Abstract
Scavenging of carrion is an important ecological process that influences ecological communities and food webs. The competitive inter- and intra-specific interactions in terrestrial vertebrate scavenger communities are likely to limit access to carrion for some scavengers, having direct impacts on their fitness by limiting energetic intake. Striped hyenas (Hyaena hyaena) are well-known facultative scavenger that co-exists with other carnivores (i.e., tigers, Panthera tigris; leopards, Panthera pardus; and golden jackals, Canis aureus) across the landscape. In this study, we assessed the competitive interactions among large carnivores having a large-sized body (i.e., striped hyenas, tigers, leopards) and a social group foraging mesocarnivore (i.e., golden jackal) through their carrion acquisition with special reference to striped hyenas’ carrion acquisition in the semi-arid region of Rajasthan, India. We deployed camera traps at 14 carcass sites from 2020 to 2022 and considered three main aspects of scavenging behavior (presence, total feeding time, and mean feeding bout duration). We used generalized linear models (GLMs) to understand the competitive interactions among large carnivores and mesocarnivores through their carcass consumption and scavenging efficiency. Our analyses showed that the tigers are dominant scavengers that monopolize carrion resources and reduce consumption time, and hence nutrition gained, of striped hyenas and the other carnivorous scavengers. But leopards and striped hyenas did not show any major negative interactions at carcasses, while both jackals and striped hyenas negatively affected each other’s carrion acquisition at carcasses; as a result, striped hyena’s presence affected the carrion acquisition by jackals, and also jackals affected the mean feeding bout duration of striped hyenas. Our results highlight the importance of functional traits in intraguild interactions and the potential effects of competition on carrion acquisition. Specifically, large carnivores that have specific traits, i.e., large body size, negatively affected the subordinate carnivores at carcasses, while the mesocarnivores that have specific traits, i.e., social group foraging, negatively affect the carrion acquisition and energetics of solitary feeding striped hyenas.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Large carnivores may affect scavengers by influencing the distribution of carrion resources (Elbroch and Wittmer 2012; Moleón et al. 2014; Perrig et al. 2017) and by limiting access to carrion through competition (Moleón et al. 2014; Allen et al. 2015). Carnivores that hunt live prey are also often efficient scavengers (Mateo-Tomás et al. 2015; Moleón et al. 2015), and the competitive interactions between scavengers may be as complex as prey-predator relationships (Selva and Fortuna 2007; Pereira et al. 2014) since they affect food web dynamics (Barton et al. 2013). Large carnivores often control carcasses that they have killed or found, and as a result, limiting the carrion acquisition by other obligate and facultative scavengers (Wilmers et al. 2003; Hunter et al. 2007; DeVault et al. 2011; Cortés-Avizanda et al. 2012; Pereira et al. 2014; Allen et al. 2014). However, in some cases, dominant scavengers limit the consumption of carcasses of subordinate scavengers (Krofel et al. 2012; Allen et al. 2014, 2015; Inagaki et al. 2020). In addition to providing the carrion to scavengers from their kills, large carnivores often open large carcasses, allowing other species to access edible parts of the carcass that they would not be able to open on their own. (Selva et al. 2003).
The direct interactions between large carnivores and other species can lead to indirect consequences further down the food web via trophic cascades (Ripple et al. 2016). For example, increases in gray wolves (Canis lupus) in Minnesota resulted in decreases in coyotes (Canis latrans), which in turn allowed an increase in smaller red foxes (Vulpes vulpes; Levi and Wilmers 2012). Yet, large carnivores do not always suppress mesopredators (Lesmeister et al. 2015; Lyly et al. 2015). In Africa, lions (Panthera leo) and spotted hyenas (Crocuta crocuta) coexist over food resources and also show competitive behavior (Amorós et al. 2020). Similarly, in India, leopards (Panthera pardus) and striped hyenas (Hyaena hyaena) have been reported feeding together on a carcass (Mandal et al. 2018). One important aspect of this may be the facilitation of carrion resources that large carnivores provide to other scavengers (Allen et al. 2014), but the responses of subordinate scavengers to larger carnivores may also be species-specific. Striped hyenas are solitary large-sized carnivore (Califf et al. 2020) that is widely distributed in arid and semi-arid landscapes from East Africa to South Asia (Akash et al. 2021; Singh et al. 2010), but are subordinate to tigers (Panthera tigris) and leopards, but larger than golden jackals in semi-arid landscapes in India (Menon 2014). Apex carnivores such as lions, tigers, and leopards frequently kill wild ungulates (i.e., sambar Rusa unicolor, chital Axis axis, wild boar Sus scrofa, nilgai Boselaphus tragocamelus) as well domestic livestock (cow, buffalo, sheep, goat) that are attractive to scavengers. Striped hyenas and golden jackals are facultative scavengers, but rarely kill the ungulates or domestic animals and instead opportunistically feed on the remains of animals predated by larger carnivores and domestic livestock carcasses discarded by humans (Singh et al. 2010).
Both tigers and leopards are ecologically, behaviorally, and morphologically similar. Tigers (with body weights of 100–260 kg) are about double the size of leopards (30–90 kg) and much larger than striped hyenas (22–55 kg) and golden jackals (7–11 kg). Owing to their larger body size, tigers are typically dominant over leopards and other carnivores; and while tigers occasionally kill and eat leopards (Karanth and Sunquist 1995), no such incidents have been reported for striped hyenas. Tigers typically kill medium-sized prey species (weighing between 50 and 200 kg) (Biswas and Sankar 2002), and a tiger needs to kill between 50 and 60 large animals in a year to meet their energetic requirements (Miller et al. 2013). With this regular killing of wild ungulates and domestic livestock, tigers and leopards act as an important source of carrion for scavengers (Singh et al. 2014, 2015; Panda et al. 2022).
