Abstract
Novel nanocomposite sorbent was produced by depositing nanostructured water treatment residual (nWTR) onto zeolite (Ze) using high-energy ball milling process. The physicochemical properties of nanocomposite (Ze-nWTR) prior and after Cd adsorption were analyzed by SEM–EDX, FTIR, BET and XRD. A batch study of cadmium adsorption (Ze-nWTR) was performed at various process parameters (sorbent dose, contact time, solution pH, competing ions, initial concentration and temperature). The obtained data were fitted to various equilibrium and kinetics models. The Langmuir and power function models successfully described Cd adsorption equilibrium and kinetic processes, respectively. The maximum adsorption capacity (qmax) value of Cd by Ze-nWTR nanocomposite (147 mgg−1) was 3 and 5.9 times higher than those of nWTR and zeolite sorbents, respectively. Increasing temperature from 287 to 307 K has resulted in increasing the maximum Cd adsorption capacity (qmax) of the nanocomposite from 147.9 to 270 mgg−1. The calculated thermodynamics parameters suggested physical and chemical attraction between Cd and Ze-nWTR and the association of dissociative mechanism in Cd(II) sorption process. The excellent reusability and Cd removal ability of Ze-nWTR nanocomposite (98%) from industrial wastewater confirm its potential as promising adsorbent for wastewater treatment applications.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Recently, surface water and groundwater contamination with heavy metals is worldwide serious environmental concern (Kumar et al. 2021; Hussain et al. 2019a). Sewage from different industries such as cadmium–nickel batteries, plastic manufacturing, melting and casting industries, electroplating, pesticides, mining, petroleum refining processes, and photography is considered the main resource of heavy metals release in the environment. At extremely low concentrations, heavy metal can seriously damage public health and aquatic life (Mehta et al. 2015). Cadmium (Cd2+) has caught the attention of academia and industry because of its carcinogenic effect and common use in industrial applications (Rangel-Porras et al. 2010; Pinto 2019). Therefore, seeking a suitable approach to remediate Cd-contaminated wastewater is urgently needed.
Because adsorption technique is considered the best choice over further traditional techniques in wastewater treatment field (Jia et al. 2019; Tavker et al. 2021), optimization of inexpensive and effective sorbent materials is a major challenge in developing the most efficient technology for heavy metals removal (Hussain et al. 2017; Ahmed et al. 2020; Liu et al. 2022). Zeolite is a cheap natural aluminum silicate mineral that widely studied because of its high affinity to adsorb heavy metals (Li et al. 2011; Ngah et al. 2013; Peng et al. 2021). Similarly, millions of tons of water treatment residual (WTR), byproducts of drinking water industry, are generated every day and released to the environment from water treatment facilities. Recently, these bulk waste materials have converted to nanoparticles by physical approaches (Elkhatib et al. 2015) and proven to be one of the low cost and efficient sorbents due to their specific and strong binding to Hg and Cr (Elkhatib et al. 2017, 2019; Moharem et al. 2019). The WTR nanoparticles also demonstrated high removal efficiency for Cd from aqueous solution as the adsorption capacities of nWTR were 17 and 10 times higher than those of bulk WTR in single- and multi-element systems, respectively (Elkhatib et al. 2016). However, flow rate through nWTRs system in fixed bed column is low because of separation difficulty of solid–liquid phase as a result of its ultrafine particle size (El-Kammah et al. 2022).
In recent years, the role of composites in water purification have drawn the attention of researchers and environmentalist due to their high selectivity and decontamination ability in removal of various pollutants (Herrera-Barros et al. 2020; Ajala et al. 2022; Hussain et al. 2019b). Pandey et al. (2017) have used zeolite-supported microscale zero-valent iron (Z-mZVI) to remove Cd and Cr from aqueous solution. They indicated that the adsorption capacity of Z-mZVI composite for Cr and Cd much exceeded the removal capacity of either nanoscale zero-valent iron (nZVI) or zeolite. Similarly, Angaru et al. (2021) reported that fly ash-based zeolite decorated with nano-ZVI and Ni-bimetallic have been proven to be highly efficient in removal of heavy metals from industrial effluents. Furthermore, coating zeolite surface with Al–iron oxides nanoparticles enhanced the reactivity and zeolite adsorption affinity for heavy metals (Nguyen et al. 2015). Thus, zeolite can be easily modified to improve the relative adsorption capacity toward heavy metals (Bowman 2003; Akhigbe et al. 2016; Li et al. 2019; Khulbe and Matsuura 2018). However, information on the capability of zeolite-supported nWTR (Ze-nWTRs) in heavy metals removal under different environmental conditions in batch and in column adsorption systems are not available and need investigation. In this study, a novel composite sorbent (Ze-nWTR) was produced by depositing nWTR onto zeolite to get the maximum benefit of the two sorbents for removal of Cd from wastewater. Thus, the current study objectives were to: (1) synthesize and characterize a zeolite–nWTR nanocomposite (Ze–nWTR), (2) assess Ze–nWTR nanocomposite efficiency for Cd removal from wastewater through batch and column experiments, (3) demonstrate Cd sorption behavior by Ze-nWTR under various conditions of solution pH, coexist competing ions, sorbent dose, temperature and contact time and (4) elucidate removal mechanism of Cd from aqueous solution by the nanocomposite.
Materials and methods
Source/synthesis of (Ze-nWTR) composite
The bulk drinking water treatment residual (WTR) was collected from Kafr El-Dawar water purification plant in El-bohera, Egypt. Physio-chemical characteristics of WTRs are presented in Table (S1) (Supplementary materials). The WTR samples were air-dried, ground and passed through two different 2 mm and 51 μm diameter stainless steel sieves. The WTRs were milled into nanoscale particles using the Fritsch planetary mono mill technique (Elkhatib et al. 2015). The natural zeolite was purchased from Delta Biotec company (Borg Alarab, Egypt). The natural zeolite certified analysis is shown in Table (S2). The zeolite samples were dried at 80 °C for 24 h., ground and sieved with a 100 mesh screen before use. The composites were prepared by ionizing 1.0 g zeolite with NaNO3 0.1 M for one week, and finally dried. The coating process of clay/nWTR (w/w) mixture was in a ratio of 2:1 and 4:1. The mixture was treated with ultrasound for 30 min and then vigorously stirred at room temperature for 120 min. The black solid product was washed five times with a (1:1) ethanol /water solution, dried and then stored.
