Abstract
The present study was undertaken to investigate the extent of pollution load at Port Qasim coastal area, the second largest port in the country. The samples of sea water were collected from 23 locations, including the samples from Bundal Island situated in the Port Qasim area. Samples were analyzed for physico-chemical characteristics to determine the extent of pollution load. Samples were also analyzed for heavy metals including As, Cr, Cu, Cd, Pb and Ni. The investigation revealed that the Port Qasim Area is grossly polluted from inorganic and organic loads mainly coming from the nearby industrial setup. Extensively high organic load was found at the site receiving the wastewater from the Landhi Cattle colony. PQ-6 receives untreated wastewater from oil industries as exhibited by its high oil content. PQ-14 is receiving untreated industrial discharges from the nearby chemical industries and represents a very high pollution load. The sites closed to Bundal Island are relatively less polluted. The mangroves in the area are constantly under threat owing to the indiscriminate discharge of both domestic and industrial effluents. The concentration of DO although not on higher side but at least prevents the anoxic condition in the area. Out of all the heavy metals the concentration of Cr and Pb was exceptionally higher. The distribution of heavy metals demonstrated a typical diffusion pattern from the land to the sea. Principal component analysis and cluster analysis of the data showed the major gradients, similarities of localities and the variables of importance in terms of overall variability in the pollution characteristics. It is concluded that the level of pollution is increasing at an alarming rate, which is detrimental to the biodiversity of the coastal area.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The coastline of Pakistan is about 1050 km which is divided into Sindh and Balochistan coast. The length of Sindh coast is approximately 250 km whereas Balochistan coast comprises of 800 km. Karachi coastline is about 60 km. Karachi is the seventh largest city in the world. The city is recognized as the financial hub of the country which comprises of 45% industries of the country. It is also the most populous city of the country having an estimated population of 23.5 million. The total area of Karachi is approximately 3527 km2.
The major industrial zones of Karachi are SITE(Sindh Industrial Trading Estate), LITE (Landhi Industrial Estate), HITE (Hub Trading Estate) and KIA (Korangi Industrial area). It has been estimated by Khan and Shaukat (2008), and the city is producing > 350 MGD of domestic and industrial wastewater. Only a small fraction of this wastewater is partially treated at a few functional wastewater treatment plants of the City District Government (Sajjad et al. 2009). The remaining untreated wastewater is discharged annually in the coastal area (Saleem 2002; Siddique 2006). Needless to say that the remaining untreated wastewater is discharged into the coastal area through Lyari and Malir rivers (Saleem 2002; Siddique 2006).
Karachi coast is heavily polluted due to industrial, municipal, port and transportation activities (Qari et al. 2005; Rizvi et al. 1988; Saifullah et al. 2002) with the consequence that Karachi coast is one of the most deteriorated and polluted area in Pakistan (Khan and Khan 2001).
Indiscriminate disposal of untreated industrial and domestic effluent accumulates at the coast as heavy metals, xenobiotics, persistent organic contaminants, and excessive nutritional loadings (Akhtar et al. 1997; Alamgir et al. 2017; Beg et al. 1992; Bissen & Frimmel 2003; Bleninger et al. 2011). Several studies described the doom of coastal pollution as well as the EEZ of the country, counting Karachi’s coastal area (Ahmad 1997; Ahmed 1977, 1979; Beg et al. 1975; Haq 1976). This situation is alarming because of the huge pollution load that is routinely discharged into the sea without being treated, harming aquatic biodiversity and tainting seafood. In contrast, the majority of the untreated wastewater in Karachi is dumped into the sea at a shallow depth.
There are two major ports in Karachi namely Karachi Port and Port Qasim while in addition to these ports, two fish harbors namely Karachi and Korangi fish harbors are also operative. Arabian Sea passes through these ports. Port Qasim is a deep-water seaport and the second busiest port along Arabian sea of the country catering 35% of the cargo while Karachi Port handles more than 60% of all outward trade of Pakistan. This paper focuses on anthropogenic impact on Port Qasim.
Materials and methods
Study area
The total area of Port Qasim is about 12,000 acres. Gharo, Kadiro and Phiiti creeks constitute the approach channel to Port Qasim. These creeks are linked with mangrove patches and mud flat ecosystems (Amjad and Khan 2015). The creek system is approximately 28 km long and its width ranges from 250 m to 2.5 km. Within the jurisdiction of the Port Qasim, many industrial zones are operating out of which approximately 80% of the Pakistan’s automotive industry is located at Port Qasim. Two other major industrial setups located at Port Qasim are Pakistan Steel Mills (PSM) and K- Electric Power Plant. The port is located adjacent to the Bin Qasim town. The navigation route at Port Qasim is in fact the old channel of Indus River. The shipping vessels up to 75,000 tons (dead weight) reach to the port through a 45-km long Navigation Channel. On the other hand, the port is connected to national highway which is located at a distance of 15 km. The national railway network is also present at about 14 km from the port. Rapid industrialization and urbanization in the past few decades stimulate the generation of industrial effluent in the study area.
The PQA region is also marked with an island commonly known as Bundal Island which is one of the biggest island in the study area. Economically that island may be used as landing place, however, this aspect has not been investigated in detail by the concerned authorities. The total length of the island is 8.0 km. Its northern side is bit wider (4 km), while its southern portion is approximately 1.0 km which is occupied by thick mangrove swamps. Geomorphologically, the island mostly comprises of sand dunes with sparse vegetation. At the south of Bundal island, there is a small area comprises of dry sand which is recently emerged and known as Buddo island (Haider 1991; Siddiqui and Maajid 2004).
