Abstract
The extension of chest wall resection for the treatment of primary and secondary tumours is still widely debated. The reconstructive strategy after extensive surgery is challenging as well as chest wall demolition itself. Reconstructive surgery aims to avoid respiratory failure and to guarantee intra-thoracic organs protection. The purpose of this review is to analyse the literature on this issue focusing on the planning strategy for chest wall reconstruction. This is a narrative review, reporting data from the most interesting studies on chest wall demolition and reconstruction. Representative surgical series on chest wall thoracic surgery were selected and described. We focused to identify the best reconstructive strategies analyzing employed materials, techniques of reconstruction, morbidity and mortality. Nowadays the new “bio-mimetic” materials in “rigid” and “non-rigid” chest wall systems reconstructive represent new horizons for the treatment of challenging thoracic diseases. Further prospective studies are warranted to identify new materials enhancing thoracic function after major thoracic excisions.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The chest wall (CW) is a complex structure, including a bony skeletal part (the rib cage) and a muscular as well as a fascial parts. The CW has important functions including the protection of thoracic (i.e., heart, lungs) and abdominal (i.e., liver and spleen) organs, stabilizing the actions of the shoulder and arm, and promoting respiratory movement. CW can be affected by several types of tumours. CW malignancies might be classified into primary (tumours that originate in the chest wall) and secondary tumours. Those latter might spread (metastasize) to the chest wall directly from surrounding organs or due to haematogenous dissemination [1]. In the field of surgical oncology, resection of the CW represents a challenging procedure.
It is well-known that surgical treatment of CW malignancies might require wide radical resection with the aim to achieve free margins. In most cases, those wide resections are associated with complex reconstructions through the adoption of prosthetic materials (e.g., synthetic or biological/cadaveric) and/or myocutaneous or muscular flaps [2,3,4,5]. Furthermore, several authors reported that the extension of CW resection may impact negatively respiratory mechanics [6,7,8,9]. Planning the correct reconstruction strategies is necessary as well as planning the resection phase to preserve lung function [10,11,12,13,14,15,16,17,18,19,20].
According to the principles of “biomimesis”, the ideal prosthesis should respect and reconstruct the original anatomy and the organ function. The major difficulty is to preserve a functional and rigid chest wall structure, which is a protective system able to create a negative pressure to breathe. A perfect reconstruction is rigid and flexible at the same time. In addition, materials used for CW reconstruction should undergo these rules: malleability, radiolucency and inertness. Specifically, it should guarantee the protection of the mediastinum and lungs, with adequate biocompatibility anatomical structures (without allergies or inflammation/reject from foreign material) and it should allow physiological pulmonary expansion. According to these principles, a variety of prosthetic materials (combined or not with a multitude of reconstructive approaches) have been proposed in literature, with different postoperative outcomes. In this narrative review, we performed a critic analysis of the literature to assess the pros and cons of various reconstructive strategies, especially after extensive and challenging CW resection. Data on the most pertinent studies on this issue were collected and discussed [1,2,3,4,5,6,7,8,9,10,11,12,13,14,15,16,17,18,19,20,21,22,23,24,25,26,27,28,29,30,31,32,33,34,35,36,37,38,39,40,41,42,43,44,45,46,47,48,49,50,51,52,53,54,55,56,57,58,59,60,61,62,63,64,65,66,67,68,69,70,71,72,73,74,75]. Hence, we describe surgical techniques focusing on various materials that might be used during the reconstructive phases. Moreover, we focused our discussion on postoperative outcomes, including recovery, morbidity, and anatomical/functional modifications.
