Summary
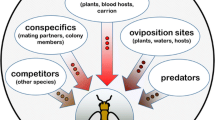
Odorants provide insects with crucial information about their environment and trigger various insect behaviors. A remarkably sensitive and selective sense of smell allows the animals to detect extremely low amounts of relevant odorants and thereby recognize, e.g., food, conspecifics, and predators. In recent years, significant progress has been made towards understanding the molecular elements and cellular mechanisms of odorant detection in the antenna and the principles underlying the primary processing of olfactory signals in the brain. These findings show that olfactory hairs on the antenna are specifically equipped with chemosensory detector units. They contain several binding proteins, which transfer odorants to specific receptors residing in the dendritic membrane of olfactory sensory neurons (OSN). Binding of odorant to the receptor initiates ionotropic and/or metabotropic mechanisms, translating the chemical signal into potential changes, which alter the spontaneous action potential frequency in the axon of the sensory neurons. The odor-dependent action potentials propagate from the antennae along the axon to the brain leading to an input signal within the antennal lobe. In the antennal lobe, the first relay station for olfactory information, the input signals are extensively processed by a complex network of local interneurons before being relayed by projection neurons to higher brain centers, where olfactory perception takes place.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
We all remember spending warm summer evenings outdoors and finding that one’s own skin is“irresistible” to mosquitos. And we all know the apparently magical attraction over-ripe fruit in the kitchen holds for fruit flies. The cause of these“animal attacks” lies in odorants emitted by our bodies or by fruit which indicate a potential blood donor or offer the prospect of food or a suitable place to lay eggs. The attractant chemical signals are detected from afar by the animals’ highly sensitive sense of smell and trigger characteristic search behavior programs in the brain, ultimately leading the animal to the source of the odor.
As these everyday examples illustrate, olfaction plays a central role for most insects in the registration of small, volatile compounds in the environment. In this context, the ability to sensitively and specifically recognize odors is often vital for their survival, since the chemical signals can provide essential information about sources of food, predators, or conspecifics. In terms of mate detection, and thus also of survival of the species, the registration of intraspecies chemical signals (pheromones) is of crucial importance. The existence of species-specific substances emitted by female animals to attract their male counterparts was first identified in 1879 by the Swiss natural scientist Auguste Farbre in his experiments with moths [14]. However, it was not until the late 1950s that Alfred Butenandt and his colleagues were able to successfully isolate 1 mg Bombykol, the first sex pheromone identified, from approximately 500,000 abdominal glands of female chinese silkworms (Bombyx mori) [7]. At around the same time, detailed research into the molecular and cellular basis of the recognition and processing of olfactory stimuli in insects was initiated. Still today, silkworms and other moths are preferred objects of research due to their highly sensitive and species-specific pheromone systems, their characteristic pheromone-induced behavior, as well as the often considerable dimensions of their antennae. The honey bee (Apis mellifera), in contrast, has established itself as a model organism for the investigation of olfactory signal processing in the brain due to its marked ability to classify and learn odors. Moreover, over the past 2 decades the fruit fly Drosophila melanogaster has become a very important model organism in insect olfactory research, which enables unique experimental approaches due to the simplicity of its olfactory system and, in particular, due to its genetic manipulability. In addition, olfactory research is currently focusing on insect species which represent severe food and plant pests or pose a threat to our health. Given the enormous significance of olfactory information in the search for food and in host detection, the insect sense of smell is under intensive investigation for being a promising target for being the development of alternative strategies to defend and fight against insect pests.
Over the last 20 years, ground-breaking advances in our understanding of insect olfaction have been made by combining biochemical, molecular biological, and cell-physiological approaches. The present article gives a short overview of our current knowledge of the peripheral and primary brain processes of odor perception. In particular, we discuss the molecular elements and mechanisms responsible for the recognition of olfactory information in the antennae and present the currently discussed models for the transduction of a chemical signal into an electrical response in the olfactory sensory neurons (OSNs). Furthermore, we will focus on the processing steps in the antennal lobe, the equivalent to the vertebrate olfactory bulb, paying particular attention to the function and interconnection of the neuronal types involved and the significance of the neuronal network for coding olfactory signals.
Structure of the peripheral olfactory system in insects
In contrast to mammals, insects have a less complex olfactory system, which is also made up of significantly fewer cells in the periphery. While there are several million OSNs in the mammalian nose, only thousands to tens of thousands of olfactory receptor cells are found in the antennae and maxillary palps of insects. Despite its lower number of OSNs, the insect olfactory system is by no means inferior to the mammalian olfactory system in terms of its sensitivity to particular odorants, often even surpassing its mammalian counterparts.
In terms of their peripheral architecture, the insect and mammalian olfactory systems are surprisingly similar. In mammals, the sensory cilia of the OSNs are embedded in the aqueous nasal mucous covering the olfactory epithelium in the nasal cavity, whereas the axon of the olfactory neuron projects directly to the olfactory bulb in the brain. Similarly, the sensory dendrites of insect OSNs lie in cuticular hair-like structures filled with aqueous lymph, the so called sensilla (Fig. 1); and also the OSN axons project directly–without any switching stations–to the brain, in this case the antennal lobe (see Fig. 4). A distinction is made between several olfactory hair types on the basis of their form and surface structure: slender trichoid sensilla, cone-shaped basiconic sensilla, and pitted coeloconic sensilla can often be found. Cuticular pores through which odorant molecules can enter the interior of the sensillum, and thereby reach the sensory cell membrane, are common to all olfactory hair types. Neurons responsive to“general” odorants are found in all hair types. In contrast, pheromone-sensitive cells have only been found in trichoid sensilla to date. In adaptation to highly sensitive pheromone detection, their dendrites, and thus also the corresponding pheromone hairs, are often extremely long (Fig. 1), greatly enlarging the antenna’s total receptive surface. Furthermore, sensory hairs involved in the detection of special odorants (pheromones, host odorants) are often very abundant and generally contain between one and three OSNs. The number of reactive neurons in sensory hairs for“general” odorants, however, can go up to 30 and more cells. Irrespective of their number, the cell bodies of the OSNs are surrounded by three supporting cells, whereby the innermost, thecogen supporting cell covers the neuron, much like a glial cell. This is followed by a tormogen and a trichogen supporting cell, both of which are involved in maintaining the composition of the sensilla lymph.
