Abstract
The unique physico-chemical properties of fullerene C60 have aided in its application in various fields along with several ecological impacts as an emerging pollutant. So, there is a significant need to demonstrate the ecological risk and toxic impact of fullerene C60 in the freshwater ecosystem. The present study focused on the toxic impact of fullerene C60 on behavioral and hematological changes in the freshwater fish, Anabas testudineus. The acute toxicity of fullerene C60 in Anabas testudineus determined by Probit analysis was found to be 50 mg/L. For the analysis, the fish were exposed to two sublethal concentrations of 5 mg/L (one-tenth of LC50-96 h) and 10 mg/L (one-fifth of LC50-96 h) for short-term (24, 48, 72 and 96 h) and long-term (7, 15, 30 and 60 days) durations. Fullerene C60 exposure showed prominent changes in behavior of the fish that comprises changes in swimming activity, disruption of schooling behavior, air engulping and surfacing along with morphological alterations such as descaling, slight hemorrhage and mucous secretion on the body surface, which was correlated to significant (P < 0.05) decrease in the activity of acetylcholinesterase enzyme in brain tissue. Hematological changes includes significant (P < 0.05) reduction in the erythrocyte and leukocyte counts, hemoglobin concentration, percentage of the packed cell volume (hematocrit) and the levels of serum albumin, globulin and total protein, whereas serum glucose concentration and the activities of the serum alanine and aspartate aminotransferases were significantly (P < 0.05) increased during the sublethal exposures. Severe degeneration in the columnar epithelial cells and lamina propria, along with increased number of mucous cells observed in the intestine of the fish indicates sublethal toxicity of fullerene C60. The present findings led to the conclusion that the sublethal concentrations of fullerene C60 have a toxic impact on the fish A. testudineus by affecting the normal physiology, and thus the presence of this nanomaterial in the environment may affect the health status of the ecosystem.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Nanostructured materials are unique groups that have distinctive structural features with at least one dimension of 100 nm or less, which includes zero-dimensional quantum dots or nanoparticles, one-dimensional nanowires, nanotubes, nanorods, nanobelts, and nanoribbons, two-dimensional nanosheets, nanowalls, nanoprisms, nanoplates, and nanodisks and three-dimensional materials such as nanoballs, nanocoils, nanopillars, nanocones, and nanoflowers (Tiwari et al. 2014). Novel carbon-derived nanomaterials such as carbon nanotubes, graphene, crystalline diamond, and diamond-like carbon have gained significant attention in multiple disciplines including physics, chemistry and nanomedicines. One of the carbon nanomaterials, fullerene C60, discovered by Kroto et al. (1985) is one of the most common and stable members in the fullerene family possessing unique properties including a large surface-to-volume ratio, high conductivity, and electron mobility at room temperature that distinguish it from other carbon nanomaterials. Fullerene C60 with a truncated icosahedron structure having 12 pentagons and 20 hexagons used its specific features to introduce promising new applications in the fields of material science (Coro et al. 2016), medicine (Pan et al. 2015), personal care products (Lens 2011) and solar cells (Mohajeri and Omidvar 2015). Biomedical applications of fullerene ranged from the treatment of the several diseases related to oxidative stress (Injac et al. 2013), neurodegenerative disorders (Dugan et al. 1997), cancer (Shi et al. 2014), HIV (Friedman et al. 1993) and influenza (Shoji et al. 2013).
Humans were exposed to fullerene at low concentrations mainly in geological materials dating back to 85 billion years (Becker et al. 1994). Fullerenes exist naturally in coal deposits (Fang et al. 2006), lava pillows (Jehlicka et al. 2000), shungites (Parthasarathy et al. 1998) and fulgurites (Daly et al. 1993). Recently, the presence of fullerenes in the environment increased drastically due to the anthropogenic activities mainly during emission from the working environment, transportation and use, at the time of disposal, and relatively small amounts of soluble and dispersed fullerenes emit into the water during the manufacture, depending on poor treatment facility (Farre et al. 2010). During sewage treatment, most of the fullerene particles are likely to form deposits, but soluble fullerenes are emitted into the aquatic environment (Dhawan et al. 2006). The release of nanomaterials into the aquatic environment is expected to form unstable colloidal suspensions. The stability of colloid under natural conditions depends upon various factors including pH, ionic strength and the presence of natural organic matter (Lead and Wilkinson 2006). Nanomaterials released into the environment is affected by several environmental factors such as light, oxidants or microorganisms that result in chemical or biological modification, degradation of the surface functionalization or the embedding matrix which may result in the formation of free nanoparticles (Nowack and Bucheli 2007). The potential mobility of nanomaterials under natural conditions depends on the surface modification, while the adsorption of nanomaterials to organic compounds depends on the dispersion state (Cheng et al. 2004).
Nanomaterials are able to cross the cell membrane and become internalized into a variety of mammalian cell types (Rothen-Rutishauser et al. 2006). One of the direct aquatic exposure routes of fullerene C60 is from the cosmetic wastewater effluents mainly released from the skin care products at a detectable range from 0.04 to 1.1 μg/g (Benn et al. 2011). Fullerene C60 and C70 were detected directly in 8 of 11 cosmetic sample products with the concentration of 10–340 ng/g (Zakaria et al. 2018). Fullerenes were noticed in trace amounts in the wastewater treatment plants with the highest concentration reaching μg/L in the suspended solids of effluents (Farre et al. 2010). Thus, domestic wastewater forms the main source that brings fullerene C60 into the aquatic environment and the estimated maximum concentration released from the skin care products is about 26 μg/kg body weight (Hansen et al. 2008). Noticeable concentrations of pristine fullerenes such as C60 and C70 were found in pond water at the range of 9–330 pg/L, while in sediments it ranged between 0.1 and 7.2 ng/Kg, whereas the concentration of fullerene C60 derivatives ranged from 1.5 to 8.5 pg/L in pond water samples (Astefanei et al. 2014). Besides these, certain hydrological conditions and heavy rains also contribute to the high concentration of nanomaterial contamination in the aquatic ecosystems.
Aggregation and size-dependent sedimentation are the main parameters that determine the uptake of nanomaterials into the body of the organisms (Limbach et al. 2005). Reports have suggested that fullerenes form stable nanoscale aggregates such as nano-C60 by the action of certain natural constituents such as dissolved natural organic matter found in the aquatic bodies (Xie et al. 2008). The suspended form of C60 fullerene aggregates exists in water bodies even for months at a concentration of 100 mg/L (Fortner et al. 2005). However, poor solubility and the propensity for aggregation of fullerenes in the water bodies limit the accessibility to living organisms to a certain extent (Brant et al. 2005). Usually, the uptake of nanomaterials occurs through endocytosis or phagocytosis in specialized cells where the coating of nanomaterials by protein in the growth medium causes conformational changes in the protein structure, which stimulates the uptake of nanomaterials into the cell by limiting it below 120 nm (Lynch et al. 2006). Nanomaterials are then stored within the vesicles or mitochondria of the cells to exert a toxic response.
Most of the toxicity studies have been carried out on aquatic organisms including fish (Oberdorster 2004; Oberdorster et al. 2006; Zhu et al. 2006), daphnids (Lovern and Klaper 2006; Lovern et al. 2007), bacteria (Fortner et al. 2005; Lyon et al. 2006; Lyon and Alvarez 2008) and aquatic invertebrates (Pakarinen et al. 2011; Waissi-Leinonen et al. 2015). Information available regarding the toxicity of fullerene C60 in fish population seems to depend on several factors that cause potential hazardous effects including generation of reactive oxygen species (Nel et al. 2006; Sumi and Chitra 2017c), cytotoxicity (Oberdorster et al. 2006), respiratory and cardiovascular diseases (Moore 2006), and carcinogenicity (Armstrong et al. 2004). Nanomaterials can be transferred from lowest to the highest trophic levels of the food chain and this has generated significant interest in aquatic toxicology, since fish are the key element of aquatic ecosystems that convey the reliable effects to other vertebrates including humans (Holbrook et al. 2008).