Surgical technique
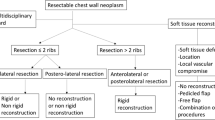
CW resection is required to treat patients with a primary or secondary tumour invading the CW. Other indications for resection range from non-oncological conditions (being radionecrosis the most common disease) to a variety of congenital malformations. A specific approach to CW resection and subsequent reconstruction begins with the assessment of the size and location of the defects, the depth of tumoral invasion, the quality of adjacent tissues, the prognosis of disease (and related expected long-term and disease-free survival) and the need for curative or palliative surgery. In this scenario, the most used reconstructive technique is a combination of mesh and muscular flaps. The rigid reconstruction (with or without myocutaneous flap) is required according to the surgeons’ experiences. It can be employed for repairing large CW defects usually more than four ribs, and more frequently in anterior and sternal defects, with the aim to avoid flail chest and to decrease the post-operative non-invasive ventilatory support [7, 21,22,23,24]. Given the presence of trunk muscles and scapula, posterior and apical defects as well as those near the spine can be reconstructed without the application of rigid prosthesis. In selected cases, limited anterior defects can be covered with pectoralis muscle, with or without soft reconstruction. After the first reports on metal prosthetic application [25, 26], many new materials have been developed: biologic, alloplastic and synthetic [43,44,45,46,47,48, 53]. Methyl-methacrylate (MM), polyglactin (Vycril), polypropylene, polytetrafluoroethylene (PTFE), titanium represent the most common synthetic materials employed in clinical practice.
Materials: soft vs. rigid
The meshes and patches are easy to deploy, handle and anchor to adjacent ribs around the defect. They can be permeable or impermeable to fluid and applied using a single or double layer to guarantee further resistance. The Vicryl and Polypropylene are the most common flexible materials used in those patients with a high risk of infection because of the extreme permeability to fluids that avoids the occurrence of seroma. Contrariwise, although its worldwide use, PTFE is a waterproof non-absorbable material associated with a higher risk of infection (ranging from 4.3% and 9% in the literature) [48, 49, 60].
Concerning rigid reconstruction, Methyl-methacrylate (MM) is one of the most common materials utilized especially after extended CW resections, since it allows increased CW stability as well as a mechanical and protective function. Also, it can be sandwiched between two layers of other meshes (i.e. polypropylene, Surgimesh-PET, etc.) [8, 27, 28]. This “in situ” modeled technique has the advantage of rigid reconstruction that avoids paradoxical motion. On the other hand, its extreme rigidity is associated with higher rates of seromas, hematomas, infections, fracture, and risk of prosthesis removal (up to 5% of patients) (8; 23).
We presented in 2011 our experience about Methyl-methacrylate “rib-like” technique [68] modelling the two cyanoacrylate resins on a chest aluminium cast obtained from a plaster mould reproducing costal arches and the sternum.
Materials: osteosynthetic materials
Ideal characteristics for CW rigid materials include high-tensile strength and resistance, good biocompatibility and bio-incorporation, as well as low infection rates. In this setting, titanium is a solid highly biocompatible metal, inert, resistant to infections and fully adaptable to the shape of CW. For these reasons, it is considered a better metallic implant system in surgery compared to ceramic [72] and stainless materials and can be used with excellent results both in oncological and non-oncological diseases (i.e., traumatic or malformity conditions) [63, 65, 67]. Specifically, there are two different systems for titanium implantation. The first refers to the application of titanium bar on the titanium clips screwed in ribs or to the titanium bridge bars directly screwed in ribs. In this context, Berthet et al. described the association of this system with PTFE mesh and reported a significant failure risk (44%) [29, 30]. Similarly, Fabre et al. experienced implant failure in 44% of cases (83.3% for broken mesh and 16.7% for displaced prosthetic replacement); anterior defects and more than 3 implants are associated with significant surgical failure (p = 0.02) [73].
Materials: bioscaffolds
Nowadays, a great interest is focused on biological tissues such as those derived from the cadaveric human dermis, from porcine small intestine submucosa, from the porcine dermis and bovine pericardium matrix [31,32,33,34,35,36,37,38, 55]. These bioprosthetic meshes are composed of decellularised scaffolds and matrices that allow fibrous tissue ingrowth. To avoid rejection or digestion [32], nearby vascularized tissues or mhe yo-cutaneous flaps are necessary. Other advantages of these scaffolds are the low risk of infection, and the possibility to employ in case of infection after rigid mesh reconstruction [33]. However, they should not be used for the reconstruction of large sternal resections (including bilateral costal cartilages and ribs) because there is an increased risk of anterior paradoxical motion and respiratory compromise [55]. Similarly to the titanium system, another limitation is the high cost. In selected cases, the cadaveric cryopreserved sternal allograft (combined with titanium bars) for sterno-chondral replacement showed encouraging results with a very low rate of respiratory impairment or flail chest [36, 66]. However, the possible further applications of these new biomaterials should be better addressed in future prospective studies.
