponding to those provinces are single, mutually isolated units (Ramírez 2005, Parra-H et al 2006). Not represented in the analysis, but distinguishable by regional richness (most likely because diversity is low relative to the area those communities comprise (Chessman 1929), are the Caribbean Province and Gorgona Island Province. Species from the savannah (Llanos) are excluded from most of the neighboring Provinces, constituting the Orinoquia Province sensu lato. The Piedmont Province emerges as a transition between the communities of the Andes and the “Llanos,” sharing certain species but lacking an exact match at any hierarchical level. The biogeographic Chocó region has an orchid bee assemblage that tends to be segregated into two divisions: North Chocó Province, linked to Central America (Panama), and South Chocó Province, where the assemblage is related to the Peruvian–Ecuadorian Amazonian euglossines. The species that belong to these two communities are distributed around the piedmont of the western cordillera and occur only up to 1,700 m asl.
Natural provinces proposed for Colombia. 1 (all in dense black representing the cordillera) [and(h)]: high Andes (elevations above 2,000 m asl), 2 [ama(s)]: South Amazon, 3 [ama(n)]: Northeastern Amazon, 4 [and(c)]: Inter-Andean valleys–Cauca valley, 5 [and(m)]: Inter-Andean valleys–Magdalena valley and Sierra Nevada, 6 [atl]: Caribbean, 7 [gor-pac]: Gorgona Island, 8 [ori]: Orinoquia, 9 [ori(p)]: Eastern llanos foothills, 10 [pac(n)]: North Chocó, and 11 [pac(s)]: South Chocó.
Discussion
Data reliability and the link between statistical criteria and ecology
The morphological and behavioral variety of the tribe Euglossini reflects its diversification within a changeable ecosystem. Unless it is already well studied, the taxonomy of orchid bees needs to be reviewed from novel perspectives (see Roubik 2004), consensus on taxonomic status, and emendations [e.g. Euglossa azureoviridis Friese was synonymized under Euglossa milenae Bembé unless both species are cited as valid with distinct distributions: (see Bembe 2007, Moure et al 2008)]. In addition, surveys of poorly explored regions are needed (González & Engel 2004, Ramírez 2005, Parra-H et al 2006). Nevertheless, specific studies of the Colombian bee fauna (Bonilla-Gómez & Nates-Parra 1992, Ospina-Torres 1998, Roubik 2004, Ramírez et al 2002, 2010, Ramírez 2005, Parra-H et al 2006, Ospina-Torres et al 2006) and a good representation in the museums whose collections were assessed for this study (76% of all the specimens included in the present inventory are deposited at LABUN) allowed us to form an accurate picture of the occurrence of orchid bee species (but not their abundance). This background is related to the validity of the scheme that we have used to organize our data. The background allowed us to determine that our sample was adequate to define the incidence of the species of interest according to the locality and, therefore, according to province. However, in some cases, species seasonality was not adequately represented in the sample.
The assumption of monophyly for the corbiculate clade for behavioral categorization could represent a bias because it does not necessarily justify treating the species as independent. The disparity of sampling effort found in the museum records and references used could represent a drawback and produce sampling artifacts. Correspondence analysis solves such difficulties by converting data distributions into a continuous set. It derives simplified and improved patterns from the first ordination axis and relegates noise to later axes. This approach yields meaningful grouping patterns because uninformative data will be separated from meaningful data (e.g., Smith & Bermingham 2005).
The consistency of the data analysis was also reflected by the high efficiency of the test procedure used. The results of the correspondence analysis and ordination explained more than 72% of the variance at the tenth histogram division (Table 1). Because reproducibility is frequently difficult in practice (Casey & Blackburn 2006), we expect that the benefit of our approach will be its utility as a method for interpreting traits, but only if relevant and timely environmental ecological criteria are involved. The biogeographic delimitation of species throughout and following vegetation units and from a taxonomic perspective will merely reflect contingencies and be of value only for mapping (Mackey et al 2008).