Organization of the peripheral olfactory system in insects. A Drosophila melanogaster (fruit fly). B Head of the fruit fly with olfactory appendages: antennae (Ant) and maxillary palps (Mp). The olfactory organs are visualized by expression of a green fluorescent protein (GFP) in the olfactory cells. C Head of the moth Heliothis virescens with segmented antennae (Ant). D Scanning electron microscope image of two segments of a male H. virescens antenna. The surface is covered with hundreds of both long and short sensilla hairs. Pheromone-reactive neurons are found predominantly in the long trichoid sensilla (reproduced with kind permission from [20]). E Immunohistochemical visualization of HR13 expression, the receptor for the main component of the female sex pheromone blend of H. virescens in sensory neurons of the long trichoid sensilla. Fluorescence-labeling on a tissue section of the male antenna. F Structure of a sensilla hair. Olfactory sensory neurons (OSNs) are surrounded by supporting cells. The OSN dendrites project into the aqueous sensillum lymph. Airborne odorant molecules (odorant, pheromone) are dissolved by binding proteins in the lymph and transported to the dendritic membrane. [Images A and B courtesy of Veit Grabe (MPI, Jena)]
Odorant- and pheromone-binding proteins
In insects, odorant detection begins when signal molecules penetrate the cuticular pores in the sensilla hair. Since odorants, and in particular pheromones, are usually highly hydrophobic molecules, their solubility in the aqueous sensilla lymph is extremely low and their transfer via hydrophilic fluid to specific receptor proteins in the OSN membrane is not straightforward. Nature has apparently solved this problem by coming up with odorant-binding proteins (OBPs) and special pheromone-binding proteins (PBPs). These small globular proteins (12–16 kDa) are synthesized by the tormogen and trichogen supporting cells and secreted in the sensilla lymph, where they are found at extremely high concentration (10–20 mM). Structurally, binding proteins are characterized by six highly conserved cysteines in their amino acid sequence. These form three disulphide bridges, thereby stabilizing a predominantly α-helical protein structure [48]. Binding studies as well as nuclear magnetic resonance (NMR) and crystallographic structure analyses suggest that OBPs and PBPs are able to transport lipophilic olfactory molecules after rendering them practically water-soluble by nestling them in hydrophobic binding pockets. In this way, they likely protect the olfactory molecules from decomposition or modification by degrading or transforming enzymes in the sensilla lymph.
Biochemical analyses of the sensilla lymph as well as investigations of the genome and the genes expressed in the antenna have shown a multitude of OBP types and often several PBP types in all species investigated to date [53, 64]. In addition, different binding protein types were found at the same time in the lymph of a single sensilla hair. This suggests that various binding proteins transport a variety of odorants, thereby contributing to the specificity of the olfactory system. Indeed, binding studies on various OBP types have detected distinct yet partially overlapping ligand spectra [43]. Moreover, investigations into the moth and Drosophila pheromone systems have shown that PBPs are more strongly“tuned” to special ligands than OBPs, and that they interact in a specific way only with particular pheromone components. Using receptor-expressing cell lines and purified binding proteins, it was demonstrated that there is an interplay between a particular PBP and a distinct pheromone receptor in the recognition of a species-specific pheromone component [17, 22]. Correspondingly, functional studies on Drosophila mutants provide evidence that an interplay between the pheromone receptor Or67d and the PBP Lush enables recognition of the pheromone 11-cis-vaccenyl acetate [23, 62].
Comparative investigations into the structure of pheromone-free and pheromone-laden PBPs also indicate that binding of the“correct” ligand induces a specific conformational change [36, 38]. However, it remains unclear whether the complex formed by the ligand and the binding protein then directly activates the receptor, or whether the pheromone needs to be released from the PBP first via a further conformational change in order to then activate the receptor alone. There are currently supporting findings for both possible mechanisms: while in Drosophila the PBP Lush can activate the corresponding sensory neuron in the pheromone-bound conformation [36], a release of the pheromone preceding receptor activation is supported by studies showing a pH value-dependent conformational change of moth PBPs. The latter may be effected by a low pH value near the plasma membrane [61]. Binding of the free pheromone to the receptor is also supported by functional studies of pheromone and odorant receptors expressed in cell cultures and in oocytes of the clawed frog Xenopus laevis where receptor activation even in the absence of a binding protein could be seen [21, 37, 40].
Sensory neuron membrane proteins (SNMPs)
The precise mechanism of the interaction between pheromones and pheromone/PBP complexes and OSNs has not been definitively elucidated. Recent studies revealed additional and compelling evidence that the well-known sensory neuron membrane proteins (SNMPs) may also play an important role in pheromone recognition. The first SNMP was discovered back in the mid 1990s in the dendritic membrane of pheromone-sensitive neurons of the Antheraea polyphemus moth [44]. SNMPs belong to the diverse CD36 family, the members of which are all characterized by two transmembrane domains and a large extracellular binding domain. A common feature in the hitherto functionally characterized CD36 proteins is that they recognize, bind, and transport hydrophobic molecules such as cholesterin and fatty acids, as well as lipid–protein complexes [51]. It is therefore assumed that SNMPs may represent docking sites for PBP–pheromone complexes near pheromone receptors (PRs), where they function as co-receptors that“catch” the pheromone and“pass it on” to the neighboring PR [53]. Indeed, recent studies on Drosophila mutants show that SNMP1 is crucial for the pheromone response of a sensory neuron, and that the protein lies within the membrane in direct proximity to the pheromone receptor (Or67d) [3]. In addition, SNMP1 was also found in the supporting cells surrounding the neuron in Drosophila. Hence, it is assumed that the protein in the supporting cell membrane fulfils a different function, possibly playing a role in the elimination of hydrophobic pheromones or pheromone–PBP complexes from the sensilla lymph.
In the Heliothis virescens moth, in contrast, exclusive expression in OSNs and co-expression with the sex pheromone receptor HR13 was found for the SNMP1 type. Interestingly, however, a further SNMP2 subtype, which is only expressed in the non-neuronal supporting cells, was identified in H. virescens [16]. This cell type-specific expression of the two Heliothis SNMP subtypes suggests that the two proteins serve different functions and could indicate an advanced specialization in the moth pheromone system: while for SNMP1 in the dendritic membrane of the OSN an interaction with pheromone/PBP complexes or pheromones is possible, SNMP2 in the membrane of supporting cells could be involved in sensilla lymph clearance.