Characterization
Emission scanning electron microscopy (SEM) equipped with energy-dispersive X-ray spectra (EDX) was used for characterization of nWTR, zeolite and Ze–nWTR nanocomposite. Surface area of the three studied sorbents was measured by N2 adsorption using surface area and porosimetry (SAP) analyzer (Brunauer et al. 1938). The KBr pellets were used for recording Fourier transform infrared (FTIR) spectra of the sorbent powders before and after Cd sorption.
Kinetics of Cd adsorption
Adsorption experiments were performed in polyethylene tubes (50 mL) by adding 0.2 g zeolite, nWTR or Ze-nWTR composite into 20 mL Cd2+ solution of certain concentrations at 25 °C and placed on a shaker of 170 rpm for different time intervals (5 min–24 h). Samples were withdrawn at each time intervals, centrifuged for 10 min at 4000 rpm and filtered through a Millipore filter (0.45 μm). The total amount of Cd concentrations in the filtered solution was determined using atomic absorption spectrometer (contrAA 300). The sorption data were applied to different kinetic models (First-order, Elovich, Intraparticle diffusion model, and modified Freundlich).
Adsorption isotherms
Equilibrium sorption experiments were performed by adding 0.2 g of zeolite, nWTR, or Ze-nWTR composite to 20 mL of (CdCl2·2H2O) solution with a concentration range of 40–800 mgL−1. After stirring at 170 rpm at 25 °C for 20 h, solution samples were collected, centrifuged, filtered, and analyzed for total Cd as previously stated in the kinetics section. Adsorption data were fit seven isotherm mathematical models (Langmuir, Freundlich, Elovich, Temkin, Fowler–Guggenheim (FG), Kiselev, and Hill de Boer). The thermodynamic parameters of the Ze-nWTR composite were calculated by applying the Arrhenius equation to the sorption data obtained at three different temperatures (287, 297 and 307 K), three pH values (4, 7 and 9) and different Cd concentrations (100), 250, 500 and 1000 mg L−1).
To evaluate the effect of coexisting ions on Cd adsorption by Ze-nWTR, a similar series of adsorption experiments were performed in the presence of three competing cations (Ni, Zn, Cu) at an initial Cd(II) concentration of 500 mg L−1. Additionally, to investigate the effect of adsorbent dose on Cd removal, the same adsorption experiments were performed using three different adsorbent weights (0.1, 0.2, and 0.3 g) of each adsorbent at an initial Cd concentration of 500 mg L−1.
The thermodynamic study
The changes in Gibbs free energy (ΔG°, J mol−1), enthalpy (ΔH°, J mol−1) and entropy (ΔS°, J mol−1 K−1) were determined to grasp the explanatory effect of temperature on Cd adsorption process. These parameters were determined by the following equations:
where R = gas constant [8.314 kJ/(molK)], Kc = equilibrium constant, Cqe = amount of Cd(II) adsorbed from solution to adsorbent at equilibrium (mg L−1), CS = equilibrium concentration of Cd(II) in solution (mg L−1).
To obtain Cqe and CS, we used qe from the Langmuir model.
ΔH° and ΔS° were calculated from the ΔG° and T plot (Fig. 8) using the following equations:
Reuse of Ze-nWTR adsorbent composite
The reusability of the Ze-nWTR nanocomposites was evaluated for six cycles using 0.01 M HCl solution. The nanocomposites loaded with Cd were dried, soaked in 50 mL of HCl (0.01 M) and stirred for 120 min at room temperature. The suspension was then filtered, dried, and the recovered nanocomposite was reused for Cd removal. The adsorption–desorption process was repeated for six cycles.
The Cd adsorptive removal efficiency of nanocomposite
Bach study
The efficiency of Ze-nWTR nanocomposite for Cd(II) removal was evaluated by set up a batch experiment using real wastewater from Al-Bilali agricultural drainage and industrial effluents of paper manufacturing factory (Rakta). The chemical analysis of the wastewaters used in the study is presented in Table (S3). Because the agricultural drainage sample was free of cadmium, it was spiked with 5 mg L−1 Cd.
The efficiency of Cd removal by nanocomposites
Bach study
The effectiveness of Ze-nWTR nanocomposites for removal of Cd(II) was evaluated by conducting batch experiments using real wastewater from Al-Bilali agricultural wastewater and industrial wastewater from a paper mill (Rakta). The chemical analysis of the wastewater used in the study is shown in Table (S3). Because agricultural drainage samples were Cd free, 5 mg L−1 Cd was added to the industrial effluent samples.
Column experiment
Column tests were presented to investigate the efficiency of nanocomposites for Cd removal from real wastewater. Experiments were conducted in a flow reactor contained a mixture of Ze-nWTR composite and sand. A PVC column with a height of 20 cm and an inner diameter of 2.5 cm was used to test the filter medium, i.e., a homogeneous mixture of Ze-nWTR composite and sand at certain mass ratios. A peristaltic pump continuously supplied the pollutant containing solution to a reservoir connected to the column. The solution passed through the column by gravity, and the effluent was periodically collected and analyzed. A schematic diagram of the experimental column is shown in Figure (S1) (Supplementary Material).
Results and discussion
Surface appearance and chemical structure of the adsorbent
nWTR
The nWTR particles are predominantly spherical in the size range 45–96 nm, as shown in Fig. S2; elemental analysis (SEM–EDX) showed that nWTR contained Fe, Si, and Al in proportions of 49.57, 21.77, and 6.3% of the total elements, respectively. In addition, X-ray diffraction analysis confirmed that amorphous iron, aluminum (hydr)oxide, and silicon oxide mainly existed in the crystalline iron-Al(hydr)oxide-free nWTR (Fig. S2).