Richness, diversity, and the natural clusters
The values of richness obtained from all the methods used were greater than the known values. The tendency of the methods to produce such estimates independently clearly shows that the diversity in the region analyzed is greater than that currently supposed. Within the limits of resolution of our methods, SHE analysis allowed us to elucidate how the community is sensitive to structuring across the resulting gradient (provinces). The method described possible discontinuities in the distribution of a sample depending on richness, evenness, and diversity. When both richness and diversity increase simultaneously as species evenness decreases, structuring events should be expected (Fig 2); (Buzas & Hayek 1998).
Applying this criterion to the community array obtained from our analysis, we find evidence of maximum levels of interaction before structuring for provinces with ca. 25 species, 50 species and 70 species each (Fig 3). Moreover, in conjunction with the evenness of taxa and the increase in diversity per sampled locality, if we exclude adverse habitats such as the high Andes, isolated Gorgona Island, and the homogeneous Caribbean region, the value of regional beta diversity was found to be high compared with other similar surveys of bees in Colombia (see Nates-Parra & Parra-H 2006). These findings reinforce the idea that all the biogeographic provinces identified interact ecologically, particularly in the South Amazon (Fig 3).
Our clusters appear to represent units where species richness is limited by ecological tradeoffs resulting from interactions in the community. This classification should not be totally in agreement with those barriers representing rigid delimitations of ecosystems and between natural units. Our clusters are areas of transitional and continual interfaces. Our “lines” represent a possible breakpoint in the distribution of traits. More importantly, they reveal the niche response surface for which the community will have a maximum probability of occurrence as a result of the interactions produced by adaptation.
Ecological and biogeographic significance
The biogeographic regions previously considered for the distribution of orchid bees in Colombia are not consistent with our results. Nevertheless, the resulting classification is very similar to those obtained with other criteria, e.g., the biogeographic representation of districts according to conservation criteria (Vásquez & Serrano 2009, Hernández et al 1991). The overlap found between orchid bee communities and ecosystems indicates that the features analyzed are indeed those that mediated the adaptation of the bees to these particular habitats. This overlap does not represent a direct correlation between bee traits (e.g., long tongues) and vegetation characteristics (e.g., deep corollas).
he behavioral assumptions used in this study should be viewed with caution because, as stated above, further observations on euglossine nesting biology are needed. Regardless of the organizational advantages offered by CA, the correct interpretation of the high inertia attributed to social structure by the analysis is that nesting biology is both a keystone and a checkpoint for orchid bees’ evolutionary success through the availability of different nesting substrates along spatial, temporal, and environmental gradients.
The development of an accurate distributional framework for organisms that resembles their natural history relationships is complex. An improved understanding and a clarified consensus on taxonomic identity are needed. In this case, the ecology of this outstanding group of neotropical pollinators appears to reflect not only the morphology of the organisms but also other properties of the group. Ecological approaches will demonstrate that the absence of records of occurrence in certain areas can be addressed in terms of habitat affinity, thereby predicting the linkage between species and habitats (the linkage between ecology and biogeography) and the occurrence of species. For example, Eufriesea bare (González & Gaiani) and Eufriesea danielis (Schrottky) were considered to occur exclusively in the Venezuelan Amazon basin. However, these species are actually collected in juxtaposed areas of the northeastern Colombian Amazon.
Another concern is the status of sibling species (see Roubik 2004). A recent species description affirms that Euglossa sovietica Nemésio occurs exclusively in the Brazilian Amazon basin. However, our criteria indicate that the species should also be expected in the southern Colombian Amazon. This result suggests that such species may not be distinct; E. sovietica is exactly identical to Euglossa igniventris Friese (see Nemesio 2007a, b for discussion). E. igniventris has a broad distribution through Central America, North Chocó region, and the Eastern “Llanos” foothills of Colombia. Because this distribution represents a typical (but infrequent) disjunct array, the species should also be expected in the southern Colombian Amazon basin. The situation may involve sibling species, as described by Roubik (2004) for other members of the genera Glossura (Cockerell) and Glossuropoda (Moure) or it could represent an effective taxonomic divergence.