In a study on male Fischer rats, water-soluble fullerenes were reported to penetrate the blood–brain barrier, and 90% of the particles retained in the body for a long period (Yamago et al. 1995). Accumulation of fullerene in kidney, spleen and liver of rats indicated the translocation and accumulation of nanomaterials, which were enabled through the blood vascular system (Chen et al. 1998). A similar study reported that the penetration of fullerene C60 nanoparticles from the gastrointestinal tract of rats into the bloodstream occurs after intragastrical administration, which then translocated into liver, lungs, spleen and kidneys without showing lethality in the exposed animal (Hendrickson et al. 2014). Rats exposed to C60 fullerene nano- and microparticles for 10 days showed alteration in hematological parameters along with a prominent increase in the pulmonary deposition of nanoparticles than the microparticles (Baker et al. 2008). However, oral administration of polyvinylpyrrolidone-wrapped fullerene C60 for 7 days in mice did not exhibit any effects in hematological parameters (Yamashita et al. 2013). Similarly, a dietary exposure of the rainbow trout fish, Oncorhynchus mykiss to fullerene C60 for 6 weeks showed no effect on growth or hematology (Fraser et al. 2011). A previous report from our laboratory showed that C60 fullerene induced oxidative imbalance in gonads and caused reproductive dysfunction in the freshwater fish, Anabas testudineus (Sumi and Chitra 2019). The available literature provides both safety and toxic effects of fullerene C60 in various animal models. Although there are several toxicological endpoints to detect the adverse effects of fullerene, studies on hematological parameters are used as a valuable diagnostic tool to assess the health and physiological status of an organism against the injury, lesion, deficit, stress and disease condition (Tavares-Dias and Moraes 2007). As there is lack of knowledge on the effect of fullerene C60 nanomaterial in the complete blood count and plasma chemistry profile of teleostian fish, the present study was conducted to address the gap of knowledge in behavioral and hematological parameters of the freshwater fish, Anabas testudineus. Blood profiling is considered as an essential tool in establishing the challenges exhibited by fish to overcome toxicant-related stress conditions. Therefore, the study performed provides new insights into the sublethal toxicity of fullerene C60 nanomaterials in the aquatic ecosystems.
Materials and methods
Model organism
Healthy freshwater fish, Anabas testudineus of weight 11 ± 1 g and length 8 ± 1 cm were collected from Pulimugham hatcheries, Alappuzha district, Kerala, India. Fish were transported to the laboratory in well-aerated polythene bags with least disturbance and acclimatized in the laboratory conditions for 2 weeks prior to the experiment. During acclimatization, fish were properly fed three times a day with standard fish food pellets and maintained in dechlorinated water under a photoperiod of 12 h light: 12 h dark and the health of fish was continuously monitored. The test tanks were covered with mono-filament netting to avoid the fish from jumping out of the experimental tanks. The preliminary tests were carried out in tap water following standardized procedures as prescribed by the APHA guidelines (1998) thus maintaining the water temperature at 28 ± 2 °C, pH 6.5–7.5 and oxygen saturation (70 and 100%) throughout the experiment durations.
Test chemicals
Fullerene C60 (purity > 98%, Product Number: 483036, CAS No. 99685-96-8) was purchased from Sigma-Aldrich, Germany and was dispersed in dimethyl sulfoxide (DMSO) (Himedia, CAS No. 67-68-5) by sonicating for 3 h to ensure an even size distribution. Previous studies reported from our laboratory (Asifa and Chitra 2017; Sumi and Chitra 2017c, 2019), and other studies reported in zebrafish in vivo (Usenko et al. 2007) and embryonic stem cells in vitro (Adler et al. 2006) have suggested that the use of 1% DMSO, as vehicle solvent, did not elicit toxic effects in exposed fishes, and therefore, that concentration was used in the present study. The concentration of nanomaterial selected in the present study was chosen to evaluate the sublethal effects of fullerene C60 in the fish model, and not to mimic the environmental concentration and its exposure scenario. All other chemicals used in the present study were of analytical grade, which were purchased from local commercial sources.
Characterization
Fullerene C60 nanomaterial was characterized by X-ray diffraction (XRD) with Cu Kα radiation exposed at 1.54 α wavelength, 40 kV and 30 mA current. The analysis was done using PanAlytical X’pert-PRO MRD diffractometer system, Eindhoven, Netherlands. Further, the average crystalline size was determined by Scherrer’s formula, D = 0.94 kλ/β cos θ, where D, K, λ, β and θ are the average crystal size, Scherrer coefficient (0.94), X-ray wavelength, Bragg’s angle and the full width at half maximum in radian, respectively. The size and morphological nature of the fullerene C60 nanomaterial were determined using a high-resolution transmission electron microscope (JEOL-JEM-200 CX) having 0.23 nm point-to-point resolution, 0.14 nm lattice resolution and 2000 X–1500000 X magnification.
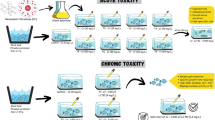
Acute toxicity test
Acute toxicity of fullerene C60 was tested preliminarily to find the median lethal concentration (semi-static; 96 h-LC50) according to the guideline of OECD 203 (OECD 1992). Fish were stopped from feeding 24 h prior to the experiment to reduce the contamination of the test solution with excess food and feces. Different concentrations of the test chemical (20, 30, 40, 50, 60, 70 and 80 mg/L) were added to separate tanks having a capacity of 50 L holding ten healthy fish per tank maintaining triplicates under the same conditions. A control tank without toxicant and solvent, and vehicle tank with 1% DMSO were also maintained along with the test tanks. The mortality and behavioral changes of fish from the controls and treatment tanks were recorded at every 24 h up to 96 h duration. The concentration at which 50% mortality of fish represent the median lethal concentration (96 h-LC50) was analyzed using the Probit tool of regression analysis with a confidence limit of 5% level (Finney 1971).
Sublethal test
After estimating the median lethal concentration (50 mg/L), two sublethal concentrations, i.e., one-tenth (5 mg/L) and one-fifth of the LC50 (10 mg/L) were selected for further analysis and the fish were grouped as follows:
Group I: Negative control group (without solvent and toxicant).
Group II: Vehicle control group (1% DMSO).
Group III: Treatment groups.
Group IIIA: Fullerene C60 at 5 mg/L exposed for 24, 48, 72 and 96 h.
Group IIIB: Fullerene C60 at 5 mg/L exposed for 7, 15, 30 and 60 days.
Group IIIC: Fullerene C60 at 10 mg/L exposed for 24, 48, 72 and 96 h.
Group IIID: Fullerene C60 at 10 mg/L exposed for 7, 15, 30 and 60 days.
Monitoring of fish behavior
The behavioral modifications were monitored continuously for 30 min in each experimental tank at every 24 h interval up to 96 h in acute toxicity tests and short-term sublethal exposure groups. Similarly, the changes in the behavioral pattern were observed at 24 h interval for long-term sublethal exposure groups for 60 days. The observation time changed from morning to evening to avoid the changes in the behavior due to the diurnal fluctuations. The behavioral changes observed in the treatment groups were noted and compared with the control groups.
Histomorphology of intestine
After the end of the 96 h and 60 days of both sublethal treatment groups, fish were sacrificed and the intestines of the fish were dissected out, cleaned with physiological saline (0.9%) and finally fixed in buffered formalin (10%) for 24–48 h. The tissues were then dehydrated with ascending grades of alcohol, cleared in xylene and then dipped in molten paraffin wax for an hour for complete infiltration so as to prepare the tissue blocks. Serial sections were made using a rotary microtome with a thickness of 4–6 μm and the sections were double stained with haematoxylin and eosin, and finally mounted with DPX (Roberts and Smail 2001). The slides were observed under the Carl Zeiss Axioscope-2 plus Trinocular Research Microscope and microphotographs were taken using a Canon shot camera fitted to the microscope.
