Abstract
Polycyclic aromatic hydrocarbons (PAHs) are hazardous organic micropollutants that are ubiquitous as well as recalcitrant to degradation. They are generally found in wastewater bodies with proximity to gas production, oil exploration, and other processing industries. Therefore, their clean-up and removal techniques are of high importance. In this work, a novel adsorbent was developed by modification of palm date seed-derived activated carbon (AC) with acrylic acid-co-crotonic acid polymer. The effectiveness of the produced polymer-modified AC (PM-AC) in the simultaneous aquatic removal of naphthalene and fluorene was studied systematically. The physicochemical characteristics of the developed PM-AC adsorbent were assessed by FTIR, SEM, and EDX. The equilibrium data are well in agreement with the Langmuir isotherm model and the adsorption capacity of the PM-AC was determined as 2.76 mg g−1 by considering this model. The adsorption mechanism of both PAHs onto the PM-AC adsorbent was well described by the pseudo-second-order kinetic model. The thermodynamic ΔG parameter showed the increased spontaneity of the PAHs adsorption with raising the temperature as the ΔH value confirmed the endothermic nature of the adsorption process. The π–π interactions were the key mechanisms of PAHs adsorption onto the prepared adsorbent. The PM-AC also demonstrated good adsorption performance in the case of a model wastewater sample containing some toxic heavy metals. The obtained findings revealed that the prepared PM-AC composite could be a promising adsorbent for developing cost-effective filters for the removal of PAHs from urban wastewaters. The PAHs levels were determined by using gas chromatography tandem mass spectrometry (GC–MS/MS). Recovery values were found nearly quantitative (≥ 95%).
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
1 Introduction
Wastewater treatment procedures are very important for reducing environmental pollution in urban and industrial areas. Polycyclic aromatic hydrocarbons (PAHs) are important organic pollutants which are containing two or more aromatic rings [1]. PAHs are mutagenic, toxic, and carcinogenic and listed as priority pollutants by the US-EPA and the EU [2]. PAHs are sourced from fossil fuels, petroleum, and industrial spills, and they are released into the atmosphere, environment, and water [3]. The carcinogenicity of PAHs is related to their molecular weight. PAHs cause an increased risk in various organs. They are also suppressing the immune system and are suspected of being endocrine disrupters [4]. Environmental Protection Agency (EPA) has defined approximately sixteen PAHs compounds as the most priority pollutants [5, 6].
Several methods have been used for the treatment of PAHs from contaminated water and soil samples. The main treatment methods for PAHs polluted water, soil, and sediments are physical, chemical, thermal, biological, and phytoremediation process such as thermal desorption, ion exchange, photolysis, oxidation, electrolysis, chemical precipitation, adsorption, and phytoextraction [7,8,9,10,11]. Conventional water treatment techniques such as filtration and sedimentation are based on the assumption that several organic contaminants are easily untaken by attaching to particulates. Nevertheless, there are some reports on significant portions of organic contaminants such as PAHs to reside in the dissolved and colloidal phases in wastewater [12, 13].
Adsorption procedure is a promising technology for the removal of pollutants due to its simplicity, high efficacy, and accessibility to several sorbents. Carbon-based sorbents including carbon nanotubes, fullerenes, graphene, and activated carbon have been reported to display good performance in removing pollutants from water [14]. Adsorption and desorption mechanisms of PAHs are important for their transport from aquatic environments. Adsorption mechanisms of PAHs may be π-π interaction, electron donor–acceptor interaction, and hydrogen bond formation on various adsorbents [15]. The adsorption process has some advantages such as simple operation, low cost, high efficiency, and easy to find various synthetic and natural adsorbents [16]. Several adsorbents such as detritus of green tide algae [17], marine algae [18], porous carbon obtained from petroleum coke [15], and activated carbon produced from palm shell [19] have been used in the removal of PAHs from waters. These adsorbents are not useful for adsorption and removal of PAHs because of their low adsorption capacity.