Sampling
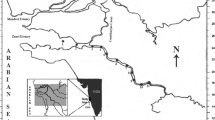
For the present study, the biochemical survey was performed during 2015. The sampling campaign was conducted from pre-designated locations. The sampling points were carefully chosen to cover the whole study area. The sampling points correspond to those sites from where the untreated domestic and industrial effluents enter into the port area. Twenty-three surface water samples were collected from the study area (Table 1). A map of the sampling locations is shown in Fig. 1.
Collection of samples
Sea water samples were collected from the surface (approx. 10 cm) with metal free Niskin bottle from the sites approachable through feet. A fishing boat was also hired for the collection of samples from the creek area. The collected water samples were preserved in sterilized amber glass bottles for later analysis. Periodical quality controls checks were undertaken to obtain a reliable analytical dataset. All samples were grab collection.
Physical parameters
pH, Dissolved Oxygen (DO) and salinity were measured in situ using portable meters. pH was recorded using pH 330i/SET, WTW, Germany. Salinity was measured by Inolab 320i WTW, Germany. DO was estimated using Jenway 630i DO meter. The DO probe was immersed in the sample stream to an adequate depth and in a manner to ensure sufficient sample movement across the probe-sensing element.
Biochemical and chemical parameters
The organic pollution of the study area was determined through BOD5 (Biochemical oxygen demand and COD (Chemical oxygen demand). The former was determined through azide modification method while later was estimated through dichromate reflux method using HACH COD reactor. The nutrient parameters such as total phosphate (TP) and total Kjeldahl nitrogen (TKN) were estimated by molybdo-phosphoric blue and distillation methods, respectively. Total cyanide was ascertained by distillation method using Drechsel gas washing bottle. Direct photometric method was employed for the Phenol estimation. Oil and grease (n-Hexane extract) estimation was performed by gravimetric technique. The above-mentioned parameters were determined in accordance with the prescribed methods of (APHA 2005).
Sea water samples were also analyzed for As, Cu, Cr, Ni and Pb through selective kits of Merck (NOVA 60), Germany.
Data analysis
Descriptive statistics were computed for each variable including mean, median, minimum, maximum, standard deviation and standard error. PCA (Principle Component analysis) was performed and eigenvalues, and eigenvectors were extracted from the original variables. The eigenvalues of the Principle component represent their associated variance, the contribution of the original variables in the PCs is given by the factor loadings (eigen vector coefficients), and the individual transformed interpretations are called scores (Singh et al. 2004). Cluster analysis was accomplished on the normalized data set by Wards method. The function of Cluster analysis is to gather objects based on the characteristics they have. In this multivariate analysis, the objects are classified on the basis of similar characteristics in the cluster with regards to a predetermined selection criterion. The subsequent clusters of objects showed high internal (within-cluster) homogeneity and high external (between cluster) heterogeneity (Wang and Zhang 2007). All the statistical analysis were performed using STATISTICA (99 Edition) software.
Results and discussion
PQA is receiving a very huge amount of untreated domestic and industrial wastewater that is affecting creek system, adjacent mangroves and its associated biodiversity. Tables 2 presents’ descriptive statistics of physical, chemical and metal analysis of samples collected from PQA.
pH values presented significant differences in the PQA area, wherein those sites which are under the influence of anthropogenic activities represent higher pH values, while the sites that represent relatively less polluted seawater have relatively lower pH values. Overall, pH values of the samples ranged from 7.1 to 7.8 with a mean value of 7.36. The highest pH value (7.8) was recorded at PQ-1 and PQ-2 which receive industrial effluents from Landhi industrial area.
The physiology and multiplicative capabilities of marine biota are greatly influenced by the salinity (Kinne 1970). Qasim (1982) reported that the surface salinity of Arabian Sea varied from 35.4 to 36.6 ‰ and declined from north to south. From Table 2, the mean salinity value in PQA was 37.07 ‰ that fluctuated in a range of 26.1 (PQ-1)–39.5 ‰ (PQ-12). The results of salinity are in accordance with Khatoon and Hussain (2006). However, salinity is expected to increase during summer due to continuous evaporation of water because of high temperature.
The two pertinent factors that express the waste assimilative ability of the coastal waters are DO and BOD (Thomann and Mueller 1987). The mean DO concentration of the samples was 3.917 mg/l. The lowest DO (1.2 mg/l) was recorded at PQ-10. This is the site that receives cooling water from Bin Qasim Thermal power plant (Khattak et al. 2012). This power plant generates 220 kV and situated about 50 km east of Karachi. The effluent generated from the power plant is discharged into the PQA at a location about 3 km from the plant. The temperature of PQ-10 was as high as 60-65OC which could be the major cause of low DO value. High temperature is negatively related with surface seawater (Ghamrawy 1982). Mahmood (1990) also described that DO concentration of seawater is inversely related to the temperature. The highest DO (5.3 mg/l) value was recorded at PQ-23 (Buddo Island). Khan and Saleem (1988) reported DO values between 3.0 and 6.55 mg/l from Karachi fish harbor at Karachi Port Trust area, whereas Hussain and Samad (1995) reported DO concentration of 2.4 to 6.7 mg/l from Manora channel included in Karachi Port Trust area. It can be argued that low DO concentration could be due to high BOD values, which indicates the extent of stress on the ecosystem (Dhage et al. 2006). DO is greatly affected by the elevated organic load responsible for exhaustion of DO in the process of biodegradation (Babu et al. 2006). Aerobic and anaerobic stabilization processes at water and sediment interface also deplete DO that in turn regenerates nutrients (Jørgensen 1983). It can be noticed that all the sites except PQ-5 and PQ-10 are still not facing hypoxic condition which normally occur when DO concentration is < 2.0 mg/l (Diaz and Rosenberg 1995). Lower DO (< 4.0 mg/l) is likely to occur in summer in both estuary and adjacent coastal waters (Yin et al. 2004).