A specific examination of the relevant phenotypic traits from an ecological and evolutionary perspective will be needed to relate the trait to tradeoff mechanisms and the way in which drivers operate to produce adaptation. Nevertheless, it will not be possible to wait for accurate information or to obtain inferences about aspects of an organism’s biology. The groups resulting from the implementation of our methodological approach present a foundation for determining the geographical scale on which the evolutionary framework provides a basis for diversification.
References
Ackerman JD (1985) Euglossine bees and their nectar hosts. In: D’Arcy WG, Correa MD (eds) The botany and natural history of Panama. Missouri Botanical Garden, St. Louis, pp 225–233
Ackerman JD, Mesler MR, Lu KL, Montalvo AM (1982) Food-foraging behavior of male Euglossini (Hymenoptera: Apidae): vagabonds or trapliners? Biotropica 14:24–248
Armbruster WS, Berg EE (1994) Thermal ecology of male euglossine bees in a tropical wet forest: fragrance foraging in relation to operative temperature. Biotropica 26:50–60
Augusto SC, Garófalo CA (2004) Nesting biology and social structure of Euglossa (Euglossa) townsendi Cockerell (Hymenoptera, Apidae, Euglossini). Insect Soc 51:400–409
Augusto SC, Garófalo CA (2009) Bionomics and sociological aspects of Euglossa fimbriata (Apidae, Euglossini). Genet Mol Res 8:525–538
Augusto SC, Garófalo CA (2010) Task allocation and interactions among females in Euglossa carolina nests (Hymenoptera, Apidae, Euglossini). Apidologie 42:162–173
Bembé B (2007) Revision der Euglossa cordata-gruppe und untersuchungen zur funktionsmorphologie und faunistik der euglossini (Hymenoptera, Apidae). Entomofauna Suppl 14:1–146
Bennett FD (1965) Notes on a nest of Eulaema terminata Smith (Hymenoptera: Apoidea) with a suggestion on the occurrence of a primitive social system. Insect Soc 1:81–92
Bonilla-Gómez MA, Nates-Parra G (1992) Abejas euglossinas de Colombia (Hymenoptera: Apidae). I. Claves ilustradas. Caldasia 17:149–172
Borrell BJ (2004) Suction feeding in orchid bees (Apidae: Euglossini). Proc R Soc B 271:164–166
Borrell BJ (2007a) Mechanics of nectar feeding in the orchid bee Euglossa imperialis: pressure, viscosity and flow. J Exp Biol 209:4901–4907
Borrell BJ (2007b) Scaling of nectar foraging in orchid bees. Am Nat 169:569–580
Borrell BJ, Krenn HW (2006) Nectar feeding in long proboscid insects. In: Herrel A, Speck T, Rowe N (eds) Ecology and biomechanics: a mechanical approach to the ecology of animals and plants. CRC, Boca Raton, pp 185–212
Borrell BJ, Medeiros MJ (2004) Thermal stability and muscle efficiency in hovering orchid bees (Apidae: Euglossini). J Exp Biol 207:2925–2933
Buzas MA, Hayek LC (1998) SHE analysis for biofacies identification. J Foramin Res 28:233–239
Burke K (1988) Tectonic evolution of the Caribbean. Annu Rev Earth Planet Sci 16:201–230
Cameron SA (2004) Phylogeny and biology of Neotropical orchid bees (Euglossini). Annu Rev Entomol 49:377–404
Cassey P, Blackburn TM (2006) Reproducibility and repeatability in ecology. Bioscience 56:958–959
Cheesman LE (1929) Hymenoptera collected on the “St. George” Expedition in Central America and the W. Indies. Trans Entomol Soc Lond 77:141–154
Cocom-Pech ME, W-de-J M-I, Medina-Medina LA, Quesada-Euán JJG (2008) Sociality in Euglossa (Euglossa) viridissima Friese (Hymenoptera, Apidae, Euglossini). Insect Soc 55:428–433
Colinvaux PA, de Oliveira PE, Bush MB (2000) Amazonian and Neotropical plant communities on glacial time scales: the failure of the aridity and refuge hypotheses. Quat Sci 19:141–169