Hematological and biochemical parameters
Fish were exposed to sublethal concentrations of fullerene C60 for short-term (24, 48, 72 and 96 h) and long-term (7, 15, 30 and 60 days) durations. At the end of every tenure of treatments, fish were gently removed from the experimental tanks for hematological and biochemical analyses. Anesthesia was not given to fish before collecting the blood samples, because it could affect the blood parameters and also hemolyze the tissues (McKnight 1966).
RBC and WBC counts
Blood was collected by cardiac puncture and the dilutions were made with appropriate diluting fluids such as Hayem’s (for RBC) and Turk’s fluid (for WBC) and immediately used for the estimation of erythrocyte and leukocyte count using the Neubauer counting chamber (Rusia and Sood 1992). The RBC and WBC counts were expressed as 106/cu. mm and 103/cu. mm, respectively, which were obtained by the given calculations:
where the dilution factor = 200, the area counted = 0.2 square mm and the depth of fluid = 0.1 mm.
where the dilution factor = 20, the area counted = 4 square mm and the depth of fluid = 0.1 mm.
Hemoglobin content and packed cell volume
The hemoglobin content was measured by the cyanmethaemoglobin method according to Blaxhall and Daisley (1973). Briefly, 20 µL of blood was mixed thoroughly with 4 mL of Drabkin’s reagent and kept for 10 min to allow complete conversion of hemoglobin into cyanmethaemoglobin, which was measured spectrophotometrically at 540 nm, and was expressed as g/dL. The packed cell volume was measured using a micro-haematocrit according to Hesser (1960), and was expressed in percentage (%).
Collection of blood serum
The blood sample collected from fish, without adding anticoagulant, was kept undisturbed for 30–60 min at room temperature to clot, which was then centrifuged at 1000g for 15 min to obtain the serum, and stored at − 80 °C until biochemical analysis were performed.
Total protein
The total protein was analyzed by Lowry et al. (1951) using bovine serum as standard. An aliquot of serum is mixed with 4.5 mL of alkaline copper reagent and kept at room temperature for 10 min. After incubation, the sample was mixed with 0.5 ml of Folin–Ciocalteau reagent and kept at room temperature for 20 min, then a blue color developed which was measured at 620 nm and the result was expressed as mg/mL.
Albumin and globulin
Albumin was estimated with the bromocresol green binding method described by Doumas et al. (1997). Briefly, 5 μL of sample was added to bromocresol green reagent (0.6 mM), and allowed to stand at room temperature for 10 min, and the absorbance was measured at 620 nm using UV–visible spectrophotometer. The concentration of serum albumin was expressed in mg/mL.
Serum globulin was calculated from the known value of protein and albumin concentration using the formula:
Glucose
Glucose was estimated according to Trinder (1969). Briefly, the serum sample was mixed with sodium sulfate–zinc sulfate reagent, centrifuged at 800g for 5 min, and the supernatant collected was mixed with 5 mL of glucose oxidase reagent. The reagent mixture was incubated at room temperature for 10 min, and absorbance was measured at 520 nm in UV–visible spectrophotometer and the unit was expressed in mg/dL.
Alanine aminotransferase
The activity of serum alanine aminotransferase was measured according to the method of Reitman and Frankel (1957). Briefly, the reaction mixture containing DL-α-alanine (0.2 M) and 2-oxoglutarate (2 mM) dissolved in phosphate buffer (0.1 M; pH 7.4) was vortexed and incubated at 37 °C for 1 h. After incubation, 250 µL of 2,4-dinitrophenyl hydrazine was added and incubated at room temperature for 20 min. Finally, sodium hydroxide (0.4 N) was added to stop the reaction, mixed and incubated at room temperature for 10 min. The absorbance was read at 510 nm against the blank. A standard calibration was prepared using different concentrations of sodium pyruvate. The results were expressed as µM pyruvate formed per mL.
Aspartate aminotransferase
The activity of aspartate aminotransferase was assayed by the method as described by Reitman and Frankel (1957). The reaction mixture containing aspartate (0.1 M) and 2-oxoglutarate (2 mM) dissolved in phosphate buffer (0.1 M; pH 7.4) was vortexed and incubated at 37 °C for 1 h. After incubation, 250 µL of 2,4-dinitrophenyl hydrazine was added and incubated at room temperature for 20 min. Finally, to stop the reaction, sodium hydroxide (0.1 N) was added, mixed and incubated at room temperature for 10 min. The absorbance was read at 510 nm against the blank. A standard calibration was prepared using different concentrations of sodium pyruvate and the results were expressed as μM glutamate formed/ml protein.
Acetylcholinesterase in brain
The supernatant of brain tissue homogenate (1% w/v) from all durations of both sublethal concentrations was used to analyze the activity of acetylcholinesterase enzyme (Ellman et al. 1961). Briefly, the supernatant of tissue homogenate in phosphate buffer (0.1 M, pH 8.0) was dissolved in sodium bicarbonate and dithiobisnitrobenzoic acid (DTNB; 0.01 M). The enzyme activity was calculated by measuring the yellow color indicator produced from thiocholine on reaction with dithiobisnitrobenzoate ion. The absorbance was measured at 405 nm against the reagent blank and the activity of enzyme was expressed as nmol acetylthiocholine hydrolysed/min/mg protein.
Statistical analysis
The median lethal concentration was analyzed by Probit regression analysis with 95% confidence limit, and then the correlation between the mortality versus concentration was plotted for the best fit line.
The hematological and biochemical data were analyzed using the statistical package SPSS 21.0 by one-way analysis of variance (ANOVA). The differences between the mean of control versus treatment groups were determined using the Duncan’s multiple range as the post hoc test, in which the significance level was denoted in asterisk as P < 0.05 against the control groups. Data were expressed as mean ± standard deviation (SD) for ten fish in each group.
Results
Characterization of fullerene C60 using X-ray diffraction (XRD) and high-resolution transmission electron microscope (HR-TEM)
The purity of fullerene C60, obtained from the manufacturer was analyzed using the X-ray diffraction technique (Fig. 1). The face-centered cubic (FCC) lattice of the fullerene C60 demonstrated three distinct reflections showing diffraction peaks approximately at 10°, 17° and 20° angles corresponding to 111, 220 and 311 crystal planes of FCC, respectively. All the peaks obtained were distinct without any peaks for impurities, and the XRD results of fullerene C60 were analogous to that of the standard cards (JCPDS file No.44-0558). The size of the crystalline form of fullerene C60 confirmed by XRD was further estimated using the Scherrer’s formula measuring the full width at half maximum (FWHM) of the Bragg’s angle, and the average size calculated was 34.95 nm.
High-resolution transmission electron microscopy (HR-TEM) was used to analyze the size and morphological structure of fullerene C60 (Fig. 2). The average particle size obtained ranged between 30 and 60 nm, and the selected area electron diffraction (SAED) pattern of fullerene C60 used to identify the morphology of nanomaterial confirmed the crystalline structure, which was in agreement with the XRD results (Fig. 2).
Median lethal concentration (96 h-LC50) of fullerene C60
No mortality was observed in the control and vehicle control groups throughout the experiment durations. Exposure to fullerene C60 at 20 mg/L concentration did not show mortality for 96 h. Fish when exposed to 30, 40, 50 and 60 mg/L concentrations showed 10, 40, 50 and 60% mortality, respectively. When the concentration of fullerene C60 was increased to 70 and 80 mg/L, 80% mortality was shown at 96 h and 72 h, respectively (Table 1). The graph of concentration against mortality plotted using MS Excel showed a high degree of positive correlation, r = + 0.978 (Fig. 3). The median lethal concentration, i.e., 96 h-LC50 of fullerene C60 calculated using Probit analysis was 50 mg/L in concentration. Two sublethal concentrations, i.e., 5 mg/L (one-tenth of 96 h-LC50) and 10 mg/L (one-fifth of 96 h-LC50) were chosen as sublethal concentrations to evaluate the toxicity of the nanomaterial.