Materials: flaps and tissue engineering
As reported by the majority of studies, muscle, myo-cutaneous and omentum flaps play a significant role in the reconstructive setting after CW demolition. The vascularized flaps optimize the intake of the prosthesis, reduce the risk of wound infections, obliterate the dead space (avoiding the occurrence of seromas), cover and separate the synthetic materials reducing the risk of prosthesis removal [39] The choice of flap depends mainly on the type of resection and on the defect’s position. The greater omentum plays a crucial role in reconstruction especially in the contaminated field because it can be transposed in any CW location, supporting leukocyte and blood cells supply. On the other hand, its use may be associated with significant intra-abdominal morbidity such as abdominal and trans-diaphragmatic hernia. Furthermore, to prevent postoperative complications, attention should be paid to preserving the arterial blood supply to avoid excessive tension, which may be related to necrosis, retraction of the flap or instability of the overlying skin graft [18].
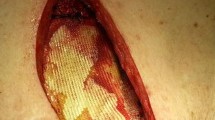
A new engineered method described to fill up the dead space and to improve the intake of the prosthesis is the implantation of stem adipocyte cells on the inner surface of the mesh. This method implies the creation of a new tissue that improves the vascularisation of in vivo implants. Other interesting results were reported by applying hybrid scaffolds composed of soft or bone/cartilagineous tissues derived from engineering amniotic mesenchymal stem cells [34, 35, 39, 43]. These specific materials lead to an enhanced recruitment of hematopoietic cells, supporting the regeneration of the defect site. Furthermore, these hybrid scaffolds are stable, flexible and easy-to-use and can be molded to the shape of the defect with good results. We deem that these interesting features will be employed to develop future clinical applications (Fig. 1).
Postoperative outcomes
CW is a complex and challenging procedure. The risk of developing morbidity and mortality is not neglectable after complex CW resection and reconstruction. In the present chapter, we discussed the risk prevalence of surgery-related complications and their impact on patients’ recovery. Additionally, we discussed how CW resection/reconstruction affect anatomical and functional features of the CW.
Outcome: overall complications and risk of prosthetic removal
Despite the advances in surgical techniques and prosthetic materials, general complications after CW resection/reconstruction are common, occurring in 24% to 46% of patients (Table 1). They are related to major surgery such as haemorrhages, atrial fibrillation, deep vein thrombosis, renal failure and, respiratory complications depending on either a poor pulmonary toilet or paradoxical respiratory movement (flail chest). Specifically, as already described in the literature, main lung complications are respiratory failure, pneumonia and acute respiratory distress syndrome (ARDS) (46; 3) with related long hospital and ICU stay as well as increased mortality rate (ranging from 0% to 15.3% of patients, Table 1). Although data on mortality are still debated, generally it has been reported a worse outcome in the case of lung resection associated with CW surgery. In this setting, Weyant et al. [23] reported a mortality rate of 44% in patients undergoing CW resection and combined pneumonectomy. Similar results were reported by Doddoli et al. [65] in patients undergoing lung and CW surgery (mortality of 5.7% for lobectomy, 33.3% for bilobectomy and 12.7% for pneumonectomy). On the contrary, a multicentre retrospective study on CW resection [54] evidenced an acceptable mortality rate after pneumonectomy (2.9%). According to these interesting data, it has been suggested that a careful knowledge of comorbidity and accurate cardio-respiratory assessment can help to identify patients with a higher risk for unfavourable outcomes. In addition, as already reported by other authors, another critical factor is the experience of the institution in dealing with such complex surgery [20].
In the literature, other common morbidities are those related to local complications (from 4 to 20% of cases) such as wound infection, seroma, flap hematoma or necrosis, wound erosion, and prosthetic dislocation, erosion or infection [48, 49, 56, 60]. General management implies conservative treatment (such as antibiotics, Vacuum Assisted Closure Therapy and positioning of silicone tube for suction) as well as surgical procedures (debridement of necrotic/infected tissue, prosthetic removal). Surgical treatment remains the main option in case of prosthetic dislocation/rupture/erosion. These complications range from 2 to 44% in different series and are more often are associated with the use of MM and PTFE (Table 1) [8, 9, 14, 15, 27, 46, 51, 52, 55, 72, 73]. Numerous studies tried to identify possible predictors of prolonged hospital stays and morbidities. Although it is hard to analyse and stratify the risk for such a heterogeneous population (different histologies and techniques for CW resection/reconstructions), significant predictors of local complications are male gender, large tumour size, associated lung resection, large resection area, use of prosthesis, R1/R2 resection and secondary malignant tumours [7, 14, 54, 64].