Craw RC (1983) Panbiogeography and vicariance cladistics: are they truly different? Syst Zool 32:431–438
Crisci JV, Morrone JJ (1992) A comparison of biogeographic models: a response to Baston Wilson. Glob Ecol Biogeogr 2:174–176
Danforth BN, Sipes S, Fang J, Brady SG (2006) The history of early diversification based on five genes plus morphology. PNAS 103:15118–15123
Darwin C (1859) On the origin of species by means of natural selection. Murray, London
Dick CW, Roubik DW, Gruber KF, Bermingham E (2004) Long-distance gene flow and cross-Andean dispersal of lowland rainforest bees (Apidae: Euglossini) revealed by comparative mitochondrial DNA phylogeography. Mol Ecol 13:3775–3785
Dudley R (1995) Extraordinary flight performance of orchid bees (Apidae: Euglossini) hovering in heliox (80% He/20% O2). J Exp Biol 198:1065–1070
Eberhard WG (1989) Group nesting in two species of Euglossa bees (Hymenoptera: Apidae). J Kansas Entomol Soc 61:406–411
Engel MS (1999) The first fossil of Euglossa and phylogeny of the orchid bees (Hymenoptera: Apidae: Euglossini). Am Mus Novit 3272:1–14
Engel MS (2000) Monophyly and extensive extinction of advanced eusocial bees: insights from an unexpected Eocene diversity. PNAS 98:1661–1664
García-Barros E, Gurrea P, Luciáñez MJ, Cano JM, Munguira ML, Moreno JC, Sainz H, Simón JC (2002) Parsimony analysis of endemicity and its application to animal and plant geographical distributions in the Ibero-Balearic region (western Mediterranean). J Biogeogr 29:109–124
Garófalo CA (1994) Biologia de nidificação dos Euglossinae (Hymenoptera: Apidae). Encontro Sobre Abelhas 1:17–26
Garófalo CA (2006) Comportamento social nos Euglossini (Hymenoptera, Apidae). Encontro Sobre Abelhas 7:24–27
González VH, Engel MS (2004) The tropical Andean bee fauna (Insecta: Hymenoptera: Apoidea), with examples from Colombia. Entomol Abhandlungen 62:65–75
Gregory-Wodzicki KM (2000) Uplift history of the Central and Northern Andes: a review. Geol Soc Am Bull 112:1091–1105
Hernández JA, Ortiz R, Walschburger T (1991) Unidades biogeográficas de Colombia. In: Hernández JA, Ortiz R, Walschburger T (eds) Estado de la biodiversidad en Colombia. Informe final. Instituto Colombiano para el Desarrollo de Ciencia y Tecnología “Francisco José de Caldas”–Colciencias, Bogotá
Inouye DW (1975) Flight temperatures of male Euglossine bees (Hymenoptera: Apidae: Euglossini). J Kansas Entomol Soc 48:366–370
Kato M, Roubik DW, Inoue I (1992) Foraging behavior and concentration preferences of male Euglossine bees (Hymenoptera: Apidae). Tropics 1:259–264
Kimsey LS (1982) Systematics of bees of the genus Eufriesea (Hymenoptera, Apidae). Univ Calif Publ Entomol 95:1–125
Mackey BG, Berry SL, Brown T (2008) Reconciling approaches to biogeographical regionalization: a systematic and generic framework examined with a case study of the Australian continent. J Biogeogr 35:213–229
Michel-Salzat A, Cameron SA, Oliveira ML (2004) Phylogeny of the orchid bees (Hymenoptera: Apinae: Euglossini): DNA and morphology yield equivalent patterns. Mol Phylogenet Evol 32:309–323
Michener CD (1979) Biogeography of the bees. Ann Mo Bot Gard 66:277–347
Michener CD (2007) The bees of the world. Johns Hopkins University Press, Baltimore
Morrone JJ (2001) Homology, biogeography and areas of endemism. Divers Distrib 7:297–300
Moure JS, Melo GAR, Faria Jr LRR (2008) Euglossini Latreille, 1802. In: Moure JS, Urban D, Melo GAR (Orgs). Catalogue of bees (Hymenoptera, Apoidea) in the neotropical region. Universidade Federal de Paraná. Online