Zeolite
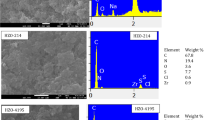
As seen in the SEM image (Fig. 1a), the zeolite particles are mostly spherical in the size range of 20–28 nm. As a result of SEM–EDX elemental analysis, O, Si, Al, and Fe accounted for about 91% of all zeolitic elements, and C, Ca, K, Fe, and Mg accounted for 9%.The XRD pattern of zeolite (Fig. 1b) shows strong characteristic peaks at 2θ = 4.5 and 67.861, indicating that the zeolite sample contains a large proportion of silica (SiO2). Additionally, the peaks appearing at 2θ values of 9.4 and 25 indicate the presence of potassium sodium aluminum silicate, while the peaks appearing at 2θ values of 21.3 and 27.3 belong to potassium sodium calcium aluminum. Moreover, peaks with 2θ values of (31.8 and 40), (35.021 and 46.5) and (41.9 and 76.5) indicate the presence of calcium iron oxide, iron oxide and potassium oxide, respectively.
Ze-nWTR nanocomposite
The morphology of the surface microstructure of the nanocomposite is shown in Fig. 2a. SEM images show that zeolite flakes and nWTR spherical nanoparticles are dispersed on the zeolite surface. The diameter of the spherically dispersed particles is in the nanoscale range (51.02–71.43 nm), and the presence of these nanoparticles on the surface of the zeolitic clay can prevent the agglomeration of these particles. According to EDX analysis, O, Si, C, Al and Fe, K and Na are the main elements in the nanocomposite, accounting for about 98% of the total element concentration. The minor components of the nanocomposite are Ca, Mg, Cu, and Cr, which are 0.81, 0.51, 0.83, and 0.51%, respectively. The presence of Al and Fe indicates the presence of nano-WTR and may increase the Cd removal capacity of the Ze-nWTR nanocomposite. SEM–EDX elemental analysis of Cd-loaded Ze-nWTR nanocomposite is shown in Fig. 2b. Among the detected elements, the appearance of the cadmium peak (3.08%) is evidence of the successful loading of Cd into the Ze-nWTR nanocomposite. XRD pattern of Ze-nWTR (Fig. 2c) shows characteristic peaks at 2θ = 9.48 and 12.81, indicating the presence of potassium and sodium aluminosilicate in the Ze-nWTR sample. The appearance of peaks at 2θ values of 12.7 and 28.6 indicates the presence of sodium potassium manganese oxide, while the peaks at 2θ values of 22 and 27.7 belong to calcium aluminum silicate. Iron, aluminum, chromium oxide and iron, aluminum, chromium, copper have 2θ values (36.1 and 58.3) and (44.7 and 64.7), respectively.
Specific surface area (SSA)
The nWTR and the zeolite specific surface area (SSA) were determined. The nWTR SSA (129 m2g−1) is much higher than that of the zeolite sample (39 m2g−1). The SSA of the Ze-nWTR nanocomposite was found to be 89 m2g−1. Certainly, nWTR can provide additional available surface sites for Cd adsorption on high SSA nanocomposites.
Fourier transmission infrared spectroscopy
FTIR spectrum of Ze-nWTR (Fig. 3a) showed a strong broad band at 3424 cm−1 attributed to the O–H bending vibration and a small band at 1638 cm−1 associated with the H–O–H molecular bending mode (Ruan et al. 2002). In addition, the main band at 1044 cm−1 assigned to the FeOH vibration of feroxyhyte and the band at 466 cm−1 assigned to the O–AL–O stretching vibration are shown in Fig. 3a (Carlson and Schwertmann 1981). The retention of Cd on the Ze-nWTR surface loaded with Cd results in significant spectral changes. The shift of the band from 3424 to 3438 cm−1 and the increase in intensity confirm the participation of surface hydroxyl groups in the Cd adsorption process. Similarly, the band at 1638 cm−1 shifts to 1634 cm−1 and increases in intensity. The band shift and intensity increases from 1044 to 1045 cm−1 and the band shift from 466 to 475 cm−1 are clear evidences of specific molecular interactions. Therefore, OH, O–Al–O, FeOH and FeO(OH) structures are proposed to participate in the Cd adsorption process by Ze-nWTR nanocomposites. The FTIR spectra of nWTR or Cd-loaded/unloaded zeolites are shown in Fig. 3b, c, respectively. After Cd adsorption, two new FeOOH labeled bands appeared at 794 cm−1 and 683 cm−1 and (3633 cm−1 and 729.37 cm−1) in nWTR and zeolite, respectively, indicating the role of iron oxyhydroxide on Cd adsorption process (Fig. 3b, c).
Adsorption kinetics
Understanding the kinetics and mechanisms of Cd adsorption reactions is a prerequisite for quantifying Cd sequestration by the studied adsorbents. A kinetic study was performed to estimate the equilibrium time. The Cd/zeolite or Ze-nWTR composite systems reached equilibrium within 2 h, while the nWTR-Cd system reached equilibrium after 8 h. These data are important because the equilibration time is a key parameter for the economics of wastewater treatment (Kadirvelu and Namasivayam, 2001). In addition, the amount of Cd adsorbed by the Ze-nWTR composite was higher than that of zeolite and nWTR (Fig. 4a). For all investigated adsorbents, Cd adsorption was very fast during the first 30 min. Cd was 99% adsorbed, followed by a slow adsorption step. Elkhatib et al. (2023) reported that the rapid adsorption of Cd is a surface phenomenon in which the pores on the adsorbent surface are rapidly filled in the initial stage, followed by slow diffusion and migration.