Outcome: lung and spine anatomical/functional modifications
After CW resection/reconstruction, data on lung function compromise are still debated in the literature. Some authors [7] found no significant difference in pre/post-operative force expiratory volume (FEV1) values among patients undergoing CW reconstruction with or without a synthetic prosthesis. Similar results were reported by other authors [8] that treated patients with (or without) a non-rigid prosthesis [48, 50], also in terms of postop ventilation need support. Otherwise, some authors [63] evidenced up to 11% of partial paradoxical movement with non-rigid reconstruction, although it did not impair significantly respiratory function.
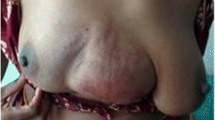
In the literature, very few studies reported data on anatomical change after CW surgery. In our experience on extended CW demolitions/reconstructions, we observed modification in the shape and in the external appearance of the reconstructed hemithorax in all patients, a worsening of left-convex scoliosis (50% of cases) and a progressive approximation of the prosthesis to the mediastinum (25% of patients) [28, 69, 70]. Concerning the quality of life, no patients experienced chronic chest pain, upper girdle dysfunction, shortness of breath or digestive disorders, suggesting a specific body adaptation after CW reconstruction.
Conclusion
This is a narrative review evaluating techniques for CW demolition and reconstruction. CW surgery should be carefully planned considering histology, previous treatment (chemo/radiotherapy), size of resection, experience in the application of specific prosthetic materials and expertise in managing myo-cutaneous/muscolar flaps. A better local control results in improved overall survival in cancer patients. Optimal oncological and functional outcomes may be achieved through a combination of multimodal therapies and planned surgical reconstructive strategies, especially in experienced centres. The discovery of new “bio-mimetic” materials and more appealing techniques has allowed to treat larger tumours in more challenging patients who could not be treated at all in past years. The major extended CW resections (including combined multi-organ excision) should be performed only in institutions with a dedicated multi-disciplinary team with expertise in managing general and specific postoperative complications through the use of conservative (e.g., advanced medications non-invasive ventilation) and more aggressive approaches (e.g., re-operation for prosthesis removal, prosthetic re-implant, re-harvesting/re-implant of myocutaneous flaps). The thoracic prosthetic reconstruction should combine flexibility, protection and bio-compatibility, with high bio-compatibility and low risk of infections and failure [36, 75] Actually, bio-scaffolds represent the future step for auto-regeneration, however, the “classical” (rigid or soft) prosthetic reconstruction is still the mainstay in this kind of surgery. Given the high heterogeneity in terms of patients’ features and surgical techniques, we recommend the need for further comparative and multicenter studies to better understand the outcome differences between”past” and “new” materials and their application.
