Abstract
Purpose of Review
Individuals with dementia (IwD) have unique characteristics related to movement quality, motor learning strategies, and interpersonal needs. Without an understanding of these issues, physical therapists (PTs) treat these patients as they would cognitively intact older adults and become frustrated or disheartened when patients do not respond to therapy as expected. The purpose of this article is to facilitate the success of therapeutic interventions with IwD by suggesting therapeutic strategies that play to the strengths of this population.
Recent Findings
Classic and recent literature related to motor control and motor learning in IwD is reviewed, and relevant clinical applications are highlighted. Key components related to successful interventions in existing evidence are presented.
Summary
Armed with an understanding of the unique qualities related to movement disorders and motor learning, PTs will be better equipped to exploit the strengths of IwD for optimal rehab outcomes.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
One in ten Americans over age 65 is living with Alzheimer’s disease (AD), the most common of the dementias [5]. As baby boomers age, rehab professionals are more and more likely to treat clients with dementia. A substantial proportion of patients receiving care in orthopedic outpatient, acute care, and sub-acute patient settings are older adults, so an appreciation of the intricacies of individuals with dementia (IwD) is important not only for those physical therapists (PTs) who work exclusively in long-term care or geriatrics but in almost all practice settings.
Physical therapy and exercise can be of great benefit to IwD, but PTs must recognize that the needs of these patients are different than those in whom cognition is intact. When PTs treat IwD as they would their cognitively intact age-matched peers, they are unlikely to get the results they anticipate. PTs often become frustrated and a sense of therapeutic nihilism may overshadow future efforts at rehab with this population. This is evident when patients who do not follow commands become “low priority” on the therapy list! Even therapists who work routinely with IwD feel as though they could benefit from further education related to optimizing interventions with this group [16, 82].
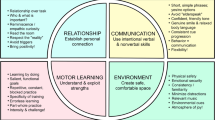
The purpose of this article is to facilitate the understanding of the unique needs of IwD within a rehabilitation context. If PTs design their therapeutic interactions to exploit the strengths of IwD, they will have greater rehabilitation success. To do this, therapists must acknowledge and understand [1] the movement disorders often seen in dementia, [2] the intricacies of motor learning in this population, and [3•] the importance of the therapeutic relationship. When rehabilitation efforts are put in a context that respects these issues, as well as the basic physiological principles behind effective rehabilitation (e.g., appropriate level of challenge and intensity), therapeutic success is much more likely. Given the potential impact of maintaining functional independence and decreasing burden of care, IwD should be among our highest priorities.
Brief Overview of Types of Dementia
Dementias are a group of chronic, progressive, irreversible conditions that result in the death of CNS neurons. There is no effective treatment and no cure for any of the dementias. Alzheimer’s disease (AD) is by far the most common of the dementias. It generally has a slow steady trajectory with memory complaints routinely being the initial symptom associated with the predictable initial pathology in the hippocampus and medial temporal lobe. Vascular dementia (VaD), the second most common type of dementia, can display more of a step-wise progression, with onset of new symptoms linked to new vascular insults. While memory complaints may be the initial symptom, individuals with vascular dementia will demonstrate difficulty based upon the brain regions impacted by vascular pathology, so initial complaints may include cognitive or language issues. Lewy body dementia (LBD) is the third most common dementia and clinically contrasts AD and VaD in its fluctuations in signs and symptoms over time. Individuals with Parkinsons disease dementia (PDD) share the same dementia pathology as LBD and often, individuals with LBD have some of the movement disorders familiar to PD. Mixed pathology dementias, commonly AD or LBD combined with VaD, are likely a common occurrence in dementia in older adults. Fronto-temporal dementia (FTD) is the most common of the young-onset dementias (diagnosed under age 60), and the hallmark symptoms of FTD are personality and behavior changes or language issues, with sparing of memory.
Movement Disorders
While dementia is considered a cognitive disorder, the definition of dementia includes a component of functional decline. The functional deterioration is certainly related to cognition, but there are also movement disorders that are observed in IwD that impact function. These come in the form of gait changes, diminished postural control, apraxia, bradykinesia, and paratonia.
Gait Changes
There is substantial evidence that cognition and gait are intricately related [7•, 8, 9, 93, 105], and several longitudinal studies have demonstrated that gait changes are predictive of cognitive decline in older adults [7•, 46•, 50, 98, 104]. The changes in gait parameters that are seen in typical aging are magnified in IwD. As compared to older adults with cognition intact, IwD display decreased gait speed; decreased step and stride length; increased single limb stance time and double limb support time; and increased variability of stride length, width, and time [6, 7•, 8, 62, 101, 109]. There is some evidence that etiology of dementia may impact the severity of gait and balance issues. This is an area under continuous study, but gait disturbances may be more predictive of and pronounced in individuals with non-AD dementias, such as LBD and FTD [7•, 30, 72]). Although, some researchers have found that gait speed is related to executive function irrespective of dementia type [42, 90].
Anticipating and recognizing decline in gait function is a staple of geriatric PT. Gait speed is considered a “vital sign” of the older adult [31], and the implications of slowing gait, given the relationship of gait speed with functionality and mortality, are a key component of the assessment and treatment of all older adults, with or without cognitive impairment. Encouraging targeted bouts of fast walking in the context of PT or exercise interventions provides practice and confidence in this skill. Educating caregivers about the importance of walking at speeds greater than “mosey” or “stroll” can make a difference in everyday activity of IwD and help them to maintain gait speed over time. Gait speed has been shown to be responsive to training in this population [3•, 12•, 54].
Diminished Postural Control (Balance and Falls)
It is conservatively estimated that IwD fall at least two to three times more than older adults without dementia, regardless of setting [4, 24, 27]. Decreased gait speed and increased gait variability have been associated with falls [2, 61], and there is some evidence of balance deficit preceding dementia diagnosis [51, 53]. Executive function has been repeatedly associated with gait slowing and with falls [11, 44•, 65] and has been identified as a reasonable predictor of falls in community-dwelling older adults [64]. In a study demonstrating the deterioration of balance control with cognitive decline in individuals with AD, executive function was determined to be the most relevant of cognitive measures to all aspects of balance control [88]. PTs do not routinely assess executive function as a part of falls screening. In fact, a recent study identified that fewer than 33% of PTs surveyed included cognitive screening as a component of falls risk assessment, and when they did screen cognition, most commonly, it was in the form of assessing orientation or administering the Mini Mental State Exam, neither of which is a strong indicator of executive function [10]. Perhaps the easy addition of the Mini-Cog [13] as a cognitive screen for older adults without any diagnosis of cognitive deficit would be appropriate. Mini-Cog is a 3-min assessment tool that assesses three-word registration (i.e., can the individual repeat the words?), clock drawing test (i.e., provided with a blank circle, can the individual create a clock face and register the time as 11:10?), followed by remembering the three words (i.e., can the individual recall the three words after being distracted by the clock drawing test?). A simple algorithm for scoring identifies whether an individual requires further work up for cognitive deficit.