Odorant receptors
Receptor proteins in the dendritic membrane of olfactory cells are key elements in the molecular recognition and discrimination of odorants. They interact in a specific manner with the relevant odorants or pheromones, and initiate the transduction of the chemical signal into an electrical response of the OSN.
Odorant receptors (ORs) were first discovered in vertebrates. In their ground-breaking work in 1991, for which they later received a Nobel prize in 2004, Linda Buck and Richard Axel found a giant family of (meanwhile) more than a thousand identified genes in the rodent genome, which encode G-protein coupled receptors (GPCRs) with typically seven transmembrane domain (7TMD) proteins, each of which is expressed in subpopulations of OSNs [6]. Subsequently, very large GPCR gene families that corresponded functionally to those found by Buck and Axel were identified in the nematode Caenorhabditis elegans; in addition, GPCRs encoding putative pheromone receptors in the vomeronasal organ of rodents were described.
Identifying insect ORs proved to be very challenging. All early approaches based on the homology of OR sequences in vertebrates and nematodes were unsuccessful. A breakthrough came, however, in 1999 following the availability of the Drosophila melanogaster genome sequence and the application of special search program for proteins with 7TMD combined with the extensive sequencing of differentially expressed antennal cDNAs [9, 54]. This led to the discovery of a diverse family of 7TMD receptors, whose members are specifically expressed in OSNs in the antennae and maxillary palps, where they are concentrated in the sensory dendrites of the cells.
Compared with the more than thousand functional OR genes found in the genome of some vertebrates, Drosophila with its 62 ORs has a much lower number. Meanwhile, the genomes of other insects have been sequenced and investigated for the presence of OR genes using bioinformatic methods. Relatively few OR genes were also found in the malaria mosquito Anopheles gambiae (79 ORs) and in the silkworm Bombyx mori (48 ORs). In contrast, however, significantly more have been identified in other insects: 163 OR candidates in the honeybee genome (Apis melifera), 225 ORs in the jewel wasp (Nasonia vitripennis), 265 ORs in the red flour beetle (Tribolium castaneum), and recently more than 400 possible ORs in the fire ant (Solenopsis invicta). Thus, extremely large numbers of different ORs are clearly not exclusive to the vertebrate olfactory system, but rather can also be found in the insect olfactory system. Why the OR gene family in some insect species (e.g., the fire ant) has experienced specific expansion is presently unclear. It is assumed, however, that the variety of OR types in some species reflect evolutionary adaption to certain ecological and physiological demands in the search for food or the major importance of odorants in social communication between insects living in colonies.
Functional analyses have meanwhile shown for most of the 62 Drosophila ORs and many Anopheles receptors that these are indispensable for the response of OSNs to odorants and they represent the molecular basis for the specificity of the various OSN types populating the antenna, as detected in electrophysiological studies [8, 25, 30, 57].
In terms of the bandwidth of their odorant spectra, insect and vertebrate ORs are similar. The protein sequences of insect and vertebrate ORs also share the 7TMD typical to GPCRs. However, the commonalities end here. OR proteins in both animal groups display no sequence similarities and are not phylogenetically related. Furthermore, in vitro and in vivo structural analyses demonstrated a surprising inverse membrane topology in insect receptor proteins: in contrast to vertebrate ORs, the N-terminal end in insects is located intracellularly, the C-terminal end extracellularly [2]. In addition to these fundamental differences in their ORs, insect OSNs differ from their vertebrate counterparts in a further significant point: while vertebrate OSNs display only one OR subtype, current studies indicate that, in the majority of insect OSNs, a specific ligand-binding OR type forms a heteromer with a second common co-receptor from the OR gene family [2]. According to the most recent nomenclature, this protein is referred to as Orco (formerly Or83b in Drosophila, OR2, or OR7 in other insects). The Orco protein is unusual in many ways: Firstly, it is the only member of the OR family which is highly conserved between insect species and it possesses a remarkably large intracellular domain. Secondly, it does not bind any odorants itself and is necessary for the transfer of specific OR types to the dendritic membrane. Finally, the Orco protein forms a non-selective cation channel which, according to recent studies, is involved in signal transduction (see below) [2, 29, 31, 35].
Pheromone receptors
Genes for special pheromone receptors (PR) in insects were first identified in the tobacco budworm (Heliothis virescens) [32]. As in many moth species, the female insect releases a complex mixture of pheromones made up of a main component and several secondary components in order to attract males. This suggests that pheromone receptors are predominantly expressed on male antennae. By combining genome analyses using known OR sequences with“screening” of an antennal cDNA library, it was possible to identify sequences for several 7TMD receptors in Heliothis that are exclusively expressed in male antennae and which occur in sensory neurons of the pheromone-sensitive olfactory hair. In functional studies employing receptor-expressing cell lines, it was demonstrated that the HR13 receptor type is activated by the main component of the female sex pheromone (Z)-11-hexadecenal [22]. Subsequently, the bombykol receptor in the Chinese silkworm Bombyx mori was successfully identified [47, 33]. Since then, PR sequences for a number of other moth species have been elucidated. Pheromone-receptors are similar in structure to odorant receptors for general odorants (Fig. 2) and are also found together with the Orco protein in olfactory cells. Remarkably, PRs form a separate group of more conserved proteins within the otherwise widely diverse moth OR family [32, 58]. This conservation of PR amino acid sequences in various species may be explained by the chemical similarity of their pheromone ligands or, alternatively, reflect the high negative evolutionary selection pressure which is exerted on the highly specific pheromone recognition system, prohibiting certain mutations in the receptor protein for the purposes of successful reproduction.