The high manufacturing cost makes sorbents less economically feasible to be used in wastewater purification. The use of cheap biosorbents attained from natural wastes such as agricultural for water purification can protect the environment by both removing the pollutants from water and reducing solid waste disposal [7]. Biochars, carbon-rich porous materials resulting from the agricultural-wastes pyrolysis, have been reported for the sorption of PAHs from water [20]. Magnetic biochar has been reported to enhance biochar’s adsorption efficacy toward removal of PAHs, naphthalene or phosphate, in a single phase removal process [21]. Byproduct coconut coir fiber (attained from the outer husk of a coconut) is a lignocellulosic biomass that consists of around 40% cellulose, 30% lignin, and 16% hemicellulose. Interestingly, cellulose comprises phenolic and carboxylic acids having functional groups that are accountable for the adsorption of polar pollutants, whereas lignin aromatic rings are accountable for the adsorption of organic contaminants [22, 23]. Nevertheless, some carbon-based materials with irreversible adsorption characteristics may result in poor regeneration. Hence, hybrid materials consisting of for example polymer and carbon could be a promising alternative. Polymeric materials have been lately getting importance due to their unique characteristics and enhanced surface properties enabling efficient adsorption and reusability [24]. The production of palm dates is an important agricultural activity in the arid regions of East and North Asia [25]. They can also be found in southern Europe, Asia, the African continent, Australia, South America, and the USA, especially in southern California, Texas, and Arizona [26]. Palm dates are used both in the food sector for humans and animals and in the construction sector including textiles [27]. Removal of organic pollutants including nitrophenols, chlorophenol, bisphenol, and monocrotophos has been performed by using activated carbon derived from palm shells [28]. Green synthesis of zinc oxide nanoparticles from palm dates was used for dye degradation and antibacterial performance in wastewater treatment [29]. According to our literature survey, there is no study about simultaneous removal of polyaromatic hydrocarbons from wastewaters by using polymer modified carbon derived from palm date seeds powder. In industry, where such pollutants are every day produced, it is highly required to remove them from wastewater before discard the water into environment or before the possible reuse.
Recently, further efforts have been drifting toward green chemistry where the emphasis is on zero waste as a tool to manage waste materials. Thus, agricultural waste, date seed-based activated carbon, is of high interest to be used for adsorption of organic pollutants, such as PAHs, from contaminated water. Within this framework, the activated carbon derived from the date seed was produced and then modified with poly (acrylic acid-co-crotonic acid) copolymer. The π–π interactions and carboxyl groups of the copolymer grafted to the surface of AC played an important role in significantly increasing the simultaneous adsorption of the naphthalene and fluorene as PAH model compounds in aqueous media.
The aim of this study was to preparation a new and novel adsorbent which has effective and high adsorption capacity for removal of PAHs. So, polymer-modified AC (PM-AC) was prepared and effectively used for simultaneous adsorption and removal of PAHs from wastewaters by using GC–MS/MS.
2 Experimental
2.1 Materials
Nitric acid, acrylic acid, crotonic acid, tetrahydrofuran (THF), azobisisobutyronitrile (AIBN), hexane (≥ 97% GC grade), dichloromethane (DCM), naphthalene (Naph), and fluorene (Fl) with the purity of 99% were from Sigma Aldrich US. The stock concentrations of 1000 µg L−1 PAHs were prepared by DCM. The stock solution was used to prepare working concentration daily by diluting it in an appropriate amount of hexane.
2.2 Synthesis
The carbon derived from the palm date seeds (which is available in several countries) were oven-dried then ball milled into powder. The obtained powder was heated in a closed horizontal furnace under the flow of N2, 200 mL min−1) to create an inert atmosphere. After cooling, the obtained carbon was treated with zinc acetate (i.e., ratio 2:1), which is comparatively less harsh, a weak Lewis acid. Six grams of date seed powder was mixed with 3 g of zinc salt. The system was kept under stirring for 4 h in order to ensure homogeneity. The obtained activated carbon (AC) was collected dried and then further activated with nitric acid, 1 M, for 1 h. It was then washed until neutral pH and then dried in a vacuum oven. The obtained carbon was then modified with polymer as per the following steps.