Fullerene C60-induced behavioral modifications
Fish exposed to fullerene C60 showed behavioral modifications in both the acute toxicity and sublethal tests when compared with the control groups. In the acute toxicity test, soon after fullerene C60 exposure, disruption was seen in the schooling behavior, while surfacing and air engulping increased. After 24 h of exposure, the fish showed vigorous swimming activity, banged with each other and also the walls of the treatment tanks, and the movement gradually declined leading to lethargy. Slight hemorrhage, increased mucous secretion and loss of equilibrium were also observed at the end of exposure period immediately before the mortality. The death of the fish was confirmed by the cessation of the opercular movements and the failure of animal to respond to gentle prodding with a glass rod.
Sublethal exposure groups also showed prominent behavioral modifications particularly in the long-term exposure groups. The main abnormalities included the fish preferring to stay at the bottom of the tank all the time but intermittently surfacing to engulp atmospheric air. The morphological changes observed were slight hemorrhage on the body surface, increased descaling and mucous secretion and interestingly, black aggregates were observed in the intestinal lumen of fullerene C60-exposed fish when dissecting the animal (Fig. 4). However, no mortality was observed throughout the treatment period. Fullerene C60-induced intestinal abnormalities were further confirmed by histological analysis and the changes in the behavioral pattern were compared for alteration in the activity of acetylcholinesterase enzyme in brain tissue of sublethal exposed groups and with the control groups.
Histopathological changes in the intestine of fish
The transverse section of intestine of fish obtained from the control and vehicle control groups showed normal histoarchitecture with serosa, muscularis, lamina propria and mucosal layer. Several villous occupied the mucosal layer with the basal lining of tall columnar cells and centrally located nuclei. Mucous-secreting goblet cells were more prominent in continuity with the intestinal lumen (Fig. 5a). The intestine of fish exposed to sublethal concentrations of fullerene C60 for 96 h and 60 days showed significant alterations including severe degeneration of lamina propria, degeneration of columnar epithelial cells and increased number of prominent mucous cells (Fig. 5b). The severity observed in the treatment groups were found to increase with increase in the time and concentration of fullerene.
Photomicrographs of the intestine of Anabas testudineusa–d (H&E). a, b control; c, d vehicle control (1% DMSO) showing normal histoarchitecture with serosa (S), muscularis (ML), submucosa (SM), mucosa with lamina propria (LP), columnar epithelial cells (CEC) and intestinal villi. b Photomicrographs of the intestine of Anabas testudineus exposed to fullerene C60e–l (H&E). e, f 5 mg/L-C60 fullerene-exposed group for 96 h; g, h 10 mg/L-C60 fullerene-exposed group for 96 h; i, j 5 mg/L-C60 fullerene-exposed group for 60 days; k, l 10 mg/L-C60 fullerene-exposed group for 60 days. The treatment groups showing shortened and fused intestinal villi with disintegration of the submucosa (SM), lamina propria (LP) and columnar epithelial cells (→), increased number of gastric glands (G) and vacuole formation (V)
Alteration in the brain acetylcholinesterase enzyme activity after sublethal exposure to fullerene C60
Exposure of fish to both sublethal concentrations of fullerene C60 in short-term treatment groups showed significant (P < 0.05) decrease in the activity of acetylcholinesterase enzyme only after 96 h in the brain tissue (Fig. 6). However, in the long-term exposure groups, there was a significant (P < 0.05) decrease in the activity of acetylcholinesterase enzyme in a time-dependent manner when compared to the control brain tissues (Fig. 6).
Hematological changes after sublethal exposure to fullerene C60
Short-term exposure to both sublethal concentrations of fullerene C60 did not show any significant changes in the erythrocyte count, packed cell volume and hemoglobin content, except at 10 mg/L concentration exposed for 96 h (Figs. 7, 8, 9). However, long-term exposure to fullerene C60 showed a time-dependent significant (P < 0.05) reduction in the erythrocyte count, packed cell volume and hemoglobin content in a time-dependent manner (Figs. 7, 8, 9). Fullerene C60 exposure significantly (P < 0.05) decreased the leukocyte count after 96 h at 5 mg/L concentration, and after 72 h at 10 mg/L concentration in a time-dependent manner (Fig. 10).
Changes in serum biochemistry after sublethal exposure to fullerene C60
Exposure to fullerene C60 at 5 and 10 mg/L concentrations showed significant (P < 0.05) reduction in serum total protein and serum albumin after 96 h of high sublethal concentration, i.e., 10 mg/L (Figs. 11 and 12). While the serum total protein and serum albumin showed significant (P < 0.05) reduction after 7 days at 5 mg/L, i.e., low sublethal concentration (Figs. 11 and 12). Serum globulin showed significant (P < 0.05) increase only after 96 h at 10 mg/L concentration, whereas a low sublethal concentration did not show any significant changes (Fig. 13). In the long-term exposure groups, there was a significant (P < 0.05) increase in the serum globulin after 7 and 15 days, while it declined significantly (P < 0.05) after 30 and 60 days of low sublethal exposure group (Fig. 13). Fullerene C60 exposed at 10 mg/L concentration showed a time-dependent significant (P < 0.05) reduction in the serum globulin of long-term exposure groups (Fig. 13). Exposure to fullerene C60 at 5 mg/L concentration showed a significant (P < 0.05) and time-dependent increase in the serum glucose after 96 h onwards, while at 10 mg/L concentration, the level of serum glucose was found to be increased significantly (P < 0.05) after 72 h in a time-dependent manner (Fig. 14). The activities of alanine and aspartate aminotransferases showed significant (P < 0.05) increase in a time-dependent manner in the long-term exposure groups at both sublethal concentrations (Figs. 15 and 16). However, no significant changes in the activities of enzymes were noted in short-term exposure groups other than a significant (P < 0.05) increase after 96 h of fullerene C60 exposure at 10 mg/L concentration (Figs. 15 and 16).
Discussion
The present study showed a negative impact of fullerene C60 on the behavior and hematology of the freshwater fish, Anabas testudineus. The unique physico-chemical properties of fullerene C60 inspired researchers in nanotechnology, and simultaneously, the proven toxic impact gained the attention of the nanotoxicologists. The anticipated production of nanomaterials on a large scale and their possible release into the environment has led to widespread concerns about the potential toxicity. Several literatures have stated that fullerenes have an impact on aquatic life such as algae, fungi, bacteria, aquatic invertebrates, fishes and other higher aquatic vertebrates, though relatively small effects were reported at environmentally relevant concentrations. In the natural environment, fullerenes are expected to settle in the bottom sediments even up to 100 mg/L concentration for months or end up in the lipids of aquatic organisms. The present study focused on the toxic effect of fullerene C60 in the fish, Anabas testudineus by applying a sublethal concentration rather than the environmental relevant concentration. The water solubility of fullerene C60 is very peculiar showing a dualistic character of insoluble property to water and other polar solvents having less than 10−9 mg/L solubility (Ruoff et al. 1993). However, fullerenes have a characteristic to transform the poor solubility property by modifying as water-soluble colloidal clusters or agglomerates, either by vigorous stirring or sonication in the laboratory, or by natural processes like water flow and mixing in the natural environmental condition (Scharff et al. 2004). Fullerene C60 was subjected to sonication in the present study so as to improve homogeneity and stability of the nanosuspension. Homogeneity brings a much narrower particle size distribution, whereas stability prevents from bottom sediment settlement thereby ensuring uniform dispersion of fullerene C60 nanomaterial in the exposed experimental tanks.