BOD is generally used to determine the oxygen demanding potential of the wastewater. The mean BOD5 value of PQA area ranged from 58 to 876 mg/l. High BOD (> 600 mg/l) values are recorded at PQ-5 (Cattle colony waste), PQ-14 (Near Sindh coastal highway bridge) and PQ-15 (Gharo creek). Landhi cattle colony is one of the world’s largest cattle colony spread over an area of 6.5 km2. The colony has more than 0.4 million livestock in which 95% are buffaloes. The daily milking yield is 4.0 million liters while the daily cow dung production is about 7200 tons. The colony fulfills 70% of the milk demand of Karachi. Only a small portion of cow dung (250 tons per day) is being picked up and used as an organic manure, while the liquid waste is drained into the main sewer thorough which it discharges into PQA area. This liquid waste contains high organic loading as depicted in the BOD value of PQ-5. PQ-14 and PQ-15 receive the waste from Landhi industrial area, which represents high organic and inorganic loads. Khan et al.,(2003) already reported high BOD values from Gharo creek. Khan et al.,(2012) also described high BOD values from Gizri creek area (PQ-18). A high BOD concentration tends to reduce DO concentration as oxygen is utilized by bacteria creating anoxic conditions detrimental to marine life.
There is no palpable linear relationship between BOD and COD in relatively clean seawater samples away from the shore (Jin et al. 2009). A direct correlation exists between BOD and COD in estuary heavily contaminated with sewage. The COD regime depicted similar trend as that of BOD. COD values of all the samples ranged between 656 mg/l (PQ-22, Buddo Island) and 2654 mg/l (PQ-1 Landhi industrial area effluent). There are several sources of industrial waste that eventually discharge into seawater of PQA area. Moreover, untreated domestic wastewater from the nearby settlements also finds its way to the sea. The quantity of untreated effluent is sufficiently high that would possibly adversely affect the PQA ecosystem. COD, in fact indicates the level of water pollution by reductive pollutants (Jin et al. 2004). Ahipathy and Puttaiah (2006) while working on Vrishabhavathy River in Bangalore (India) reported that direct discharge of untreated domestic and industrial effluents into the river are responsible for the high organic pollution that is represented by high BOD and COD values.
The anthropogenic and agriculture activities are main sources of nutrient parameters in an aquatic ecosystem that mainly emerge from non-point sources therefore, difficult to manage, regulated and treated (Carpenter et al. 1998).
Orthophosphoric acid is the principal form of dissolved inorganic phosphate that exists in the sea. Martin (1972) suggested that the average concentration of dissolved orthophosphate in the seawater is 73 µg/l. Boto (1982) opined that the concentration of 31 µg/l is considered as low. The mean concentration of total phosphate of the samples was 3.21 mg/l (0.86–7.87 mg/l). Minimum concentration (0.86 mg/l) was found at PQ-10 (Bin Qasim thermal power plant). Low concentration of PO4− at PQ-10 is due to high temperature of seawater which does not allow phytoplanktonic activity. Highest concentration (7.87 mg/l) was recorded at PQ-3. This is the site which receives domestic effluent from Landhi town and Rehri goth. PQ-6, PQ-7 and PQ-20 represent the area close to the mangroves. At these sites, the phosphate concentration was 4.72, 4.11 and 6.9 mg/l. Vazquez et al. (2000) reported 18 µg/l of phosphates from mangrove areas that seems to be quite low as compared to the phosphate concentration of the samples in the present study. High concentration of phosphate is mainly attributed to phytoplanktonic activity. Excessive input of nutrients in the form of nitrate and phosphate is responsible for eutrophication. In the Mediterranean coastal lagoons, large input of nutrients (i.e., N and P) causes eutrophication in summer. High concentration of phosphate is deposited in the sediments of these lagoons that can serve as a reservoir of phosphate (Mesnage and Picot 1995).
Land-based Nitrogen (N), mainly of anthropogenic origin, can be attributed to variation in estuaries and coastal waters all over the world that eventually leads to eutrophication. It has been reported that the exchange of nutrients in the tidal waters is strongly influenced by seasonal fluctuations (Rivera-Monroy et al. 1995). However, the seasonal fluctuation with regards to direction or magnitude of nitrogen fluxes has not been clearly differentiated in mangrove ecosystems (Valiela and Cole 2002). The coastal nitrogen cycling in mangroves area may bear similarity to saltmarsh ecosystems that exhibits the process of nitrogen exchange (Whiting et al. 1989).
In coastal lagoons, the predominant form of inorganic nitrogen is ammonium (Serpa et al. 2007). In the present study, the attention was, however, focused on the available nitrogen in the form of Total Kjedahl nitrogen (TKN). The average TKN of all the samples was 60.87 mg/l. The highest concentration was recorded at PQ-5 (125 mg/l). This is the site receiving untreated discharge from Landhi cattle colony which contains a very high organic load that is responsible for high TKN concentration.