About 6 g crotonic acid and 6 g acrylic acid were mixed with 70 mL distilled toluene in a flask with mechanical agitation under the flow of nitrogen at 100 mL/min flow rate. Then, the system was heated to 75 °C for 15 min. After that, 0.05 g AIBN as an initiator was introduced under nitrogen [30]. After 4 h, 3 g of the obtained AC was introduced onto the obtained polymer [31] with mechanical agitation for 4 h. The formed material was then separated by magnetic fields and clearly washed 3 times using 200 mL anhydrous ethanol. After that, it was dried under vacuum at 70 °C. The synthesized poly (acrylic acid-co-crotonic acid) copolymer modified AC was abbreviated as PM-AC (Scheme 1).
2.3 Instrumentations
The structures of the developed adsorbent were characterized by FTIR Spectrometer (a Thermo Scientific Nicolet 6700 model). The microstructure of the composite was studied using scanning electron microscopy, SEM/EDX (JSM-6610LV Scanning Electron Microscope from JEOL) coupled with energy dispersive X-ray (EDX).
The GC–MS/MS (GCMS TQ 8030 Shimadzu) was utilized for the PAHs analysis. The Rxi-1 ms column (Restek USA) has an internal diameter 30 mm × 0.25 mm and a film thickness of 0.25-µm film thickness. The injection temperature was 300 °C, and the flow rate of helium as carrier gas (99%) was set to 1.0 mL.min−1. The splitless mode with a volume injection of 1.0 µL was used. The oven temperature was programmed as listed. It was ramped from 40 to 250 °C at a rate of 10 °C/min without any hold, then another ramping from 250 to 300 °C at a rate of 5 °C min−1 and holding for 5 min. The gas flow rate was set at 1.5 mL min−1, and the temperature of the detector was 230 °C. The detection limit of PAH analysis method is about 1 μg L−1, while the limit of quantification is 10 μg L−1 with ≈ 97% recovery. Inductively coupled plasma optic emission spectrometry (ICP-OES) (PlasmaQuant® PQ 9000) was used for the determination of metal ions. The recovery measurement was determined through comparing samples spiked before and after extraction.
2.4 Batch adsorption experiments
In order to evaluate the adsorption efficiency of the prepared adsorbent, experiments were conducted to obtain information on the rate of naphthalene and fluorene adsorption. The effect of several parameters was optimized as well including the adsorbent dosage (range between 0.01 and 0.6 g, initial concentration (range between 0.1 and 120 µg/L), contact time (from 0 to 60 min), and temperature (range between 298 and 3628). For the temperature effect, the batch adsorption experiments were carried out at various temperatures of 298, 308, and 318, as well as 328 K. The percentage removal was calculated by Eq. 1, and the capacity of adsorption was calculated by Eq. 2:
where Co (mg L−1) and Cexp (mg L−1) refer to initial concentrations of the PAHs (naphthalene and fluorene) before and after the removal. M in g is the mass of the adsorbent and V in L is the volume of the PAHs solutions. The internal standard (Otho-Terphenyl) was introduced to the aliquot. The remnant PAHs together with the internal standard were extracted by the method of liquid–liquid extraction. The GC–MS/MS was utilized for the determination of PAHs.
2.5 Isotherm modeling of adsorption data
The Langmuir isotherm theorizes that the adsorption process of the pollutants is carried out by specific homogeneous sites with equal energy on the surface of the adsorbent. Thus, the adsorptive sites were covered as homogenous monolayer by pollutant species [32, 33]. This model is given in non-linear equation form, respectively [34]:
where qe are the total adsorbed quantity of two PAH species per unit mass of the PM-AC adsorbent (mg g−1) as Ce is the total PAH concentration (mg L−1) at equilibrium conditions. qmax is the monolayer adsorption capacity of the adsorbent (mg g−1), and KL (L mg−1) is an adsorption energy-associated constant.
The Freundlich model is based on heterogeneous multilayer adsorption by adsorptive sites with non-unequal energy. The isotherm model is expressed in non-linear form as [34]:
where Kf (L.g−1) and n are model constants that are related to saturation capacity and intensity of adsorption, respectively. The 1/n value between 0.1 and 1.0 specifies favorable adsorption [35].