Postural control mechanisms are thought to be effected in many ways in dementia; most notably individuals with dementia experience visual perceptual changes that can impact their balance control systems, but they are also slower to process sensory data and less agile in their motor responses [59]. IwDs demonstrate poorer postural stability and more rapid decline than do their age-matched peers as evidenced by computerized posturography and clinical static and dynamic balance measures [85,86,87]. Efforts at systematic review of the literature on the topic of postural stability in dementia have been complicated by the variability in methodology of studies [17]. A recent review of postural control in AD suggests that individuals with mild to moderate dementia display impaired static and functional (in task context) postural stability as compared to healthy peers, and the most influential factors contributing to this postural instability are increased attentional demand (dual tasking) and decreased visual input [63]. There are obvious implications here for training.
Fall determinants in dementia overlap with the multitude of intrinsic and extrinsic risks identified for older adults without cognitive impairment. As with all older adults, a history of falls, polypharmacy, and balance and gait deficits increase the risk of future falls in IwD. Some unique risks for IwD include behavioral issues (e.g., disruptive, attention-seeking behaviors), visual perceptual problems, caregiver burden, and, specific to the long-term care environment, psychotropic drugs and restraints [26, 49]. Comprehensive falls screening and a dedicated multidisciplinary team approach is likely the best way to address fall risk. Rehabilitation professionals bring expertise in physical and functional training, but medical and nursing staff may be pivotal to troubleshooting polypharmacy and behavioral issues.
Balance has been demonstrated to be responsive to training in this population [3•, 12•, 57, 71]. Across the spectrum of mild to severe dementia, combined with any level from mild to significant balance impairment, PTs must introduce the appropriate level of therapeutic balance challenge. If patients do not experience loss of balance in the context of therapy, the training is too easy.
Apraxia
Normal motor planning, resulting in skilled movement, may be framed within the conceptual-production systems model, the components of which are the sensory-perceptual system which processes sensory input (auditory, visual, tactile), the conceptual system which stores knowledge of motor actions and tools, and the production system which retrieves appropriate motor responses based upon the first two components [56, 81]. These functions are thought to be housed in discrete anatomical brain regions. Apraxia, or difficulty with motor planning, was historically considered one of the defining features of AD, and it is seen with increased prevalence as severity of AD increases [81]. Apraxia is evident in 32–35% of individuals with AD [68, 80] and is found in other dementias as well, particularly FTD (which includes the disorder of primary progressive aphasia). Limb apraxia is more common in AD whereas buccofacial apraxia is more common in FTD [1, 43]. Limb apraxia is often evident in self-care skills when patients struggle to use a shoe horn or a toothbrush or some other implement they have been using effectively for decades. There is some indication that providing verbal cuing to an apraxic patient may be counterproductive and may overload an already engaged left hemisphere (i.e., dominant hemisphere for right-handed movement and language comprehension) [81]. Apraxia impacts an individual’s ability to pantomime or imitate gestures as well, so PTs may have greater success with guidance and tactile cuing over demonstration.
Bradykinesia
Bradykinesia, an extrapyramidal motor sign, is an anticipated finding in Parkinson’s disease dementia (PDD) and Lewy body dementia (LBD) [30], but it also may be present in AD, especially with advanced disease and/or individuals experiencing neuropsychiatric symptoms (e.g., hallucinations) [95, 96]. Bradykinesia in the context of dementia likely has a large component of bradyphrenia (slowness of thought and processing). Integrating fast and/or large amplitude motions in the context of rehab for this population seems justifiable, but has not been studied.
Paratonia
Paratonia is a unique presentation of hypertonia characterized by involuntary resistance to passive movement or pressure that increases with the progression of dementia [39, 97]. Paratonia, sometimes referred to as Gegenhalten tone, may be present in any of the dementias, although individuals with dementia of vascular etiology or diabetes mellitus appear to be at higher risk [40]. It is distinctly different than rigidity or spasticity, and characteristics of paratonia include involuntary variable resistance during passive movement (may be in any direction), degree of resistance correlates to speed of movement, resistance felt in two movement directions of one limb or two separate limbs, and absence of clasp-knife phenomenon [38]. As it progresses, paratonia can complicate the ability to assist functional movements and can ultimately lead to immobility and contractures. Mobilizing individuals with advanced dementia and paratonia can be difficult; however, it is possible to exploit this unique tone to facilitate movement. For example, in assisting an IwD to move forward in a chair in preparation for standing, the PT may intuitively pull the individual forward with hands placed on the posterior aspect of the shoulders. An individual with paratonia will respond by pushing backward into the cue of the hands so he or she is pushing back into the chair rather than moving the upper body forward; the PT and IwD are effectively fighting each other (Fig. 1a). If the therapist modifies hand placement to be on the anterior aspect of the individual’s shoulders, the IwD will respond by pushing into the pressure of the hands, facilitating movement in the proper direction to achieve the forward weight shift in preparation for mobility (Fig. 1b). This tactic may work for supine to sit transitions—counterintuitively, the PT will give the cue of pushing the person back into the bed they are trying to get them to rise from. In transitioning sit to stand, this manual technique can work if the PT strategically starts with posteriorly directed pressure, causing the patient to lean anterior, and transitions to inferiorly directed pressure, to facilitate the motion to stand (Fig. 1c). While these techniques work on only a select group of patients with significant paratonia, when they do work, they can substantially reduce the burden of care for caregivers responsible for mobilizing them.
a If the PT tries to “pull” the patient with paratonia forward in the chair, the patient will push backward. b If the PT places hands anteriorly to gently “push” the patient with paratonia back, the patient will lean forward. c Strategic use of force first posteriorly and then downward can facilitate standing in a patient with paratonia.
Excess Disability
Excess disability is a construct that suggests that individuals with dementia often appear more functionally disabled than they should, given their physical impairments [15, 92]. Likely, this is a function of both the patient and the caregiver. It takes longer and is more tenuous to allow an IwD independence in performing tasks; thus, caregivers often step in, with the noble intent of enhancing safety and efficiency and perhaps avoiding frustration for all. The unintended result is that individuals lose the opportunity to practice tasks and over time lose the ability to perform the tasks altogether. Caregivers are often quick to provide assistance with tasks such as walking to and getting into the car (e.g., offering a hand while walking, opening the car door) or walking while carrying things (e.g., a bag of groceries from the car to the house, a plate of food from the counter to the table). Without the opportunity to practice these tasks, skill proficiency suffers. Fortunately, with mindful strategies, excess disability is an avoidable or reversible phenomenon [77]. PTs should provide pointed education to caregivers related to allowing opportunities to carry out functional tasks as independently as possible, while being sensitive to the reality that life is busy and oftentimes caregivers are forced to assist, given time constraints. Excess disability may help explain the potential for great success with exercise and rehabilitation interventions in IwD. If IwDs are provided with physical and functional challenges and are given the opportunity to work to their potential, they may regain lost abilities.
Motor Learning
Motor learning in dementia is not well-studied, but there are some relevant and important considerations of which therapists should be aware to provide the most promising interventions to IwD. A summary of memory and learning systems and strategies to enhance practice sessions are presented below with a summary and examples presented in Table 1.