Kinetic modeling
It is important to select a mathematical model that satisfactorily fits the data and provides a reasonable adsorption mechanism (Oualid and Mahdi 2007). The kinetic data of Cd(II) sorption by the three investigated adsorbents were analyzed using four kinetic models: first order (Elkhatib et al.1992), Elovich (Elkhatib et al. 1984), intraparticle diffusion (Elkhatib and Hearn 1988) and modified Freundlich (Hamadeen and Elkhatib 2022a, b). The model parameters, coefficient of determination (R2) and standard error (SE) values for Cd retention of the studied adsorbents are presented in Table 1. The consistency between the experimental data and the values predicted by the model is expressed by the coefficient of determination (R2) and the standard error (SE) of the estimated value. The model with the highest R2 values (close to 1) and the lowest SE values was considered the best model that best represents the adsorption kinetics of Cd(II) (Fig. 4b). The R2 and SE mentioned values belong to the average values of the three adsorbents. The adsorption rate (ka) from the power function model was used to compare the Cd adsorption rates of the three investigated adsorbents. As shown in Table 1, ka appeared in the order of nanocomposite (49,306.03 min−1) > nWTR (46,902.93 min−1) > zeolite (45,729.87 min−1).
Adsorption isotherm
Figure 5a shows the Cd(II) adsorption isotherms by nWTR, zeolite and Ze-nWTR composite. A continuous increase in the amount of Cd adsorbed by the three studied adsorbents was observed with the increase of Cd concentration from 40 to 640 mg L−1. The Cd adsorption capacity of the three adsorbents studied was in the following order: nanocomposite (Ze-nWTR) > nWTR > Zeolite). Interestingly, the shapes of the Ze-nWTR composite and nWTR sorption isotherms are L-shaped isotherms according to the Giles classification (Giles et al. 1974), indicating an initial slope that does not increase with increasing pollutant concentration. The shape of the Cd adsorption isotherm by zeolite is an S-type isotherm, which is characterized by an increase in the slope as the pollutant concentration increases.
The adsorption equilibrium model
Cd adsorption parameters for three adsorbents (nWTR, zeolite and Ze-nWTR nanocomposites) studied using seven adsorption isotherm models (Langmuir, Freundlich, Elovich, Temkin, Fowler–Guggenheim (F.G.), Kiselev and Hill de Boer) were used for data interpretation (Table 2). When describing the adsorption data, R2 and SE values (Table 2) were used to determine the best fit for the isotherm. The R2 and SE values of the studied models (Table 2 and Fig. 5b) indicate that the Langmuir model is the most successful model in describing the Cd adsorption data. Therefore, it is assumed that the removal of Cd from the surface of the three investigated adsorbents is a monolayer adsorption process. The maximum adsorption capacity (qmax) of Ze-nWTR (147.9 mgg-1) was 3 and 5.9 times higher than the qmax values of nWTR and zeolitic adsorbent, respectively (Table 2). The zeolite coating process using nWTR and the synergistic action between clay and iron nanoparticles are responsible for the high adsorption capacity of the nanocomposite (Hamadeen et al. 2022). Therefore, it is recommended to use the Ze-nWTR nanocomposite as an effective adsorbent for the removal of Cd from polluted water sources (Arancibia-Miranda et al. 2016).
Operating conditions for Cd removal
Effect of solution pH and temperature
The Cd sorption process was monitored over a solution pH range of 4–9, adsorbent dose of 0.2 g and three different temperatures (287,297,307 K). Figure 6 shows that Cd removal increases with increasing solution pH and peaks at pH 9. The influence of different pH on the Cd ion removal rate of the studied adsorbents can be explained by the surface charge characteristics of the adsorbents and the ionization state of Cd (Mittal 2006). Therefore, the zero point charge (pHzpc) of the nanocomposite was determined to be 7.2 (Fig. S3). In solutions with a pH greater than 7.2, the repulsion between positively charged Cd and the surface charge of nanoparticles is minimized, resulting in increased Cd adsorption (Elkhatib et al. 2016). Additionally, the Langmuir adsorption capacity of the nanocomposites increased from 147.9 to 270 mgg−1 as the temperature increased from 279 to 308 K(Al-Qodah et al. (2007) reported that the adsorbate diffusion rate in the internal pores of adsorbent particles increases with increasing temperature.
The effect of competing cations
Industrial wastewater contains many metal ions that can interact with adsorbents and compete for binding sites. Therefore, it is important to study the competition between heavy metals for Cd adsorption by Ze-nWTR. To evaluate the proportion of Cd adsorbed by Ze-nWTR in single and multi-element systems, a series of adsorption studies were performed in the presence and absence of Zn, Cu and Ni at different Cd concentrations (20, 60,180 mg L−1). As shown in Fig. 7a, the presence of Zn2+,Cu2+,Ni+2 cations reduced the removal of Cd ions from solution by the Ze-nWTR nanocomposite because of the antagonism between the charged metal for the available sorption sites on Ze-nWTR nanocomposite surface and the increase in ionic strength of the solution (Hezarjaribi et al. 2021; Liu Y et al. 2020).
Effect of sorbent dose
The capacity of an adsorbent for a given initial concentration is highly dependent on the capacity of the adsorbent. The effect of Ze-nWTR nanocomposite, nWTR and zeolite bulk on Cd(II) removal from aqueous solution was determined by adding different doses (0.1–0.3 g) of each adsorbent using 20 mL of 500 mg Cd L−1. Focus once every 60 min. Figure 7b shows the effect of adsorbent weight on Cd removal. The results show that there is a significant difference in the adsorption capacity of Cd between different doses. For example, by increasing the dosage of Ze-nW nanocomposite, the removal rate of Cd increased from 33,225 to 91,310 mg/kg. For a given Cd2+ concentration, increasing the adsorbent weight increases the surface area and the number of active sites available for Cd interaction.