Structure, appearance, and ligands of various olfactory receptor types in insects. A OSNs of basiconic sensilla and trichoid sensilla, which respond to general odorants each express a distinct odorant receptor (ORx) and the general olfactory co-receptor (Orco). Recent studies show that ORx and Orco form heteromers, whereby the ORx subunit binds the ligand while Orco functions as an ion channel. B Pheromone-sensitive OSNs have hitherto been found only in trichoid sensilla. They possess a specific pheromone receptor type (PRx) and also express the Orco protein. ORs, PRs, and Orcos each have seven transmembrane domains. The N-terminal end of the polypeptide chain is located intracellularly, the C-terminal end extracellularly. C Two types of ionotropic olfactory receptors (IRx and IRy) are expressed in OSNs of coeloconic sensilla, and likely form heteromers. Their basic structure resembles that of ionotropic glutamate receptors. Most IRs (like IRx), however, have a shorter N-terminal. Three transmembrane domains, a pore region, and venus fly-like extracellular S1 and S2 ligand-binding domains are characteristic of all IRs. A–C Possible receptor ligand-binding sites are marked with a yellow circle. PM plasma membrane
Ionotropic receptors
A completely new class of putative olfactory receptors was recently discovered in Drosophila melanogaster antennae [4], as well as in a number of other insects [10]. These proteins belong to a subfamily of so called ionotropic receptors (IRs) and differ fundamentally in structure from ORs and PRs (Fig. 2). IRs are related to ionotropic glutamate receptors (iGluRs) in terms of their sequence and structure. However, they lack certain amino acids which interact with glutamate in iGluRs. In addition, the corresponding ligand binding region of iGluRs in IRs, in line with a possible function as a binding site for various odorants, is extremely variable. Current studies demonstrate the expression of IRs particularly in OSNs of coeloconic sensilla which do not express other 7TMD-ORs, as well as a concentration of the IR protein in the dendritic processes of the sensory neurons. By means of“miss”-expression of IRs in a different OSN type, which usually does not possess these receptors, the direct involvement of IRs in the recognition of distinct odorants could be demonstrated. Interestingly, between two and five IR types are expressed in a single OSN. As with the Orco protein in OR cells, there are also especially conserved IR types which heterodimerize with variable IR types [1].
Olfactory signal transduction
Odorant-specific ORs, PRs, and IRs are the receptive proteins in the dendritic membrane of OSNs. But how, after binding of a suitable ligand, is a chemical odorant signal transduced into an electrical cell response of the OSN that is first of all a change in the membrane potential?
To date, functional studies on special OSNs of the coeloconic sensilla equipped with IRs have shown that IRs themselves form odorant-activated ion channels from combinations of between two and three different IR types. In addition to an IR type which determines ligand specificity, the heteromer contains one or two (often the same) IR types, which function as co-receptors [1]. In OSNs with IRs, it is assumed that the binding of odorants to the receptor/ion channel complex, analogous to the IR-related ionotropic glutamate receptors, results in a cation influx into the sensory neuron and depolarization of the cell.
However, the molecular processes involved in olfactory signal transduction in those OSNs with 7TMD odorant or pheromone receptors, which are prevalent on the antennae, are largely unclear and the literature reports partially controversial findings. More recent functional studies by two research groups who carried out electrophysiological investigations on mammalian cells heterologously expressing combinations of a ligand-specific Drosophila OR and the Orco protein unanimously point to an odorant-activated heteromeric receptor–ion channel complex made up of one variable OR subunit and the conserved Orco protein [49, 59]. However, there is disagreement as to whether ORs are only subunits of ligand-activated OR/Orco channels which open upon odorant binding and directly cause OSN depolarization (ionotropic signal pathway). Alternatively, insect ORs are also G-protein-coupled receptors which, following their activation, trigger downstream intracellular reaction cascades and lead to the formation of second messengers, which in turn indirectly open the Orco ion channel (metabotropic signal pathway).
The results obtained by Sato et al. gave no support for any involvement of G-protein-activated reaction cascades, pointing to a purely“ionotropic signal pathway” (Fig. 3 A). These findings were consistent with the view that insect ORs could not be GPCRs, since they have no sequence similarities with vertebrate and nematode ORs, or other classic GPCRs, and are furthermore characterized by an inverse membrane topology. However, studies by Wicher et al. [59] demonstrate that odorant binding to the OR subunit also activates a Gs protein and that, additionally, Orco is an ion channel which can be opened by cAMP, a classic second messenger. This group’s model thus proposes the existence of dual (combined) ionotropic and metabotropic signal transduction (Fig. 3 B), according to which a direct OR–Orco interaction occurs in the presence of high odorant concentrations, hence a rapid opening of the Orco channel, while in the presence of low odorant concentrations, the ligand-specific OR triggers a G-protein cascade, leading to the formation of cAMP, which in turn opens the Orco channel. This slower G-protein-mediated amplification may be crucial for the highly sensitive recognition of odorants and would represent an interesting analogy to the vertebrate olfactory system, where the binding of odorants to ligand-specific ORs, also via G-protein activation (Golf) and an adenylyl cyclase, leads to cAMP synthesis, which opens a cAMP-gated cation channel.
Models of olfactory signal transduction in insects. A A specific odorant receptor (ORx) and the common co-receptor (Orco) form a ligand-activated receptor–ion channel complex in OSNs which respond to general odorants. An odorant molecule is transferred by an odorant-binding protein and binds to the ORx subunit, thereby causing the non-selective cation channel to open [49]. B According to Wicher et al., the variable ORx subunit is a G-protein coupled receptor, while Orco is a direct and cyclic nucleotide-activated ion channel [59]. In the presence of high concentrations of a general odorant, a direct ORx/Orco interaction causes the Orco channel to open. In the case of low odorant concentrations, the model proposes an indirect opening of the channel. Firstly, the receptor activates a G-protein (G s), which leads to cAMP production via an adenylyl cyclase (AC). This second messenger in turn leads to the opening of the Orco channel. C Findings in pheromone-sensitive OSNs suggest the involvement of a sensory neuron membrane protein (SNMP) in signal transduction. The SNMP may serve as a docking site for the ligand-laden pheromone-binding protein and/or be involved in the release of the pheromone. The Orco channel opens following binding of the pheromone to the PRx subunit. Alternatively, it has been proposed that binding of the PBP–pheromone complex to SNMP reverses an inhibition of the pheromone receptor (PR x)–Orco complex, thus triggering an influx of cations into the cell [24]. D Several lines of evidence indicate a G-protein-mediated pathway in pheromone recognition [52]. Receptor activation could thus activate a phospholipase C (PLCβ) via a Gq-protein, which converts phosphoinositol-(4,5)-biphosphate (PIP 2) into inositol trisphosphate (IP 3) and diacylglycerol (DAG). IP3 then opens a calcium-selective ion channel (CaC); the intracellular increase in calcium concentration subsequently activates other non-selective cation channels (CC)
With regard to the transduction of distinct pheromone signals, investigations on Drosophila mutants have shown that a specific pheromone receptor, the Orco protein, and SNMP1 are essential for the olfactory cell’s electrophysiological response. On the basis of the most recent Drosophila data, an exclusively“ionotropic signalling pathway” has been suggested, which includes involvement of the SNMP1 protein but rejects G-protein activation. In contrast, biochemical, electrophysiological, and genetic investigations carried out in the last 20 years also suggest the existence of a G-protein-mediated metabotropic mechanism in pheromone signal transduction: in this context, studies on various moth species support a pheromone-activated IP3 signal cascade in pheromone-sensitive OSNs [39, 52]. A corresponding model which attempts to combine experimental findings obtained to date (Fig. 3 D) proposes that the binding of a pheromone molecule to a specific pheromone receptor mediates the activation of a G-protein and, subsequently, a phospholipase C (PLC); this enzyme catalyzes the hydrolysis of phosphatidyl-4,5-bisphosphate to yield inositol trisphosphate (IP3) and diacylglycerol (DAG). IP3 then opens a calcium (Ca2+)-selective ion channel, which in turn“gates” further non-selective cation channels via the calcium influx [52]. How the expression of the cAMP-activatable Orco channel in pheromone-responsive OSNs is reconcilable with this signal transduction cascade remains as yet unclear. However, it has been suggested that Orco in pheromone-responsive neurons is not directly involved in the transduction of odorant signals, but may be relevant for setting and modulating the spontaneous activity in OSNs [52].