Prior to the initiation of any toxicological experiments, it is always necessary to understand the characterization of the nanomaterial, since the particle size and structure influence the biological or toxicological effects. In the present study, fullerene C60 was characterized by the X-ray diffraction technique, and the size and morphological structure further confirmed using high-resolution transmission electron microscopy was found to be an average size of 34.95 nm, as compared to the manufacturer’s data. The toxic nature of fullerene C60 nanomaterial was initially determined by the conventional acute toxicity test conducted for 96 h using eight different concentrations of nanomaterial in a semi-static condition maintaining temperature, light intensity and other factors at a constant range. The median lethal concentration that killed 50% of the test animals was 50 mg/L, and the detected concentration was the expected level of concentration that could cause adverse effects in the field population. In another study, the acute toxicity of fullerene C60 was determined in Daphnia magna by preparing the test solution in two methods like sonication and dissolution in tetrahydrofuran and filtration. The study has reported that acute toxicity (48 h-LC50) of filtered C60 in tetrahydrofuran was 460 ppb, whereas the acute toxicity of sonicated C60 was only at 7.9 ppm (Lovern and Klaper 2006). Similarly, exposure of tetrahydrofuran-nC60 at 0.5 ppm concentration in the adult male fathead minnow, Pimephales promelas showed 100% mortality within the initial 6–18 h, while the same concentration when exposed for 48 h after sonication did not show significant toxicity (Zhu et al. 2006) thereby proving the risk of using tetrahydrofuran as solvent. In the present study, dimethyl sulfoxide (DMSO; 1%) was used as vehicle solvent to prepare uniform suspension of the test solution by sonication and the results presented in this study showed no toxicity in the vehicle control group. Acute toxicity of aqueous suspension of fullerene C60 has also been reported in other aquatic organisms such as Oryzias latipes, Daphnia magna and Pseudokirchneriella subcapitata at greater than 2.15, 2.25 and 2.27 mg/L concentrations, respectively (Seki et al. 2008). The results of the reported studies were not comparable to the present observation, as the median lethal concentration of sonicated fullerene C60 was 50 mg/L, stating that either surface modification by sonication or less toxicity of DMSO could have contributed conflict of nanomaterial toxicity in the test organism, Anabas testudineus.
The behavior of an organism serves as a link between the ecology and physiology, hence the behavioral alterations can be used as an end point to study the adverse effects of toxicants. In the present investigation, the fish, Anabas testudineus were exposed to fullerene C60 which resulted in behavioral alterations including disruption of schooling, increased mucous secretion, descaling, increase in surfacing and altered swimming activity. The disruption in the schooling behavior was observed as the lethal and sublethal effects of nanomaterial which resulted in the change of normal swimming activity (Murthy 1987). Similarly, the increased surfacing behavior of fish during the long-term exposures of fullerene C60 might be due to treatment-related hypoxia and decreased oxygen level in the treatment tanks. Fish exposed to fullerene C60 also preferred to stay at the bottom of the tank for sometime, which was in agreement with another study when fathead minnow, Pimephales promelas, exposed to 20 ppm of hydroxylated fullerene by intraperitoneal injection showed lethargy, clustered schooling behavior and spent more time at the aquarium’s bottom (Jovanovic et al. 2014). Exposure to C60 carbon nanoparticles at 1 and 2 mg/L concentrations in adult zebrafish showed behavioral alterations such as hypoactivity, reduced aggression or fear, anxiety and impairment in circadian rhythm (Sarasamma et al. 2018). Fullerene also induced behavioral changes in Daphnia magna as observed with increased hopping frequency, heart rate, appendage movement and curling of post-abdominal claw (Lovern et al. 2007). In another study, water-stirred C60 and sonicated carboxylic acid-functionalized fullerenes have been shown to alter swimming behaviors which seriously affected other vital behaviors such as predator avoidance and predation risk in Daphnia magna (Brausch et al. 2011).
Fullerene C60-exposed fishes showed thick mucous deposition throughout the body surface, which is considered as a protective mechanism to avoid the absorption of nanomaterial through the skin. In addition, descaling was also observed after chronic exposure of the treated fish, and this could be due to the passage of lipophilic nanomaterial through the thick mucous of skin. It may ultimately lead to skin and muscle tissue damage in the fish as observed in our previous study in the fish, Pseudetroplus maculatus (Sumi and Chitra 2017a). In the present study brain acetylcholinesterase enzyme activity was used as a biomarker of neurotoxicity which was found to be associated with behavioral alterations. Chronic exposure of fullerene C60 inhibited acetylcholinesterase enzyme in a time-dependent manner and this could be the reason for the abnormal locomotion and loss of equilibrium of exposed fish. Accumulation of fullerene C60 in the form of black aggregates was observed in the intestinal lumen of exposed fish indicating adverse effects of nanomaterial on the histoarchitecture of intestine. Histopathological alterations such as degeneration in the columnar epithelial cells, lamina propria and stratum compactum along with increased number of mucous cells observed in this study further confirmed sublethal toxicity of fullerene C60, which in turn could obstruct the intestinal absorption. The histomorphological changes were in agreement with another study on exposure to hydroxylated fullerene at 20 ppm concentration for 72 h has been shown to cause lesions in the intestinal lumen of fathead minnow, Pimephales promelas (Jovanovic et al. 2014). Accumulation of single walled nanotubes in the gut tract after 45 min and 1 h of exposure, which was followed by precipitated clumps of nanotubes after 20 h of exposure, has been reported in Daphnia magna (Roberts et al. 2007).
There arises a serious concern that after accumulation of C60 fullerene in intestine, it could cross the intestinal barriers to reach other vital organs through blood circulation, therefore, analysis of hematological parameters is the most significant aspect. Fullerene nanoparticles have been known to penetrate from the gastrointestinal tract into the bloodstream and translocate into secondary organs (Hendrickson et al. 2015). In the present study, hematological alterations were examined in the fish, Anabas testudineus exposed to sublethal concentrations of fullerene C60. A significant reduction in erythrocyte and leukocyte counts, hemoglobin content and packed cell volume observed in a time-dependent manner reflected the anemic state of fish as a result of fullerene C60-induced haemolysis. Changes in the hematological profile indicated the stress condition of fish on exposure to fullerene nanomaterial, which could obviously affect the normal physiology and metabolism (Kaya et al. 2014). In a study, C60 nanoparticles prepared by solvent exchange method (nC60THF) caused shrinkage, crenation and, eventually, hemolysis of human RBC and the erythrocyte damage that was mediated through oxidative stress (Trpkovic et al. 2010). Infiltration of C60 nanoparticle has reported to affect the mechanical properties of RBC membrane and weakened the tensile resistance of lipid bilayers in human RBC (Zhang et al. 2013).
The haemolytic property of fullerene C60 was further confirmed by serum biochemical analysis, which could reflect the physiological state of the fish, and hence was used as an important tool to diagnose the sublethal toxicity of nanomaterial. Fullerene C60 exposure at sublethal concentrations showed reduction in serum total protein, albumin and globulin with an increase in the level of glucose, which was concentration and time dependent. The decline in the levels of serum total protein and albumin could occur due to several reasons including protein degradation, utilization for the metabolic processes, inhibited hepatic synthesis of blood protein and the destruction of subcellular protein synthesizing structures (Fontana et al. 1998). The ability of nanoparticle to bind strongly with the protein may contribute to loss of protein function and also lead to protein denaturation (Roach et al. 2005). The binding affinity of one of the nanoparticles, carboxyfullerene with different proteins such as fullerene specific antibody, HIV protease, bovine serum albumin and human serum albumin has been documented using several docking models (Benyamini et al. 2006). The rise in the level of serum glucose observed in the present study indicated the release of glucose from stored glycogen owing to the toxicant-induced high energy demand. A similar observation was reported on inhalation of fullerene nanoparticles in the serum of rat (Baker et al. 2008). The activities of serum transaminases namely alanine and aspartate aminotransferases were recorded as the conventional indicator of liver damage in fish, exposed at two sublethal concentrations of fullerene C60 nanomaterial. Serum transaminases activities are found to be in a high level in heart, liver, skeletal muscle, kidney and erythrocytes and any damage to these organs raises the level of enzyme activities in blood serum (Zilva et al. 1992). The elevated activities of transaminase enzymes in the serum reflected membrane damage of any vital organs, which leads to extensive leakage of enzymes into the blood serum. The findings in the present study gain conformity with the subsequent tissue damage reported after fullerene C60 exposure in the freshwater fish, Pseudetroplus maculatus (Sumi and Chitra 2017b). The overall alteration in the hematological and biochemical parameters indicated toxic metabolic stress induced by fullerene C60 in the fish, Anabas testudineus.