Effect of temperature
Thermodynamic parameters for Cd retention in Ze-nWTR nanocomposites were calculated at different initial Cd concentrations (100, 250, 500 and 1000 mgL−1) and different pH values (4,7,9) to determine adsorption nature (Al-Anber 2011). At initial concentration (100 mg L−1) and pH 9, standard free energy changes (ΔG°) for Cd(II) sorption were observed by Ze-nWTR to be − 22.302, − 27.252 and − 32.455 kJ mol−1 at 14 °C, 24 °C and 34 °C, respectively (Table 3). The negative values of ΔG° indicate the feasibility of the Cd adsorption process and explain the spontaneous response of Cd on Ze-nWTR nanocomposite adsorbents (Bayuo 2021). As shown in Fig. 8 and Table 3, a decrease in the ΔG° value (i.e., an increase in negativity) means that the amount of adsorption increases with increasing temperature. This result is consistent with the results of Fawzy et al. (2022), who found a decrease in ΔG° with increasing temperature for the biosorption of Cd on alginate-immobilized T. ornata biomasses. In addition, the ΔG° value increased negatively with increasing pH value, indicating that there were more adsorption sites as the pH value increased from 4 to 9 (Qadeer 2005; Chen and Wang 2006). In contrast, ΔH° values were positive at different initial solution concentrations, indicating the endothermic nature of Cd adsorption by Ze-nWTR (Suresh et al. 2010) (Table 3). The data in Table 4 showed that the ΔH° value decreases with the increase of the initial concentration, which means that less energy is required for the Cd adsorption reaction in Ze-nWTR due to the increase of the initial Cd concentration. Typically, ΔH° between 2.1 and 20.9 kJ mol −1 indicate physical adsorption, while values between 20.9 and 408 kJ mol −1 confirm chemisorption. (Tuzen et al. 2009). Therefore, physical and chemical attractions between Cd and Ze-nWTR are suggested due to the different large values of ΔH° (123,292–18,600 J mol−1) for Cd adsorption on Ze-nWTR at different initial solution concentrations. The negative values of ΔS° indicate that the dissociation mechanism is involved in the Cd(II) adsorption process (Suresh et al. 2010).
Cd adsorption mechanism
The Cd adsorption mechanism of Ze-nWTR nanocomposites was revealed by FTIR, XRD and EDX analyses. The XRD pattern confirmed the sorption reaction of Cd on the nanocomposite evidenced by cadmium peak (3.08%) detected in Cd-loaded Ze-nWTR nanocomposite (Fig. 2b). The FTIR analysis of zeolite showed that the band at 3420 cm−1 (OH bending vibration) completely disappeared after Cd was trapped on the zeolite surface, indicating the role of the OH functional group in Cd adsorption. Due to the presence of OH groups, cadmium can form surface complexes with the outer sphere zeolite surface (Peng et al. 2021; Ma et al. 2022). Therefore, ion exchange between H and Cd2+, which corresponds to the OH group of the zeolite, is the proposed adsorption mechanism, as shown in the following equation:
For the nWTR adsorbent, the FTIR spectrum of nWTR after Cd adsorption shows that the band associated with the O–H bending vibration at 4012 cm−1 has completely disappeared. Increasing intensities and changing the location of O–H bending vibrations band from 3416 to 3377 cm−1, the H2O bending vibrations band from 1636 to 1629 cm−1, the FeOH modes of peroxygite bending vibration from 1091 to 1029 cm−1 are identified. Moreover, the shift and enhancement of the two bands at 794 cm−1 and 683 cm−1 compared to FeOOH are a clear indication of real molecular interactions. Therefore, the six-coordinate structure of Cd suggests that it can form a bidentate complex on the surface of FeOOH and the inner sphere of nWTR, as shown in Fig. 9. It is also noteworthy that the Ca ratio decreased from 7.8 to 4.2% after Cd-saturated nWTR according to EDX analysis, as shown in Fig. (S2) (Supplementary Material). This result indicates the ability of Cd to replace Ca inside the octahedral FeOOH structure of nWTR, which can be attributed to the partial similarity between the ionic radii of Cd2+ (0.97 Å) and Ca2+ (0.99 Å) (Ji et al. 2020). Therefore, the general predicted mechanism of Cd adsorption in Ze-nWTR nanocomposites is initially through the electrostatic attraction between Cd ions in solution and zeolite OH. Subsequently, the adsorbed Cd can be specifically adsorbed on FeOOH-nWTR by inner-sphere complexation. Additional reactions may occur due to Cd occlusion of the internal FeOOH structure.
Reusability
The regeneration of the functional groups of the nanocomposites used by frequent movements corresponds to their stability, which is very important for industrial wastewater treatment. Therefore, the reusability and stability of Ze-nWTR nanocomposites were investigated for six consecutive adsorption/desorption cycles using 0.01 M HCl solution to desorb loaded Cd(II). The cumulative Cd absorbed by Ze-nWTR nanocomposites at initial Cd concentrations of 10.0 mg L−1 and/or 100.0 mg L−1 are shown in Fig. 10.The adsorption efficiency of Ze-nWTR nanocomposites for Cd(II) decreased by 1.7%, 1.9%, 3.6% and 3.7% during the second, third, fifth and, the sixth cycle, respectively. The results showed that the Ze-nWTR nanocomposite could be efficiently reused for up to six adsorption cycles with little change in the adsorbed Cd, indicating the high stability of the nanocomposite-loaded Cd. This makes the sorption process economical, reliable and technically attractive due to the low generation of solid waste.
Cadmium removal efficiency of nanocomposites
Batch study
The efficiency of nanocomposites for Cd(II) removal was investigated using batch experiments on real wastewater. Cd-contaminated industrial wastewater from the Rakta paper mill was treated with Ze-nWTR to remove Cd. The results showed that Ze-nWTR successfully removed 98.45% of Cd from Cd-contaminated industrial wastewater. Batch experiments showed that the Cd removal potential of Ze-nWTR was not affected by the presence of different anions (e.g., SO4, CO3, HCO3, Cl-) in industrial wastewater. In addition, the nanocomposite removed 97.53% of cadmium (Cd) from Al-Bilali agricultural drainage. These results demonstrate the suitability and efficiency of Ze-nWTR for Cd removal from real wastewater.