Cyanide is widely used in electroplating, metal refining, organic chemical production and many other processes (Nagaraja et al. 2002). These industries are located both in formal and informal sectors of PQA area. Cyanide forms complex compounds which are often less toxic as compared to free cyanide (Fuller and Randle 1984). Cyanide is also widely used for fish collection in many countries of the world. It is reported that acute toxicity to cyanide may cause death within 96 h at a concentration of 0.1–0.3 mg/l (Doudoroff 1980). The concentration of cyanide ranging between 0.005 and 0.01 mg/l exhibits adverse effect on fishes in the form of abridged growth, compromised swimming enactment, augmented metabolism inhibition and increased respiratory rates (Leduc 1984). Cyanide also caused desquamation of filamental epithelium at the concentration of 0.05 mg/l in freshwater fish Gnathonemus petersii (Alazemi et al. 1996). As reported by some workers, very little scientific research has been published on the effects of cyanide on marine fish (Rubec and Pratt 1984). The mean concentration of cyanide was 0.661 mg/l (range 0.05–1.76 mg/l) with the highest concentration recorded at PQ-5. It is interesting to note this site receives untreated discharge from Landhi cattle colony where there is no apparent source of cyanide; noticeably, this needs further investigation. Continuous accumulation of cyanide in the study area could be lethal to the marine life.
Oil pollution is a major contributor that deteriorates marine water quality (Abdullah et al. 1996). Dispersed oil causes an increased toxicity to marine life therefore, lethal for marine biodiversity. (Anderson et al. 2009; Bartoli et al. 2001). The mean concentration of oil and grease was 137 mg/l. The lowest concentration (54 mg/l) was recorded at PQ-19. This site is adjacent to the Bundal Island where there is no apparent source of oil and grease. It may be due to the fact that oil and grease accumulated at the area because of wind direction. At PQ-6, the highest concentration (212 mg/l) was estimated. The site receives wastewater from nearby oil refineries.
The presence of significant amount of oil and oil products to any water body causes an immediate rise in the BOD5 due to the activities of hydrocarbon degraders and the consequent blockage of oxygen dissolution. Oil and grease also hamper the dissolution of atmospheric oxygen in water thereby creating hypoxic conditions detrimental to marine life. Routine fishing and navigation operations in the study area are also a major source of oil and grease. A sizeable amount of oil also tends to sink in the bottom and continues to deposit where they undergo microbial degradation owing to anoxic environment. Relatively high values of oil and grease have been reported in the Chinna creek close to the study area (Khan and Shaukat 2008).
Phenol is often considered as one of the principal component of industrial wastewater particularly chemical industry. Phenol is also present in appreciable quantity in the discharges of metal industry, pharmaceutical, paint and varnish industries, textile industries, etc., (Navarro et al. 2008). However, the information about the phenol toxicity at ecosystem level is limited (Saha et al. 1999). It has been reported that photolysis is the primary transformation process for the polychlorinated phenols (Hwang et al. 1986). In this context, Eklund and Kautsky (2003) found that macroalgae are more sensitive than other aquatic organisms to phenol. The mean phenol values of the samples were 1.29 mg/l. The highest phenol concentration (3.76 mg/l) was observed at PQ-4 (Rehri goth Jetty). Elevated concentrations of phenol are mainly due to the untreated discharges of industrial origin from the PQA and adjoining industrial area. Limited concentration of phenol is also attributed to the use of janitorial agents. In the vicinity of the study, a large number of tanneries are also located where phenol is one of the constituent in their effluents. Phenol is detrimental to marine life forms even at concentration as low as 5 µg/l (Navarro et al. 2008).
Table 2 represents the mean concentration of all the metals present in samples. The availability of metal concentration was in the order of Ni > Cr > Cu > Pb > Cd > As. Nickel is primarily used in the manufacturing of stainless steel but also as a catalyst in industrial processes and in oil refining (Kara 2005). The mean Ni concentration of all the sample was 10.59 mg/L. The minimum concentration of Ni (4.54 mg/l) was found at PQ-20 (Bundal Island), while the highest concentration (15.84 mg/l) was observed at PQ-3 (effluent from Landhi and Rehri goth). Qari and Siddiqui (2008) reported Ni concentration of 0.07–1.86 mg/l from the sea water samples collected from Nathia Gali at Karachi coast. Low concentration of Ni at Nathia Gali is due to the fact that this area does not receive any industrial discharge directly. Saifullah et al., (2002) reported much lower concentration of Ni (0.11 and 5.01 ppb) from Sandspit and Korangi creek along the Karachi coast. Even at harbor they reported the values of Ni close to 1 ppb.
In Karachi, Cr is used in a variety of industries (Saha et al. 2011). There are several tanning industries in Karachi converting raw hides or skins into leather where a huge quantity of chrome sulfate is used, which prevents penetration of water into leather pores and provides thermal stability (Baral et al. 2009). Hexavalent chromium is often considered toxic mainly originating from human activities (Saha and Orvig 2010). In addition, a number of paint industries are also located in PQA area used lead chromate for paints, inks and plastics. The mean concentration of Cr in all the samples was 10.15 mg/l. The minimum concentration of Cr (2.67 mg/l) was recorded at PQ-20 (Mangroves area at Bundal island), while maximum concentration was at PQ-3 (18.54 mg/l). High concentration of Cr is also attributed to the textile industries effluent where Cr is mainly used in dyes. Cr salts are also used as an anticorrosive agents in many industrial setups. Qari and Siddiqui (2008) reported Cr concentration of 0.05–1.04 mg/l form the Karachi coast which is much lower than the concentration reported in the present study.
