Abstract
Four hitherto unknown polysubstituted isoflavonoids, including three isoflavans: 7,4′-dihydroxy-8,2′,3′-trimethoxyisoflavan (1), 7,2′,4′-trihydroxy-8,3′-dimethoxyisoflavan (2), and 7,2′,4′-trihydroxy-5-methoxyisoflavan (3), and one prenylated isoflavone cudraisoflavone M (4) were isolated from the ethanol extracts of Spatholobus suberectus (for 1 and 2), Flemingia macrophylla (for 3), and Cudrania cochinchinensis (for 4), respectively. Their structures were established on the basis of extensive spectroscopic analysis. Compounds 1 and 4 exhibited weak cytotoxic activity against five human cancer cell lines (HL-60, A-549, SMMC-7721, MCF-7, and SW-480).
Graphical Abstract

Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
Isoflavonoids are a large group of secondary metabolites with diverse biological activities that occur widely in plants, which can be subdivided into isoflavones, isoflavans, isoflavanones, rotenoids, pterocarpans, etc. An overwhelming number of isoflavonoids reported come from the Leguminosae family, and some non-leguminous families such as Moraceae are also relatively abundant in isoflavonoids [1, 2]. Some isoflavans and prenylated isoflavones possess potent biological activities, especially in cytotoxicity [3,4,5,6], anti-inflammatory [7], and neuroprotective activity [8]. As part of a BioBioPha (http://www.chemlib.cn) objective to assemble a large-scale natural product library valuable in the discovery of new drug leads from nature [9,10,11,12], the phytochemical investigations on Spatholobus suberectus (Leguminosae), Flemingia macrophylla (Leguminosae), and Cudrania cochinchinensis (Moraceae) led to the isolation of three new isoflavans 7,4′-dihydroxy-8,2′,3′-trimethoxyisoflavan (1), 7,2′,4′-trihydroxy-8,3′-dimethoxyisoflavan (2), and 7,2′,4′-trihydroxy-5-methoxyisoflavan (3), and one new prenylated isoflavone namely cudraisoflavone M (4), respectively (Fig. 1). Herein we report the structure elucidation of new isoflavonoid and their cytotoxicity evaluation against five human cancer cell lines (HL-60, A-549, SMMC-7721, MCF-7, and SW-480).
2 Results and Discussion
Compound 1 was obtained as white amorphous powder, and its molecular formula was determined to be C18H20O6 from the positive HRESIMS at m/z 355.1163 [M+Na]+ (calcd. for C18H20O6Na, 355.1158) with nine degrees of unsaturation. The 1H NMR spectrum (Table 1) displayed five aliphatic proton signals due to two methylene groups [δ H 2.86 (1H, ddd, J = 15.8, 5.3, 1.9 Hz) and 2.94 (1H, dd, J = 15.8, 11.3 Hz), 4.00 (1H, t, J = 10.5 Hz) and 4.37 (1H, ddd, J = 10.5, 3.7, 1.9 Hz)], and one methine [δ H 3.53 (1H, dddd, J = 11.3, 10.5, 5.3, 3.7 Hz)], which were characteristic of an isoflavan [13]. The 1H NMR spectrum also exhibited two pairs of ortho-coupled aromatic doublets [δ H 6.52 (1H, d, J = 8.3 Hz), 6.72 (1H, d, J = 8.3 Hz), 6.71 (1H, d, J = 8.5 Hz), and 6.75 (1H, d, J = 8.5 Hz)], three methoxy groups [δ H 3.87, 3.92 and 3.93 (each 3H, s)], and two phenolic hydroxy protons [δ H 5.66 and 5.71 (each 1H, s)]. The 13C NMR spectrum (Table 2) showed a total of 18 carbon signals, including 12 aromatic carbons for two phenyl units (rings A and B), the ring C carbons at δ C 31.4 (d), 31.5 (t), 70.5 (t), and three methoxy carbons at δ C 60.6, 60.8, 61.0 (each q). The above NMR spectroscopic features were very similar to those of isoduartin (=7,2′-dihydroxy-8,3′,4′-trimethoxyisoflavan) [14], except for an obvious downfield shift for H-5′ (Δ = +0.27 ppm), which hinted a different substituted pattern in ring B. The pattern can be determined as 4′-hydroxy-2′,3′-dimethoxy by the following HMBC correlations (Fig. 2): from 4′-OH (δ H 5.71) to C-3′ (δ C 139.8), C-4′ (δ C 148.5) and C-5′ (δ C 110.5); from H-3 (δ H 3.53) and 2′-OCH3 (δ H 3.87) to C-2′ (δ C 150.8); and from 3′-OCH3 (δ H 3.93) to C-3′. The particular downfield shifts (Δ ≈ +5 ppm) of aromatic methoxy carbons also implied the location of these groups. According to the empirical rule, the carbon signals of the methoxy groups with substituents in both ortho positions will appear at 60–62 ppm, while those sterically non-hindered at 55–57 ppm [15]. On the basis of the above analysis, the structure of 1 was established as 7,4′-dihydroxy-8,2′,3′-trimethoxyisoflavan.