References
Hasse J (1991) Surgery for primary, invasive and metastatic malignancy of chest wall. Eur J Cardiothorac Surg 5:346–351
Incarbone M, Pastorino U (2001) Surgical treatment of chest wall tumors. World J Surg 25(2):218–230
Mansour KA, Thourani VH, Losken A, Reeves JG, Miller JI Jr, Carlson GW, Jones GE (2002) Chest wall resections and reconstruction: a 25-year experience. Ann Thorac Surg 73(6):1720–1725
Arnold PG, Pairolero PC (1996) Chest-wall reconstruction: an account of 500 consecutive patients. Plast Reconstr Surg 98:804–810
Chapelier AR, Missana M, Couturaud B et al (2004) Sternal resection and reconstruction for primary malignant tumors. Ann thorac Surg 77:1001–1007
Chapelier A, Fadel E, Macchiarini P, Lenot B, Le Roy LF, Cerrina J, Dartevelle P (2000) Factors affecting long-term survival after en-bloc resection of lung cancer invading the chest wall. Eur J Cardiothorac Surg 18(5):513–518
Leuzzi G, Nachira D, Cesario A, Novellis P, PetraccaCiavarella L, Lococo F, Facciolo F, Granone P, Margaritora S (2015) Chest wall tumors and prosthetic reconstruction: a comparative analysis on functional outcome. Thorac Cancer 6(3):247–254
Lardinois D, Muller M, Furrer M, Banic A, Gugger M, Krueger T, Ris HB (2000) Functional assessment of chest wall integrity after methylmethacrylate reconstruction. Ann Thorac Surg 69(3):919–923
Daigeler A, Druecke D, Hakimi M, Duchna HW, Goertz O, Homann HH, Lehnhardt M, Steinau HU (2009) Reconstruction of the thoracic wall-long-term follow-up including pulmonary function tests. Langenbecks Arch Surg 394(4):705–715
Coonar AS, Wihlm JM, Wells FC, Qureshi N (2011) Intermediate outcome and dynamic computerised tomography after chest wall reconstruction with the STRATOS titanium rib bridge system: video demonstration of preserved bucket-handle rib motion. Interact Cardiovasc Thorac Surg 12(1):80–81
Coonar AS, Qureshi N, Smith I, Wells FC, Reisberg E, Wihlm JM (2009) A novel titanium rib bridge system for chest wall reconstruction. Ann Thorac Surg 87(5):e46–e48
Hamad AM, Marulli G, Bulf R, Rea F (2009) Titanium plates support for chest wall reconstruction with Gore-Tex dual mesh after sternochondral resection. Eur J Cardiothorac Surg 36(4):779–780
Wiegmann B, Zardo P, Dickgreber N, Länger F, Fegbeutel C, Haverich A, Fisher S (2010) Biological materials in chest wall reconstruction: initial experience with the Peri-Guard repair patch. Eur J Cardiothorac Surg 37(3):602–605
Novoa N, Benito P, Jiménez MF, de Juan A, Luis Aranda J, Varela G (2005) Reconstruction of chest wall defects after resection of large neoplasms: ten-year experience. Interact Cardiovasc Thorac Surg 4(3):250–255
Chang RR, Mehrara BJ, Hu QY, Disa JJ, Cordeiro PG (2004) Reconstruction of complex oncologic chest wall defects: a 10-year experience. Ann Plast Surg 52(5):471–479 (discussion 479)
Burkhart HM, Allen MS, Nichols FC 3rd, Deschamps C, Miller DL, Trastek VF, Pairolero PC (2002) Results of en bloc resection for bronchogenic carcinoma with chest wall invasion. J Thorac Cardiovasc Surg 123(4):670–675
Watanabe A, Watanabe T, Obama T, Ohsawa H, Mawatari T, Ichimiya Y, Takahashi N, Abe T (2003) New material for reconstruction of the anterior chest wall, including the sternum. J Thorac Cardiovasc Surg 126(4):1212–1214
Skoracki RJ (2006) Chang DW reconstruction of the chest wall and thorax. J Surg Oncol 94(6):455–465
Akan M, EkerUluçay G, Kargi B, Yildirim S, Aköz T (2006) Combined reconstruction of complex defects of the chest wall. Scand J Plast Reconstr Surg Hand Surg 40(2):93–100
Pastorino U, Duranti L, Scanagatta P, Leo F, Piccioni F, Collini P, Gronchi A (2014) Thoracopleuropneumonectomy with riblike reconstruction for recurrent thoracic sarcomas. Ann Surg Oncol 21:1610