Cu and Pb are related to the concentration of nutrients and their concentration varied seasonally. Copper, however, is the most commonly tested metal followed by zinc, cadmium, mercury, and lead in order of number of test results (Eklund and Kautsky 2003). Copper is an extensively used in many applications like transportation, manufacturing, currency, transportation of electricity, construction and agriculture field (Barceloux and Barceloux 1999). The major sources of Cu in the study area are the nearby chemical industries. The urban runoff appears to be the major source of Cu in the area. Urban runoff plays an important role in copper inputs to natural waters (Sodré et al. 2005). The concentration of Cu varies from 3.76 (PQ-22. Buddo Island) to 11.22 mg/L (PQ-3).
In the PQA area, Pb and its compounds are used in electroplating, construction materials, dyes, surface coatings, batteries and plastics as reported by (Kara 2005). The mean concentration of Pb in all the samples was 5.45 mg/l. The lowest concentration of Pb was found at PQ-23 (1.67 mg/l), while the maximum concentration was found at PQ-5 (8.97 mg/l). Another possible source of Pb in the samples is the fuel used for operating the fishing vessels. The fishing vessels in the area are mostly operated through petrol or diesel. These defective boats render the spillage of the fuel, which is a common problem in the area.
Cadmium is a relatively rare heavy metal, and its occurrence in sea water mainly owes to anthropogenic activities (Nriagu 1988), metal smelting and refining, fossil fuel burning, application of phosphate fertilizers, sewage sludge (Kabata-Pendias 2004). Cadmium also occurs in road dust due to its presence in automobile fuel and in soil (Faiz et al. 2009). Minimum and maximum concentration of Cd ranged between 0.98 mg/l (PQ-23), while maximum (2.45 mg/l) was found at PQ-2. This would mean that Cd is mainly coming from the fuel used for navigation purposes and also from oil industries.
The toxicity of different arsenic species varies in the order: arsenite > arsenate > monomethylarsonate (MMA) > dimethylarsinate (DMA) (Sturgeon et al. 1989). The concentration of arsenic in natural waters depends on the geological composition and the degree of pollution of the environment (Jain and Ali 2000). The anthropogenic activities lead to the release of As in the environment may include use of arsenical pesticides, wood preservative and smelting of metal ores (Bhattacharya et al. 1995). The mean concentration of As of all the samples is 0.49 mg/l. The lowest value was recorded at PQ-22 (0.14 mg/l), while the maximum value was found at PQ-21 (1.45 mg/l). It seems that the possible source of As in the study area is anthropogenic rather than natural.
Apparently, the sources of these metals are mainly the untreated industrial discharges. Maximum concentration of all the metals found at PQ-3. This was mainly due to the fact that the site receives untreated wastewater of both domestic and industrial origin. The health risks associated with these metals are also of great concern and noticed by many workers (Eriyamremu et al. 2005; Muchuweti et al. 2006).
Principal component analysis of seawater samples of PQ coastal area is presented in Table 3 and Fig. 2. The magnitude of variance covered by each PC depends on the comparative value of its eigenvalue to the total sum of eigenvalues (Wang and Zhang 2007). The first three components of seawater (Table 3) explained 70.10 percent of the cumulative variance. The first component that accounted for 45.35 percent of total variance is governed by As, TP, Cyanide, Salinity and TKN. The second component that explained 16.67 percent variance is controlled by pH, Pb, DO, Phenol, and COD. The third component exhibited 8.08 percent of the total variance is attributed to cyanide, COD, As, Oil and grease and Cu.
The dendrogram derived from cluster analysis shows two main groups (Fig. 3). The group on the left shows less polluted samples with lesser values of BOD and COD as well as other pollutants. By contrast, the group on the right comprising of 15 samples essentially represents polluted sites. To a great extent, the results of cluster analysis accord with the PCA ordination.
Conclusions
On the basis of the extent of the pollution load, the sampling stations can be divided into two main groups. The sampling stations that receive industrial discharges directly from the non-point sources are more polluted than the sampling stations found within the creek area. PQ-5 is receiving wastewater from a Landhi cattle colony which exhibits a very high-organic load. In addition, the heavy metals concentration was also relatively higher than other sampling stations. At PQ-10, the temperature of seawater is very high owing to the dumping of cooling water from the Bin Qasim thermal power plant, which has rendered the area deprived of marine biodiversity. PQ-1 and PQ-6 are receiving untreated industrial effluent from the Landhi industrial state, which exhibits an extremely high pollution load both in term of organic and inorganic forms. The sampling stations closed to the Bundal Island are relatively less polluted. This could be mainly due to the dilution factor. In general, the concentration of COD, TKN and oil and grease is exceptionally higher in most samples, which is detrimental to marine biodiversity. Although, DO concentration was low, but still it may prevent anoxic conditions. From the ecological viewpoint, the concentration of phenol and cyanide was much higher although no significant point source of these pollutants was identified. Nutrient parameters were relatively higher in concentration in the samples collected from the mangroves area. Out of heavy metals, the concentration of Ni was exceptionally higher followed by Cr which is an indication of industrial discharges coming from tanneries. The presence of high concentration of lead indicates the use of lead containing fuel used in the defective motorboats particularly used for fishing. Principal component analysis and cluster analysis were found to be useful techniques to disclose the trends of pollution load inherent in the data.