Taken together, findings to date show an inhomogeneous and complex picture of olfactory signal transduction in insect olfactory sensory cells. Further experiments in the future are needed to clarify to what extent ionotropic and metabotropic mechanisms, either alone or in combination, mediate the transduction of odorant and pheromone stimuli.
The olfactory pathway
As mentioned above, the interaction between an odorant molecule and a specific odorant receptor is transduced into neuronal excitation of the OSN. This sequence of action potentials is transmitted via the antennal nerve to the first relay station in the brain, the antennal lobe. The antennal lobe represents the primary olfactory neuropil for the integration and coding of olfactory information and is structurally similar to the primary olfactory center in vertebrates, the olfactory bulb [26]. Both brain centers consist of discrete round structures known as olfactory glomeruli (Fig. 4), within which OSN axons are synaptically interconnected to projection neurons and a network of local interneurons. It has already been shown in many insect species that the axons of OSNs expressing the same odorant receptor types converge in one glomerulus and that, in most cases, each glomerulus receives only the afferent input of one receptor type [55]. It could also be demonstrated for some species that the number of glomeruli corresponds approximately 1:1 to the number of different odorant receptor types. In this way, the activity of an OSN population which responds to certain odorants is concentrated in one glomerulus. Thus, olfactory glomeruli represent not only structural but also functional units of olfactory coding. The number of glomeruli is genetically determined and specific to each species: the fruit fly has approximately 50 glomeruli, the moth around 60, and the honeybee approximately 160. More than 400 glomeruli have been found in some ant species. Not only is the number of glomeruli genetically determined, but also their size and position in the antennal lobe, making it possible to compile a digital, 3D atlas of the antennal lobe using morphological data [34, 5]. Regarding the processing of female sex pheromone signals, the males of some insect species, in particular many moth species such as the moth Heliothis virescens, exhibit an interesting feature: a comparison of male and female antennal lobes demonstrates specific glomeruli in the male which are located separately at the entrance of the antennal nerve and are significantly larger than other glomeruli (Fig. 4 B,C). This macroglomerular complex (MGC) receives and encodes the activity of pheromone-sensitive OSNs. Various components of the sex pheromone mixture have been identified and their coding could be attributed to distinct MGC glomeruli in many moth species. In the fruit fly Drosophila, however, only one substance, 11-cis-Vaccenyl-acetate, could be identified as a sex pheromone to date. Moreover, the antennal lobe in the latter shows no MGC comparable with that seen in any moth species. However, there is consensus that other pheromone components exist also for Drosophila. Thus, pheromone detection is not necessarily linked to the presence of an MGC.
Organization of the antennal lobe in Heliothis virescens. A Frontal view of the head with antennae connected to the head capsule above the compound eyes. The white box denotes the area in which the left antennal lobe lies within the head. B Glomerular structures in the antennal lobe of a male moth. These round neuropil structures are formed from the endings of OSNs and the projections of projection neurons and local neurons. In addition to the macroglomerular complex (MGC) (comprising four glomeruli) where incoming pheromone signals are processed, many“ordinary glomeruli,” where“general” odorants are processed, are visible. To visualize glomerular structures, the antennal lobe has been fluorescence stained with an antibody against a synaptic protein and an“optical section” was imaged with a confocal laser scanning microscope. C Three-dimensional reconstruction of the antennal lobe of a male moth, generated by using optical section series of the antennal lobe seen in B. The MGC is marked in gray, while the various“ordinary glomeruli” are marked in different colors. [Images courtesy of P.H. Olsen (A), B.B. Løfaldli (B and C) and H. Mustaparta (all from the NTNU, Norway)]
The antennal lobe receives input from sensory neurons and accommodates a complex neuronal network in which a vast receptor-neuron input is transferred to comparatively few output neurons, the projection neurons. The OSN terminals and projection neuron terminals in each individual glomerulus are connected with a further neuron type in the antennal lobe, the local interneurons. These limit their branches to the antennal lobe, forming a multitude of connections between the various glomeruli. Both inhibitory (GABAergic) and excitatory (cholinergic) local interneurons have been described in Drosophila. To date, only inhibitory local interneurons have been identified in other insect species, such as the moth and honeybee. Local interneurons process and transform incoming olfactory information from the antennal OSNs and strongly modulate the outgoing signals from individual glomeruli. This integrated olfactory information is received by uni- and multiglomerular projection neurons, which innervate only one or several glomeruli. The projection neurons convey the olfactory information to higher brain centers, such as the lateral protocerebrum and the mushroom bodies, where odor perception takes place.