The interactions between tigers and leopards have been well studied (Karanth et al. 2017), as leopards sometimes opportunistically scavenge on tiger kills (Prater 1965), but interactions with other carnivores have not been as well studied. Being a large carnivore and apex predator, tigers require large amounts of food, consuming up to 10–34 kg of meat per day (Tamang 1982). However, leopards and hyenas are generalist species, and their diet niches overlap considerably, which may lead to greater competition for food (Heptner and Sludjkij 1982; Arivazhagan et al. 2007). It has been reported that striped hyenas chase leopards (Pocock 1941), and leopards have also been documented killing striped hyenas in the Sariska Tiger Reserve (Mandal et al. 2018). Chourasia et al. (2010) reported a significant dietary overlap of 67% between striped hyenas and golden jackals in semi-arid habitats, suggesting the high competition due to the mutual dependence on ungulate carcasses. Moreover, golden jackals coexist with larger carnivores in some areas, which may affect their diet through competition, increased intraguild predation risk, or facilitation through jackals scavenging the carcasses of larger carnivore kills (Hayward et al. 2017). However, depending on food availability, golden jackals may be solitary or group hunters (Macdonald 1979), and in India, packs of 5 to 18 jackals have been observed scavenging on the carcasses of large ungulates (Jhala et al. 2004).
The potential competition for carrion acquisition among tigers, leopards, golden jackals, and striped hyenas is understudied in India. In this study, first, we evaluated how solitary large carnivores (tigers and leopards) and a social mesocarnivore (golden jackal) affect the acquisition of carrion by striped hyenas. Second, we evaluated the effects of tigers on leopards and golden jackals and the effects of striped hyenas on leopards and golden jackals carrion acquisition. To address this question, we deployed camera traps at wild and domestic carcasses in and around Sawai Mansingh Wildlife Sanctuary (SMS WLS), Rajasthan, India. We focused on three aspects of carrion acquisition exhibited by carnivores (based on Allen et al. 2015): (1) presence (a proxy for a carnivore’s ability to access the carrions), (2) total feeding time (a proxy for the amount of energy gained by a carnivore from a given carcass), and (3) mean feeding bout duration (a proxy for the effects of predation risk perceived by carnivores, as carnivores exhibit shorter feeding events at carcasses with higher perceived risk; Charnov 1976). As a dominant apex carnivore, we expected tigers to limit all three aspects of carrion acquisition by the other carnivores. As a potentially dominant competitor, we expected leopards to limit the total feeding time and mean feeding bout duration of by striped hyenas, but not exclude hyenas from carcasses. Although golden jackals are smaller mesocarnivores, we hypothesized that scavenging on the carcasses by large groups of golden jackals could also negatively affect the carrion acquisition of striped hyenas.
Material and methods
Study area
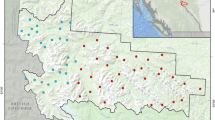
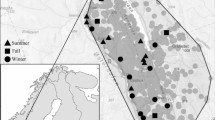
We conducted this study in and around the Sawai Mansingh Wildlife Sanctuary (SMS WLS), Rajasthan, India (Fig. 1). The SMS WLS is a part of the tiger conservation and management unit of the Ranthambhore Tiger Reserve (Singh et al. 2013). The total area of SMS WLS is 127.6 km2, while another adjacent forested area (Qualji area) is 7.58 km2 and another forested area is 132.96 km2 (Jhala et al. 2011). The entire Ranthambhore landscape forms a transition zone between the true desert and seasonally wet peninsular India (Singh et al. 2013). The region’s average annual rainfall is 800 mm, of which 500 mm falls in the monsoon season. The temperatures can be ≤ 2 °C in January and ≥ 47 °C in May. The landscape is undulating and dominated by humans; there are 75 villages within a 5 km buffer of SMS WLS with more than 104,261 people inhabiting the area. The rolling landscape mosaic is interspersed with forest, scrublands, grasslands, riverine areas, and agricultural lands. The residents are mostly engaged in agriculture, livestock farming, cutting grass, grazing livestock, lopping trees, and mining (illegally) to supplement household incomes. All of the villages are primarily dependent on agriculture for their livelihood, and their economy is supplemented by animal husbandry. They have numerous cows, buffaloes, and goats, but very few herds of sheep or camels. The villagers tend to graze their animals in the fallow agricultural lands and the village commons during the lean periods of the year (viz., January to June). However, the villagers enter the peripheral forest area to graze their animals throughout the year. The area is dominated by northern tropical, dry deciduous, and thorny forests. The forests are mainly of edaphic climax and belong to subgroup 5B (northern tropical dry deciduous forests) and subgroup 6B (DS1 – zizyphus scrub) (Champion and Seth 1968). The degradation stages are DS1 – dry deciduous scrub and SS4 – dry grasslands (Champion and Seth 1968). The vegetation was representative of a typical dry deciduous dhok forest (Anogeissus pendula). Apart from dhok, the species commonly found are kadaya (Sterculia urens), salai (Boswellia serrata), raunj (Acacia leucophloea), amaltas (Cassia fistula), Palash (Butea monosperma), tendu (Diospyros melanoxylon), gurjan (Lannea coromandelica), and Jamun (Syzygium cumini). Away from the human-dominated landscape are carnivores including the striped hyena, tiger, leopard, sloth bear (Melursus ursinus), golden jackal, Bengal fox (Vulpes bengalensis), and wild ungulates including sambar, chital, nilgai, chinkara (Gazella gazelle), and wild boar.