Column research
Using a bed reactor under continuous flow conditions (flow rate 3 mL min−1) by Ze-nWTR nanocomposites, the Cd removal efficiency reached 95.5% and 98% for agricultural drainage and industrial discharge, respectively. The highest removal rate of the Ze-nWTR nanocomposite indicates that the nanocomposite can be used as a potential adsorbent for the removal of various toxic pollutants from industrial and agricultural wastewater. Overall, the results of this study showed that the Ze-nWTR nanocomposite is a very environmentally friendly and reusable adsorbent for the effective removal of Cd from wastewater.
Comparison of various adsorbents for Cd removal
The effective removal of Cd(II) from wastewater by the Ze-nWTR nanocomposites was further evaluated by comparing the qmax values with the adsorbents shown in Table 4. Clearly, Ze-nWTR nanocomposites have higher Cd(II) adsorption capacity compared to other nano-adsorbents such as SiO2@DOPP nanocomposites, plant magnetic nanoparticles, hydroxyapatite coated zinc ferrite nanocomposites, gum iron modified oxide, and sodium-exchanged montmorillonite (Saravanan et al. 2012; Bunhu et al. 2017; Ali et al. 2019; Das and Dhar 2020; Saini et al. 2021). These results confirmed that the Ze-nWTR nanocomposite performed better than the aforementioned nanocomposites in removing Cd(II) from wastewater.
Conclusion
Ze-nWTR nanocomposites were prepared by the grinding method. Subsequently, SEM–EDX, XRD and FTIR analyzes were used to study the nanocomposites. The spherical nWTR nanoparticles distributed on the zeolite surface of the film are clearly visible in the size range of 51.02–71.43 nm. The synergistic behavior between zeolitic clay and iron-nWTR nanoparticles was evident as the maximum value of adsorption capacity (qmax) of the nanocomposite (147.9 mg g−1) was 3 and 5.9 times higher than that of nWTR and zeolite, respectively, at 287 K. In addition, the adsorption rate (ka) from the power function model was higher for nanocomposite (49,306.03 min−1) than for nWTR (46,902.93 min−1) and zeolite (45,729.87 min−1). The optimal conditions for Cd removal by nanocomposites are pH 9 and 307 K. The suggested mechanism of the Cd adsorption in the nanocomposite is started by the traction of static electricity between the Cd and the OH-zeolite, and then fixed into FeOOH-nWTRs internal surface. A slight change in the Cd adsorbed on the nanocomposite was observed as a result of the high stability of the nanocomposite loaded with Cd. In addition, Cd removal from agricultural drainage and industrial discharge are efficiently emitted at 95% and 98%, respectively. The final results confirmed the effectiveness, reusability and sustainability Ze-nWTR nanocomposite in removing Cd from contaminated industrial wastewater.
Data availability
All data generated or analyzed during this study are included in this published article and its supplementary information files.
References
Ahmed S, Aktar S, Zaman S, Jahan RA, Bari ML (2020) Use of natural bio-sorbent in removing dye, heavy metal and antibiotic-resistant bacteria from industrial wastewater. Appl Water Sci 10:107. https://doi.org/10.1007/s13201-020-01200-8
Ajala MA, Abdulkareem AS, Tijani JO, Kovo AS (2022) Adsorptive behaviour of rutile phased titania nanoparticles supported on acid-modifed kaolinite clay for the removal of selected heavy metal ions from mining wastewater. Appl Water Sci 12:19. https://doi.org/10.1007/s13201-021-01561-8
Akhigbe L, Ouki S, Saroj D (2016) Disinfection and removal performance for Escherichia coli and heavy metals by silver-modified zeolite in a fixed bed column. Chem Eng J 295:92–98
Al-Anber MA (2011) Thermodynamics-interaction studies-solids liquids and gases. Intech Open, London, pp 738–764
Ali I, Peng C, Naz I (2019) Removal of lead and cadmium ions by single and binary systems using phytogenic magnetic nanoparticles functionalized by 3-marcaptopropanic acid. Chin J Chem Eng 27:949–964. https://doi.org/10.1016/j.cjche.2018.03.018
Al-Qodah Z, Lafi WK, Al-Anber Z, Al-Shannag M, Harahsheh A (2007) Adsorption of methylene blue by acid and heat treated diatomaceous silica. Desalination 217:212–224
Angaru GKR et al (2021) Facile synthesis of economical feasible fly ash-based zeolite-supported nano zerovalent iron and nickel bimetallic composite for the potential removal of heavy metals from industrial effluents. Chemosphere 267:128889. https://doi.org/10.1016/j.chemosphere.2020.128889
Arancibia-Miranda N, Baltazar SE, García A, Muñoz-Lira D, Sepúlveda P, Rubio M, Altbir AD (2016) Nanoscale zero valent supported by zeolite and montmorillonite: template effect of the removal of lead ion from an aqueous solution. J Hazard Mater 301:371–380
Bayuo J (2021) Decontamination of cadmium(II) from synthetic wastewater onto shea fruit shell biomass. Appl Water Sci 11:84. https://doi.org/10.1007/s13201-021-01416-2
Bowman RS (2003) Applications of surfactant-modified zeolites to environmental remediation. Micropor Mesopor Mat 61:43–56
Brunauer S, Emmett PH, Teller E (1938) Adsorption of gasses in multimolecular layers. J Amer Chem Soc 60:309–319
Bunhu T, Tichagwa L, Chaukura N (2017) Competitive sorption of Cd2+ and Pb2+ from a binary aqueous solution by poly (methyl methacrylate)-grafted montmorillonite clay nanocomposite. Appl Water Sci 7:2287–2295. https://doi.org/10.1007/s13201-016-0404-5