Conclusions
The present findings proved that sublethal exposure to fullerene C60 resulted in adverse effects in the freshwater fish, Anabas testudineus as evident by the prominent behavioral, hematological and biochemical changes, and pathological alteration in intestinal tissue. The translocated fullerene from the intestine into blood elicited significant toxicity which advocated changes in the hematology and serum biochemistry of the fish. The release of fullerene C60 into the aquatic ecosystem even at a sublethal concentration could seriously affect the health and existence of the aquatic organisms. Therefore, the discharge of fullerene C60 from the manufacturing sites or from industries into the environment, especially in aquatic ecosystems, should be strictly monitored.
References
Adler S, Pellizzer C, Paparella M, Hartung T, Bremer S (2006) The effects of solvents on embryonic stem cell differentiation. Toxicol In Vitro 20:265–271. https://doi.org/10.1016/j.tiv.2005.06.043
APHA (1998) Standard methods for the examination of water and wastewater, 20th edn. American Public Health Association, American Water Works Association and Water Environmental Federation, Washington, DC
Armstrong B, Hutchinson E, Unwin J, Fletcher T (2004) Lung cancer risk after exposure to polycyclic aromatic hydrocarbons: a review and meta-analysis. Environ Health Perspect 112:970–978. https://doi.org/10.1289/ehp.6895
Asifa KP, Chitra KC (2017) Hepatic biotransformation of chlordecone and induction of hepatotoxicity in the cichlid fish, Pseudetroplus maculatus (Bloch, 1795). J Appl Res 3:521–526
Astefanei A, Nunez O, Galceran MT (2014) Analysis of C60-fullerene derivatives and pristine fullerenes in environmental samples by ultrahigh performance liquid chromatography-atmospheric pressure photoionization-mass spectrometry. J Chromatogr A 1365:61–71. https://doi.org/10.1016/j.chroma.2014.08.089
Baker GL, Gupta A, Clark ML, Valenzuela BR, Staska LM, Harbo SJ, Pierce JT, Dill JA (2008) Inhalation toxicity and lung toxicokinetics of C60 fullerene nanoparticles and microparticles. Toxicol Sci 101:122–131. https://doi.org/10.1093/toxsci/kfm243
Becker L, Bada JL, Winans RE, Hunt JE, Bunch TE, French BM (1994) Fullerenes in the 1.85-billion-year-old Sudbury impact structure. Science 265:642–645. https://doi.org/10.1126/science.11536660
Benn TM, Westerhoff P, Herckes P (2011) Detection of fullerenes (C60 and C70) in commercial cosmetics. Environ Pollut 159:1334–1342. https://doi.org/10.1016/j.envpol.2011.01.018
Benyamini H, Shulman-Peleg A, Wolfson HJ, Belgorodsky B, Fadeev L, Gozin M (2006) Interaction of C60-fullerene and carboxyfullerene with proteins: docking and binding site alignment. Bioconjugate Chem 17:378–386. https://doi.org/10.1021/bc050299g
Blaxhall PC, Daisley KW (1973) Routine haematological methods for use with fish blood. J Fish Biol 5:771–781. https://doi.org/10.1111/j.1095-8649.1973.tb04510.x
Brant J, Lecoanet H, Wiesner MR (2005) Aggregation and deposition characteristics of fullerene nanoparticles in aqueous systems. J Nanopart Res 7:545–553. https://doi.org/10.1007/s11051-005-4884-8
Brausch KA, Anderson TA, Smith PN, Maul JD (2011) The effect of fullerenes and functionalized fullerenes on Daphnia magna phototaxis and swimming behavior. Environ Toxicol Chem 30:878–884. https://doi.org/10.1002/etc.442
Chen HHC, Yu C, Ueng TH, Chen S, Chen BJ, Huang KJ, Chiang LY (1998) Acute and subacute toxicity study of water-soluble polyalkylsulfonated C60 in rats. Toxicol Pathol 26:143–151. https://doi.org/10.1177/019262339802600117
Cheng X, Kan AT, Tomson MB (2004) Naphthalene adsorption and desorption from aqueous C60 fullerene. J Chem Eng Data 49:675–683. https://doi.org/10.1021/je030247m
Coro J, Suarez M, Silva LSR, Eguiluz KIB (2016) Fullerene applications in fuel cells: a review. Int J Hydrog Energy 41:7944–17959. https://doi.org/10.1016/j.ijhydene.2016.08.043
Daly TK, Buseck PR, Williams P, Lewis CF (1993) Fullerenes from a fulgurite. Science 259:1599–1601. https://doi.org/10.1126/science.259.5101.1599
Dhawan A, Taurozzi JS, Pandey AK, Shan W, Miller SM, Hashsham SA, Tarabara VV (2006) Stable colloidal dispersions of C60 fullerenes in water: evidence for genotoxicity. Environ Sci Technol 40:7394–7401. https://doi.org/10.1021/es0609708
Doumas BT, Watson AW, Biggs HG (1997) Albumin standards and the measurement of serum albumin with bromocresol green. Clin Chim Acta 258:21–30. https://doi.org/10.1016/s0009-8981(96)06447-9
Dugan LL, Turetsky DM, Du C, Lobner D, Wheeler M, Almli CR, Shen CK-F, Luh T-Y, Choi DW, Lin T-S (1997) Carboxyfullerenes as neuroprotective agents. Proc Natl Acad Sci USA 94:9434–9439. https://doi.org/10.1073/pnas.94.17.9434
Ellman GL, Courtney KD, Anders V, Featherstone RM (1961) A new and rapid colorimetric determination of acetylcholinesterase activity. Biochem Pharmacol 7(2):88–95. https://doi.org/10.1016/0006-2952(61)90145-9
Fang PH, Chen F, Tao R, Ji B, Mu C, Chen E, He Y (2006) Fullerene in some coal deposits in China. Developments in fullerene science. Springer, Berlin, pp 257–266. https://doi.org/10.1007/1-4020-4135-7_12
Farre M, Perez S, Gajda-Schrantz K, Osorio V, Kantiani L, Ginebreda A, Barcelo D (2010) First determination of C60 and C70 fullerenes and N-methylfulleropyrrolidine C60 on the suspended material of wastewater effluents by liquid chromatography hybrid quadrupole linear ion trap tandem mass spectrometry. J Hydrol 383:44–51. https://doi.org/10.1016/j.jhydrol.2009.08.016
Finney DJ (1971) Probit analysis, 3rd (Ed.), Cambridge University Press, London, 333
Fontana L, Moreiram E, Torres M, Fernandez I, Rios A, de Medina FS, Gil A (1998) Dietary nucleotides correct plasma and liver microsomal fatty acids alterations in rats with liver cirrhosis induced by oral intake of thioacetamide. J Hepatol 28:662–669. https://doi.org/10.1016/s0168-8278(98)80291-2
Fortner JD, Lyon DY, Sayes CM, Boyd AM, Falkner JC, Hotze EM, Alemany LB, Tao YJ, Guo W, Ausman KD, Colvin VL, Hughes JB (2005) C60 in water: nanocrystal formation and microbial response. Environ Sci Technol 39:4307–4316. https://doi.org/10.1021/es048099n
Fraser TWK, Reinardy HC, Shaw BJ, Henry TB, Handy RD (2011) Dietary toxicity of single-walled carbon nanotubes and fullerenes (C60) in rainbow trout (Oncorhynchus mykiss). Nanotoxicology 5:98–108. https://doi.org/10.3109/17435390.2010.502978