Monitoring of carcasses
In this study, we monitored a total of 14 carcasses (domestic = 7, wild = 7; Supplementary Table S1) using camera traps with white flash (Cuddeback C1 type; WI, USA digital camera) between October and March of the winter season of 2020–2022. We generally determined the age and cause of mortality for domestic animals based on reports received from villagers and for wild ungulates based on reports from park authorities in < 24 h after death, and the coordinates of the carcasses were collected using a Garmin GPS (eTrac 20). We categorized mortalities as natural (i.e., old age, disease), predation (killed by tigers or leopards), and accidental (i.e., vehicle collision). The body weight of each carcass was assessed based on its age category, ranging from 40 to 180 kg (Supplementary Table S1). Domestic carcasses (n = 7) were monitored at carcass dumping sites as well as in forests close to the road, while the wild ungulates (n = 7) carcasses were monitored inside the forest (open forest < 30% vegetation covers). We installed a single camera trap per carcass site to record the activity of scavengers at the carcass. We set the camera traps 50–60 cm above the ground and at a distance of approximately 3 m from the carcass. We set camera time according to coordinated universal time (UTC) as the time standard, and time and date stamps were imprinted on each image when the camera was triggered. We set the camera trap to record the maximum amount of activity with an FAP (fast as possible) mode, which implies minimum delays between two consecutive captures. The camera traps were active 24 h/day until the carcasses were completely consumed, that is, when only bones, tissues, and ribs remained (mean = 18.1 days ± 3.4 SE, range 5–51). The livestock carcasses were monitored daily due to the proximity to the human settlements, while the wild ungulate carcasses were monitored via camera trap images to check the decomposition stage of carcasses.
Data analysis
Tigers and striped hyenas are both considered to be competitors with leopards (Karanth et al. 2017; Mandal et al. 2018), so we considered tigers and striped hyenas as independent variables to model the carrion used by leopards. Similarly, golden jackals are subordinate to tigers and striped hyenas and also potential competitors, so we considered tigers and striped hyenas as independent variables to model the carrion used by golden jackals (Figs. 2 and 3). Striped hyenas are also subordinate to tigers and leopards, and golden jackals are a potential competitor to striped hyenas (Figs. 2 and 3; Chourasia et al. 2012), so we modeled the carrion use of striped hyenas against the tigers, leopards, and golden jackals. We considered three aspects of carrion acquisition: presence (proportion of days scavenging), mean feeding bout duration, and total feeding time. We calculated presence as the proportion of days at the carcass each carnivore was recorded, calculating the proportion of days by dividing the number of days an animal was captured at carcasses by the total number of days available for scavenging. We calculated the duration of feeding bouts by subtracting the starting time of visits from the ending time of visits at carcasses, and we calculated mean feeding bout duration as the mean of all feeding bouts at carcasses. The two feeding bouts were considered independent if separated by at least a 5-min time gap. The total time at carcasses was calculated as the sum of all feeding bouts. Species spending a minimum of 1 min at carcasses were considered in our analysis. We also recorded some small mammals (mongoose, civets, honey badgers etc.) and bird species such as cattle egrets, crows, and rufous treepies, at some carcasses during the daytime, except for the red-headed vulture, which was only recorded at one carcass for a single day with a short period of < 30 min. So, we did not consider small mammals and birds in the analyses due to their unreliable detection with camera traps.
We fitted generalized linear models (GLMs) using the “MASS” package to analyze the competitive interaction among carnivores by testing the effects of tigers, leopards, and jackals on the striped hyenas, the effects of tigers and hyenas on leopards, and the effects of hyenas on leopards and jackals’ carrion acquisition. We assigned each of the three aspects of carrion acquisition as our dependent variables for striped hyenas, leopards, and jackals and chose the appropriate distribution based on error residuals. We also standardized predictor variables by centering and scaling them around a mean of 0 with a standard deviation of 1. We examined the multicollinearity among variables using IBM SPSS Statistics (ver. 21.0; SPSS Inc., Chicago, IL., USA), and we included variables with VIF (variance inflation factor) < 5 in the analyses (Shrestha 2020). VIF ranged from 1.02 to 4.78 (Supplementary Table S2); therefore, all variables were retained in the analyses. We tested each model for overdispersion using the “AER” package (Kleiber and Zeileis 2008), which tests the null hypothesis of equi-dispersion in Poisson GLMs against the alternative of overdispersion and under dispersion (a significant p-value indicates the model is either over or under dispersed; Cameron and Trivedi 1990). Because each dataset was determined to be over-dispersed, we found the Poisson distribution to be inappropriate and instead used a negative binomial distribution with a log link function for total feeding time, mean feeding bout, and presence. We fitted separate GLM models (n = 21 total) for each aspect of carrion acquisition by striped hyenas, leopards, and jackals. The list of all possible models was created to examine the competitive interactions among large- and mesocarnivores at carcasses. As an additional measure of model fit, we also calculated adjusted deviance:
which represents the percent deviance explained by each model (Franklin 2010). In each analysis, we considered P ≤ 0.05 to be significant and P ≤ 0.10 to be marginally significant. We performed all analyses in program R v.4.0.2 (R Development Core Team 2021).
Results
We recorded a total of 2332 photos of our target carnivores (tiger, leopard, striped hyena, and golden jackal) at the carcasses (Table 1). Striped hyenas were present in 12 carcasses, followed by jackals (n = 8), tigers (n = 8), and leopards (n = 6), respectively (Supplementary Table S3). Striped hyenas had the highest mean total feeding time at the carcasses, followed by tigers, golden jackals, and leopards, respectively (Table 1). Tigers had the highest mean feeding bout duration, followed by golden jackals, striped hyenas, and leopards, respectively (Table 1).