The representation of odorants in the brain
How are odorants represented and processed at the various processing levels in the antennal lobe? Functional calcium imaging studies in honey bees have made an important contribution to our understanding of olfactory coding in the brain [18, 19, 28, 46]. This method is based on staining the brain with a calcium-sensitive dye which changes its fluorescence in a calcium-dependent manner. Calcium is a very good indicator of neuronal activity since it is involved in a series of signal cascades as a second messenger substance and also plays an important role in synaptic transmission. By using a highly sensitive camera, functional imaging enables neuronal activity at different sites in the brain to be measured simultaneously. In this way, the spatial and temporal activity of neurons involved in encoding an olfactory impression can be visualized. Studies on honeybees have been able to show that odorant stimulation evokes a reproducible spatio-temporal activity pattern in the antennal lobe [46]. All odorants activate a specific combination of glomeruli, whereby each glomerulus can be activated by several odorants. Thus, the olfactory system has developed a combinational strategy with which the enormous variety of odorants can be represented with only a few coding units, the glomeruli. These odorant-specific patterns are bilaterally symmetric and conserved between different individuals of the same species [19], i.e., odorant patterns are genetically determined.
In recent years, functional imaging has also been applied in the olfactory system of the fruit fly Drosophila melanogaster [15]. Various molecular genetic techniques have been established in recent years in the fruit fly, which have contributed to making it a genetic model organism. With the help of the GAL-UAS system, for example, specific gene products can be ectopically expressed in subpopulations of individual neurons. This system makes it possible for a calcium-sensitive protein, such as the GFP derivate G-CaMP, to be selectively expressed in OSNs or projection neurons of the Drosophila olfactory system (Fig. 5). Thus, it enables the representation of odorants at various processing levels of the antennal lobe to be visualized. Results to date show that odorants can also evoke a reproducible and combinatorial activity pattern in the fruit fly, both in sensory and projection neurons (Fig. 5), highlighting the fact that combinatorial odorant coding is a general principle in the insect olfactory system.
Odorant representation in the input and output neurons of the Drosophila antennal lobe (AL). The calcium-sensitive protein G-CaMP was selectively expressed in olfactory sensory neurons (OSN, bottom) or projection neurons (PN, top). Calcium signals for two distinct odorants were superimposed onto morphological images of the antennal lobe. Red indicates a strong and yellow a medium increase in the intracellular calcium concentration (weaker calcium signals were cropped). Both odorants activate a specific combination of glomeruli. The patterns of the right and left antennal lobe are bilaterally symmetric. A comparison of the activity patterns between the two processing levels shows similar yet non-identical patterns
Processing in the antennal lobe network
Since the antennal lobe is a complex network of excitatory and inhibitory neuronal circuits, the question arises as to how odorant representation of the input, i.e., the sensory neurons, is transformed into an output representation in the projection neurons, which comprise only a fraction of the number of neurons compared with the vast number of OSNs. When comparing odorant representation in the sensory input with representation in the projection neurons (Fig. 5), we see that the activity patterns are similar yet not identical. Numerous studies have addressed the question of the input–output relationship in the antennal lobe, reaching partially controversial results. Whereas imaging data from the honeybee show that the activity patterns of projection neurons are contrast-enhanced and sharpened compared to the input pattern of OSNs [45], other studies suggest that calcium signals from sensory neurons and output neurons in Drosophila are identical [41, 56]. However, a further study in Drosophila in which individual projection neurons and individual OSNs on the antennae were electrophysiologically recorded showed that projection neuron responses were more unspecific, i.e., broader, than those of OSNs which innervate the same glomerulus [60]. Furthermore, recent studies on Drosophila show that OSNs are also presynaptically inhibited by GABA, suggesting a“gain control“ mechanism which modulates neural transmission from OSNs to the downstream projection neurons [42]. Thus, it can be seen in Drosophila that various glomeruli are modulated differently in the network according to the odorant [50].
With regard to the synaptic connections in a single glomerulus, basic studies have been carried out in cockroaches [11, 12, 13]. Supported by findings from current research on other insects, it could be shown that all neuron types in the antennal lobe are synaptically interconnected (Fig. 6): OSNs are cholinergic neurons and transmit olfactory information to both projection neurons and local interneurons. Moreover, OSNs receive the above-mentioned presynaptic inhibition from inhibitory, i.e., GABAergic, local neurons. In turn, local interneurons are connected to OSNs, other local interneurons, and projection neurons. Ultimately, projection neurons are generally cholinergic, i.e., excitatory, and connected to both other neuronal types. The only synaptic connection that has not been found so far is a connection from projection neurons back to OSNs.
Connectivity model of the antennal lobe. A Morphological images of the three neuron types in the Drosophila antennal lobe, in each of which sensory neurons (bottom), inhibitory local interneurons (middle), or projection neurons (top) are marked in green; the remaining neuropil is marked in red. B A model of the hitherto identified synaptic connections in the antennal lobe between sensory neurons (green), excitatory and inhibitory local interneurons (blue; iLN, eLN), and projection neurons (red). Sensory neurons form chemical excitatory synapses with projection neurons and both types of local interneurons. Inhibitory local interneurons inhibit projection neurons, sensory neurons (presynaptic inhibition), as well as other glomeruli, via chemical inhibitory synapses. Excitatory local interneurons excite other glomeruli with chemical excitatory synapses. They also possess electrical synapses with which they are able to modulate projection neurons and inhibitory local interneurons
Interestingly, it could be shown recently that excitatory local neurons in Drosophila excite projection neurons and inhibitory local neurons via electrical synapses, leading to lateral excitation between glomeruli in the antennal lobe [27, 63] (Fig. 6). These new findings explain, firstly, why odorant representation in the projection neurons of Drosophila is broader compared to input neurons. Secondly, this lateral excitation could increase sensitivity to low-concentration odorants, whereas strong odorants could be specifically weakened by the inhibitory“gain control” mechanism.
In conclusion, a homogeneous model of how the antennal lobe processes the afferent input of OSNs cannot be developed as yet on the basis of previous studies, which used a variety of techniques, methods of analysis, and animal models, nor on the basis of their partially controversial results. However, recent findings already impressively show how the antennal lobe network extensively transforms the input signal from sensory neurons via intra- and interglomerular interactions to ensure reliable recognition, discrimination, and perception of odors.