Friedman SH, De-Camp DL, Sijbesma RP, Srdanov G, Wudl F, Kenyon GL (1993) Inhibition of the HIV-1 protease by fullerene derivatives: model building studies and experimental verification. J Am Chem Soc 115:6506–6509. https://doi.org/10.1021/ja00068a005.6/j.biomaterials.2014.03.071
Hansen SF, Michelson ES, Kamper A, Borling P, Stuer-Lauridsen F, Baun A (2008) Categorization framework to aid exposure assessment of nanomaterials in consumer products. Ecotoxicology 17:438–447. https://doi.org/10.1007/s10646-008-0210-4
Hendrickson OD, Morozova OV, Zherdev AV, Yaropolov AI, Klochkov SG, Bachurin SO, Dzantiev BB (2014) Study of distribution and biological effects of fullerene C60 after single and multiple intragastrical administrations to rats. Fuller Nanotub Car N 23:658–668. https://doi.org/10.1080/1536383x.2014.949695
Hendrickson OD, Morozova OV, Zherdev AV, Yaropolov AI, Klochkov SG, Bachurin SO, Dzantiev BB (2015) Study of distribution and biological effects of fullerene C60 after single and multiple intragastrical administrations to rats. Fuller Nanotub Car N 23:658–668. https://doi.org/10.1080/1536383x.2014.949695
Hesser EF (1960) Methods for routine fish hematology. Prog Fish-Cult 22:164–171. https://doi.org/10.1577/1548-8659(1960)22%5b164:mfrfh%5d2.0.co;2
Holbrook RD, Murphy KE, Morrow JB, Cole KD (2008) Trophic transfer of nanoparticles in a simplified invertebrate food web. Nat Nanotechnol 3:352–355. https://doi.org/10.1038/nnano.2008.110
Injac R, Prijatelj M, Strukelj B (2013) Fullerenol nanoparticles: toxicity and antioxidant activity. Methods Mol Biol 1028:75–100. https://doi.org/10.1007/978-1-62703-475-3_5
Jehlicka J, Ozawa M, Slanina Z, Obarsawa E (2000) Fullerenes in solid bitumens from pillow lavas of Precambrian age (MÍTOV, Bohemian Massif). Fuller Sci Technol 8:449–452. https://doi.org/10.1080/10641220009351424
Jovanovic B, Whitley EM, Palic D (2014) Histopathology of fathead minnow (Pimephales promelas) exposed to hydroxylated fullerenes. Nanotoxicology 8:755–763. https://doi.org/10.3109/17435390.2013.828794
Kaya H, Celik ES, Yılmaz S, Tulgar A, Akbulut M, Demir N (2014) Hematological, serum biochemical, and immunological responses in common carp (Cyprinus carpio) exposed to phosalone. Comp Clin Path 24:497–507. https://doi.org/10.1007/s00580-014-1930-x
Kroto HW, Heath JR, Obrien SC, Curl RF, Smalley RE (1985) C60 Buckminsterfullerene. Nature 318:162–163. https://doi.org/10.1038/318162a0
Lead JR, Wilkinson KJ (2006) Aquatic colloids and nanoparticles: current knowledge and future trends. Environ Chem 3:159–171. https://doi.org/10.1071/en06025
Lens M (2011) Recent progresses in application of fullerenes in cosmetics. Recent Pat Biotechnol 5:67–73. https://doi.org/10.2174/187220811796365707
Limbach LK, Li Y, Grass RN, Brunner TJ, Hintermann MA, Muller M, Gunther D, Stark WJ (2005) Oxide nanoparticle uptake in human lung fibroblasts: effects of particle size, agglomeration, and diffusion at low concentrations. Environ Sci Technol 39:9370–9376. https://doi.org/10.1021/es051043o
Lovern SB, Klaper R (2006) Daphnia magna mortality when exposed to titanium dioxide and fullerene (C60) nanoparticles. Environ Toxicol Chem 25:1132–1137. https://doi.org/10.1897/05-278R.1
Lovern SB, Strickler JR, Klaper A (2007) Behavioral and physiological changes in Daphnia magna when exposed to nanoparticle suspensions (Titanium Dioxide, NanoC60, and C60HxC70Hx). Environ Sci Technol 4:4465–4470. https://doi.org/10.1021/es062146p
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193:265–275
Lynch I, Dawson KA, Linse S (2006) Detecting cryptic epitopes created by nanoparticles. Sci Signal 327:14. https://doi.org/10.1126/stke.3272006pe14
Lyon DY, Alvarez PJJ (2008) Fullerene water suspension (nC60) exerts antibacterial effects via ROS-independent protein oxidation. Environ Sci Technol 42:8127–8132. https://doi.org/10.1021/es801869m
Lyon DY, Adams LK, Falkner JC, Alvarez PJJ (2006) Antibacterial activity of fullerene water suspensions: effects of preparation method and particle size. Environ Sci Technol 40:4360–4366. https://doi.org/10.1021/es0603655
McKnight IM (1966) A hematological study on the mountain whitefish, Prosopium williamsoni. J Fish Res Board Can 23:45–64. https://doi.org/10.1139/f66-005
Mohajeri A, Omidvar A (2015) Fullerene-based materials for solar cell applications: design of novel acceptors for efficient polymer solar cells-a DFT study. Phys Chem Chem Phys 17:22367–22376. https://doi.org/10.1039/c5cp02453f
Moore MN (2006) Do nanoparticles present ecotoxicological risks for the health of the aquatic environment? Environ Int 32:967–976. https://doi.org/10.1016/j.envint.2006.06.014
Murthy AS (1987) Sub lethal effects of pesticides on fish. Toxicity of pesticide to fish, vol 4. CRS Press, Boca Roton, pp 55–100
Nel A, Xia T, Madler L, Li N (2006) Toxic potential of materials at the nanolevel. Science 311:622–627. https://doi.org/10.1126/science.1114397
Nowack B, Bucheli TD (2007) Occurrence, behavior and effects of nanoparticles in the environment. Environ Pollut 150:622–627
Oberdorster E (2004) Manufactured nanomaterials (Fullerenes, C60) induce oxidative stress in the brain of juvenile largemouth bass. Environ Health Perspect 112:1058–1062. https://doi.org/10.1289/ehp.7021
Oberdorster E, Zhu SQ, Blickley TM, McClellan-Green P, Haasch ML (2006) Ecotoxicology of carbon based engineered nanoparticles: effects of fullerene (C60) on aquatic organisms. Carbon 44:1112–1120. https://doi.org/10.1016/j.carbon.2005.11.008
OECD (1992) Guidelines for the testing of chemicals 203. Fish, acute toxicity test. OECD, Paris. https://doi.org/10.1787/9789264069961-en
Pakarinen K, Petersen EJ, Leppeanen MT, Akkanen J, Kukkonen JVK (2011) Adverse effects of fullerenes (nC60) spiked to sediments on Lumbriculus variegatus (Oligochaeta). Environ Pollut 159:3750–3756. https://doi.org/10.1016/j.envpol.2011.07.014
Pan M, Annamalai KP, Tao Y (2015) Applications of nanocarbons in bio-medical devices. Rec Innov Chem Eng 8:67–74. https://doi.org/10.2174/2405520408666160509165356
Parthasarathy G, Srinivasan R, Vairamani M, Ravikumar K, Kunwar AC (1998) Occurrence of natural fullerenes in low grade metamorphosed Proterozoic shungite from Karelia, Russia. Geochim Cosmochim Acta 62:3541–3544. https://doi.org/10.1016/s0016-7037(98)00242-7
Reitman S, Frankel S (1957) A colorimetric method for the determination of serum glutamic-oxaloacetic and glutamic-pyruvic transaminases. Am J Clin Pathol 28:56–63. https://doi.org/10.1093/ajcp/28.1.56
Roach P, Farrar D, Perry CC (2005) Interpretation of protein adsorption: surface induced conformational changes. J Am Chem Soc 127:8168–8173. https://doi.org/10.1021/ja042898o