Effects of tigers on carrion’s acquisition of other carnivores
We found that tigers had adverse effects on the carrion acquisition of other species. Our results showed that the total feeding time of leopards was negatively affected by the mean feeding bout duration and total feeding time of tigers (Table 2). However, the leopard’s presence was negatively affected by the total feeding time of tigers (Table 2). In the absence of tigers, leopards increased their total feeding time by 327% and their mean feeding bout duration by 3.4 times (Table 3). The mean feeding bout duration of tigers also had a negative effect on the presence and total feeding time of golden jackals at carcasses but did not significantly limit the mean feeding bout duration of jackals (Table 4). However, the tiger’s presence significantly affected the presence, total feeding time, and mean feeding bout duration of golden jackals (Table 4). Golden jackals increased the total feeding time by 940% and their mean feeding bout duration by 1.5 times at tiger-free carcasses (Table 3). Our results also showed that the presence of tigers and their total feeding time and mean feeding bout duration on carcasses significantly affected the carrion acquisition of striped hyenas (Fig. 3). The results of the GLM showed that the total feeding time of tigers considerably limited the presence, total feeding time of striped hyenas (Table 5). Also, the mean feeding bout duration and presence of tigers had a negative effect on the presence, total feeding time, and mean feeding bout duration of striped hyenas (Table 5). At carcasses where tigers were absent, the mean total feeding time for scavenging striped hyenas was 165.6 (95% CI = − 43 to 374.8) min, 566% longer than where tigers were present. Also, the mean feeding bout duration of striped hyenas was 8.88 min (95% CI = 3.1 to 14.6) (Table 3 and Supplementary Table S4), an increase of 2.64 times over where tigers were absent.
Effects of leopards on carrion acquisition of striped hyenas
Our analyses showed that leopards had mixed effects on the carrion acquisition of striped hyenas. At carcasses where leopards were present, the mean total feeding time for scavenging striped hyenas was 158 min (95% CI = − 57 to 373.4), 416% longer than where leopards were absent (Supplementary Table S4, Fig. 3). Also, their mean feeding bout duration was 7.3 min (95% CI = 1.6 to 13.1), 1.6 times longer where leopards were present (Supplementary Table S4). But leopards did not have any significant negative effects on the presence, total feeding time, and mean feeding bout duration of striped hyenas (Table 5). This suggests that the presence of leopards did not have any major effects on the carrion acquisition of striped hyenas (Fig. 3), although leopards feeding for long periods did limit the carrion acquisition of striped hyenas.
Effects of golden jackals on carrion acquisition of striped hyenas
Our analyses showed that jackals had mixed effects on the carrion acquisition of striped hyenas. The total feeding time of jackals had a positive effect on the total feeding time and presence of striped hyenas (Table 5). But, the total feeding time of jackals did not significantly limit the mean feeding bout of striped hyenas (Table 5). Also, the presence of jackals did not significantly limit the presence, total feeding time, and mean feeding bout duration of striped hyenas (Table 5). However, the mean feeding duration of jackals had a negative effect on the presence, total feeding time, and mean feeding bout duration of striped hyenas (Table 5). At carcasses where jackals were present, the mean total feeding time for scavenging striped hyenas was 108.1 (95% CI = − 51.9 to 228.2) min, 98% longer than when jackals were absent. Also, the mean feeding bout duration was 4.5 min (95% CI = 0.03 to 9.8) (Supplementary Table S4), 1.3 times shorter than jackal-free carcass (Fig. 3).
Effects of striped hyenas on carrion acquisition of leopards and golden jackals
Our analysis showed that striped hyenas also affected the carrion acquisition of leopards and golden jackals. The carrion acquisition of striped hyenas did not significantly limit the carrion acquisition of leopards (Table 2). In the presence of hyenas, leopards decreased their total feeding time by 43.3%. Our results also showed that striped hyenas affected the carrion acquisition of golden jackals. The presence of striped hyenas also had a negative effect and considerably limited the presence, total feeding time, and mean feeding bout duration of jackals (Table 4). In the presence of hyenas, the total feeding time of jackals decreased by 29.6% and the mean feeding bout duration decreased by 1.07 times.
Discussion
Our study shows that species with specific functional traits, including large body mass (Monterroso et al. 2020) and group living social animals (Sebastián-González et al. 2021), can limit the acquisition of carrion by subordinate solitary scavengers. Many competitive interactions among carnivore species are dictated by the dominance of species with larger body sizes (Chaudhary et al. 2020; Vissia and van Langevelde 2022; Allen et al. 2016), and our results highlighted that tigers are dominant over other carnivore species (leopards, striped hyenas, and jackals) for shared carrion resources in our study system. This dominance was highlighted by tigers reducing the consumption time, and hence, nutrition gained, of all of the other carnivorous scavengers we studied. But tigers were not alone in this characteristic, as we found that leopards and jackals also limited different aspects of carrion acquisition by striped hyenas, and striped hyenas limited the carrion acquisition by leopards and jackals. This study is the first to our knowledge to assess the potential interactions among carnivores on the carrion acquisition at carcasses in a semi-arid landscape of India.