Procedural Vs. Declarative Memory and Learning
In training/retraining motor skills in individuals with cognition intact, we often use both procedural (implicit) and declarative (explicit) memory and learning systems. Though simplified, it is helpful to consider that procedural learning is “learning by doing” and develops through repetition or skill practice without the need for deliberate cognitive oversight (e.g., attention, awareness, reflection), whereas declarative learning requires more conscious knowledge and awareness (e.g., articulation of processes, reflection on performance, intentional comparison to previous skill performance) [76, 106]. When cognition is intact, learners can “learn by doing” and enhance their learning by intentionally articulating their strategies and/or evaluating and reflecting upon their performance, thus employing both systems. It has been well documented that procedural and declarative memory are functions of different neuroanatomical brain regions. The medial temporal lobe (i.e., hippocampus and related structures) is vital to declarative memory and learning. This region is implicated early and progressively in AD particularly, rendering declarative memory impaired and ultimately unavailable to most IwD. By default, the use of procedural (implicit) memory and learning is pivotal to success in skill acquisition/reacquisition in this population [69, 100, 106]. The burden is on the PT to design treatment tasks that will elicit desired motor activities without the need for substantial cognitive oversight. For example, in training walking on altered terrain, a PT may create a walkway using yoga mats with towels underneath creating irregularly shaped and spaced bumps. In a patient with cognition intact, the PT may direct the patient to attend to the mat while walking and to think about the feel of the mat and how it is different than level ground. After a couple trials, the PT may ask the patient to articulate what went well and what they might do differently on future attempts. These strategies require cognitive oversight. For training the same activity in an IwD, the PT may set up the same walkway but give no instruction about the walkway itself but give the patient a target on the far end of the walkway (e.g., “Touch the red flag”); thus, the environment demands practice of the task, but there is no added cognitive demand in the context of training. With repetition of the task, the IwD uses procedural memory and learning to master the skill.
Errorless Learning
Because IwDs do not typically monitor and learn from their mistakes like their cognitively intact peers, errorless learning strategies may benefit learning/relearning of skills. There is some discrepancy in the literature as to whether errorless learning strategies are, by nature, involved in implicit or explicit learning, or can be applied to both [45, 58, 102, 103], but perhaps that matter is academic. These strategies are designed to stop the learner from practicing the task incorrectly, because if the learner performs the task incorrectly, then that most recent incorrect performance is likely to be their default performance upon the next attempt. A systematic review of errorless learning strategies applied to functional/ADL tasks in dementia demonstrated errorless learning to be superior to errorful learning [19]; however, these findings are not undisputed. A recent well-designed multi-site randomized controlled trial (with some shared authors of the review study) compared structured relearning of ADLs with errorless vs. trial-and-error learning in IwD and determined that both strategies were equally effective and sustaining [107]. Another recent but smaller study comparing trial-and-error learning and errorless learning strategies with mild to moderately severe AD found the interventions to be equally effective in retraining instrumental ADLs [14]. Not surprisingly, these authors demonstrated the learning effects were greater for implicit understanding of the task (i.e., being able to perform the trained task) than explicit knowledge of the task (i.e., ability to organize pictures or written description of steps of task).
The errorless learning principle is rather new to the literature and as more therapists become aware of the concept, there is the risk of misapplication to broad rehab training. While avoiding errors may be important in the context of ADL and IADL retraining, or practicing the steps to a functional training task (e.g., components of safe transfer, remembering to use assistive device for gait), there are some situations where allowing the patient to experience some “error” is appropriate and necessary. For example, in the context of balance training, patients must experience their limits of stability in the protected environment of rehab in order to improve their balance—giving the patient the opportunity to experience “near falls” (some of which are saved by the PT and some of which are saved by the patient) is an invaluable component to balance training.
Part-Whole Practice
Deconstructing a task into its component parts has recently been used within an errorless learning paradigm for ADL/IADL activities and for sit-to-stand transfers with some success in individuals with mild to moderate dementia [14, 110]. Forward chaining is a useful strategy in part-whole practice in which the therapist adds the next component part of a task when the learner masters the previous part. Retention of learned tasks is promising in delayed post-tests; however, the more important and as-yet unstudied variable would be the ability to integrate these learned activities into the context of daily life. Whenever utilizing part-whole practice, it is essential that the practice session finishes by putting the parts back into the context of the entirety of the task. Spaced retrieval training (an errorless learning strategy) might be used to do this; the therapist strategically increases or decreases guidance and cues based upon patient performance (large intervals between cues when the patient is performing well, brief intervals when the patient needs more instruction). The goal, over time, is to taper guidance and cues.
Practice Schedules (Variable Vs. Constant, Random Vs. Blocked)
Cognitively intact older adults benefit from variable (vs. constant) and random (vs. blocked) practice, as these serve to broaden their motor program creating flexibility and adaptability which bodes well for real-world success where task conditions can be unpredictable. With a goal of independent sit-to-stand transfers in a patient with intact cognition, a sound intervention would be to practice sit-to-stand in variable (e.g., integrating transfer surfaces of different heights, compliancy, and stability) and random practice conditions (e.g., interspersed among other therapeutic activities). In contrast, this would not be the best treatment session design for an IwD. Classic studies by Dick and colleagues and summary studies from the early to mid 2000s lay a foundation for our understanding of creating optimal practice sessions for individuals with dementia [21,22,23, 69, 100]. In looking at both gross motor (tossing) and fine motor (rotary pursuit) skills, Dick demonstrated that individuals with AD learn better with constant practice as they are unable to engage in the relational processing required with variable practice—they are unable to compare and contrast their efforts and outcomes and encode and store the information for later retrieval and use [22]. Consistency of training becomes an important parameter in IwD. Whereas broadening a motor program for real-world adaptability is a realistic goal in patients with intact cognition, this is effective because of these patients’ ability to transfer training to related tasks. Transfer of training is much more limited in IwD [21, 22, 69, 100]; specificity of training becomes necessary in this population with intentional narrowing or focusing of the motor program. If sit-to-stand training is a goal of therapy, it would be important to know the specific sit-to-stand needs of the patient’s living environment and to re-create the task as close to the real-life demands as possible for therapeutic practice. Repetitive and deliberate practice is instrumental in motor learning in both normal and pathological conditions, but consistency of the training is key to IwD.
Feedback
The use of feedback during motor learning is not well studied in IwD. There is some evidence that older adults with AD rely more heavily than do their cognitively intact peers on constant visual feedback while learning new motor tasks [20, 100]. Assuring proper eyewear for corrected vision is therefore important. In terms of feedback schedules, cognitively intact adults benefit from strategic extrinsic feedback (summary or interval feedback) that fades over time to encourage intrinsic reliance on the learner. The cognitive abilities of IwD may not support the ability to internalize feedback; however, a small study comparing learning of a visuomotor computer task in participants with mild to moderate AD compared feedback of 100% knowledge of results (KR) vs. 33% KR during acquisition phase, and those participants who had the reduced frequency of feedback had better retention and transfer of the task than did those who received feedback after every trial [70]. PTs should be careful not to bombard patients with feedback yet to provide relevant information that patients may need for optimal safety and function. Additionally, it is important to remember that PT comments on performance can be motivating and encouraging and IwDs may thrive on this attention.