Compound 2, isolated as white amorphous powder, had a molecular formula of C17H18O6 by the positive HRESIMS at m/z 341.1002 [M+Na]+ (calcd. for C17H18O6Na, 341.1001). Comparison of the 1H and 13C NMR spectral data (Tables 1, 2) with those of 1 revealed that a methoxy group in the ring B was replaced by a hydroxy. From the HMBC correlations of the OH signal at δ H 5.72 (1H, s) to C-1′ (δ C 119.9), C-2′ (δ C 147.1), and C-3′ (δ C 134.5), we could conclude that the emerging hydroxy was positioned at C-2′ (Fig. 2). Hence, the structure of 2 was established as 7,2′,4′-trihydroxy-8,3′-dimethoxyisoflavan.
Compound 3 was isolated as white amorphous powder, and its molecular formula was determined to be C16H16O5 according to the positive HRESIMS at m/z 289.1075 [M+H]+ (calcd. for C16H17O5, 289.1076). Five diagnostic ring C proton signals [δ H 2.59 (1H, dd, J = 16.3, 10.8 Hz), 2.76 (1H, ddd, J = 16.3, 5.5, 1.9 Hz), 3.34 (1H, dddd, J = 10.8, 10.2, 5.5, 3.1 Hz), 3.86 (1H, dd, J = 10.2, 10.2 Hz), and 4.16 (1H, ddd, J = 10.2, 3.1, 1.9 Hz)], an ABX-type aromatic proton system [δ H 6.30 (1H, d, J = 2.4 Hz), 6.24 (1H, dd, J = 8.3, 2.4 Hz), and 6.85 (1H, d, J = 8.3 Hz)], two meta-coupled aromatic doublets [δ H 5.88 (1H, d, J = 2.1 Hz) and 5.99 (1H, d, J = 2.1 Hz)], and one methoxy signal [δ H 3.74 (3H, s)] were observed in the 1H NMR spectrum (Table 1), which revealed that 3 was a monomethyl ether derivative of 5,7,2′,4′-tetrahydroxyisoflavan. The HMBC correlations from the methoxy and H-4 proton signals to C-5 (δ C 160.1) confirmed the methoxy group at C-5 (Fig. 2). Therefore, the structure of 3 was established as 7,2′,4′-trihydroxy-5-methoxyisoflavan.
Absolute configurations of the isoflavans (1–3) were established by comparison of the CD curves with those reported earlier for closely related analogues. All the CD spectra showed a negative Cotton effect in the transition region (215–240 nm), similar with those of 7-O-methylisomucronulatol, (3R)-isomucronulatol, and abruquinone L [16,17,18,19], which indicated the absolute configurations at C-3 of 1–3 being R-form. And optical rotation measurements were also taken to assign the absolute configuration. We carefully examined the specific rotation values of some 2′,3′,4′-trisubstituted isoflavans (Table 3), and the results suggested that the methoxy group substituted at C-2′ had a huge impact on the specific rotation, probably on account of the spatial proximity. For 2′-methoxyisoflavans, R-/S-form result in positive/negative specific rotation values, respectively, but for 2′-hydroxyisoflavans, R-form gives negative values [14, 17, 20,21,22,23,24,25]. Our current research results were consistent with the above empirical rule.