As we expected, our results showed that tigers negatively affected the presence, total feeding time, and mean feeding bout duration of striped hyenas at carcasses, similar to how other apex carnivores limit the total feeding time, mean feeding bouts, and presence of subordinate scavengers (Allen et al. 2015). Previous studies have stated that large carnivores do not necessarily have to eat to full gastric capacity per day or do not need to eat every day, which in turn reduces kill frequencies or drives other ecological processes such as scavenging, kleptoparasitism, and partial carcass consumption (De Cuyper et al. 2019). We observed that tigers exhibit longer feeding bout durations but that their frequency of presence at carcasses was less than the other scavengers. There was a pattern of most of the striped hyena’s feeding occurring when tigers were absent, but the spatio-temporal interactions between tigers and striped hyenas at carcasses or in general are not well known. Previous studies reported that gray wolves and striped hyenas share a common niche (Mukherjee et al. 2021), and Nayak et al. (2015) reported that wolves and striped hyenas in Ranthambore Tiger Reserve shared a carcass without showing any kind of aggression. The interactions of striped hyenas with tigers may affect their feeding habits and population dynamics since fitness and reproductive ability can be influenced by energy intake from carrion (Watson et al. 1992). Similarly, we found tiger’s mean feeding bout duration also affected the jackal’s presence at carcasses. Thus, our results support our prediction that top predators like tigers would affect the carrion acquisition of striped hyenas and jackals, with the large body size of tigers likely playing a major role in the suppression of subordinate hyenas and jackals. Although jackals are social and facultative scavengers that are known to exploit large carnivore kills (Hayward et al. 2017), jackals may limit their scavenging time due to their smaller body size compared to tigers. At carrion, the main effects are likely because of their ability to consume large quantities of food and dominant interactions with competitors due to their larger body size (Allen et al. 2016; Inagaki et al. 2020; Prugh and Sivy 2020).
Tigers and leopards are both considered to be large carnivores (Löe and Röskaft 2004) with a great overlap in their dietary niche across their distribution (Wang and Macdonald 2009; Harihar et al. 2011; Mondal et al. 2011; Ramesh et al. 2012). Large carnivores both provide and distribute carrion among scavengers while also limiting their access through competition (DeVault et al. 2011; Cortez-Avizanda et al. 2012; Moleón et al. 2014; Pereira et al. 2014; Allen et al. 2014). Our results highlight that tigers affected the carrion acquisition of leopards by limiting the total feeding time and mean feeding bout duration at carcasses, similar to how the presence of larger bears (Ursus sp.) reduces the acquisition of carrion of solitary felids (Krofel et al. 2012; Allen et al. 2015). Despite, or possibly because of, their dietary niche overlap, tigers and leopards show spatial and temporal segregation to reduce the interspecific encounters which could lead to intraguild predatory killing (Mondal et al. 2012; Karanth et al. 2017; Vissia and van Langevelde 2022; Rayan and Linkie 2016). A previous report stated that the reintroduction of tigers in Sariska Tiger Reserve affected the leopard population and activity pattern (Mondal et al. 2012), and our results suggest that tigers and leopards also likely exhibit avoidance behavior of each other at carcasses.
Leopards and striped hyenas coexist with tigers across most of the latter’s distribution in the Indian subcontinent (Mandal et al. 2018). Our results suggested that the presence and total feeding time of leopards had a positive effect on the carrion acquisition of striped hyenas. And striped hyenas total feeding time negatively affected the total feeding time of leopards. However, leopards are known for both hunting and scavenging behavior (i.e., seen accessing tiger kills; Prater 1948), and acting as a direct competitor of hyenas (Heptner and Sludskii 1992). Sometimes, carnivores pose a threat to subordinate scavengers and carnivores at their kills via direct predation (Allen et al. 2015; Mandal et al. 2018), but carnivores can also coexist over food resources while showing competitive behavior (Amorós et al. 2020). In one instance, we observed a 15-min-long interaction between a striped hyena and a leopard on a cattle carcass, with neither species showing obvious signs of aggression. Because carrion is an ephemeral food resource that can only be exploited for a finite time, sharing the resource might not be negative for a species, especially when the carcass is large relative to the scavenger’s body size (Sebastián-González et al. 2021). As our results suggested, leopards and hyenas did not show any major negative effects on each other’s carrion acquisition, so the coexistence between leopards and striped hyenas at carcasses may be explained through the cascading effects of tigers. Specifically, leopards and striped hyenas are subordinate to tigers, and to avoid the risk of predation, both species were mainly feeding on tiger-free carcasses, which may result in interactions between leopards and hyenas. This suggests that, despite leopards having larger body sizes than striped hyenas, striped hyenas and leopards can coexist and share resources.
In human-dominated ecosystems that are devoid of major mammalian predators, mesocarnivores can potentially monopolize carcasses (DeVault et al. 2011; Sebastián-González et al. 2021), but especially social foragers that can outnumber other scavengers. We found that jackals’ mean feeding bout duration also affected striped hyenas’ mean feeding bout duration at carcasses. We observed that jackals were consuming carcasses in groups of 2–6 individuals, similar to the sighting recorded by Jhala et al. (2004). Social species tend to search for and guard food collaboratively and use their conspecifics as signs that food is nearby (Dermody et al. 2011), which helps to defend themselves from potential predators (Krause et al. 2002; Allen and Krofel 2017). Furthermore, group foraging often strengthens the dominance hierarchy, allowing social foragers to outcompete other scavenger species (Kendall 2013), as similar studies found that gray wolves were dominant over pumas due to their social structure (Ruth and Murphy 2010; Elbroch and Kusler 2018). This suggests that social foragers like jackals can dominate over solitary striped hyenas at carcasses which may affect the carcass acquisition of striped hyenas. As mesocarnivores are also highly adapted species with species-specific adaptations that include strategy, aggressiveness, and weapons that may influence the outcome of encounter competition contests (Martin and Ghalambor 2014; Allen et al. 2016). Furthermore, we also found that the jackal’s carrion acquisition was also affected by the presence of striped hyenas. At carcasses, interactions between jackals and hyenas frequently occurred, which resulted in competitive interactions with their feeding bouts frequently overlapping. As striped hyenas are slightly larger than jackals and both are generalist species, their diet overlaps greatly (Chourasia et al. 2012). This leads to suppression that can occur both ways, with jackals’ groups limiting the feeding bout of solitary hyenas and larger hyenas, sometimes limiting the presence of jackals.