Carlson LT, Schwertmann U (1981) Natural ferrihydrites in surface deposits from Finland and their association with silica. Geochim Cosmochim Acta 45:421–429
Chen C, Wang X (2006) Adsorption of Ni (II) from aqueous solution using oxidized multiwall carbon nanotubes. Ind Eng Chem Res 45:9144–9149
Das KC, Dhar SS (2020) Removal of cadmium (II) from aqueous solution by hydroxyapatite-encapsulated zinc ferrite (HAP/ZnFe2O4) nanocomposite: kinetics and isotherm study. Environ Sci Pollut Control Ser 27:37977–37988
El-Kammah M, Elkhatib E, Aboukila E (2022) Ecofriendly nanoparticles derived from water industry byproducts for effective removal of Cu (II) from wastewater: adsorption isotherms and kinetics. Inorg Chem Commun. https://doi.org/10.1016/j.inoche.2022.110062
Elkhatib EA, Hern JL (1988) Kinetics of phosphorus desorption from appalachian soils1. Soil Sci 145:222–229. https://doi.org/10.1097/00010694-198803000-00010
Elkhatib EA, Bennett OL, Wright RJ (1984) Kinetics of arsenite sorption in soils. Soil Sci Soc Am J 48:758–762
Elkhatib EA, Elshebiny GM, Balba AM (1992) Kinetics of lead sorption in calcareous soils. Arid Land Res Manag 6:297–310. https://doi.org/10.1080/15324989209381324
Elkhatib EA, Mahdy AM, Salama KA (2015) Green synthesis of nanoparticles by milling residues of water treatment. Environ Chem Lett 13:333–339
Elkhatib E, Mahdy A, Sherif F, Elshemy W (2016) Competitive adsorption of cadmium (II) from aqueous solutions onto nanoparticles of water treatment residual. J Nanomater 2016:8496798. https://doi.org/10.1155/2016/8496798
Elkhatib EA, Moharem ML, Mahdy A, Mesalem M (2017) Sorption, release, and forms of mercury in contaminated soils stabilized with water treatment residual nanoparticles. Land Degrad Develop 28:752–776
Elkhatib E, Moharem M, Hamadeen H (2019) Low-cost and efficient removal of mercury from contaminated water by novel nanoparticles from water industry waste. Desalin Water Treat 144:79–88
Elkhatib EA, Moharem ML, Saad AF, Farida AA (2023) Using nano-magnesium oxide/bentonite composite for cadmium removal from industrial wastewater. Environ Eng Res 28:210545. https://doi.org/10.4491/eer.2021.545
Fawzy MA et al (2022) Process optimization and modeling of Cd2+ biosorption onto the free and immobilized Turbinaria ornata using Box-Behnken experimental design. Sci Rep 12:3256
Giles CH, Smith D, Huitson AA (1974) General treatment and classification of the solute adsorption isotherm. I Theor J Colloid Interface Sci 47:755–765
Gong J, Chen L, Zeng G, Long F, Deng J, Niu Q, He X (2012) Shellac-coated iron oxide nanoparticles for removal of cadmium(II) ions from aqueous solution. J Environ Sci 24:1165–1173. https://doi.org/10.1016/S1001-0742(11)60934-0
Hamadeen H, Elkhatib E (2022a) New nanostructured activated biochar for effective removal of antibiotic ciprofloxacin from wastewater: adsorption dynamics and mechanisms. Environ Res 210:112929
Hamadeen H, Elkhatib E (2022b) New nanostructured activated biochar for effective removal of antibiotic ciprofloxacin from wastewater: adsorption dynamics and mechanisms. Environ Res 210:112929
Hamadeen HM, Elkhatib EA, Moharem ML (2022) Optimization and mechanisms of rapid adsorptive removal of chromium (VI) from wastewater using industrial waste derived nanoparticles. Sci Rep 12:14174
Herrera-Barros A, Tejada-Tovar C, Villabona-Ortíz A, Gonzalez-Delgado AD, Benitez-Monroy J (2020) Cd (II) and Ni (II) uptake by novel biosorbent prepared from oil palm residual biomass and Al2O3 nanoparticles. Sustain Chem Pharm 15:100216. https://doi.org/10.1016/j.scp.2020.100216
Hezarjaribi M, Bakeri GH, Sillanp M, Chaichi MJ, Akbari S, Rahimpour A (2021) Novel adsorptive PVC nanofibrous/thiol-functionalized TNT composite UF membranes for effective dynamic removal of heavy metal ions. J Environ Manage 284:111996
Hussain A, Maitra J, Khan KA (2017) Development of biochar and chitosan blend for heavy metals uptake from synthetic and industrial wastewater. Appl Water Sci 7:4525–4537. https://doi.org/10.1007/s13201-017-0604-7
Hussain A, Priyadarshi M, Dubey S (2019a) Experimental study on accumulation of heavy metals in vegetables irrigated with treated wastewater. Appl Water Sci 9:122. https://doi.org/10.1007/s13201-019-0999-4
Hussain A et al (2019b) Development of crosslinked chitosan membrane networks for uptake of copper and nickel metal ions. Desalin Water Treat 146:257–265
Ji Z, Su L, Pei Y (2020) Synthesis and toxic metals (Cd, Pb, and Zn) immobilization properties of drinking water treatment residuals and metakaolin-based geopolymers. Mater Chem Phys 242:122535
Jia J, Fu Z, Wang L, Huang Z, Liu C (2019) Conversion of waste polystyrene foam into sulfonated hyper-crosslinked polymeric adsorbents for cadmium removal in a fixed-bed column. Chem Eng Res Des 142:346–354
Kadirvelu K, Thamaraiselvi K, Namasivayam C (2001) Removal of heavy metals from industrial wastewaters by adsorption onto activated carbon prepared from an agricultural solid waste. Bioresour Technol 76:63–65
Khulbe KC, Matsuura T (2018) Removal of heavy metals and pollutants by membrane adsorption techniques. Appl Water Sci 8:19. https://doi.org/10.1007/s13201-018-0661-6
Koju NK, Song X, Wang Q (2017) Effective remediation of low-concentration cadmium in groundwater using nano-scale magnesia. Environ Sci Pollut Res 24:10819–10832