References
Abdullah A, Woon W, Bakar R (1996) Distribution of oil and grease and petroleum hydrocarbons in the Straits of Johor. Penins Malaysia Bull Environ Contam Toxicol 57:155–162
Ahipathy M, Puttaiah E (2006) Ecological characteristics of vrishabhavathy River in Bangalore (India). Environ Geol 49:1217–1222
Ahmad M (1997) Natural and human threats to biodiversity in the marine ecosystem of Coastal Pakistan BT-coastal zone management imperative for maritime developing nations. In: Haq BU, Haq SM, Kullenberg G, Stel JH (eds) Coastal zone management imperative for maritime developing nations. Springer, Netherlands, pp 319–332. https://doi.org/10.1007/978-94-017-1066-4_17
Ahmed M (1977) An assessment of the magnitude of coastal pollution in Pakistan through a survey of its marine fauna and fisheries. Thalassia, Jugosalvaica, THJUAP 13(3/4):395–412
Ahmed M (1979) The present status of marine pollution in Pakistan. In: Nat. regional seminar on protection of marine environment and related ecosystems, pp 1–16
Akhtar W, Ali I, Zaidi SSH, Jilani S (1997) The state of pollution levels of Karachi Harbor and adjoining coastal water. Water Air Soil Pollut 94(1):98–107. https://doi.org/10.1023/A:1026430528690
Alamgir A, Khan MA, Shaukat SS, Kazmi SJH (2017) A quantitative appraisal of Lyari river effluent, Karachi, Pakistan. Desalination Water Treat 62:175–184. https://doi.org/10.5004/dwt.2017.20131
Alazemi B, Lewis J, Andrews E (1996) Gill damage in the freshwater fish Gnathonemus petersii (family: Mormyridae) exposed to selected pollutants: an ultrastructural study. Environ Technol 17:225–238
Amjad S, Khan MA (2015) Marine ecological assessment for LNG terminal at Port Qasim. Pakistan J Eng Technol Sci 1
Anderson BS, Arenella-Parkerson D, Phillips BM, Tjeerdema RS, Crane D (2009) Preliminary investigation of the effects of dispersed Prudhoe Bay Crude Oil on developing topsmelt embryos, Atherinops Affinis. Environ Pollut 157:1058–1061
APHA (2005) Standard methods for the examination of water and wastewater, 21st edn. American Public Health Association Washington DC, USA
Babu M, Das VK, Vethamony P (2006) BOD–DO modeling and water quality analysis of a waste water outfall off Kochi, west coast of India. Environ Int 32:165–173
Baral S, Das N, Ramulu T, Sahoo S, Das S, Chaudhury GR (2009) Removal of Cr (VI) by thermally activated weed Salvinia cucullata in a fixed-bed column. J Hazard Mater 161:1427–1435
Barceloux DG, Barceloux D (1999) Copper. J Toxicol Clin Toxicol 37:217–230
Bartoli M, Nizzoli D, Viaroli P, Turolla E, Castaldelli G, Fano EA, Rossi R (2001) Impact of Tapes philippinarum farming on nutrient dynamics and benthic respiration in the Sacca di Goro. Hydrobiologia 455:203–212
Beg MAA, Mohammad SN, Yousufzai AHK (1975) Industrial effluents, their nature and disposal in Karachi region, Part 1: survey of the pollution Lyari River. Pakistan Acad Sci 12:125–130
Beg MAA, Mahmood SN, Yousufzai AHK (1992) Heavy metals pollution in the coastal environment of Karachi. Pakistan J Mar Sci 1(2):117–126
Bhattacharya P, Chatterjee D, Jacks G (1995) Arsenic contamination in groundwater within multilevel aquifer system of Indo-gangetic delta plains: a case study from West Bengal, Eastern India. In: 5th seminar on hydrogeologyand environmental geochemistry, B. Bölviken Seminar-95; Geological Survey of Norway, pp 8–9
Bissen M, Frimmel FH (2003) Arsenic—a review. Part I: occurrence, toxicity, speciation, mobility. Acta Hydrochim Et Hydrobiol 31(1):9–18. https://doi.org/10.1002/aheh.200390025
Bleninger T, Jirka GH, & Roberts PJW (2011) Mixing zone regulations for marine outfall systems. In: Proceedings of the international symposium on outfall systems, Mar Del Plata, Argentina, pp 1–20. http://www.osmgp.gov.ar/symposium2011/Papers/59_Bleninger.pdf
Boto KK (1982) Nutrient and organic fluxes in mangroves. Mangrove Ecosyst Aust Struct, Funct Manag 239–258
Carpenter SR, Caraco NF, Correll DL, Howarth RW, Sharpley AN, Smith VH (1998) Nonpoint pollution of surface waters with phosphorus and nitrogen. Ecol Appl 8:559–568
Dhage S, Chandorkar A, Kumar R, Srivastava A, Gupta I (2006) Marine water quality assessment at Mumbai West Coast. Environ Int 32:149–158
Diaz RJ, Rosenberg R (1995) Marine benthic hypoxia: a review of its ecological effects and the behavioural responses of benthic macrofauna. Oceanogr Mar Biol Ann Rev 33:3
Doudoroff P (1980) A critical review of recent literature on the toxicity of cyanides to fish. American Petroleum Institute, USA
Eklund BT, Kautsky L (2003) Review on toxicity testing with marine macroalgae and the need for method standardization––exemplified with copper and phenol. Mar Pollut Bull 46:171–181