Our results highlight the importance of functional traits in intraguild interactions at wild and domestic carcasses and the potential effects of this competition on carrion acquisition in human-dominated landscapes. Future studies should be done at a larger scale to test the relationships and interactions between functional traits and the carrion use of large carnivores and mesocarnivores. Large carnivores provide carrion through their kills, attracting scavengers and smaller carnivores (Moleón et al. 2014), but also clearly have competitive interactions with these scavengers. In the semi-arid region of India, the striped hyena is a major scavenger that provides important ecosystem services by removing carrion from the ecosystem (Panda et al. 2022). Previous studies in this area reported the loss of tigers in Ranthambore Tiger Reserve (RTR) due to the poisoning of carcasses by locals, which could affect the other scavengers’ assemblages. We suggest the proper protection and management of wild and domestic carcasses by forest officials, which affect the species assemblage and functions through complex interaction at carcasses (Sebastián-González et al. 2016) and play a major role in the energy transfer between tropic levels (Barton et al. 2013; Barton 2015).
Data availability
The data used in this study are provided in Supplementary information.
References
Akash M, Dheer A, Dloniak SM, Jacobson AP (2021) The faded stripes of Bengal: a historical perspective on the easternmost distribution of the striped hyena. Eur J Wildl Res 67(6):1–12
Allen ML, Elbroch LM, Wilmers CC, Wittmer HU (2014) Trophic facilitation or limitation? Comparative effects of pumas and black bears on the scavenger community. PLoS ONE 9(7):e102257
Allen ML, Elbroch LM, Wilmers CC, Wittmer HU (2015) The comparative effects of large carnivores on the acquisition of carrion by scavengers. Am Nat 185(6):822–833
Allen ML, Wilmers CC, Elbroch LM, Golla JM, Wittmer HU (2016) The importance of motivation, weapons, and foul odors in driving encounter competition in carnivores. Ecology 97(8):1905–1912
Allen ML, Krofel M (2017) Resource holding potential. In: Vonk J, Shackelford T (eds) Encyclopedia of animal cognition and behavior. Springer, Cham
Amorós M, Gil-Sánchez JM, López-Pastor BDLN, Moleón M (2020) Hyaenas and lions: how the largest African carnivores interact at carcasses. Oikos 129(12):1820–1832
Arivazhagan C, Arumugam R, Thiyagesan K (2007) Food habits of leopard (Panthera pardus fusca), dhole (Cuon alpinus) and striped hyena (Hyaena hyaena) in a tropical dry thorn forest of southern India. J Bombay Nat Hist Soc 104:178–187
Barton PS, Cunningham SA, Lindenmayer DB, Manning AD (2013) The role of carrion in maintaining biodiversity and ecological processes in terrestrial ecosystems. Oecologia 171(4):761–772
Barton PS (2015) The role of Carrion in ecosystems. In: Benbow ME, Tomberlin JK, Tarone AM (eds) Carrion ecology, evolution, and their applications, 1st edn. CRS Press, Boca Raton, pp 273–286. https://doi.org/10.1201/b18819
Biswas S, Sankar K (2002) Prey abundance and food habit of tigers (Panthera tigris tigris) in Pench National Park, Madhya Pradesh India. J Zool 256(3):411–420
Califf KJ, Green DS, Wagner AP, Scribner KT, Beatty K, Wagner ME, Holekamp KE (2020) Genetic relatedness and space use in two populations of striped hyenas (Hyaena hyaena). J Mammal 101(2):361–372
Cameron AC, Trivedi PK (1990) Regression-based tests for overdispersion in the Poisson model. J Econometr 46(3):347–364
Champion HG, Seth SK (1968) A revised survey of the forest types of India. Manager of Publications, Delhi
Charnov EL (1976) Optimal foraging, the marginal value theorem. Theor Popul Biol 9(2):129–136
Chaudhary R, Zehra N, Musavi A, Khan JA (2020) Spatio-temporal partitioning and coexistence between leopard (Panthera pardus fusca) and Asiatic lion (Panthera leo persica) in Gir protected area, Gujarat India. Plos One 15(3):e0229045
Chourasia P, Mondal K, Sankar K, Qureshi Q (2012) Food habits of golden jackal (Canis aureus) and striped hyena (Hyaena hyaena) in Sariska Tiger Reserve Western India. World J Zool 7(2):106–112
Cortés-Avizanda A, Jovani R, Carrete M, Donázar JA (2012) Resource unpredictability promotes species diversity and coexistence in an avian scavenger guild: a field experiment. Ecology 93(12):2570–2579
De Cuyper A, Clauss M, Carbone C, Codron D, Cools A, Hesta M, Janssens GP (2019) Predator size and prey size–gut capacity ratios determine kill frequency and carcass production in terrestrial carnivorous mammals. Oikos 128(1):13–22
Dermody BJ, Tanner CJ, Jackson AL (2011) The evolutionary pathway to obligate scavenging in Gyps vultures. PLoS ONE 6(9):e24635
DeVault TL, Olson ZH, Beasley JC, Rhodes OE Jr (2011) Mesopredators dominate competition for carrion in an agricultural landscape. Basic Appl Ecol 12(3):268–274
Elbroch LM, Wittmer HU (2012) Table scraps: inter-trophic food provisioning by pumas. Biol Let 8(5):776–779
Elbroch LM, Kusler A (2018) Are pumas subordinate carnivores, and does it matter? PeerJ 6:e4293
Franklin J (2010) Mapping species distributions: spatial inference and prediction. Cambridge University Press, Cambridge
Harihar A, Pandav B, Goyal SP (2011) Responses of leopard Panthera pardus to the recovery of a tiger Panthera tigris population. J Appl Ecol 48(3):806–814