References
Abuin L, Bargeton B, Ulbrich MH, Isacoff EY, Kellenberger S, Benton R (2011) Functional architecture of olfactory ionotropic glutamate receptors. Neuron 69:44–60
Benton R, Sachse S, Michnick SW, Vosshall LB (2006) Atypical membrane topology and heteromeric function of Drosophila odorant receptors in vivo. PLoS Biol 4:e20
Benton R, Vannice KS, Vosshall LB (2007) An essential role for a CD36-related receptor in pheromone detection in Drosophila. Nature 450:289–293
Benton R, Vannice KS, Gomez-Diaz C, Vosshall LB (2009) Variant ionotropic glutamate receptors as chemosensory receptors in Drosophila. Cell 136:149–162
Berg BG, Galizia CG, Brandt R, Mustaparta H (2002) Digital atlases of the antennal lobe in two species of tobacco budworm moths, the Oriental Helicoverpa assulta (male) and the American Heliothis virescens (male and female). J Comp Neurol 446:123–134
Buck L, Axel R (1991) A novel multigene family may encode odorant receptors: a molecular basis for odor recognition. Cell 65:175–187
Butenandt A, Beckmann R, Stamm D, Hecker E (1959) Über den Sexuallockstoff des Seidenspinners Bombyx mori, Reindarstellung und Konstitution. Zeitschrift für Naturforschung 14:283–284
Carey AF, Wang G, Su CY, Zwiebel LJ, Carlson JR (2010) Odorant reception in the malaria mosquito Anopheles gambiae. Nature 464:66–71
Clyne PJ, Warr CG, Freeman MR, Lessing D, Kim J, Carlson JR (1999) A novel family of divergent seven-transmembrane proteins: candidate odorant receptors in Drosophila. Neuron 22:327–338
Croset V, Rytz R, Cummins SF, Budd A, Brawand D, Kaessmann H, Gibson TJ, Benton R (2010) Ancient protostome origin of chemosensory ionotropic glutamate receptors and the evolution of insect taste and olfaction. PLoS Genet 6:e1001064
Distler PG, Boeckh J (1996) Synaptic connection between olfactory receptor cells and uniglomerular projection neurons in the antennal lobe of the American cockroach, Periplaneta americana. J Comp Neurol 370:35–46
Distler PG, Boeckh J (1997a) Synaptic connections between identified neuron types in the antennal lobe glomeruli of the cockroach, Periplaneta americana: I. Uniglomerular projection neurons. J Comp Neurol 378:307–319
Distler PG, Boeckh J (1997b) Synaptic connections between identified neuron types in the antennal lobe glomeruli of the cockroach, Periplaneta americana: II. Local multiglomerular interneurons. J Comp Neurol 383:529–540
Fabre JH (1879) Hochzeitsflüge der Nachtpfauenaugen. In: Bilder aus der Insektenwelt: Übersetzung aus “Souvenirs Entomologiques”I. X. Kosmos, Gesellschaft der Naturfreunde, Paris, Stuttgart, p 80
Fiala A, Spall T, Diegelmann S, Eisermann B, Sachse S, Devaud JM, Buchner E, Galizia CG (2002) Genetically expressed cameleon in Drosophila melanogaster is used to visualize olfactory information in projection neurons. Curr Biol 12:1877–1884
Forstner M, Gohl T, Gondesen I, Raming K, Breer H, Krieger J (2008) Differential expression of SNMP-1 and SNMP-2 proteins in pheromone-sensitive hairs of moths. Chem Senses 33:291–299
Forstner M, Breer H, Krieger J (2009) A receptor and binding protein interplay in the detection of a distinct pheromone component in the silkmoth Antheraea polyphemus. Int J Biol Sci 5:745–757
Galizia CG, Menzel R (2000) Odour perception in honeybees: coding information in glomerular pattern. Curr Opin Neurobiol 10:504–510
Galizia CG, Sachse S, Rappert A, Menzel R (1999) The glomerular code for odor representation is species specific in the honeybee Apis mellifera. Nat Neurosci 2:473–478
Gohl T, Krieger J (2006) Immunolocalization of a candidate pheromone receptor in the antenna of the male moth, Heliothis virescens. J Inv Neurosci 6:13–21
Grosse-Wilde E, Svatos A, Krieger J (2006) A pheromone-binding protein mediates the bombykol-induced activation of a pheromone receptor in vitro. Chem Senses 31:547–555
Grosse-Wilde E, Gohl T, Bouche E, Breer H, Krieger J (2007) Candidate pheromone receptors provide the basis for the response of distinct antennal neurons to pheromonal compounds. Eur J Neurosci 25:2364–2373
Ha TS, Smith DP (2006) A pheromone receptor mediates 11-cis-vaccenyl acetate-induced responses in Drosophila. J Neurosci 26:8727–8733
Ha TS, Smith DP (2009) Odorant and pheromone receptors in insects. Front Cell Neurosci 3:10
Hallem EA, Carlson JR (2006) Coding of odors by a receptor repertoire. Cell 125:143–160
Hildebrand JG, Shepherd GM (1997) Mechanisms of olfactory discrimination: converging evidence for common principles across phyla. Annu Rev Neurosci 20:595–631
Huang J, Zhang W, Qiao W, Hu A, Wang Z (2010) Functional connectivity and selective odor responses of excitatory local interneurons in Drosophila antennal lobe. Neuron 67:1021–1033
Joerges J, Küttner A, Galizia CG, Menzel R (1997) Representation of odours and odour mixtures visualized in the honeybee brain. Nature 387:285–288
Jones WD, Nguyen TAT, Kloss B, Lee KJ, Vosshall LB (2005) Functional conservation of an insect odorant receptor gene across 250 million years of evolution. Current Biology 15:R119–R121
Kreher SA, Kwon JY, Carlson JR (2005) The molecular basis of odor coding in the Drosophila larva. Neuron 46:445–456
Krieger J, Klink O, Mohl C, Raming K, Breer H (2003) A candidate olfactory receptor subtype highly conserved across different insect orders. J Comp Physiol A Neuroethol Sens Neural Behav Physiol 189:519–526
Krieger J, Grosse-Wilde E, Gohl T, Dewer YME, Raming K, Breer H (2004) Genes encoding candidate pheromone receptors in a moth (Heliothis virescens). Proc Natl Acad Sci U S A 101:11845–11850
Krieger J, Grosse-Wilde E, Gohl T, Breer H (2005) Candidate pheromone receptors of the silkmoth Bombyx mori. Eur J Neurosci 21:2167–2176