2.6 Modeling of kinetic data
In this framework, the most widely used kinetic models, pseudo-first-order (PFO) and pseudo-second-order (PSO), were adopted for the experimental data. These model equations are given as following [4, 36, 37]:
where qe and qt are the total amount of PAHs on the surface of PM-AC adsorbent at equilibrium time and any t time, respectively. k1 and k2 are the PFO rate constant and PSO rate constant, respectively.
3 Results and discussion
3.1 Characterization
The SEM images (Fig. 1a–f) reveal the morphology of the obtained carbon from dates and the polymer-modified carbon PM-AC. SEM images of the AC (Fig. 1a–c) reveal slides shape with grooves surfaces. The SEM images of and the PM-AC reveal some small branches grafted on carbon, and the surface is smoother and a uniform shape. The EDX elemental analysis indicates the presence of the elements forming the AC (Fig. 2a) and the PM-AC (Fig. 2b).
The chemical structure was evaluated by FTIR analysis. Bands at around 3200 to 3400 cm−1 are assigned to the stretching vibrations of hydroxyl groups on the surface of the polymeric branches. The band at about 1650 cm−1 is assigned to the C = O stretching [38]. The bands between 2840 and 2920 cm−1 can be attributed to the symmetric as well as asymmetric stretching vibrations of the -CH2 group of the polymeric branches. The bands at 1410, 1480, and 1584 cm−1 can be attributed to the vibrations in the aromatic structure. In addition, the band at around 1720 cm−1 can be attributed to the carboxylic groups on the polymer. The presence of such functional groups in addition to the aromaticity of the activated carbon plays a key role in the removal of PAHs. This allow the adsorbent to interact with PAHs pollutants via covalent and non-covalent bond formation such as hydrogen bonding, electrostatic forces, π-π stacking, van der Waals forces, and hydrophobic interactions [39].
3.2 Optimization of PAHs removal
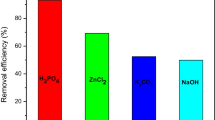
The effects of the batch parameters on the removal of PAHs using the prepared adsorbents were optimized and the obtained results were compared. The results depicted in Fig. 3 indicate that PM-AC adsorbents show better removal of PAHs which can be ascribed by the presence of more active sites on the adsorbent in addition to the carbon structure. The removal of single PAH pollutants (naphthalene or fluorene) was compared also with the simultaneous adsorption of both naphthalene and fluorene with respect to the change in the dosage of the AC and PM-AC adsorbents (Fig. 4). The results indicated that the material showed excellent simultaneous removal of both PAHs pollutants which can be attributed to the π–aromatic system interacting with the active sites on the adsorbent. It should be mentioned that the experiments were repeated three times and the average of the percentage removal was presented. The error bars represent the standard division of the measurements which indicate good repeatability of the experiments.
3.3 Isotherm modeling results
The non-linear model curves obtained for simultaneous adsorption of the PAHs, naphthalene, and fluorene by PM-AC adsorbent are demonstrated in Fig. 5. As seen from the correlation coefficient value R2, 0.998, the Langmuir model well describes the monolayer adsorption behaviors of the PAHs onto all of the PM-AC adsorbents as well as satisfactorily reflects the relation between the adsorption equilibrium data (qe and Ce). Similar isotherm modeling results were reported for PAHs adsorption using Al-MCM-41 [32] and zeolite [33] from wastewaters. Moreover, the Langmuir monolayer adsorption capacity (qm) of PM-AC adsorbent was estimated as 2.76 mg g−1, respectively, for simultaneous adsorption of the PAHs. Additionally, the model constant KL was found as 8.19 × 10−3 L mg−1.
The non-linear Freundlich isotherm plots obtained for simultaneous adsorption of the PAHs by PM-AC are also demonstrated in Fig. 5. As seen from the relatively lower R2 value, 0.989, this model does not well designate the relation between the adsorption equilibrium data. The Kf and n constants were determined in the range of 48.88 L g−1 and 0.70 for simultaneous adsorption of the PAHs by PM-AC.