Hayward MW et al (2017) Factors affecting the prey preferences of jackals (Canidae). Mamm Biol 85(1):70–82
Heptner VH, Sludskii AA (1992) Mammals of the Soviet Union. Vol. II Part 2: Carnivora (Feloidea). Vyssha Shkola, Moscow
Hunter JS et al (2007) Patterns of scavenger arrival at cheetah kills in Serengeti National Park Tanzania. Afr J Ecol 45(3):275–281
Inagaki A, Allen ML, Maruyama T, Yamazaki K, Tochigi K, Naganuma T, Koike S (2020) Vertebrate scavenger guild composition and utilization of carrion in an East Asian temperate forest. Ecol Evol 10(3):1223–1232
Jhala YV, Mukherjee S, Shah N, Chauhan KS, Dave C et al (2004) Monitoring lions. In: Jhala YV (ed) Monitoring of Gir. Technical Report, Wildlife Institute of India, Dehra Dun, India, RR, pp 55–71
Jhala YV et al (eds) (2011) Status of tigers co-predators and prey in India 2010. In: National Tiger Conservation Authority, Government of India, New Delhi, and Wildlife Institute of India, Dehradun, TR, p 302
Karanth KU, Sunquist ME (1995) Prey selection by tiger leopard and dhole in tropical forests. J Anim Ecol 64(4):439–450
Karanth KU, Srivathsa A, Vasudev D, Puri M, Parameshwaran R, Kumar NS (2017) Spatio-temporal interactions facilitate large carnivore sympatry across a resource gradient. Proc R Soc B: Biol Sci 284(1848):20161860
Kendall CJ (2013) Alternative strategies in avian scavengers: how subordinate species foil the despotic distribution. Behav Ecol Sociobiol 67(3):383–93
Kleiber C, Zeileis A (2008) Applied Econometrics with R. Springer, Berlin
Krause J, Ruxton GD, Ruxton G (2002) Living in groups. Oxford University Press, New York City
Krofel M, Kos I, Jerina K (2012) The noble cats and the big bad scavengers: effects of dominant scavengers on solitary predators. Behav Ecol Sociobiol 66(9):1297–1304
Lesmeister DB, Nielsen CK, Schauber EM, Hellgren EC (2015) Spatial and temporal structure of a mesocarnivore guild in midwestern North America. Wildl Monogr 191(1):1–61
Levi T, Wilmers CC (2012) Wolves–coyotes–foxes: a cascade among carnivores. Ecology 93(4):921–929
Löe J, Röskaft E (2004) Large carnivores and human safety: a review. AMBIO: J Human Environ 33(6):283–288
Lyly MS, Villers A, Koivisto E, Helle P, Ollila T, Korpimäki E (2015) Avian top predator and the landscape of fear: responses of mammalian mesopredators to risk imposed by the golden eagle. Ecol Evol 5(2):503–514
Macdonald DW (1979) The flexible social system of the golden jackal Canis aureus. Behav Ecol Sociobiol 5(1):17–38
Mandal D et al (2018) Behavioural observations on interaction of leopard and striped hyena western India. Cat News 67:20–21
Martin PR, Ghalambor CK (2014) When David beats Goliath: the advantage of large size in interspecific aggressive contests declines over evolutionary time. PLoS ONE 9(9):e108741
Mateo-Tomás P, Olea PP, Moleón M, Vicente J, Botella F, Selva N, Viñuela J, Sánchez-Zapata JA (2015) From regional to global patterns in vertebrate scavenger communities subsidized by big game hunting. Divers Distrib 21(8):913–924
Menon V (2014) Indian mammals: a field guide. Hachette India
Miller CS, Hebblewhite M, Petrunenko YK, Seryodkin IV, DeCesare NJ, Goodrich JM, Miquelle DG (2013) Estimating Amur tiger (Panthera tigris altaica) kill rates and potential consumption rates using global positioning system collars. J Mammal 94(4):845–855
Mondal K, Gupta S, Qureshi Q, Sankar K (2011) Prey selection and food habits of leopard (Panthera pardus fusca) in Sariska Tiger Reserve, Rajasthan, India. Mammalia 75(2):201–205
Moleón M, Sánchez-Zapata JA, Selva N, Donázar JA, Owen-Smith N (2014) Inter-specific interactions linking predation and scavenging in terrestrial vertebrate assemblages. Biol Rev 89(4):1042–1054
Mondal K, Gupta S, Bhattacharjee S, Qureshi Q, Sankar K (2012) Response of leopards to re-introduced tigers in Sariska Tiger Reserve, Western India. Int J Biodivers Conserv 4(5):228–236
Monterroso P et al (2020) Ecological traits and the spatial structure of competitive coexistence among carnivores. Ecology 101(8):e03059
Moleón M, Sánchez-Zapata JA, Sebastián-González E, Owen-Smith N (2015) Carcass size shapes the structure and functioning of an African scavenging assemblage. Oikos 124(10):1391–1403
Mukherjee T et al (2021) Indian Grey Wolf and Striped Hyaena sharing from the same bowl: high niche overlap between top predators in a human-dominated landscape. Global Ecology and Conservation 28:e01682
Nayak SP, Shah S, Borah J (2015) Going for the kill: an observation of wolf-hyaena interaction in Kailadevi Wildlife Sanctuary Rajasthan India. Canid Biol Conserv 18(7):27–29. http://www.canids.org/CBC/18/wolf-hyaena_interaction_in_india.pdf. Accessed 20 July 2022
Panda D et al (2022) High striped hyena density suggests coexistence with humans in an agricultural landscape Rajasthan. PloS one 17(5):e0266832
Pereira LM, Owen-Smith N, Moleón M (2014) Facultative predation and scavenging by mammalian carnivores: Seasonal, regional and intra-guild comparisons. Mammal Rev 44(1):44–55
Perrig PL, Donadio E, Middleton AD, Pauli JN (2017) Puma predation subsidizes an obligate scavenger in the high Andes. J Appl Ecol 54(3):846–853
Prater SH (1965) The book of Indian animals. Bombay Natural History Society, Prince of Wales Mus. of Western India, Mumbai, India