Laissue PP, Reiter C, Hiesinger PR, Halter S, Fischbach KF, Stocker RF (1999) Three-dimensional reconstruction of the antennal lobe in Drosophila melanogaster. J Comp Neurol 405:543–552
Larsson MC, Domingos AI, Jones WD, Chiappe ME, Amrein H, Vosshall LB (2004) Or83b encodes a broadly expressed odorant receptor essential for Drosophila olfaction. Neuron 43:703–714
Laughlin JD, Ha TS, Jones DN, Smith DP (2008) Activation of pheromone-sensitive neurons is mediated by conformational activation of pheromone-binding protein. Cell 133:1255–1265
Mitsuno H, Sakurai T, Murai M, Yasuda T, Kugimiya S, Ozawa R, Toyohara H, Takabayashi J, Miyoshi H, Nishioka T (2008) Identification of receptors of main sex-pheromone components of three Lepidopteran species. Eur J Neurosci 28:893–902
Mohl C, Breer H, Krieger J (2002) Species-specific pheromonal compounds induce distinct conformational changes of pheromone binding protein subtypes from Antheraea polyphemus. Invert Neurosci 4:165–174
Nakagawa T, Vosshall LB (2009) Controversy and consensus: noncanonical signaling mechanisms in the insect olfactory system. Curr Opin Neurobiol 19:284–292
Nakagawa T, Sakurai T, Nishioka T, Touhara K (2005) Insect sex-pheromone signals mediated by specific combinations of olfactory receptors. Science 307:1638–1642
Ng M, Roorda RD, Lima SQ, Zemelman BV, Morcillo P, Miesenbock G (2002) Transmission of olfactory information between three populations of neurons in the antennal lobe of the fly. Neuron 36:463–474
Olsen SR, Wilson RI (2008) Lateral presynaptic inhibition mediates gain control in an olfactory circuit. Nature 452:956–960
Qiao H, Xiaoli H, Schymura D, Ban L, Field L, Dani FR, Michelucci E, Caputo B, della Torre A, Iatrou K, Krieger J, Zhou JJ, Pelosi P (2010) Comparative interactions between odorant-binding proteins of Anophels gambiae. Cell Mol Life Sci (in press)
Rogers ME, Sun M, Lerner MR, Vogt RG (1997) Snmp-1, a novel membrane protein of olfactory neurons of the silk moth Antheraea polyphemus with homology to the CD36 family of membrane proteins. J Biol Chem 272:14792–14799
Sachse S, Galizia CG (2002) Role of inhibition for temporal and spatial odor representation in olfactory output neurons: a calcium imaging study. J Neurophysiol 87:1106–1117
Sachse S, Rappert A, Galizia CG (1999) The spatial representation of chemical structures in the antennal lobe of honeybees: steps towards the olfactory code. Eur J Neurosci 11:3970–3982
Sakurai T, Nakagawa T, Mitsuno H, Mori H, Endo Y, Tanoue S, Yasukochi Y, Touhara K, Nishioka T (2004) Identification and functional characterization of a sex pheromone receptor in the silkmoth Bombyx mori. Proc Natl Acad Sci U S A 101:16653–16658
Sandler BH, Nikonova L, Leal WS, Clardy J (2000) Sexual attraction in the silkworm moth: structure of the pheromone-binding-protein-bombykol complex. Chem Biol 7:143–151
Sato K, Pellegrino M, Nakagawa T, Nakagawa T, Vosshall LB, Touhara K (2008) Insect olfactory receptors are heteromeric ligand-gated ion channels. Nature 452:1002–1006
Silbering AF, Okada R, Ito K, Galizia CG (2008) Olfactory information processing in the Drosophila antennal lobe: anything goes? J Neurosci 28:13075–13087
Silverstein RL, Febbraio M (2009) CD36, a scavenger receptor involved in immunity, metabolism, angiogenesis, and behavior. Sci Signal 2:re3
Stengl M (2010) Pheromone transduction in moths. Front Cell Neurosci 4:133
Vogt RG (2003) Biochemical diversity of odor detection: OBPs, ODEs and SNMPs. In: Blomquist G, Vogt RG (eds) Insect pheromone biochemistry and molecular biology. The biosynthesis and detection of pheromones and plant volatiles. Elsevier Academic Press, London, pp 391–445
Vosshall LB, Amrein H, Morozov PS, Rzhetsky A, Axel R (1999) A spatial map of olfactory receptor expression in the Drosophila antenna. Cell 96:725–736
Vosshall LB, Wong AM, Axel R (2000) An olfactory sensory map in the fly brain. Cell 102:147–159
Wang JW, Wong AM, Flores J, Vosshall LB, Axel R (2003) Two-photon calcium imaging reveals an odor-evoked map of activity in the fly brain. Cell 112:271–282
Wang G, Carey AF, Carlson JR, Zwiebel LJ (2010) Molecular basis of odor coding in the malaria vector mosquito Anopheles gambiae. Proc Natl Acad Sci U S A 107:4418–4423
Wanner KW, Anderson AR, Trowell SC, Theilmann DA, Robertson HM, Newcomb RD (2007) Female-biased expression of odourant receptor genes in the adult antennae of the silkworm, Bombyx mori. Insect Mol Biol 16:107–119
Wicher D, Schafer R, Bauernfeind R, Stensmyr MC, Heller R, Heinemann SH, Hansson BS (2008) Drosophila odorant receptors are both ligand-gated and cyclic-nucleotide-activated cation channels. Nature 452:1007–1011
Wilson RI, Turner GC, Laurent G (2004) Transformation of olfactory representations in the Drosophila antennal lobe. Science 303:366–370
Wojtasek H, Leal WS (1999) Conformational change in the pheromone-binding protein from Bombyx mori induced by pH and by interaction with membranes. J Biol Chem 274:30950–30956
Xu PX, Atkinson R, Jones DNM, Smith DP (2005) Drosophila OBP LUSH is required for activity of pheromone-sensitive neurons. Neuron 45:193–200
Yaksi E, Wilson RI (2010) Electrical coupling between olfactory glomeruli. Neuron 67:1034–1047
Zhou JJ (2010) Odorant-binding proteins in insects. Vitam Horm 83:241–272
Conflict of interest
The corresponding author states that there are no conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sachse, S., Krieger, J. Olfaction in insects. e-Neuroforum 2, 49–60 (2011). https://doi.org/10.1007/s13295-011-0020-7
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13295-011-0020-7