Kroll SS, Walsh G, Ryan B, King RC (1993) Risks and benefits of using Marlex mesh in chest wall reconstruction. Ann Plast Surg 31(4):303–306
Tanaka H, Yukioka T, Yamaguti Y, Shimizu S, Goto H, Matsuda H, Shimazaki S (2002) Surgical stabilization of internal pneumatic stabilization? A prospective randomized study of management of severe flail chest patients. J Trauma 52(4):727–732
Weyant MJ, Bains MS, Venkatraman E, Downey RJ, Park BJ, Flores RM, Rizk N, Rusch VW (2006) Results of chest wall resection and reconstruction with and without rigid prosthesis. Ann Thorac Surg 81(1):279–285
Ferraro P, Cugno S, Liberman M, Danino MA, Harris PG (2010) Principles of chest wall resection and reconstruction. Thorac Surg Clin 20(4):465–473
Parham DW (1899) Thoracic resections for tumors growing from the bony chest wall. Trans South Surg Assoc 2:223
Graeber GM (1999) Chest wall resection and reconstruction. Semin Thorac Cardiovasc Surg 11(3):251–263
Girotti P, Leo F, Bravi F, Tavecchio L, Spano A, Cortinovis U, Nava M, Pastorino U (2011) The, “rib-like” technique for surgical treatment of sternal tumors: lessons learned from 101 consecutive cases. Ann Thorac Surg 92(4):1208–1216
Duranti L, Gronchi A, Stacchiotti S, Fiore M, Casali PG, Collini P, Pelosi G, Galeone C, Pastorino U (2013) Localised thoracic sarcomas: Outcome improvement over time at a single institution. Eur J Cancer 49(12):2689–2697
Berthet JP, Wihlm JM, Canaud L, Joyeux F, Cosma C, Hireche K, Alric P, Marty-Ané CH (2012) The combination of polytetrafluoroethylene mesh and titanium rib implants: an innovative process for reconstructing large full thickness chest wall defects. Eur J Cardiothorac Surg 42(3):444–453
Berthet JP, Gomez Caro A, Solovei L, Gilbert M, Bommart S, Gaudard P, Canaud L, Alric P, Marty-Ané CH (2015) Titanium implant failure after chest wall osteosynthesis. Ann Thorac Surg 99(6):1945–1952
Mirzabeigi MN, Moore JH Jr, Tuma GA (2011) The use of Permacol® for chest wall reconstruction in a case of desmoid tumour resection. J Plast Reconstr Aesthet Surg 64(3):406–408
Diaz JJ Jr, Conquest AM, Ferzoco SJ et al (2009) Multi-institutional experience using human acellular dermal matrix for ventral hernia repair in a compromised surgical field. Arch Surg 144(3):209–215
Butler CE, Langstein HN, Kronowitz SJ (2005) Pelvic, abdominal, and chest wall reconstruction with Allo-Derm in patients at increased risk for mesh-related complications. Plast Reconstr Surg 116(5):1263–1275
Gimble JM (2003) Adipose tissue-derived therapeutics. Expert Opin Biol Ther 5:705–713
Buschmann J, Yamada Y, Schulz-Schönhagen K, Hess SC, Stark WJ, Opelz C (2019) Gabriella Meier Bürgisser, Walter Weder, Wolfgang Jungraithmayr hybrid nanocomposite as a chest wall graft with improved integration by adipose-derived stem cells. Sci Rep 9:10910
McCormack P, Bains MS, Beattie EJ Jr, Martini N (1981) New trends in skeletal reconstruction after resection of chest wall tumors. Ann Thorac Surg 31(1):45–52
Fabre D, El Batti S, Singhal S et al (2012) A paradigm shift for sternal reconstruction using a novel titanium rib bridge system following oncological resections. Eur J Cardiothorac Surg 42(6):965–970
Deschamps C, Tirnaksiz BM, Darbandi R, Trastek VF, Allen MS, Miller DL, Arnold PG, Pairolero PC (1999) Early and long-term results of prosthetic chest wall reconstruction. J Thorac Cardiovasc Surg 117(3):588–591
Losken A, Thourani VH, Carlson GW, Jones GE, Culbertson JH, Miller JI, Mansour KA (2004) A reconstructive algorithm for plastic surgery following extensive chest wall resection. Br J Plast Surg 57(4):295–302
Rathinam S, Venkateswaran R, Rajesh PB, Collins FJ (2004) Reconstruction of the chest wall and the diaphragm using the inverted Y Marlex methylmethacrylate sandwich flap. Eur J Cardiothorac Surg 26(1):197–201