Compound 4, yellowish amorphous powder, had a molecular formula of C22H22O7 according to the positive HRESIMS at m/z 399.1440 [M+H]+ (calcd. for C22H23O7, 399.1444). The NMR spectra (Table 4) showed an olefinic signal (δ H 8.23, 1H, s; δ C 154.3, d) and a chelated hydroxy group at δ H 13.48 (1H, s), characteristic of a 5-hydroxyisoflavone skeleton [8, 13]. In addition, an ABX-type aromatic proton system [δ H 7.15 (1H, d, J = 2.0 Hz), 6.86 (1H, d, J = 8.2 Hz), and 6.94 (1H, dd, J = 8.2, 2.0 Hz)], a prenyl group [δ H 1.63, 1.79 (each 3H, s), 3.45 (2H, d, J = 7.1 Hz), and 5.22 (1H, br t, J = 7.1 Hz)], and a hydroxyethyl moiety [δ H 2.97, 3.90 (each 2H, t, J = 5.2 Hz)] were also detected in the 1H NMR spectrum. The 13C NMR spectrum displayed a total of 22 carbon resonances, including two methyls, three sp 3 methylenes, five sp 2 methines, and 12 sp 2 quaternary carbons. These spectroscopic features were very similar to those of cudraisoflavone L, which was recently isolated from the same genus and shared the same molecular formula with 4 [26]. Their structural difference was only due to the location of the prenyl group and the hydroxyethyl moiety. The hydroxyethyl unit was located at C-6 from the HMBC correlations of H-1″ (δ H 2.97) and 5-OH (δ H 13.48) to C-5 (δ C 158.5) and C-6 (δ C 111.2), while the prenyl group at C-8 by the correlations of H-1‴ (δ H 3.45) and H-2 (δ H 8.23) to C-9 (δ C 154.6). Thus the structure of 4 was established and named as cudraisoflavone M. It is worth mentioning that cudraisoflavone M (4) was also obtained in our phytochemical investigation on the plant Derris robusta (Leguminosae).
Considering the potent cytotoxic activity of some isoflavonoids, the cytotoxicity of these new isoflavonoids (1–4) was evaluated against five human cancer cell lines (HL-60, A-549, SMMC-7721, MCF-7, and SW-480) using the MTS method. DDP (cisplatin) and paclitaxel were used as positive controls. The results showed that 1 and 4 exhibited weak cytotoxic activity (Table 5), while 2 and 3 were inactive (IC50 values >40 µM) for all cell lines.
3 Experimental Section
3.1 General Experimental Procedures
Optical rotations were measured on Jasco P-1020 automatic digital polarimeter. CD spectra were recorded on a Chirascan spectropolarimeter (Applied Photophysics, Leatherhead, Surrey, UK). UV data were obtained from HPLC online analysis. IR spectra were obtained on a Bruker Tensor-27 infrared spectrophotometer with KBr pellets. NMR spectra were carried out on a Bruker Avance III 600 or DRX-500 spectrometer with deuterated solvent signals used as internal standards. ESIMS and HRESIMS were measured using Agilent G6230 time-of-flight mass spectrometer. Preparative MPLC was performed on a Büchi apparatus equipped with Büchi fraction collector C-660, Büchi pump module C-605 and manager C-615. Silica gel (200–300 mesh, Qingdao Marine Chemical Inc., China), MCI gel CHP-20P (75–150 μm, Mitsubishi Chemical Corporation, Japan), Chromatorex C-18 (40–75 μm, Fuji Silysia Chemical Ltd., Japan) and Sephadex LH-20 (GE Healthcare Bio-Sciences AB, Uppsala, Sweden) were used for column chromatography. Fractions were monitored and analyzed using TLC, in combination with an Agilent 1200 series HPLC system equipped by an Extend-C18 column (5 μm, 4.6 × 150 mm).
3.2 Plant Material and Isolation (Table 6)
The retention times (t R) of 1–4 on an analytical HPLC Extend-C18 column (20% → 100% MeOH in H2O over 8.0 min followed by 100% MeOH to 13.0 min, 1.0 mL/min, 25 °C) were 7.51, 6.85, 6.61 and 9.12 min, respectively.
3.2.1 7,4′-Dihydroxy-8,2′,3′-trimethoxyisoflavan (1)
White amorphous powder; UV (MeOH) λ max: 228 (sh), 278 nm; [α] 25D +11.0 (c 0.20, MeOH); IR (KBr) ν max: 3396, 2944, 1602, 1497, 1464, 1292, 1198, 1170, 1097, 1068, 1040, 1010, 963 cm−1; 1H NMR data: see Table 1; 13C NMR data: see Table 2; ESIMS (pos.): m/z 355 [M+Na]+; HRESIMS (pos.): m/z 355.1163 [M+Na]+ (calcd. for C18H20O6Na, 355.1158).