Nates-Parra G (2005) Abejas corbiculadas de Colombia (Hymenoptera: Apidae). Unibiblos, Universidad Nacional de Colombia, Bogotá, Colombia
Nates-Parra G, Parra-H A (2006) Uso de las Abejas Silvestres en la Definición de Áreas Prioritarias de Conservación en el Territorio CAR. Tacayá 14:4–7
Nates-Parra G, Parra-H A, Rodríguez A, Baquero P, Vélez D (2006) Abejas silvestres (Hymenoptera: Apoidea) en ecosistemas urbanos: Estudio en la ciudad de Bogotá y sus alrededores. Rev Colomb Entomol 32:77–84
Nemésio A (2007a) Three new species of Euglossa Latreille (Hymenoptera: Apidae) from Brazil. Zootaxa 1547:21–31
Nemésio A (2007b) The community structure of male orchid bees along the Neotropical region. Rev Bras Zool 9:151–158
Nemésio A, Silveira FA (2006) Deriving ecological relationships from geographic correlations between host and parasitic species: an example with orchid bees. J Biogeogr 33:91–97
Noll FB (2002) Behavioral phylogeny of corbiculate Apidae (Hymenoptera: Apinae), with special reference to social behavior. Cladistics 18:137–153
Noonan GR (1988) Biogeography of North American and Mexican insects, and a critique of vicariance biogeography. Syst Zool 37:366–384
Ospina-Torres R (1998) Revisión de la morfología genital masculina de Eulaema (Hymenoptera; Apidae). Rev Biol Trop 46:749–762
Ospina-Torres R, Parra-H A, González VH (2006) The male gonostylus of the orchid bee genus Euglossa (Apidae: Euglossini). Zootaxa 1320:49–55
Otero JT, Sandino JC (2003) Capture rates of male Euglossine bees across a human intervention gradient, Chocó region, Colombia. Biotropica 35:520–529
Otero JT, Ulloa-Chacón P, Silverstone-Sopkin P, Giray T (2008) Group nesting and individual variation in behavior and physiology in the orchid bee Euglossa nigropilosa Moure (Hymenoptera, Apidae). Insect Soc 55:320–328
Parra-H A, Nates-Parra G (2007) Variación de la comunidad de abejas de las orquídeas (Hymenoptera: Apidae) en tres ambientes perturbados del piedemonte llanero colombiano. Rev Biol Trop 55:931–941
Parra-H A, Ospina-Torres R, Ramírez S (2006) Euglossa natesi n. sp., a new species of orchid bee from the Chocó region of Colombia and Ecuador (Hymenoptera: Apidae). Zootaxa 1298:29–36
Pianka ER (1994) Evolutionary ecology, 5th edn. Harper Collins, New York
Poinar GO Jr, Danforth BN (2006) A fossil bee from Early Cretaceous Burmese amber. Science 314:614
Ramírez S (2005) Euglossa paisa, a new species of orchid bee from the Colombian Andes (Hymenoptera: Apidae). Zootaxa 1065:51–60
Ramírez S, Dressler RL, Ospina M (2002) Orchid bees (Hymenoptera: Apidae: Euglossini) from the Neotropical Region: a species checklist with notes on their biology. Biota Colombiana 3:7–118
Ramírez S, Roubik DW, Skov C, Pierce NE (2010) Phylogeny, diversification patterns, and historical biogeography of euglossine orchid bees (Hymenoptera: Apidae). Biol J Linn Soc 100:552–572
Roubik DW (1989) Ups and downs in pollinator populations: when is there a decline? Conserv Biol 5:1–30
Roubik DW (2004) Sibling species of Glossura and Glossuropoda in the Amazon region (Hymenoptera: Apidae: Euglosssini). J Kansas Entomol Soc 77:235–253
Roubik DW, Hanson PE (2004) Orchid bees of tropical America: biology and field guide. INBIO, Heredia
Sandino JC (1992) Primer inventario de abejas euglosinas (Hymenoptera: Apidae: Euglossini) en la vertiente del Pacífico del sur occidente de Colombia: Diversidad, Distribución altitudinal y efectos de la perturbación humana. Dissertation Thesis, Universidad del Valle, Facultad de Ciencias, Plan de estudios de Biología. Cali. Colombia