Nagayasu T, Yamasaki N, Tagawa T, Tsuchiya T, Miyazaki T, Nanashima A, Obatake M, Yano H (2010) Long-term results of chest wall reconstruction with DualMesh. Interact Cardiovasc Thorac Surg 11(5):581–584
Kuhn JA, Wagman LD, Lorant JA, Grannis FW, Dunst M, Dougherty WR, Jacobs DI (1994) Radical forequarter amputation with hemithoracectomy and free extended forearm flap: technical and physiologic considerations. Ann Surg Oncol 1:353–359
Steigman SA, Ahmed A, Shanti RM, Tuan RS, Valim C, Fauza DO (2009) Sternal repair with bone grafts engineered from amniotic mesenchymal stem cells. J Pediatr Surg 44(6):1120–1126 (discussion 1126)
Merritt RE (2017) Chest wall reconstruction without prosthetic material. Thorac Surg Clin 7(2):165–169. https://doi.org/10.1016/j.thorsurg.2017.01.010. (Epub 2017 Mar 1. PMID: 28363371 Review)
Marulli G, Duranti L, Cardillo G, Luzzi L, Carbone L, Gotti G, Perissinotto E, Rea F, Pastorino U (2014) Primary chest wall condrosarcomas: result of surgical resection and analysis of prognostic factors. Eur J Cardiothorac Surg 45(6):e194-201
Graham J, Usher FC, Perry JL, Barkley HT (1960) Marlex mesh as a prosthesis in the repair of thoracic wall defects. Ann Surg 151:469–479
McCormack PM (1989) Use of prosthetic materials in chest-wall reconstruction. Surg Clin North Am 69:965–976
McKenna RJ Jr, Mountain CF, McMurtrey MJ, Larson D, Stiles QR (1988) Current techniques for chest wall reconstruction: expanded possibilities for treatment. Ann Thorac Surg 46(5):508–512
Eschapasse H, Gaillard J, Henry F, Fournial G, Berthoumieu F, Desrez X (1981) Repair of large chest wall defects: experience with 23 patients. Ann Thorac Surg 32(4):329–336
Yang H, Tantai J, Zhao H (2015) Clinical experience with titanium mesh in reconstruction of massive chest wall defects following oncological resection. J Thorac Dis 7(7):1227–1234
Arnold PG, Pairolero PC (1984) Chest wall reconstruction. Experience with 100 consecutive patients. Ann Surg 199(6):725–732
Pairolero PC, Arnold PG (1986) Thoracic wall defects: surgical management of 205 consecutive patients. Mayo Clin Proc 61(7):557–563
Lampl LH, Loeprecht H (1988) Chest wall resection—alloplastic replacement. Thorac Cardiovasc Surg 36(3):157–158
Cardillo G, Spaggiari L, Galetta D, Carleo F, Carbone L, Morrone A, Ricci A, Facciolo F, Martelli M (2013) Pneumonectomy with en bloc chest wall resection: is it worthwhile? Report on 34 patients from two institutions. Interact Cardiovasc Thorac Surg 17(1):54–58
Miller DL, Force SD, Pickens A, Fernandez FG, Luu T, Mansour KA (2013) Chest wall reconstruction using biomaterials. Ann Thorac Surg 95(3):1050–1056
Lans TE, van der Pol C, Wouters MW, Schmitz PI, van Geel AN (2009) Complications in wound healing after chest wall resection in cancer patients; a multivariate analysis of 220 patients. J Thorac Oncol 4(5):639–643
Akiba T, Takagi M, Shioya H, Kurihara H, Sato S, Yamazaki Y (1998) Reconstruction of thoracic wall defects after tumor resection using a polytetrafluoroethylene soft tissue (Gore-Tex) patch. Jpn J Thorac Cardiovasc Surg 46(6):526–529
Picciocchi A, Granone P, Cardillo G, Margaritora S, Benzoni C, D’Ugo D (1993) Prosthetic reconstruction of the chest wall. Int Surg 78(3):221–224
Perry RR, Venzon D, Roth JA, Pass HI (1990) Survival after surgical resection for high-grade chest wall sarcomas. Ann Thorac Surg 49(3):363–368
McKenna RJ Jr, McMurtrey MJ, Larson DL, Mountain CF (1984) A perspective on chest wall resection in patients with breast cancer. Ann Thorac Surg 38(5):482–487
Hubbard SG, Todd EP, Carter W, Zeok J, Dillon ML, Luce E (1979) Repair of chest wall defects with prosthetic material. Ann Thorac Surg 27(5):440–444
