Abstract
Objective
To compare the efficacy and safety of oral iron chelators (Deferiprone and Deferasirox) when used singly and in combination in multi-transfused children with thalassemia.
Design
Prospective comparative study.
Setting
Thalassemia Center of a medical college affiliated hospital
Participants and Intervention
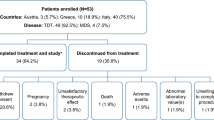
49 multi-transfused children with thalassemia with a mean (SD) age 11.6 (6.21) y received daily chelation therapy with either deferiprone alone (75 mg/kg/day in 3 divided doses), deferasirox alone (30 mg/kg/day single dose) or their daily combination (same dose as monotherapy) for 12 months.
Outcome measures
Serum ferritin levels at the start of study, after 6 months and after 12 months. MRI T2* of liver and heart initially and after 6 months of follow up. 24-hour urinary iron excretion values at the outset and after 12 months of chelation therapy. At every visit for blood transfusion, all patients were clinically assessed for any adverse effects; liver and renal functions were monitored 6-monthly.
Results
After 12 months of respective chelation therapy, serum ferritin values decreased from a mean of 3140.5 ng/mL to 2910.0 ng/mL in deferiprone alone group, 3859.2 ng/mL to 3417.4 ng/mL in deferasirox alone group and from 3696.5 ng/mL to 2572.1 ng/ mL in the combination group. The combination therapy was more efficacious in causing fall in serum ferritin levels compared to deferiprone and deferasirox monotherapy (P=0.035 and 0.040, respectively). Results of MRI T2* were equivocal. Combined drug usage produced maximum negative iron balance in the body by maximally increasing the iron excretion in urine from 61.1 μmol/ day to 343.3 μmol/day (P=0.002). No significant adverse reactions were noticed in either the monotherapy or the combination group.
Conclusion
Oral combination therapy of deferiprone and deferasirox appears to be an efficacious and safe modality to reduce serum ferritin in multi-transfused children with thalassemia.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Mourad FH, Hoffbrand AV, Sheikh-Taha M, Koussa S, Khoriaty AI, Taher A. Comparison between desferrioxamine and combined therapy with desferrioxamine and deferiprone in iron overloaded thalassaemia patients. Br J Haematol. 2003;121:187–9.
Lal A, Porter J, Sweeters N, Ng V, Evans P, Neumayr L, et al. Combined chelation therapy with deferasirox and deferoxamine in thalassemia. Blood Cells Mol Dis. 2013;50:99–104.
Olivieri NF, Koren G, Hermann C, Bentur Y, Chung D, Klein J, et al. Comparison of oral iron chelator L1 and desferrioxamine in iron-loaded patients. Lancet. 1990;336:1275–9.
Balocco M, Carrara P, Pinto V, Forni GL. Daily alternating deferasirox and deferiprone therapy for “hard-to-chelate” beta-thalassemia major patients. Am J Hematol. 2010;85:460–1.
Voskaridou E, Christoulas D, Terpos E. Successful chelation therapy with the combination of deferasirox and deferiprone in a patient with thalassaemia major and persisting severe iron overload after single-agent chelation therapies. Br J Haematol. 2011;154:654–6.
Farmaki K, Tzoumari I, Pappa C. Oral chelators in transfusion-dependent thalassemia major patients may prevent or reverse iron overload complications. Blood Cells Mol Dis. 2011;47:33–40.
Di Tucci AA, Matta G, Deplano S, Gabbas A, Depau C, Derudas D, et al. Myocardial iron overload assessment by T2* magnetic resonance imaging in adult transfusion dependent patients with acquired anemias. Haematologica. 2008;93:1385–8.
Pennell DJ. T2* magnetic resonance and myocardial iron in thalassemia. Ann N Y Acad Sci. 2005;1054:373–8.
Angelucci E, Giovagnoni A, Valeri G, Paci E, Ripalti M, Muretto P, et al. Limitations of magnetic resonance imaging in measurement of hepatic iron. Blood. 1997;90:4736–42.
Salo S, Alanen A, Leino R, Bondestam S, Komu M. The effect of haemosiderosis and blood transfusions on the T2 relaxation time and 1/T2 relaxation rate of liver tissue. Br J Radiol. 2002;75:24–7.
Chirnomas SD, Geukes-Foppen M, Barry K, Braunstein J, Kalish LA, Neufeld EJ, et al. Practical implications of liver and heart iron load assessment by T2*-MRI in children and adults with transfusion-dependent anemias. Am J Hematol. 2008;83:781–3.
Noetzli LJ, Carson SM, Nord AS, Coates TD, Wood JC. Longitudinal analysis of heart and liver iron in thalassemia major. Blood. 2008;112:2973–8.
Pepe A, Lombardi M, Positano V, Cracolici E, Capra M, Malizia R, et al. Evaluation of efficacy of oral deferiprone in beta-thalassemia major by multislice multiecho T2*. Eur J Haematol. 2006;76:183–92.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gomber, S., Jain, P., Sharma, S. et al. Comparative efficacy and safety of oral iron chelators and their novel combination in children with thalassemia. Indian Pediatr 53, 207–210 (2016). https://doi.org/10.1007/s13312-016-0821-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13312-016-0821-4