Eriyamremu G, Asagba S, Akpoborie I, Ojeaburu S (2005) Evaluation of lead and cadmium levels in some commonly consumed vegetables in the Niger-Delta oil area of Nigeria. Bull Environ Contam Toxicol 75:278–283
Faiz Y, Tufail M, Javed MT, Chaudhry M (2009) Road dust pollution of Cd, Cu, Ni, Pb and Zn along Islamabad expressway, Pakistan. Microchem J 92:186–192
Fuller SJ, Randle PJ (1984) Reversible phosphorylation of pyruvate dehydrogenase in rat skeletal-muscle mitochondria. Effects Starv Diabetes Biochem J 219:635–646
Ghamrawy M (1982) Studies on the ecology and bioiogy of penaeid shrimp in the region of Jeddah. Ph.. D. Thesis, University of Wales, vol 203
Haider S (1991) Coastal zone management around Port Qasim and the adjoining areas. In: Proceedings of the seminar on the application of remote sensing technique to land use planning and environmental surveying Karachi, Pakistan, p 77e78
Haq SM (1976) Overview on pollution in the coastal environment of Pakistan and its possible implication for the marine ecosystem. In: Meyers SP (Ed), In: Proc Int Symp Mar Poll Res, Centre for Wetland resources, LSU Baton Rouge, USA, pp 33–53. Louisiana State Univ.Baton Rouge,L.A., Centerfor Wetland Studies
Hussain S, Samad M (1995) Some physio-chemical parameters of backwater of sandspit(Northern Arabian Sea, Pakistan coast). Pakistan J Zoology 27:191–194
Hwang HM, Hodson R, Lee R (1986) Degradation of phenol and chlorophenols by sunlight and microbes in estuarine water. Environ Sci Technol 20:1002–1007
Jain C, Ali I (2000) Arsenic: occurrence, toxicity and speciation techniques. Water Res 34:4304–4312
Jin B, He Y, Shen J, Zhuang Z, Wang X, Lee FS (2004) Measurement of chemical oxygen demand (COD) in natural water samples by flow injection ozonation chemiluminescence (FI-CL) technique. J Environ Monitor 6:673–678
Jin XL, Jing M, Chen X, Zhuang ZX, Wang XR, Lee FS (2009) A study on the relationship between BOD5 and COD in coastal seawater environment with a rapid BOD measurement system. Water Sci Technol 60:3219–3223
Jørgensen B (1983) Processes at the sediment-water interface. The major biogeochemical cycles and their interactions, vol 21, pp 477–509
Kabata-Pendias A (2004) Soil–plant transfer of trace elements—an environmental issue. Geoderma 122:143–149
Kara Y (2005) Bioaccumulation of Cu, Zn and Ni from the wastewater by treated Nasturtium officinale. Int J Environ Sci Technol 2:63–67
Khan M, Hany O, Khan M (2003) Distribution of pollutants in water and sediment of Gharo creek. Pakistan J Mar Sci 12:25–33
Khan M, Shaukat SS, Alamgir A, Wafra MZ (2012) Impact of industrial effluent on the benthic biodiversity of Gizri Creek, Karachi. Int J Biol Biotechnol 9:317–326
Khan M, Khan M (2001) Impact of industrial discharge on Karachi coastal environment. Proc Eight Stat Sem2001 Karachi University 205–216
Khan SH, Saleem M (1988) A preliminary study of pollution in Karachi harbour marine science of the Arabian Sea. In: Thompson MF, Tirmizi NM (Eds.), American Institute of Biological Sciences, Washington, DC, pp 539–548
Khan MA, Shaukat SS (2008) Physico-chemical characteristics of water and sediments of Chinna creek and the associated benthic biota on its fringes. Int J Biol Biotechnol 5:2
Khatoon Z, Hussain S (2006) Some hydrographic features of Korangi–Phitti Creek. Int J Biol Biotech 3:745–752
Khattak MI, Khattak MI, Mohibullah M (2012) Study of heavy metal pollution in mangrove sediments reference to marine environment along the coastal areas of Pakistan. Pak J Bot 44:373–378
Kinne O (1970) Marine ecology. In: A comprehensive integrated treatise on life in oceans and coastal waters. Wiley, vol 1–4, Hoboken
Leduc G (1984) Cyanides in water: toxicology significance. Aquat Toxicol Series 2:153–224
Mahmood N (1990) Study on imigration of commercially important penaeid shrimp postlarvae in the estuarine area of Chakaria, Cox’s Bazar Bangladesh. Institut Akuakultur Tropika
Martin DF (1972) Marine chemistry vol 1: Analytical methods. Marcel Dekker
Mesnage V, Picot B (1995) The distribution of phosphate in sediments and its relation with eutrophication of a mediterranean coastal lagoon. Hydrobiologia 297:29–41
Muchuweti M, Birkett J, Chinyanga E, Zvauya R, Scrimshaw MD, Lester J (2006) Heavy metal content of vegetables irrigated with mixtures of wastewater and sewage sludge in Zimbabwe: implications for human health. Agric Ecosyst Environ 112:41–48
Nagaraja P, Kumar MSH, Yathirajan HS, Prakash JS (2002) Novel sensitive spectrophotometric method for the trace determination of cyanide in industrial effluent. Anal Sci 18:1027–1030
Navarro AE, Portales RF, Sun-Kou MR, Llanos BP (2008) Effect of pH on phenol biosorption by marine seaweeds. J Hazard Mater 156:405–411