Incarbone M, Nava M, Lequaglie C, Ravasi G, Pastorino U (1997) Sternal resection for primary or secondary tumors. J Thorac Cardiovasc Surg 114(1):93–99
Gonfiotti A, Santini PF, Campanacci D, Innocenti M, Ferrarello S, Caldarella A, Janni A (2010) Malignant primary chest-wall tumours: techniques of reconstruction and survival. Eur J Cardiothorac Surg 38(1):39–45
Doddoli C, D’Journo B, Le Pimpec-Barthes F, Dujon A, Foucault C, Thomas P, Riquet M (2005) Lung cancer invading the chest wall: a plea for en-bloc resection but the need for new treatment strategies. Ann Thorac Surg 80(6):2032–2040
De Palma A, Sollitto F, Loizzi D, Di Gennaro F, Scarascia D, Carlucci A, Giudice G, Armenio A, Ludovico R, Loizzi M (2016) Chest wall stabilization and reconstruction: short and long-term results 5 years after the introduction of a new titanium plates system. J Thorac Dis 8(3):490–498
Marulli G, De Iaco G, Ferrigno P, De Palma A, Quercia R, Brascia D, Schiavon M, Mammana M, Rea F (2020) Sternochondral replacement: use of cadaveric allograft for the reconstruction of anterior chest wall. J Thorac Dis 12(1):3–9
Tamburini N, Grossi W, Sanna S, Campisi A, Londero F, Maniscalco P, Dolci G, Quarantotto F, Daddi N, Morelli A, Cavallesco G, Dell’Amore A (2019) Chest wall reconstruction using a new titanium mesh: a multicenters experience. J Thorac Dis 11(8):3459–3466
Pastorino U, Scanagatta P, Girotti P, Rolli L, Gronchi A (2017) Long-term results of thoraco-pleuro-pneumonectomy (tpp) for recurrent thoracic sarcomas. Ann Surg Oncol 24(Suppl 3):551–554
Briccoli A, Manfrini M, Rocca M, Lari S, Giacomini S, Mercuri M (2002) Sternal reconstruction with synthetic mesh and metallic plates for high grade tumours of the chest wall. Eur J Surg 168(8–9):494–499
Chapelier A, Macchiarini P, Rietjens M (1994) Chest wall reconstruction following resection of large primary malignant tumors. Eur J Cardiothorac Surg 8:351–356
Fabre D, El Batti S, Singhal S, Mercier O, Mussot S, Fadel E, Kolb F, Dartevelle PG (2012) A paradigm shift for sternal reconstruction using a novel titanium rib bridge system following oncological resections. Eur J Cardiothorac Surg 42(6):965–970
Fouilloux V, Bertin F, Peltier E, Jouve JL (2019) First sternal cleft repair using a porous alumina ceramic prosthesis in a 9-year-old child European. J Pediatr Surg Rep 7(1):e20–e23. https://doi.org/10.1055/s-0039-1688775. (Epub 2019 Aug 26. PMID: 31463174)
Aghajanzadeh M, Alavy A, Taskindost M, Pourrasouly Z, Aghajanzadeh G, Massahnia S (2006) Results of chest wall resection and reconstruction with and without rigid pro-sthesis. J Thorac Cardiovasc Surg 81:279–285
Arbogast K, Maltese M, Nadkarni V, Steen P, Nysaether J (2006) Anterior-posterior thoracic force-deflection characteristics measured during cardiopulmonary resuscitation: comparison to post-mortem human subject data. Stapp Car Crash J 50:228–234
Suzuki K, Park J, BJ, Adusumilli PS, Rizk NP, Huang J, Jones DR, Bains MS, (2015) Chest Wall Reconstruction Using a Methyl Methacrylate Neo-Rib and Mesh. Ann Thorac Surg. https://doi.org/10.1016/j.athoracsur.2015.02.127
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-prof sectors.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
None of the authors have any disclosure related to the contents of this paper.
Human and animal rights
This article does not contain any studies with human participants or animals performed by any of the authors.
Informed consent
For this type of study formal consent is not required.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Duranti, L., Tavecchio, L. New perspectives in prosthetic reconstruction in chest wall resection. Updates Surg 75, 1093–1102 (2023). https://doi.org/10.1007/s13304-023-01562-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13304-023-01562-z