Kumar A et al (2021) Bio-remediation approaches for alleviation of cadmium contamination in natural resources. Chemosphere 268:128855
Li Z, Jean JS, Jiang WT, Chang PH, Chen CJ, Liao L (2011) Removal of arsenic from water using Fe-exchanged natural zeolite. J Hazard Mater 187:318–323
Li Z et al (2019) Disinfection and removal performance for Escherichia coli, toxic heavy metals and arsenic by wood vinegar-modified zeolite. Ecotox Environ Safe 174:129–136
Liu Y et al (2020) Competitive adsorption of heavy metals by anaerobic ammonium oxidizing (anammox) consortia. Chemosphere 258:127289
Liu X, Li G, Chen C, Zhang X, Zhou K, Long X (2022) Banana stem and leaf biochar as an effective adsorbent for cadmium and lead in aqueous solution. Sci Rep 1112:1584. https://doi.org/10.1038/s41598-022-05652-7
Ma Y et al (2022) Stabilization of Pb, Cd, and Zn in soil by modified-zeolite: mechanisms and evaluation of effectiveness. Sci Total Environ 814:152746
Mehta D, Mazumdar S, Singh SK (2015) Magnetic adsorbents for the treatment of water/wastewater—a review. J Water Process Eng 7:244–265
Mittal B (2006) I, me, and mine—how products become consumers’ extended selves. J Consum Behav 5:550–562
Moharem M, Elkhatib E, Mesalem M (2019) Remediation of chromium and mercury polluted calcareous soils using nanoparticles: sorption-desorption kinetics. speciation and fractionation. Environ Res 170:366–373
Ngah WW, Teong LC, Toh RH, Hanafiah MAKM (2013) Comparative study on adsorption and desorption of Cu (II) ions by three types of chitosan–zeolite composites. Chem Eng J 223:231–238
Nguyen TC, Loganathan P, Nguyen TV, Vigneswaran S, Kandasamy J, Naidu R (2015) Simultaneous adsorption of Cd, Cr, Cu, Pb, and Zn by an iron-coated Australian zeolite in batch and fixed-bed column studies. Chem Eng J 270:393–404
Oualid H, Mahdi C (2007) Removal of methylene blue from aqueous solutions by wheat bran. Acta Chim Slov 54:407–418. https://www.researchgate.net/publication/224892540
Pandey N, Shukla SK, Singh NB (2017) Water purification by polymer nanocomposites: an overview. Nanocomposites 3:47–66
Peng Z-D et al (2021) Removal of cadmium from wastewater by magnetic zeolite synthesized from natural low-grade molybdenum. Sci Total Environ 772:145355
Pinto MMSC et al (2019) Links between cognitive status and trace element levels in hair for an environmentally exposed population: a case study in the surroundings of the estarreja industrial area. Int J Environ Res Public Health 16:4560
Qadeer R (2005) Adsorption of ruthenium ions on activated charcoal: influence of temperature on the kinetics of the adsorption process. J Zhejiang Univ Sci B 6:353
Rangel-Porras G, García-Magno JB, González-Muñoz MP (2010) Lead and cadmium immobilization on calcitic limestone materials. Desalination 262:1–10
Ruan HD, Frost RL, Kloprogge JT, Duong L (2002) Infrared spectroscopy of goethite dehydroxylation: III. FT-IR microscopy of in situ study of the thermal transformation of goethite to hematite. Spectrochim Acta A Mol Biomol Spectrosc 58:967–981
Saini J, Garg VK, Gupta RK (2021) Cadmium removal from water by green synthesized nanobioadsorbent [SiO2@DOPP]: mechanism, isotherms, kinetics and regeneration studies. Sustain Chem Pharm 19:100350. https://doi.org/10.1016/j.scp.2020.100350
Saravanan P, Vinod VTP, Sreedhar B, Sashidhar RB, Kondagogu G (2012) modified magnetic nano-adsorbent: an efficient protocol for removal of various toxic metal ions. Mater Sci Eng C 32:581–586. https://doi.org/10.1016/j.msec.2011.12.015
Suresh S, Srivastav V, Mishra I (2010) Isotherm, thermodynamics, desorption, and disposal study for the adsorption of catechol and resorcinol onto granular activated carbon. J Chem Eng Data 56:811–818
Tavker N et al (2021) Removal of cadmium and chromium by mixture of silver nanoparticles and nano-fibrillated cellulose isolated from waste peels of citrus sinensis. Polymer 13:234. https://doi.org/10.3390/polym13020234
Tuzen M, Sara A, Mendil D, Uluozlu OD, Soylak M, Dogan M (2009) Characterization of biosorption process of As (III) on green algae Ulothrixcylindricum. J Hazard Mater 165:566–572
Funding
Open access funding provided by The Science, Technology & Innovation Funding Authority (STDF) in cooperation with The Egyptian Knowledge Bank (EKB). Open access funding provided by The Science, Technology & Innovation Funding Authority (STDF) in cooperation with The Egyptian Knowledge Bank (EKB). The funding was provided by Science and Technology Development Fund (Springer Nature OA agreements for Egypt).
Author information
Authors and Affiliations
Contributions
EAE contributed to concept, supervising investigation, and writing and editing. MLM involved in analysis, data validation, and writing and editing. AFS involved in supervising investigation and writing. SA involved in methodology, analysis and writing. All authors reviewed the manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors have no competing financial or personal interests.
Ethical approval
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Elkhatib, E.A., Moharem, M.L., Saad, A.F. et al. A novel nanocomposite-based zeolite for efficient remediation of Cd-contaminated industrial wastewater. Appl Water Sci 14, 75 (2024). https://doi.org/10.1007/s13201-024-02123-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13201-024-02123-4