Priming
Motor priming in neurorehabilitation is a relatively new area of study. By providing a stimulus prior to, or in conjunction with, therapy, PTs may be able to “prime” patients for motor learning and enhance outcomes. Avenues of administration include stimulation-based priming (e.g., transcranial magnetic stimulation), motor imagery and action observation, sensory priming, movement-based priming, and pharmacological priming [84]. While this adjuvant to treatment has not been studied in IwD, the rationale makes great sense for this population, as all strategies, with the exception of motor imagery, have low cognitive demands. Perceptual and sensory priming has been suggested in the context of a model for relearning ADL skills in IwD [34]. The model suggests that therapeutic interventions and environments should integrate familiar and relevant sensory and perceptual experiences during training (e.g., auditory, visual, olfactory, tactile) to enhance motor relearning of tasks. Whether this serves to prime the nervous system for learning or it simply provides a sense of comfort in the therapeutic environment, it can be a powerful tool. Integrating relevant music in the therapeutic environment may provide this comfort as well. The use of music can have a positive effect on mood and arousal which may be useful in supporting cognitive performance (attention and memory) in motor learning in IwD [74].
Dual Tasking (Real-World Preparation)
Dual tasking, or superimposing a cognitive or motor task on a base motor task, can be evidence of mature motor learning. Even cognitively intact individuals have their limits, as evidenced by movement and/or cognitive decompensation when they hit their attentional threshold. In cognitive impairment, attentional reserve is depleted, so those limits are hit earlier and the implications are often more apparent. High dual-task gait cost (i.e., substantial slowing of gait with added cognitive demand) was recently associated with progression from mild cognitive impairment to dementia [66]. In community-dwelling cognitively impaired older adults, both motor and cognitive dual-task conditions negatively impact speed and variability of gait [72, 89]. Recently, specific dual-task tests were found to have sound psychometric properties for persons with mild to moderate dementia, specifically walking while counting by 2s and walking while reciting the alphabet are suitable tests to use with this population [55].
Some authors suggest dual-task training should be avoided with IwD [100], but recognizing that there are cognitive demands to walking in anything but the most protected environments, including walking with the use of assistive device [67], it is important to prepare IwDs for real-world functioning. Additionally, given the well-documented inter-relatedness of gait and cognition, it makes sense to incorporate both cognitive and motor components in rehab efforts. In fact, recent systematic reviews demonstrate promise in using combined interventions (either dual-task training or multi-component interventions) in older adults with cognitive impairment in outcomes including cognition, functional status, and falls risk [12•, 29, 52, 108]. PTs must identify appropriate levels of dual-task challenge and create therapeutic activities that are sufficiently motivating in the context of each patient’s abilities. Therapeutic tasks should be related to real-world challenges. Technologies integrating gaming and virtual reality in rehabilitation are becoming more abundant and will likely be the subject of dual-task study with IwD. Currently, case reports describing use of computer/video gaming (Wii bowling) or virtual reality (green screen) strategies have demonstrated feasibility with IwD [25, 60].
Appropriate Challenge and Intensity
PTs working with IwD must honor basic principles of exercise and experience-driven neural plasticity, including considerable repetition, appropriate intensity of practice, and level of challenge of tasks, salience of tasks which motivates participation [47, 111]. In geriatrics, PTs often underestimate the abilities of patients—they may fail to provide an appropriate challenge or offer rest breaks too liberally. Low rehab expectations are the enemy of IwDs. It is the skill of the PT to identify the appropriate threshold for patient performance of a task and create therapeutic interventions that meet or exceed that threshold. The importance of intensity of interventions in IwD has recently been acknowledged, as evidenced by multiple publications with reference to intervention intensity in the titles [18, 41, 75, 91, 94].
Therapeutic Relationship
Creating the optimal therapeutic relationship is pivotal when working with IwD, and while detailed discussion is beyond the scope of this paper, Table 2 provides a summary of characteristics and tips for successful interpersonal interactions in rehabilitation. It is important that the rehab environment feels safe, calm, and predictable to IwDs. When there is consistency and familiarity in the setting, the people, and the time of day for exercise or rehab activities, IwDs may feel a sense of “routine” even when they have no memory of participating from day to day or week to week. Group activities are often an effective strategy for delivering exercise interventions, although tailoring an exercise or rehab program to the needs of each individual is vital to program success [99].
Conclusions
Qualitative studies with therapists working with IwD [28, 33] share the need to “be on your toes” or “think outside the box” and acknowledge some of the frustrations that come with treating this population, not the least of which are productivity requirements that are not forgiving to the needs of this group. IwDs might easily be labeled as “low priority” on a busy therapist’s schedule, and when behavioral issues or pain complaints complicate rehab efforts, it is that much more likely that therapists may feel overwhelmed. PTs who are equipped with the knowledge of the intricacies of movement and motor learning in IwD paired with strategies to foster an excellent therapeutic relationship are poised for the best possible rehab outcome.
References
Papers of particular interest, published recently, have been highlighted as: • Of importance
Ahmed S, Baker I, Thompson S, Husain M, Butler CR. Utility of testing for apraxia and associated features in dementia. J Neurol Neurosurg Psychiatry. 2016;87(11):1158–62. https://doi.org/10.1136/jnnp-2015-312945.
Allali G, Launay CP, Blumen HM, Callisaya ML, De Cock AM, Kressig RW, et al. Falls, cognitive impairment, and gait performance: results from the GOOD initiative. J Am Med Dir Assoc. 2017;18(4):335–40. https://doi.org/10.1016/j.jamda.2016.10.008.
• Allali G, Verghese J. Management of gait changes and fall risk in MCI and dementia. Curr Treat Options Neurol. 2017;19(9):29. https://doi.org/10.1007/s11940-017-0466-1. Presents the strengths of a multidisciplinary approach to managing falls in MCI and dementia.
Allan LM, Ballard CG, Rowan EN, Kenny RA. Incidence and prediction of falls in dementia: a prospective study in older people. PLoS One. 2009;4(5):e5521. https://doi.org/10.1371/journal.pone.0005521.
Alzheimer’s Association, 2017. 2017 Alzheimer’s disease facts and figures. Available at: https://www.alz.org/documents_custom/2017-facts-and-figures.pdf. Accessed 10/2/17.
Beauchet O, Allali G, Berrut G, Hommet C, Dubost V, Assal F. Gait analysis in demented subjects: interests and perspectives. Neuropsychiatr Dis Treat. 2008;4(1):155–60.
• Beauchet O, Annweiler C, Callisaya ML, De Cock AM, Helbostad JL, Kressig RW, et al. Poor gait performance and prediction of dementia: results from a meta-analysis. J Am Med Dir Assoc. 2016;17:482–90. https://doi.org/10.1016/j.jamda.2015.12.092. Suggests that poor gait performance is a stronger predictor of non-AD dementias than AD dementias.
Beauchet O, Blumen HM, Callisaya ML, De Cock AM, Kressig RW, Srikanth V, et al. Spatiotemporal gait characteristics associated with cognitive impairment: a multicenter cross-sectional study, the intercontinental “gait, cOgnitiOn and decline” initiative. Curr Alzheimer Res. 2017; https://doi.org/10.2174/1567205014666170725125621.