3.4 Kinetic investigation results
The PFO model kinetic modeling results obtained for total adsorption of two PAH species by PM-AC at 25 °C are shown in Fig. 6. As seen from the data, the correlation coefficient R2 0.863 is at a relatively low level, meaning that the PFO model can be used to characterize the kinetic modeling of the adsorption and rate-limiting step of the simultaneous adsorption mechanism of the PAHs by PM-AC. The great inconsistency between the experimental qe (1.38 mg g−1) and calculated qe data (0.46 mg g−1) also confirms this argument.
On the other hand, the experimental adsorption data are evaluated by using PSO kinetic equations, and the obtained results are presented in Fig. 7. Based on these results, it can be inferred that the PSO model equation is well appropriate to designate the kinetic mechanism and determine the rate-limiting step for simultaneous adsorption of the PAHs by PM-AC at 25 °C because of relatively high R2, 0.992. In addition, the closeness of experimental and qe,exp (1.38 mg g−1) to and calculated qe (1.32 mg g−1) verified this inference. The PSO model was indicated as the best fitting for identifying the kinetic mechanism of the various PAH adsorption onto modified organosilica, plant residue-based sorbent, and clay sediment [40].
3.5 Thermodynamic investigation results
The dependency of the adsorption yield on temperature was investigated to characterize the thermal feasibility and spontaneity of both PAHs adsorption by PM-AC. With this framework, the crucial thermodynamic functions, Gibbs free energy change ΔG°, enthalpy change ΔH°, and entropy change ΔS° were calculated under the examined temperature range. ΔG° function was calculated based on the adsorption equilibrium constant Ke (qe/Ce):
ΔH° and ΔS° are calculated by considering the plots in Fig. 8 and Eq. (8):
As clearly seen from these results, the negative ΔG° values (− 10.25, − 10.01, 11.92, and 12.66 kJ.mol−1, respectively, for 298, 308, 318, and 328 K) show that thermal probability and spontaneity of the PAHs adsorption by PM-AC are increased with temperature increase from 298 to 328 K. Moreover, the determined ΔH° (14.0 kJ mol−1) approves the endothermic progressing of the simultaneous adsorption of the selected PAHs under the considered temperature conditions. Moreover, positive ΔS°, 81.39 J mol−1 K−1 designates the reduced arbitrariness at the solid–liquid interface. The endothermic character was reported for the adsorption of naphthalene, acenaphthylene, and phenanthrene onto NH2-SBA-15 [41].
3.6 Removal evaluation using wastewater sample
The efficiency of the PM-AC was evaluated in the case of model wastewater sample containing some toxic heavy metals. The PM-AC was added to the sample, and the water was analyzed for the concentration of metal ions using inductively coupled plasma (ICP). Table 1 demonstrates the excellent performance of PM-AC for the simultaneous removal of most of the metal ions in addition to the excellent removal of PAHs under the same conditions. Therefore, the PM-AC displayed good adsorption performance [42,43,44,45,46,47,48,49].
4 Conclusions
In this study, a novel adsorbent was developed by modification of palm date seed-derived AC with acrylic acid-co-crotonic acid polymer. The effectiveness of the produced PM-AC adsorbent in the simultaneous aquatic removal of naphthalene and fluorene as PAH model compounds was investigated systematically. The microstructure and chemical structure of the synthesized PM-AC were characterized by FTIR and SEM/EDX. The equilibrium data are well in agreement with the Langmuir isotherm model and the adsorption capacity of the PM-AC was determined as 2.76 mg g−1 by considering this model. The kinetic evaluations indicated that the simultaneous adsorption of both PAHs onto the PM-AC adsorbent followed well to the PSO kinetic model. The calculated ΔG verified the increased spontaneity of the PAHs adsorption with raising the temperature as the ΔH value indicated the endothermic nature of the adsorption process. The π–π interactions were the foremost mechanisms for the adsorption of PAHs over the PM-AC adsorbent. The PM-AC showed excellent removal performance in the case of a model wastewater sample containing some toxic heavy metals. Accordingly, all of the results revealed that the PM-AC material is a hopeful adsorbent for the cost-effective adsorption of PAHs pollutants from the aquatic environments.