Prugh LR, Sivy KJ (2020) Enemies with benefits: integrating positive and negative interactions among terrestrial carnivores. Ecol Lett 23(5):902–918
Pocock RI (1941) Mammalia. The fauna of British India including Ceylon and Burma. Taylor and Francis London 2:1–503
R Development Core Team (2021) A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. http://www.R-project.org/. Accessed 8 June 2022
Ramesh T, Kalle R, Sankar K, Qureshi Q (2012) Dietary partitioning in sympatric large carnivores in a tropical forest of Western Ghats India. Mammal Study 37(4):313–321
Rayan DM, Linkie M (2016) Managing conservation flagship species in competition: tiger leopard and dhole in Malaysia. Biol Cons 204:360–366
Ripple WJ et al (2016) What is a trophic cascade? Trends Ecol Evol 31:842–849. https://doi.org/10.1016/j.tree.2016.08.010
Ruth TK, Murphy K (2009) Competition with other carnivores for prey. In: Hornocker M, Negri S (eds) Cougar: ecology and conservation. University of Chicago Press, Chicago, IL, pp 163–172
Sebastián-González E et al (2016) Nested species-rich networks of scavenging vertebrates support high levels of interspecific competition. Ecology 97(1):95–105
Sebastián-González E et al (2021) Functional traits driving species role in the structure of terrestrial vertebrate scavenger networks. Ecology 102(12):e03519
Selva N, Fortuna MA (2007) The nested structure of a scavenger community. Proc R Biol Soc London 274:1101–1108
Selva N, Jedrzejewska B, Jedrzejewski W, Wajrak A (2003) Scavenging on European bison carcasses in Bialowieza primeval forest (eastern Poland). Ecoscience 10(3):303–311
Shrestha N (2020) Detecting multicollinearity in regression analysis. Am J Appl Math Stat 8(2):39–42
Singh P, Gopalaswamy AM, Karanth KU (2010) Factors influencing densities of striped hyenas (Hyaena hyaena) in arid regions of India. J Mammal 91(5):1152–1159
Singh R, Qureshi Q, Sankar K, Krausman PR, Goyal SP (2013) Use of camera traps to determine dispersal of tigers in semi-arid landscape, western India. J Arid Environ 98:105–108
Singh R, Qureshi Q, Sankar K, Krausman PR, Goyal SP, Nicholson KL (2014) Population density of striped hyenas in relation to habitat in a semi-arid landscape, western India. Acta Theriol 59(4):521–527
Singh R, Krausman PR, Goyal SP, Chauhan NS (2015) Factors contributing to tiger losses in Ranthambhore Tiger Reserve India. Wildlife Soc Bull 39(3):670–673
Tamang KM (1982) The status of the tiger (Panthera tigris) and its impact on principal prey populations in Royal Chitwan National Park Nepal. MI State University, Lansing, MI
Vissia S, Van Langevelde F (2022) The effect of body size on co-occurrence patterns within an African carnivore guild. Wildl Biol 2022(2):e01004
Wang SW, Macdonald DW (2009) Feeding habits and niche partitioning in a predator guild composed of tigers leopards and dholes in a temperate ecosystem in central Bhutan. J Zool 277(4):275–283
Watson J, Rae SR, Stillman R (1992) Nesting density and breeding success of golden eagles in relation to food supply in Scotland. J Anim Ecol 61(3):543–550
Wilmers CC et al (2003) Trophic facilitation by introduced top predators: grey wolf subsidies to scavengers in Yellowstone National Park. J Anim Ecol 72:909–916
Acknowledgements
We are grateful to the Director, Amity Institute of Forestry and Wildlife, Amity University, Noida, for their encouragement. We are thankful to Tanuj Suryan and our field assistant Vishnu Gurjar for contributing during fieldwork and data collection.
Funding
Dr. Randeep Singh received the funds and was supported by SERB, DST, Government of India: Grant No. – EMR/2017/003580. The funder has no role in design and data collection.
Author information
Authors and Affiliations
Contributions
Conceptualization of the idea: Debashish Panda, Randeep Singh.
Data curation: Debashish Panda, Subham Mohanty, Randeep Singh.
Formal analysis: Debashish Panda, Subham Mohanty, Arjun Dheer, Randeep Singh.
Funding acquisition: Randeep Singh, Puneet Pandey, Hang Lee.
Investigation: Randeep Singh, Puneet Pandey.
Methodology: Debashish Panda, Randeep Singh.
Resources: Debashish Panda, Randeep Singh, Puneet Pandey.
Software: Debashish Panda.
Supervision: Randeep Singh, Puneet Pandey.
Validation: Randeep Singh.
Visualization: Debashish Panda, Randeep Singh.
Writing – original draft: Debashish Panda.
Writing – review and editing: Debashish Panda, Randeep Singh, Puneet Pandey, Maximilian L. Allen, Arjun Dheer, Hang Lee, Ajay Sharma.
Corresponding author
Ethics declarations
Ethics approval
This study was conducted with permission from Rajasthan Forest Department (letter no – F 19(11) permission/cwlw/2017/1678). We followed all guidelines for animal care and scientific research ethics.
Competing interests
The authors declare no competing interests.
Additional information
Communicated by: Karol Zub
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Panda, D., Mohanty, S., Allen, M.L. et al. Competitive interactions with dominant carnivores affect carrion acquisition of striped hyena in a semi-arid landscape of Rajasthan, India. Mamm Res 68, 129–141 (2023). https://doi.org/10.1007/s13364-022-00663-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13364-022-00663-1