Best JR, Liu-Ambrose T, Boudreau RM, Ayonayon HN, Satterfield S, Simonsick EM, et al. An evaluation of the longitudinal, bidirectional associations between gait speed and cognition in older women and men. J Gerontol A Biol Sci Med Sci. 2016;71(12):1616–23. https://doi.org/10.1093/gerona/glw066.
Blackwood J, Martin A. Screening for cognitive impairment as a part of falls risk assessment in physical therapist practice. J Geriatr Phys Ther. 2017;40(4):197–203. https://doi.org/10.1519/JPT.0000000000000098.
Blackwood J, Shubert T, Forgarty K, Chase C. Relationships between performance on assessments of executive function and fall risk screening measures in community-dwelling older adults. J Geriatr Phys Ther. 2016;39:89–96. https://doi.org/10.1519/JPT.0000000000000056.
• Booth V, Hood V, Kearney F. Interventions incorporating physical and cognitive elements to reduce falls risk in cognitively impaired older adults: a systematic review. JBI Database Syst Rev Implement Rep. 2016;14:110–35. https://doi.org/10.11124/JBISRIR-2016-002499. Interventions incorporating both physical and cognitive components positively impact balance, functional mobility and gait speed for older adults with cognitive impairment.
Borson S, Scanlan JM, Chen PJ, et al. The mini-cog as a screen for dementia: validation in a population-based sample. J Am Geriatr Soc. 2003;51(10):1451–4. https://doi.org/10.1046/j.1532-5415.2003.51465.x.
Bourgeois J, Laye M, Lemaire J, Leone E, Deudon A, Darmon N, et al. Relearning of activities of daily living: a comparison of the effectiveness of three learning methods in patients with dementia of the Alzheimer type. J Nutr Health Aging. 2016;20(1):48–55. https://doi.org/10.1007/s12603-015-0597-6.
Brody EM, Kleban MH, Lawton MP, Silverman HA. Excess disabilities of mentally impaired aged: impact of individualized treatment. The Gerontologist. 1971;11(2 Part 1):124–33. https://doi.org/10.1093/geront/11.2_Part_1.124.
Buddingh S, Liang J, Allen J, Koziak A, Buckingham J, Beaupre LA. Rehabilitation for long-term care residents following hip fracture: a survey of reported rehabilitation practices and perceived barriers to delivery of care. J Geriatr Phys Ther. 2013;36:39–46. https://doi.org/10.1519/JPT.0b013e3182569b4f.
Cieślik B, Jaworska L, Szczepańska-Gierach J. Postural stability in the cognitively impaired elderly: a systematic review of the literature. Dementia 2016: 1471301216663012. https://doi.org/10.1177/1471301216663012.
Dawson N, Judge KS, Gerhart H. Improved functional performance in individuals with dementia after a moderate-intensity home-based exercise program: a randomized controlled trial. J Geriatr Phys Ther. 2017; https://doi.org/10.1519/JPT.0000000000000128.
de Werd MM, Boelen D, Rikkert MGO, Kessels RP. Errorless learning of everyday tasks in people with dementia. Clin Interv Aging. 2013;8:1177–90. https://doi.org/10.2147/CIA.S46809.
Dick MB, Andel R, Bricker J, Gorospe JB, Hsieh S, Dick-Muehlke C. Dependence on visual feedback during motor skill learning in Alzheimer’s disease. Aging Neuropsychol Cogn. 2001;8(2):120–36. https://doi.org/10.1076/anec.8.2.120.840.
Dick MB, Hsieh S, Bricker J, Dick-Muehlke C. Facilitating acquisition and transfer of a continuous motor task in healthy older adults and patients with Alzheimer’s disease. Neuropsychology. 2003;17(2):202–12. https://doi.org/10.1037/0894-4105.17.2.202.
Dick MB, Hsieh S, Dick-Muehlke C, Davis DS, Cotman CW. The variability of practice hypothesis in motor learning: does it apply to Alzheimer’s disease? Brain Cogn. 2000;44(3):470–89. https://doi.org/10.1006/brcg.2000.1206.
Dick MB, Shankle RW, Beth RE, Dick-Muehlke C, Cotman CW, Kean ML. Acquisition and long-term retention of a gross motor skill in Alzheimer’s disease patients under constant and varied practice conditions. J Gerontol B Psychol Sci Soc Sci. 1996;51(2):P103–11. https://doi.org/10.1093/geronb/51B.2.P103.
Eriksson S, Strandberg S, Gustafson Y, Lundin-Olsson L. Circumstances surrounding falls in patients with dementia in a psychogeriatric ward. Arch Gerontol Geriatr. 2009;49(1):80–7. https://doi.org/10.1016/j.archger.2008.05.005.
Fenney A, Lee TD. Exploring spared capacity in persons with dementia: what WiiTM can learn. Act Adapt Aging. 2010;34(4):303–13. https://doi.org/10.1080/01924788.2010.525736.
Fernando E, Fraser M, Hendriksen J, Kim CH, Muir-Hunter SW. Risk factors associated with falls in older adults with dementia: a systematic review. Physiother Can. 2017;69(2):161–70. https://doi.org/10.3138/ptc.2016-14.
Finkelstein EE, Prabhu MM, Chen HH. Increased prevalence of falls among elderly individuals with mental health and substance abuse conditions. Am J Geriatr Psychiatry. 2007;15(7):611–9. https://doi.org/10.1097/JGP.0b013e318033ed97.
Fjellman-Wiklund A, Nordin E, Skelton DA, Lundin-Olsson L. Reach the person behind the dementia—physical therapists’ reflections and strategies when composing physical training. PLoS One. 2016;11(12):e0166686. https://doi.org/10.1371/journal.pone.0166686.
Fritz NE, Cheek FM, Nichols-Larsen DS. Motor-cognitive dual-task training in persons with neurologic disorders: a systematic review. J Neurol Phys Ther. 2015;39(3):142–53. https://doi.org/10.1097/NPT.0000000000000090.
Fritz NE, Kegelmeyer DA, Kloos AD, Linder S, Park A, Kataki M, et al. Motor performance differentiates individuals with lewy body dementia, Parkinson’s and Alzheimer’s disease. Gait Posture. 2016;2016(50):1–7. https://doi.org/10.1016/j.gaitpost.2016.08.009.
Fritz S, Lusardi M. White paper: “walking speed: the sixth vital sign”. J Geriatr Phys Ther. 2009;32(2):2–5. https://doi.org/10.1519/00139143-200932020-00002.
Haak N. Maintaining connections: understanding communication from the perspective of persons with dementia. Alzheim Care Q. 2002;3:116–28.
Hall A, Watkins R, Lang I, Endacott R, Goodwin V. The experiences of physiotherapists treating people with dementia who fracture their hip. BMC Geriatr. 2017;17(1):91. https://doi.org/10.1186/s12877-017-0474-8.
Harrison BE, Son G-R, Kim J, Whall AL. Preserved implicit memory in dementia: a potential model for care. Am J Alzheim Dis Other Demen. 2007;22(4):286–93. https://doi.org/10.1177/1533317507303761.