References
Nazir NAM, Raoov M, Mohamad S (2020) Spent tea leaves as an adsorbent for micro-solid-phase extraction of polycyclic aromatic hydrocarbons (PAHs) from water and food samples prior to GC-FID analysis. Microchem J 159:105581
Busetti F, Heitz A, Cuomo M, Badoer S, Traverso P (2006) Determination of sixteen polycyclic aromatic hydrocarbons in aqueous and solid samples from an Italian wastewater treatment plant. J Chromatogr A 1102:104–115
Ozaki N, Takamura Y, Kojima K, Kindaichi T (2015) Loading and removal of PAHs in a wastewater treatment plant in a separated sewer system. Water Res 80:337–345
Lamichhane S, Krishna KCB, Sarukkalige R (2016) Polycyclic aromatic hydrocarbons (PAHs) removal by sorption: a review. Chemosphere 148:336–353
Qi YB, Wang CY, Lv CY, Lun ZM, Zheng CG (2017) Removal capacities of polycyclic aromatic hydrocarbons (PAHs) by a newly isolated strain from oil field produced water. Int J Environ Res Public Health 14:215–227
US EPA, Priority Pollutant List. (Retrieved August 23, 2021) from https://www.epa.gov/sites/production/files/2015-09/documents/priority-pollutant-list-epa.pdf
Esfandiar N, Suri R, McKenzie ER (2021) Simultaneous removal of multiple polycyclic aromatic hydrocarbons (PAHs) from urban stormwater using low-cost agricultural/industrial byproducts as sorbents. Chemosphere 274:129812
Mortazavi M, Baghdadi M, Javadi NHS, Torabian A (2019) The black beads produced by simultaneous thermal reducing and chemical bonding of graphene oxide on the surface of amino-functionalized sand particles: application for PAHs removal from contaminated waters. J Water Process Eng 31:100798
Ge X, Wu Z, Wu Z, Yan Y, Cravotto G, Ye BC (2016) Enhanced PAHs adsorption using iron- modified coal-based activated carbon via microwave radiation. J Taiwan Inst Chem Eng 64:235–243
Mojiri A, Zhou JL, Ohashi A, Ozaki N, Kindaichi T (2019) Comprehensive review of polycyclic aromatic hydrocarbons in water sources, their effects and treatments. Sci Total Environ 696:133971
Zango ZU, Sambudi NS, Jumbri K, Ramli A, Abu Baker NHH, Saad B, Nur M, Rozaini MNH, Isiyaka HA, Osman AM, Sulieman A (2020) An overview and evaluation of highly porous adsorbent materials for polycyclic aromatic hydrocarbon and phenols removal. Water 12:2921
Björklund K, Li L (2015) Evaluation of low-cost materials for sorption of hydrophobic organic pollutants in stormwater. J Environ Manag. https://doi.org/10.1016/j.jenvman.2015.05.005
Ulrich BA, Im EA, Werner D, Higgins CP (2015) Biochar and activated carbon for enhanced trace organic contaminant retention in stormwater infiltration systems. Environ Sci Technol. https://doi.org/10.1021/acs.est.5b00376
Saleh TA, Naeemullah M, Tuzen A (2017) Sarı, Polyethylenimine modified activated carbon as novel magnetic adsorbent for the removal of uranium from aqueous solution. Chem Eng Res Des 117:218–227
Yuan M, Tong S, Zhao S, Jia CQ (2010) Adsorption of polycyclic aromatic hydrocarbons from water using petroleum coke-derived porous carbon. J Hazard Mater 181:1115–1120
Tuncaboylu DC, Abdurrahmanoglub S, Gazioglu I (2019) Rheological characterization of starch gels: a biomass-based sorbent for removal of polycyclic aromatic hydrocarbons (PAHs). J Hazard Mater 371:406–414
Zhang C, Lu J, Wu J (2019) Adsorptive removal of polycyclic aromatic hydrocarbons by detritus of green tide algae deposited in coastal sediment. Sci Total Environ 670:320–327
Arias AH, Souissi A, Glippa O, Roussin M, Dumoulin D, Net S, Ouddane B, Souissi S (2017) Removal and biodegradation of phenanthrene, fluoranthene and pyrene by the marine algae Rhodomonas baltica enriched from North Atlantic coasts. Bull Environ Contam Toxicol 98:392–399