3.2.2 7,2′,4′-Trihydroxy-8,3′-dimethoxyisoflavan (2)
White amorphous powder; UV (MeOH) λ max: 229 (sh), 277 nm; [α] 25D −13.2 (c 0.20, MeOH); IR (KBr) ν max: 3404, 2929, 1619, 1509, 1470, 1328, 1184, 1091, 1058, 1037, 1001, 804 cm−1; 1H NMR data: see Table 1; 13C NMR data: see Table 2; ESIMS (pos.): m/z 341 [M+Na]+; HRESIMS (pos.): m/z 341.1002 [M+Na]+ (calcd. for C17H18O6Na, 341.1001).
3.2.3 7,2′,4′-Trihydroxy-5-methoxyisoflavan (3)
White amorphous powder; UV (MeOH) λ max: 230 (sh), 279 nm; [α] 25D −24.0 (c 0.20, MeOH); IR (KBr) ν max: 3406, 2937, 1620, 1606, 1520, 1501, 1472, 1459, 1200, 1142, 1118, 1037, 974, 819 cm−1; 1H NMR data: see Table 1; 13C NMR data: see Table 2; ESIMS (pos.): m/z 289 [M+H]+; HRESIMS (pos.): m/z 289.1075 [M+H]+ (calcd. for C16H17O5, 289.1076).
3.2.4 Cudraisoflavone M (4)
Yellowish amorphous powder; UV (MeOH) λ max: 212, 270, 346 (sh) nm; IR (KBr) ν max: 3425, 2926, 1644, 1582, 1522, 1435, 1380, 1306, 1209, 1116, 1083, 1030 cm−1; 1H and 13C NMR data: see Table 4; ESIMS (pos.): m/z 399 [M+H]+; HRESIMS (pos.): m/z 399.1440 [M+H]+ (calcd. for C22H23O7, 399.1444).
3.3 Cytotoxicity Assays
Five human tumor cell lines (HL-60, A-549, SMMC-7721, MCF-7, and SW-480) obtained from ATCC (Manassas, VA, USA) were used in the cytotoxicity assay. All cells were cultured in RPMI-1640 or DMEM medium (Hyclone, Logan, UT, USA), supplemented with 10% fetal bovine serum (Hyclone) at 37 °C in a humidified atmosphere containing 5% CO2. Cell viability was assessed by conducting colorimetric measurements of the amount of insoluble formazan formed in living cells based on the reduction of MTS (Sigma, St. Louis, MO, USA). Briefly, 100 μL of adherent cells were seeded into each well of a 96-well cell culture plate and allowed to adhere for 12 h before drug addition, while suspended cells were seeded just before drug addition, both with an initial density of 1 × 105 cells/mL in 100 μL medium. Each cell line was exposed to the test compound at various concentrations in triplicate for 48 h, with cisplatin and paclitaxel as positive controls. After the incubation, 20 μL MTS and 100 μL medium was added to each well after removal of 100 μL medium, and the incubation continued for 2–4 h at 37 °C. The optical density was measured at 492 nm using a Multiskan FC plate reader (Thermo Scientific, USA). The IC50 value of each compound was calculated according to the Reed and Muench method.