Hauer K, Schwenk M, Zieschang T, Essig M, Becker C, Oster P. Physical training improves motor performance in people with dementia: a randomized controlled trial. J Am Geriatr Soc. 2012;60:8–15. https://doi.org/10.1111/j.1532-5415.2011.03778.x.
Heliker D. Enhancing relationships in long-term care: through story sharing. J Gerontol Nurs. 2009;35(6):43–9. https://doi.org/10.3928/00989134-20090428-04.
Hernandez S, Coelho F, Gobbi S, Stella F. Effects of physical activity on cognitive function, balance and risk of falls in elderly patients with Alzheimer’s dementia. Rev Bras Fisioter. 2010;14(1):68–74. https://doi.org/10.1590/S1413-355520.
Hobbelen JSM, Koopmans RTCM, Verhey FRJ, Habraken KM, de Bie RA. Diagnosing paratonia in the demented elderly: reliability and validity of the paratonia assessment instrument (PAI). Int Psychogeriatr IPA. 2008;20(04):840–52. https://doi.org/10.1017/S1041610207006424.
Hobbelen JSM, Koopmans RTCM, Verhey FRJ, Van Peppen RPS, de Bie RA. Paratonia: a delphi procedure for consensus definition. J Geriatr Phys Ther. 2006;29:50–6.
Hobbelen JSM, Tan FES, Verhey FRJ, Koopmans RTCM, de Bie RA. Prevalence, incidence and risk factors of paratonia in patients with dementia: a one-year follow-up study. Int Psychogeriatr. 2011;23(07):1051–60. https://doi.org/10.1017/S1041610210002449.
Hoffmann K, Sobol NA, Frederiksen KS, Beyer N, Vogel A, Vestergaard K, et al. Moderate-to-high intensity physical exercise in patients with Alzheimer’s disease: a randomized controlled trial. J Alzheimers Dis JAD. 2016;50(2):443–53. https://doi.org/10.3233/JAD-150817.
Johansson H, Lundin-Olsson L, Littbrand H, Gustafson Y, Rosendahl E, Toots A. Cognitive function and walking velocity in people with dementia; a comparison of backward and forward walking. Gait Posture. 2017;58:481–6. https://doi.org/10.1016/j.gaitpost.2017.09.009.
Johnen A, Frommeyer J, Modes F, Wiendl H, Duning T, Lohmann H. Dementia apraxia test (DATE): a brief tool to differentiate behavioral variant frontotemporal dementia from Alzheimer’s dementia based on apraxia profiles. J Alzheimers Dis JAD. 2016;49(3):593–605. https://doi.org/10.3233/JAD-150447.
• Kearney FC, Harwood RH, Gladman JRF, Lincoln N, Masud T. The relationship between executive function and falls and gait abnormalities in older adults: a systematic review. Dement Geriatr Cogn Disord. 2013;36:20–35. https://doi.org/10.1159/000350031. Highlights the relationship between executive functioning and gait speed and falls in older adults.
Kessels RPC, Hensken LMGO. Effects of errorless skill learning in people with mild-to-moderate or severe dementia: a randomized controlled pilot study. NeuroRehabilitation. 2009;25(4):307–12. https://doi.org/10.3233/NRE-2009-0529.
• Kikkert LHJ, Vuillerme N, van Campen JP, Hortobágyi T, Lamoth CJ. Walking ability to predict future cognitive decline in old adults: a scoping review. Ageing Res Rev. 2016;27:1–14. https://doi.org/10.1016/j.arr.2016.02.001. Consolidates evidence on value of gait speed to predict cognitive decline in older adults and highlights the added value of other gait parameters.
Kleim JA, Jones TA. Principles of experience-dependent neural plasticity: implications for rehabilitation after brain damage. J Speech Lang Hear Res JSLHR. 2008;51(1):S225–39. https://doi.org/10.1044/1092-4388(2008/018).
Kovach CR, Henschel H. Planning activities for patients with dementia: a descriptive study of therapeutic activities on special care units. J Gerontol Nurs. 1996;22(9):33–8. https://doi.org/10.3928/0098-9134-19960901-10.
Kröpelin TF, Neyens JCL, Halfens RJG, Kempen GIJM, Hamers JPH. Fall determinants in older long-term care residents with dementia: a systematic review. Int Psychogeriatr IPA. 2013;25(04):549–63. https://doi.org/10.1017/S1041610212001937.
Kuate-Tegueu C, Avila-Funes J-A, Simo N, Le Goff M, Amiéva H, Dartigues J-F, et al. Association of gait speed, psychomotor speed, and dementia. J Alzheimers Dis JAD. 2017;60(2):585–92. https://doi.org/10.3233/JAD-170267.
Kueper JK, Speechley M, Lingum NR, Montero-Odasso M. Motor function and incident dementia: a systematic review and meta-analysis. Age Ageing. 2017;46(5):729–38. https://doi.org/10.1093/ageing/afx084.
Law LLF, Barnett F, Yau MK, Gray MA. Effects of combined cognitive and exercise interventions on cognition in older adults with and without cognitive impairment: a systematic review. Ageing Res Rev. 2014;15:61–75. https://doi.org/10.1016/j.arr.2014.02.008.
Lee ATC, Richards M, Chan WC, Chiu HFK, Lee RSY, Lam LCW. Poor balance as a noncognitive predictor of incident dementia. J Am Geriatr Soc. 2015;63(8):1701–2. https://doi.org/10.1111/jgs.13587.
Lee HS, Park SW, Park YJ. Effects of physical activity programs on the improvement of dementia symptom: a meta-analysis. Biomed Res Int. 2016;2016:1–7. https://doi.org/10.1155/2016/2920146.
Lemke NC, Wiloth S, Werner C, Hauer K. Validity, test-retest reliability, sensitivity to change and feasibility of motor-cognitive dual task assessments in patients with dementia. Arch Gerontol Geriatr. 2017;70:169–79. https://doi.org/10.1016/j.archger.2017.01.016.
Lesourd M, Le Gall D, Baumard J, Croisile B, Jarry C, Osiurak F. Apraxia and Alzheimer’s disease: review and perspectives. Neuropsychol Rev. 2013;23(3):234–56. https://doi.org/10.1007/s11065-013-9235-4.
Lewis M, Peiris CL, Shields N. Long-term home and community-based exercise programs improve function in community-dwelling older people with cognitive impairment: a systematic review. J Physiother. 2017;63(1):23–9. https://doi.org/10.1016/j.jphys.2016.11.005.
Li R, Liu KPY. The use of errorless learning strategies for patients with Alzheimer’s disease: a literature review. Int J Rehabil Res Int Z Rehabil Rev Int Rech Readaptation. 2012;35(4):292–8. https://doi.org/10.1097/MRR.0b013e32835a2435.
Marchetti MT, Whitney SL. Postural control in older adults with cognitive dysfunction. Phys Ther Rev. 2006;11(3):161–8. https://doi.org/10.1179/108331906X144046.
McEwen D, Taillon-Hobson A, Bilodeau M, Sveistrup H, Finestone H. Two-week virtual reality training for dementia: single-case feasibility study. J Rehabil Res Dev. 2014;51(7):1069–76. https://doi.org/10.1682/JRRD.2013.10.0231.