Nriagu JO (1988) A silent epidemic of environmental metal poisoning? Environ Pollut 50:139–161
Qari R, Siddiqui SA, Qureshi NA (2005) A comparative study of heavy metal concentrations in surficial sediments from coastal areas of Karachi. Pakistan Mar Pollut Bull 50:595–599
Qari R, Siddiqui SA (2008) Heavy metal pollution in coastal seawater of Nathia Gali, Karachi (Pakistan). J Environ Res Dev 3:9–19
Qasim S (1982) Oceanography of the Northern Arabian Sea. Deep Sea Res Part A Oceanogr Res Papers 29:1041–1068
Rivera-Monroy VH, Day JW, Twilley RR, Vera-Herrera F, Coronado-Molina C (1995) Flux of nitrogen and sediment in a fringe mangrove forest in Terminos Lagoon, Mexico. Estuar, Coast Shelf Sci 40:139–160
Rizvi S, Saleem M, Baquer J (1988) Steel mill effluents: influence on the Bakran Creek environment. In: Proceedings of marine science of the Arabian sea, pp 549–569
Rubec PJ, Pratt VR (1984) Scientific data concerning the effects of cyanide on marine fish. Freshw Mar Aquar 7:4–6
Saha B, Orvig C (2010) Biosorbents for hexavalent chromium elimination from industrial and municipal effluents. Coord Chem Rev 254:2959–2972
Saha N, Bhunia F, Kaviraj A (1999) Toxicity of phenol to fish and aquatic ecosystems. Bull Environ Contam Toxicol 63:195–202
Saha R, Nandi R, Saha B (2011) Sources and toxicity of hexavalent chromium. J Coord Chem 64:1782–1806
Saifullah S, Khan S, Ismail S (2002) Distribution of nickel in a polluted mangrove habitat of the Indus Delta. Mar Pollut Bull 44:570–576
Sajjad S, Hussain B, Khan MA, Raza A, Zaman B, Ahmed I (2009) On rising temperature trends of Karachi in Pakistan. Clim Change 96:539–547
Saleem M (2002) Study of heavy metal Pollution level and Impact on the fauna and flora of the Karachi and Gawadar Coast. WWF Project Report Project No50022801, 32
Serpa D, Falcao M, Duarte P, da Fonseca LC, Vale C (2007) Evaluation of ammonium and phosphate release from intertidal and subtidal sediments of a shallow coastal lagoon (Ria Formosa–Portugal): a modelling approach. Biogeochemistry 82:291–304
Siddique A (2006) Geographical Information System (GIS) Linked Evaluation of the Ground water pollution and its Sources effecting Environment of Karachi city and its Coastal region.
Siddiqui M, Maajid S (2004) Monitoring of geomorphological changes for planning reclamation work in coastal area of Karachi. Pakistan Adv Space Res 33:1200–1205
Singh KP, Malik A, Mohan D, Sinha S (2004) Multivariate statistical techniques for the evaluation of spatial and temporal variations in water quality of Gomti River (India)—a case study. Water Res 38:3980–3992
Sodré FF, dos Anjos VE, Prestes EC, Grassi MT (2005) Identification of copper sources in urban surface waters using the principal component analysis based on aquatic parameters. J Environ Monit 7:581–585
Sturgeon RE, Siu KM, Willie SN, Berman SS (1989) Quantification of arsenic species in a river water reference material for trace metals by graphite furnace atomic absorption spectrometric techniques. Analyst 114:1393–1396
Thomann RV, Mueller JA (1987) Principles of surface water quality modeling and control. Harper & Row Publishers, Manhattan
Valiela I, Cole ML (2002) Comparative evidence that salt marshes and mangroves may protect seagrass meadows from land-derived nitrogen loads. Ecosystems 5:92–102
Vazquez P, Holguin G, Puente M, Lopez-Cortes A, Bashan Y (2000) Phosphate-solubilizing microorganisms associated with the rhizosphere of mangroves in a semiarid coastal lagoon. Biol Fertil Soils 30:460–468
Wang W, Zhang Y (2007) On fuzzy cluster validity indices. Fuzzy Sets Syst 158:2095–2117
Whiting GJ, McKellar HN, Spurrier JD, Wolaver TG (1989) Nitrogen exchange between a portion of vegetated salt marsh and the adjoining creek. Limnol Oceanogr 34:463–473
Yin K, Lin Z, Ke Z (2004) Temporal and spatial distribution of dissolved oxygen in the pearl river estuary and adjacent coastal waters. Cont Shelf Res 24:1935–1948
Funding
There is no funding source that is available for this research.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Statement on compliance with ethical standards
The authors declare that they have no conflict of interest. This article does not contain any study on the collected or captured animals. Informed consent was obtained from all individual participants of this study.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Alamgir, A., Khan, M.A., Shaukat, S.S. et al. Monitoring of anthropogenic impact on the Port Qasim coastal area, Karachi, Pakistan. Appl Water Sci 14, 4 (2024). https://doi.org/10.1007/s13201-023-02055-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13201-023-02055-5