Sandino JC (2004) Are there any agricultural effects on the capture rates of male Euglossine bees (Apidae: Euglossini)? Rev Biol Trop 52:115–118
Santos ML, Garófalo CA (1994) Nesting biology and nest re-use of Eulaema nigrita (Hymenoptera: Apidae, Euglossini). Insect Soc 41:99–110
Seberg O (1986) A critique of the theory and methods of panbiogeography. Syst Zool 35:369–380
Silveira FA, Melo GAR, Almeida EAB (2002) Abelhas Brasileiras: Sistemática e Identificação. Belo Horizonte, Brazil
Smith SA, Bermingham E (2005) The biogeography of lower Mesoamerican freshwater fishes. J Biogeogr 32:1835–1854
Soucy SL, Giray T, Roubik DW (2003) Solitary and group nesting in the orchid bee Euglossa hyacinthina (Hymenoptera: Apidae). Insect Soc 50:248–255
Thompson JN (2005) The geographic mosaic of coevolution. University of Chicago Press, Chicago
Uehara-Prado M, Garófalo CA (2006) Small-scale elevational variation in the abundance of Eufriesea violaceae (Blanchard) (Hymenoptera: Apidae). Neotrop Entomol 33:446–451
Vásquez-V VH, Serrano-G MA (2009) Las Áreas Naturales Protegidas de Colombia–Conservación Internacional Colombia & Fundación Biocolombia. Editorial Panamericana Formas e Impresos, Bogotá
Wiley EO (1988) Parsimony and vicariance biogeography. Syst Zool 37:271–290
Wilson JB (1991) A comparison of biogegraphic models: migration, vicariance and panbiogeography. Glob Ecol Biogeogr 1:84–87
Acknowledgments
The authors are grateful to José Fernando Zea for his assistance in developing the multivariate test; Brendan J. Borrell, Marcio Luiz de Oliveira, and Marco Gaiani for providing valuable literature; and David W. Roubik for kindly allowing access to his bee collection. We thank DWR, Carlos Alberto Garófalo, and anonymous reviewers for comments on a previous version of the manuscript. We also thank the Universidad Nacional de Colombia, Facultad de Ciencias, Departments of Biology and Statistics for supporting the present study and “Convocatoria Nacional Fortalecimiento de la Visibilidad de Producción Academica mediante el Apoyo para Traducción y Corrección de Estilo de Artículos de Investigación 2011-2012” center for publication support (DIB). The present work is part of the undergraduate dissertation of APH at LABUN. APH is currently a Pre Doctoral fellow of “Coordenação de Aperfeiçoamento de Pessoal de Nível S\uperior” (CAPES), “Programa de Estudantes de Convenio–Pós-Graduação” (PEC-PG).
Author information
Authors and Affiliations
Corresponding author
Additional information
Edited by Fernando B Noll – UNESP
Electronic Supplementary Material
S1. Distribution of orchid bees in Colombia. Abbreviations (specific province): ama Amazon (n north or s south), and Andes (c Cauca valley, m Magdalena valley, or h high Andes), and-atl Sierra Nevada de Santa Marta, atl Atlantic, ori Orinoquia (p piedmont), pac Pacific (n north or s south), gor-pac Gorgona, single asterisk recently described species, double asterisks new records for Colombia, white circle new locality record in Colombia, double circles new altitudinal records, expected distributions are given in parentheses.
S2. Classes resulting from the hierarchical classification.
S3. Euclidean dendrogram for species similarity.
ESM 1
(DOC 405 kb)
ESM 2
(JPEG 843 kb)
Rights and permissions
About this article
Cite this article
Parra-H, A., Nates-Parra, G. The Ecological Basis for Biogeographic Classification: an Example in Orchid Bees (Apidae: Euglossini). Neotrop Entomol 41, 442–449 (2012). https://doi.org/10.1007/s13744-012-0069-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13744-012-0069-1