McGough EL, Logsdon RG, Kelly VE, Teri L. Functional mobility limitations and falls in assisted living residents with dementia: physical performance assessment and quantitative gait analysis. J Geriatr Phys Ther. 2013;36:78–86. https://doi.org/10.1519/JPT.0b013e318268de7f.
Merory JR, Wittwer JE, Rowe CC, Webster KE. Quantitative gait analysis in patients with dementia with Lewy bodies and Alzheimer’s disease. Gait Posture. 2007;26(3):414–9. https://doi.org/10.1016/j.gaitpost.2006.10.006.
Mesbah N, Perry M, Hill KD, Kaur M, Hale L. Postural stability in older adults with Alzheimer disease. Phys Ther. 2017;97(3):290–309. https://doi.org/10.2522/ptj.20160115.
Mirelman A, Herman T, Brozgol M, Dorfman M, Sprecher E, Schweiger A, et al. Executive function and falls in older adults: new findings from a five-year prospective study link fall risk to cognition. PLoS One. 2012;7(6):e40297. https://doi.org/10.1371/journal.pone.0040297.
Montero-Odasso M, Hachinski V. Preludes to brain failure: executive dysfunction and gait disturbances. Neurol Sci Off J Ital Neurol Soc Ital Soc Clin Neurophysiol. 2014;35(4):601–4. https://doi.org/10.1007/s10072-013-1613-4.
Montero-Odasso MM, Sarquis-Adamson Y, Speechley M, Borrie MJ, Hachinski VC, Wells J, et al. Association of dual-task gait with incident dementia in mild cognitive impairment: results from the gait and brain study. JAMA Neurol. 2017;74(7):857–65. https://doi.org/10.1001/jamaneurol.2017.0643.
Muir-Hunter SW, Montero-Odasso M. The attentional demands of ambulating with an assistive device in older adults with Alzheimer’s disease. Gait Posture. 2017;54:202–8. https://doi.org/10.1016/j.gaitpost.2017.03.011.
Ozkan S, Adapinar DO, Elmaci NT, Arslantas D. Apraxia for differentiating Alzheimer’s disease from subcortical vascular dementia and mild cognitive impairment. Neuropsychiatr Dis Treat. 2013;9:947–51. https://doi.org/10.2147/NDT.S47879.
Patterson JT, Wessel J. Strategies for retraining functional movement in persons with Alzheimer disease: a review. Physiother Can. 2002;54:274–80.
Rice MS, Fertig PA, Maitra KK, Miller BK. Reduced feedback: motor learning strategy in persons with Alzheimer’s disease. Phys Occup Ther Geriatr. 2008;27(2):122–38. https://doi.org/10.1080/02703180802237715.
Ries JD, Hutson J, Maralit LA, Brown MB. Group balance training specifically designed for individuals with Alzheimer disease: impact on berg balance scale, timed up and go, gait speed, and mini-mental status examination. J Geriatr Phys Ther. 2015;38:183–93. https://doi.org/10.1519/JPT.0000000000000030.
Rucco R, Agosti V, Jacini F, Sorrentino P, Varriale P, De Stefano M, et al. Spatio-temporal and kinematic gait analysis in patients with frontotemporal dementia and Alzheimer’s disease through 3D motion capture. Gait Posture. 2017;52:312–7. https://doi.org/10.1016/j.gaitpost.2016.12.021.
Sadowsky CH, Galvin JE. Guidelines for the management of cognitive and behavioral problems in dementia. J Am Board Fam Med. 2012;25(3):350–66. https://doi.org/10.3122/jabfm.2012.03.100183.
Särkämö T. Music for the ageing brain: cognitive, emotional, social, and neural benefits of musical leisure activities in stroke and dementia. Dement Lond Engl 2017:1471301217729237. https://doi.org/10.1177/1471301217729237
Schwenk M, Zieschang T, Englert S, Grewal G, Najafi B, Hauer K. Improvements in gait characteristics after intensive resistance and functional training in people with dementia: a randomised controlled trial. BMC Geriatr. 2014;14(1):73. https://doi.org/10.1186/1471-2318-14-73.
Shumway-Cook A, Woollacott MH. Motor control: translating research into clinical practice. 5th ed. Philadelphia: LWW; 2016.
Slaughter SE, Eliasziw M, Morgan D, Drummond N. Incidence and predictors of excess disability in walking among nursing home residents with middle-stage dementia: a prospective cohort study. Int Psychogeriatr. 2011;23(01):54–64. https://doi.org/10.1017/S1041610210000116.
Small JA, Gutman G, Makela S, Hillhouse B. Effectiveness of communication strategies used by caregivers of persons with Alzheimer’s disease during activities of daily living. J Speech Lang Heart Res. 2003;46(2):353–67. https://doi.org/10.1044/1092-4388(2003/028).
Smith ER, Broughton M, Baker R, Pachana NA, Angwin AJ, Humphreys MS, et al. Memory and communication support in dementia: research-based strategies for caregivers. Int Psychogeriatr. 2011;23(02):256–63. https://doi.org/10.1017/S1041610210001845.
Smits LL, Flapper M, Sistermans N, Pijnenburg YAL, Scheltens P, van der Flier WM. Apraxia in mild cognitive impairment and Alzheimer’s disease: validity and reliability of the van Heugten test for apraxia. Dement Geriatr Cogn Disord. 2014;38(1-2):55–64. https://doi.org/10.1159/000358168.
Stamenova V, Roy EA, Black SE. A model-based approach to limb apraxia in Alzheimer’s disease. J Neuropsychol. 2013;8(2):246–68. https://doi.org/10.1111/jnp.12023.
Staples WH, Killian CB. Development of an instrument to measure attitudes of physical therapy providers working with people with dementia. Am J Alzheim Dis Other Demen. 2012;27(5):331–8. https://doi.org/10.1177/1533317512452041.
Stinson C. Caregiving tips for care of the Alzheimer’s patient. Long-term. Living. 2000;49:64–7.
Stoykov ME, Madhavan S. Motor priming in neurorehabilitation. J Neurol Phys Ther JNPT. 2015;39(1):33–42. https://doi.org/10.1097/NPT.0000000000000065.
Suttanon P, Hill KD, Said CM, Dodd KJ. A longitudinal study of change in falls risk and balance and mobility in healthy older people and people with Alzheimer disease. Am J Phys Med Rehabil Assoc Acad Physiatr. 2013;92(8):676–85. https://doi.org/10.1097/PHM.0b013e318278dcb3.
Suttanon PP, Hill KDK, Said CMC, Logiudice DD, Lautenschlager NTN, Dodd KJK. Balance and mobility dysfunction and falls risk in older people with mild to moderate Alzheimer disease. Am J Phys Med Rehabil Assoc Acad Physiatr. 2012;91(1):12–23. https://doi.org/10.1097/PHM.0b013e31823caeea.
Szczepańska-Gieracha J, Cieślik B, Chamela-Bilińska D, Kuczyński M. Postural stability of elderly people with cognitive impairments. Am J Alzheimers Dis Other Demen. 2016;31(3):241–6. https://doi.org/10.1177/1533317515602547.