Kumar JA, Amarnath DJ, Senthil PK, Kaushik CS, Varghese ME, Saravanan A (2018) Mass transfer and thermodynamic analysis on the removal of naphthalene from aqueous solution using oleic acid modified palm shell activated carbon, Desal. Water Treat 106:238–250
Reddy KR, Xie T, Dastgheibi S (2014) Evaluation of biochar as a potential filter media for the removal of mixed contaminants from urban storm water runoff. J Environ Eng (United States). https://doi.org/10.1061/(ASCE)EE.1943-7870.0000872
Chen B, Chen Z, Lv S (2011) A novel magnetic biochar efficiently sorbs organic pollutants and phosphate. Bioresour Technol. https://doi.org/10.1016/j.biortech.2010.08.067
Crisafully R, Milhome MAL, Cavalcante RM, Silveira ER, De Keukeleire D, Nascimento RF (2008) Removal of some polycyclic aromatic hydrocarbons from petrochemical wastewater using low-cost adsorbents of natural origin. Bioresour Technol. https://doi.org/10.1016/j.biortech.2007.08.041
Xi Z, Chen B (2014) Removal of polycyclic aromatic hydrocarbons from aqueous solution by raw and modified plant residue materials as biosorbents. J Environ Sci (China). https://doi.org/10.1016/S1001-0742(13)60501-X
Hayati B, Maleki A, Najafi F et al (2018) Heavy metal adsorption using PAMAM/CNT nanocomposite from aqueous solution in batch and continuous fixed bed systems. Chem Eng J 346:258–270
Lala S, Amri A, Maxted N (2018) Towards the conservation of crop wild relative diversity in North Africa: checklist, prioritisation and inventory. Genet Resour Crop Evol 65:113–124
Al-Alawi RA, Al-Mashiqri JH, Al-Nadabi JSM, Al-Shihi BI, Baqi Y (2017) Date palm tree (Phoenix dactylifera L.): natural products and therapeutic options. Front Plant Sci 8:845
Cruz-Lovera C, Manzano-Agugliaro F, Salmerón-Manzano E, Cruz-Fernández JL, Perea-Moreno AJ (2019) Date seeds (Phoenix dactylifera L.) valorization for boilers in the mediterranean climate. Sustainability 11:711
Pamidimukkala PS, Soni H (2018) Efficient removal of organic pollutants with activated carbon derived from palm shell: spectroscopic characterisation and experimental optimization. J Environ Chem Eng 6:3135–3149
Rambabu K, Bharath G, Banat F, Show PL (2021) Green synthesis of zinc oxide nanoparticles using Phoenix dactylifera waste as bioreductant for effective dye degradation and antibacterial performance in wastewater treatment. J Hazard Mater 402:123560
Bassaid S, Chaib M, Bouguelia A, Trari M (2008) Elaboration and characterization of poly (acrylic acid-co-crotonic acid) copolymers: application to extraction of metal cations Pb(II), Cd(II) and Hg(II) by complexation in aqueous media. React Funct Polym 68:483–491
Kovyazin VA, Nikitin AV, Kopylov VM (2003) Reaction of organosilicon amines with dicarboxylic anhydrides. Russ J Gen Chem 73:1072–1076
Nkansah MA, Christy AA, Barth T, Francis GW (2012) The use of lightweight expanded clay aggregate (LECA) as sorbent for PAHs removal from water. J Hazard Mater 217–218:360–365
Vidal CB, Barros AL, Moura CP, de Lima ACA, Dias FS, Vasconcellos LCG, Fechine PBA, Nascimento RF (2011) Adsorption of polycyclic aromatic hydrocarbons from aqueous solutions by modified periodic mesoporous organosilica. J Colloid and Interf Sci 357:466–473
Langmuir I (1918) The adsorption of gases on plane surfaces of glass, mica and platinium. J Am Chem Soc 40:1361–1403
Araujo RS, Azevedo DCS, Cavalcante CL, Jimenez-Lopez A, Rodrıguez-Castellon E (2008) Adsorption of polycyclic aromatic hydrocarbons (PAHs) from isooctane solutions by mesoporous molecularsieves: influence of the surface acidit. Microporous Mesoporous Mater 108:213–222