Roberts RJ, Smail DA (2001) Laboratory methods. In: Roberts RJ (ed) Fish pathology, 3rd edn. Saunders, London, pp 380–412
Roberts AP, Mount AS, Seda B, Souther J, Qiao R, Lin S, Ke PC, Rao AM, Klaine SJ (2007) In vivo biomodification of lipid-coated carbon nanotubes by Daphnia magna. Environ Sci Technol 41:3025–3029. https://doi.org/10.1021/es062572a
Rothen-Rutishauser BM, Schurch S, Haenni B, Kapp N, Gehr P (2006) Interaction of fine particles and nanoparticles with red blood cells visualized with advanced microscope techniques. Environ Sci Technol 40:4353–4359. https://doi.org/10.1021/es0522635
Ruoff RS, Tse DS, Malhotra R, Lorents DC (1993) Solubility of C60 in a variety of solvents. J Phys Chem 97:3379–3383. https://doi.org/10.1021/j100115a049
Rusia V, Sood SK (1992) Routine haematological tests. In: Mukerjee KL (ed) Medical laboratory technology. Tata McGraw Hill Publishing Co. Ltd, New York, pp 252–258
Sarasamma S, Audira G, Juniardi S, Sampurna B, Lai Y-H, Hao E, Chen J-R, Hsiao C-D (2018) Evaluation of the effects of carbon 60 nanoparticle exposure to adult zebrafish: a behavioral and biochemical approach to elucidate the mechanism of toxicity. Int J Mol Sci 19:3853. https://doi.org/10.3390/ijms19123853
Scharff P, Risch K, Carta-Abelmann L, Dmytruk IM, Bilyi MM, Golub OA, Khavryuchenko AV, Buzaneva EV, Aksenov VL, Avdeev MV, Prylutskyy YI, Durov SS (2004) Structure of C60 fullerene in water: spectroscopic data. Carbon 42:1203–1206. https://doi.org/10.1016/j.carbon.2003.12.053
Seki M, Fujishima S, Gondo Y, Inoue Y, Nozaka T, Suemura K, Takatsuki M (2008) Acute toxicity of fullerene C60 in aquatic organisms. Kankyo Kagakkaishi 21:53–62. https://doi.org/10.11353/sesj1988.21.53
Shi J, Wang L, Gao J, Liu Y, Zhang J, Ma R, Liu R, Zhang Z (2014) A fullerene-based multi-functional nanoplatform for cancer theranostic applications. Biomaterials 35:5771–5784. https://doi.org/10.1016/j.biomaterials.2014.03.071
Shoji M, Takahashi E, Hatakeyama D, Iwai Y, Morita Y, Shirayama R, Echigo N, Kido H, Nakamura S, Mashino T, Okutani T, Kuzuhara T (2013) Anti-influenza activity of C60 fullerene derivatives. PLoS One 8:e66337. https://doi.org/10.1371/journal.pone.0066337
Sumi N, Chitra KC (2017a) Fullerene (C60) induced alteration in the brain antioxidant system of the cichlid fish, Pseudetroplus maculatus (Bloch, 1795). J Global Biosci 6:4908–4917
Sumi N, Chitra KC (2017b) Histopathological alterations in gill, liver and muscle tissues of the freshwater fish, Pseudetroplus maculatus exposed to fullerene C60. Int J Fish Aquat Stud 5:604–608
Sumi N, Chitra KC (2017c) Oxidative stress in muscle tissue of the freshwater fish, Pseudetroplus maculatus (Bloch 1795): a toxic response from exposure to fullerene (C60) nanoparticles. Asian Fish Sci 30:206–214
Sumi N, Chitra KC (2019) Fullerene C60 nanomaterial induced oxidative imbalance in gonads of the freshwater fish, Anabas testudineus (Bloch, 1792). Aquat Toxicol 210:196–206. https://doi.org/10.1016/j.aquatox.2019.03.003
Tavares-Dias M, Moraes FR (2007) Haematological and biochemical reference intervals for farmed channel catfish. J Fish Biol 71:383–388. https://doi.org/10.1111/j.1095-8649.2007.01494.x
Tiwari AJ, Morris JR, Vejerano EP, Hochella MF, Marr LC (2014) Oxidation of C60 aerosols by atmospherically relevant levels of O−3. Environ Sci Technol 48:2706–2714. https://doi.org/10.1021/es4045693
Trinder P (1969) Determination of glucose in blood using glucose oxidase with an alternative oxygen acceptor. Ann Clin Biochem 6:24–27. https://doi.org/10.1177/000456326900600108
Trpkovic A, Todorovic-Markovic B, Kleut D, Misirkic M, Janjetovic K, Vucicevic L, Pantovic A, Jovanovic S, Dramicanin M, Markovic Z, Trajkovic V (2010) Oxidative stress-mediated hemolytic activity of solvent exchange-prepared fullerene (C60) nanoparticles. Nanotechnology 21:375102. https://doi.org/10.1088/0957-4484/21/37/375102
Usenko CY, Harper SL, Tanguay RL (2007) In vivo evaluation of carbon fullerene toxicity using embryonic zebrafish. Carbon 45:1891–1898. https://doi.org/10.1016/j.carbon.2007.04.021
Waissi-Leinonen GC, Nybom I, Pakarinen K, Akkanen J, Leppanen MT, Kukkonen JVK (2015) Fullerenes (nC60) affect the growth and development of the sediment-dwelling invertebrate Chironomus riparius larvae. Environ Pollut 206:17–23. https://doi.org/10.1016/j.envpol.2015.06.010
Xie B, Xu Z, Guo W, Li Q (2008) Impact of natural organic matter on the physicochemical Properties of aqueous C60 nanoparticles. Environ Sci Technol 42:2853–2859. https://doi.org/10.1021/es702231g
Yamago S, Tokuyama H, Nakamura E, Kikuchi K, Kananishi S, Sueki K, Nakahara H, Enomoto S, Ambe F (1995) In vivo biological behaviour of a water-miscible fullerene: 14C labelling, absorption, distribution, excretion and acute toxicity. Chem Biol 2:385–389. https://doi.org/10.1016/1074-5521(95)90219-8
Yamashita K, Yoshioka Y, Pan H, Taira M, Ogura T, Nagano T, Aoyama M, Nagano K, Abe Y, Kamada H, Tsunoda SI, Aoshima H, Nabeshi H, Yoshikawa T, Tsutsumi Y (2013) Biochemical and hematologic effects of polyvinylpyrrolidone-wrapped fullerene C60 after oral administration. Pharmazie 68:54–57
Zakaria S, Frohlich E, Fauler G, Gries A, Weib S, Scharf S (2018) First determination of fullerenes in the Austrian market and environment: quantitative analysis and assessment. Environ Sci Pollut Res 25:562–571. https://doi.org/10.1007/s11356-017-0213-x
Zhang X, Zhang Y, Zheng Y, Wang B (2013) Mechanical characteristics of human red blood cell membrane change due to C60 nanoparticle infiltration. Phys Chem Chem Phys 15:2473–2481. https://doi.org/10.1039/c2cp42850
Zhu S, Oberdorster E, Haasch ML (2006) Toxicity of an engineered nanoparticle (fullerene, C60) in two aquatic species, Daphnia and fathead minnow. Mar Environ Res 62:S5–S9. https://doi.org/10.1016/j.marenvres.2006.04.059
Zilva JF, Pannall PR, Mayne PD (1992) Liver disease and gall stones. In: Arnold E (Ed) Clin. Chem. Diagnosis Treat. London. pp. 287–306
Acknowledgements
Authors are thankful for the Junior Research Fellowship of CSIR-UGC, Government of India as it helped in carrying out the study.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Sumi, N., Chitra, K.C. Impact of fullerene C60 on behavioral and hematological changes in the freshwater fish, Anabas testudineus (Bloch, 1792). Appl Nanosci 9, 2147–2167 (2019). https://doi.org/10.1007/s13204-019-01041-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13204-019-01041-1