Tangen GG, Engedal K, Bergland A, Moger TA, Mengshoel AM. Relationships between balance and cognition in patients with subjective cognitive impairment, mild cognitive impairment, and Alzheimer disease. Phys Ther. 2014;94(8):1123–34. https://doi.org/10.2522/ptj.20130298.
Taylor ME, Delbaere K, Mikolaizak AS, Lord SR, Close JCT. Gait parameter risk factors for falls under simple and dual task conditions in cognitively impaired older people. Gait Posture. 2013;37(1):126–30. https://doi.org/10.1016/j.gaitpost.2012.06.024.
Taylor ME, Lasschuit DA, Lord SR, Delbaere K, Kurrle SE, Mikolaizak AS, et al. Slow gait speed is associated with executive function decline in older people with mild to moderate dementia: a one year longitudinal study. Arch Gerontol Geriatr. 2017;73:148–53. https://doi.org/10.1016/j.archger.2017.07.023.
Telenius EW, Engedal K, Bergland A. Effect of a high-intensity exercise program on physical function and mental health in nursing home residents with dementia: an assessor blinded randomized controlled trial. PLoS One. 2015;10(5):e0126102. https://doi.org/10.1371/journal.pone.0126102.
Thomas VS. Excess functional disability among demented subjects? Findings from the Canadian study of health and aging. Dement Geriatr Cogn Disord. 2001;12:206–10. https://doi.org/10.1159/000051259.
Tian Q, An Y, Resnick SM, Studenski S. The relative temporal sequence of decline in mobility and cognition among initially unimpaired older adults: results from the Baltimore longitudinal study of aging. Age Ageing. 2016; https://doi.org/10.1093/ageing/afw185.
• Toots A, Littbrand H, Lindelöf N, Wiklund R, Holmberg H, Nordström P, et al. Effects of a high-intensity functional exercise program on dependence in activities of daily living and balance in older adults with dementia. J Am Geriatr Soc. 2016;64:55–64. https://doi.org/10.1111/jgs.13880. A good example of a recent high intensity exercise intervention for IwD; effects were more evident in those with non-AD dementia.
Tosto G, Monsell SE, Hawes SE, Bruno G, Mayeux R. Progression of extrapyramidal signs in Alzheimer’s disease: clinical and neuropathological correlates. J Alzheimers Dis JAD. 2015a;49(4):1085–93. https://doi.org/10.3233/JAD-150244.
Tosto G, Monsell SE, Hawes SE, Mayeux R. Pattern of extrapyramidal signs in Alzheimer’s disease. J Neurol. 2015b;262(11):2548–56. https://doi.org/10.1007/s00415-015-7886-1.
Vahia I, Cohen CI, Prehogan A, Memon Z. Prevalence and impact of paratonia in Alzheimer disease in a multiracial sample. Am J Geriatr Psychiatry Off J Am Assoc Geriatr Psychiatry. 2007;15(4):351–3. https://doi.org/10.1097/JGP.0b013e31802ea907.
Valkanova V, Ebmeier KP. What can gait tell us about dementia? Review of epidemiological and neuropsychological evidence. Gait Posture. 2017;53:215–23. https://doi.org/10.1016/j.gaitpost.2017.01.024.
van der Wardt V, Hancox J, Gondek D, Logan P, das NR, Pollock K, et al. Adherence support strategies for exercise interventions in people with mild cognitive impairment and dementia: a systematic review. Prev Med Rep. 2017;7:38–45. https://doi.org/10.1016/j.pmedr.2017.05.007.
van Halteren-van Tilborg IADA, Scherder EJA, Hulstijn W. Motor-skill learning in Alzheimer’s disease: a review with an eye to the clinical practice. Neuropsychol Rev. 2007;17(3):203–12. https://doi.org/10.1007/s11065-007-9030-1.
van Iersel MB, Hoefsloot W, Munneke M, Bloem BR, Olde Rikkert MGM. Systematic review of quantitative clinical gait analysis in patients with dementia. Z Für Gerontol Geriatr. 2004;37(1):27–32. https://doi.org/10.1007/s00391-004-0176-7.
van Tilborg IA, Kessels RP, Hulstijn W. How should we teach everyday skills in dementia? A controlled study comparing implicit and explicit training methods. Clin Rehabil. 2011a;25(7):638–48. https://doi.org/10.1177/0269215510396738.
van Tilborg IADA, Kessels RP, Hulstijn W. Learning by observation and guidance in patients with Alzheimer’s dementia. NeuroRehabilitation. 2011b;29(3):295–304. https://doi.org/10.3233/NRE-2011-0705.
Verghese J, Wang C, Lipton RB, Holtzer R. Motoric cognitive risk syndrome and the risk of dementia. J Gerontol A Biol Sci Med Sci. 2013;68(4):412–8. https://doi.org/10.1093/gerona/gls191.
Verlinden VJA, van der Geest JN, Hofman A, Ikram MA. Cognition and gait show a distinct pattern of association in the general population. Alzheimers Dement J Alzheimers Assoc. 2014;10(3):328–35. https://doi.org/10.1016/j.jalz.2013.03.009.
Vidoni ED, Boyd LA. Achieving enlightenment: what do we know about the implicit learning system and its interaction with explicit knowledge? J Neurol Phys Ther JNPT. 2007;31(3):145–54. https://doi.org/10.1097/NPT.0b013e31814b148e.
Voigt-Radloff S, de Werd MME, Leonhart R, Boelen DHE, Olde Rikkert MGM, Fliessbach K, et al. Structured relearning of activities of daily living in dementia: the randomized controlled REDALI-DEM trial on errorless learning. Alzheimers Res Ther. 2017;9(1):22. https://doi.org/10.1186/s13195-017-0247-9.
Wajda DA, Mirelman A, Hausdorff JM, Sosnoff JJ. Intervention modalities for targeting cognitive-motor interference in individuals with neurodegenerative disease: a systematic review. Expert Rev Neurother. 2017;17(3):251–61. https://doi.org/10.1080/14737175.2016.1227704.
Webster KE, Merory JR, Wittwer JE. Gait variability in community dwelling adults with Alzheimer disease. Alzheimer Dis Assoc Disord. 2006;20(1):37–40. https://doi.org/10.1097/01.wad.0000201849.75578.de.
Werner C, Wiloth S, Lemke NC, Kronbach F, Jansen C-P, Oster P, et al. People with dementia can learn compensatory movement maneuvers for the sit-to-stand task: a randomized controlled trial. J Alzheimers Dis JAD. 2017;60(1):107–20. https://doi.org/10.3233/JAD-170258.
Winstein C, Lewthwaite R, Blanton SR, Wolf LB, Wishart L. Infusing motor learning research into neurorehabilitation practice: a historical perspective with case exemplar from the accelerated skill acquisition program. J Neurol Phys Ther. 2014;38:1–11. https://doi.org/10.1097/NPT.0000000000000046.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Julie Ries declares no conflict of interest.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
This article is part of the Topical Collection on Physical Therapy and Rehabilitation
Rights and permissions
About this article
Cite this article
Ries, J.D. Rehabilitation for Individuals with Dementia: Facilitating Success. Curr Geri Rep 7, 59–70 (2018). https://doi.org/10.1007/s13670-018-0237-1
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13670-018-0237-1