References
N.C. Veitch, Nat. Prod. Rep. 24, 417–464 (2007)
J. Reynaud, D. Guilet, R. Terreux, M. Lussignol, N. Walchshofer, Nat. Prod. Rep. 22, 504–515 (2005)
E.E. Shults, M.M. Shakirov, M.A. Pokrovsky, T.N. Petrova, A.G. Pokrovsky, P.G. Gorovoy, Nat. Prod. Res. 31, 445–452 (2017)
F. Li, S. Awale, Y. Tezuka, S. Kadota, Bioorg. Med. Chem. 16, 5434–5440 (2008)
H.D.T. Mai, T.T.O. Nguyen, V.C. Pham, M. Litaudon, F. Guéritte, D.T. Tran, V.H. Nguyen, Planta Med. 76, 1739–1742 (2010)
M.Q. Fu, S.X. Feng, N.L. Zhang, X.J. Zhou, R.M. Huang, H.J. Huang, Z.F. Xu, X.E. Li, S.X. Qiu, Helv. Chim. Acta 95, 598–605 (2012)
B. Bueno-Silva, S.M. Alencar, H. Koo, M. Ikegaki, G.V.J. Silva, M.H. Napimoga, P.L. Rosalen, J. Agric. Food Chem. 61, 4546–4550 (2013)
N.T. Hiep, J. Kwon, D.W. Kim, B.Y. Hwang, H.J. Lee, W. Mar, D. Lee, Phytochemistry 111, 141–148 (2015)
F. Wang, F.C. Ren, Y.J. Li, J.K. Liu, Chem. Pharm. Bull. 58, 1267–1270 (2010)
F. Wang, Y.J. Li, F.C. Ren, G.Z. Wei, J.K. Liu, Chem. Pharm. Bull. 59, 484–487 (2011)
F. Wang, M.F. Mao, G.Z. Wei, Y. Gao, F.C. Ren, J.K. Liu, Phytochemistry 95, 428–435 (2013)
F. Wang, X.L. Li, G.Z. Wei, F.C. Ren, J.K. Liu, Nat. Prod. Bioprospecting 3, 238–242 (2013)
G.Z. Wei, M.F. Mao, X.M. Li, F.C. Ren, F. Wang, Nat. Prod. Bioprospecting 6, 111–116 (2016)
Y. Goda, F. Kiuchi, M. Shibuya, U. Sankawa, Chem. Pharm. Bull. 40, 2452–2457 (1992)
Y. Gao, Y.D. Fang, P. Hai, F. Wang, J.K. Liu, Nat. Prod. Bioprospecting 3, 250–255 (2013)
D. Slade, D. Ferreira, J.P.J. Marais, Phytochemistry 66, 2177–2215 (2005)
A. Subarnas, Y. Oshima, H. Hikino, Phytochemistry 30, 2777–2780 (1991)
W.H. Chen, R. Wang, Y.P. Shi, J. Nat. Prod. 73, 1398–1403 (2010)
Y. Hata, S.N. Ebrahimi, M. De Mieri, S. Zimmermann, T. Mokoka, D. Naidoo, G. Fouche, V. Maharaj, M. Kaiser, R. Brun, O. Potterat, M. Hamburger, Fitoterapia 93, 81–87 (2014)
W.D. Ollis, I.O. Sutherland, H.M. Alves, O.R. Gottlieb, Phytochemistry 17, 1401–1403 (1978)
K. Kurosawa, W.D. Ollis, I.O. Sutherland, O.R. Gottlieb, A.B. De Oliveira, Phytochemistry 17, 1405–1411 (1978)
M.A. Ferreira, M. Moir, R.H. Thomson, J. Chem. Soc., Perkin Trans. 1, 2429–2435 (1974)
G.F. Spencer, B.E. Jones, R.D. Plattner, D.E. Barnekow, L.S. Brinen, J. Clardy, Phytochemistry 30, 4147–4149 (1991)
S. Yahara, T. Ogata, R. Saijo, R. Konishi, J. Yamahara, K. Miyahara, T. Nohara, Chem. Pharm. Bull. 37, 979–987 (1989)
H.A.M. Al-Ani, P.M. Dewick, Phytochemistry 24, 55–61 (1985)
H.L. Tuan Anh, D.T. Tuan, D.T. Trang, B.H. Tai, N.X. Nhiem, P.H. Yen, P.V. Kiem, C.V. Minh, T.M. Duc, H.K. Kang, Y.C. Kim, Y.H. Kim, J. Asian Nat. Prod. Res. (2016). doi:10.1080/10286020.2016.1232253
Acknowledgements
This work was financially supported by the “Large-scale Compound Library” project of National Development and Reform Commission of China.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Wang, LX., Zheng, HR., Ren, FC. et al. Polysubstituted Isoflavonoids from Spatholobus suberectus, Flemingia macrophylla, and Cudrania cochinchinensis . Nat. Prod. Bioprospect. 7, 201–206 (2017). https://doi.org/10.1007/s13659-017-0121-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13659-017-0121-2




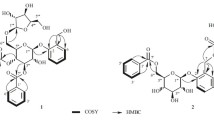


 ) correlations of 1–4
) correlations of 1–4