Chang CF, Chang CY, Chen KH, Tsai WT, Shie JL, Chen YH (2004) Adsorption of naphthalene on zeolite from aqueous solution. J Colloid and Interf Sci 277:29–34
Freundlich HMF (1906) Über die adsorption in lösungen. Zeitschrift für Physikalische (Leipzig) 57A:385–470
Zhang Z, Long J, Yang L, Chen W, Dai W, Fu X, Wang X (2011) Organic semiconductor for artificial photosynthesis: water splitting into hydrogen by a bioinspired C3N3S3 polymer under visible light irration. Chem Sci 2:1826–1830
Hu B, Gao Z, Wang H, Wang J, Cheng M (2020) Computational insights into the sorption mechanism of polycyclic aromatic hydrocarbons by carbon nanotube through density functional theory calculation and molecular dynamics simulation. Comput Mater Sci 179:109677
Schiewer S, Patil SB (2008) Pectin-rich fruit wastes as biosorbents for heavy metal removal: equilibrium and kinetics. Bioresour Technol 99:1896–1903
Xi Z, Chen B (2014) Removal of polycyclic aromatic hydrocarbons from aqueous solution by raw and modified plant residue materials as biosorbents. J Environ Sci 26:737–748
Saleh TA (2015) Mercury sorption by silica/carbon nanotubes and silica/activated carbon: a comparison study. Journal of Water Supply: Research and Technology - Aqua 64(8):892–903. https://doi.org/10.2166/aqua.2015.050
Saleh TA (2015) Isotherm, kinetic, and thermodynamic studies on Hg(II) adsorption from aqueous solution by silica- multiwall carbon nanotubes. Environ Sci Pollut Res 22(21):16721–16731. https://doi.org/10.1007/s11356-015-4866-z
Saleh TA (2015) Nanocomposite of carbon nanotubes/silica nanoparticles and their use for adsorption of Pb(II): from surface properties to sorption mechanism. Desalination and Water Treatment 57(23):10730–10744. https://doi.org/10.1080/19443994.2015.1036784
Saleh TA (2021) Protocols for synthesis of nanomaterials, polymers, and green materials as adsorbents for water treatment technologies. Environmental Technology & Innovation 24:101821. https://doi.org/10.1016/j.eti.2021.101821
Saleh TA (2020) Nanomaterials: Classification, properties, and environmental toxicities. Environmental Technology & Innovation 20:101067. https://doi.org/10.1016/j.eti.2020.101067
Saleh TA (2020) Trends in the sample preparation and analysis of nanomaterials as environmental contaminants. Trends in Environmental Analytical Chemistry 28:e00101. https://doi.org/10.1016/j.teac.2020.e00101
Saleh TA (2020) Characterization, determination and elimination technologies for sulfur from petroleum: Toward cleaner fuel and a safe environment. Trends in Environmental Analytical Chemistry 25:e00080. https://doi.org/10.1016/j.teac.2020.e00080
Saleh TA, Mustaqeem Mujahid, Khaled Mazen (2021) Developing water treatment technologies in removing heavy metals from wastewater: A review. Environmental Nanotechnology, Monitoring & Management 100617. https://doi.org/10.1016/j.enmm.2021.100617
Acknowledgements
The authors thank DSR unit of KFUPM for financial support (Project No. DF181001). Dr. A. Sarı thank KTU owing to instrumentation feasibilities. Dr. M. Tuzen also thanks Turkish Academy of Sciences due to financial support.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Saleh, T.A., Sarı, A. & Tuzen, M. Simultaneous removal of polyaromatic hydrocarbons from water using polymer modified carbon. Biomass Conv. Bioref. 14, 567–576 (2024). https://doi.org/10.1007/s13399-021-02163-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13399-021-02163-9