Abstract
Yamuna river is one of the most important rivers of India and is highly polluted. The river water and the adjacent soil are contaminated with various pollutants including heavy metals. Soil enzymes play important role in various bio-geochemical cycles and help in maintaining nutrient availability of soil. They are very sensitive toward changing environment and therefore act as a key indicator of soil health. In the present study, effects of seasonal variations and heavy metals contamination in Yamuna river water were observed on physicochemical properties and enzyme activities of soil. Soil samples were collected from ten different locations, along the course of the Yamuna river in Delhi–NCR, in three different seasons, i.e., summer, rainy and winter. These sampling sites were divided into three segments, up, middle and lower streams. The concentrations of heavy metals were found to be higher than their safe limit at all the ten sampling points. The total organic carbon content and soil respiration was significantly higher in lower stream. Dehydrogenase enzyme activity was higher in winter season, while urease and arginine deaminase enzyme activity was higher in summer season. Dehydrogenase, arginine deaminase and nitrate reductase enzyme activities were higher in lower streams, while urease enzyme shows maximum activity in the up stream. Thus, we conclude that the continuous application of Yamuna water for irrigation may degrade the soil quality.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Yamuna River plays an important role in day to day life of many people living along its bank and nearby areas. With the rapid increase in population and industrial revolution during the last few decades, it became one of the most polluted rivers in the world. Industrial effluents and household wastes through various major and minor drains are mainly responsible for its current state. In Delhi alone, there are 22 drains that submerge in Yamuna River (Dhillon et al. 2013). Pesticides leaching from agricultural soil and heavy metals from industrial and household wastes are often found in alarming concentration in the Yamuna River. Though few heavy metals act as a cofactor for various enzymes, many are toxic in nature, even at very low concentration (Singh et al. 2011). Since most of the agriculture practices in and around Delhi use Yamuna water for irrigation purpose, there exists a certain risk of these toxic chemicals entering our food chain and causing serious health problems.
Enzymes are vital components of soil, responsible for maintaining soil health (Datt and Singh 2019). Deterioration of soil, and thereby soil health, is one of the major concerns for human, animals and plant health because air and water consumed by them can be adversely affected by contaminated soil (Singer and Ewing 2000). Microorganisms (living or dead), plants roots and residues, and animals are the main source of soil enzymes. About 40–60% of soil enzyme activity comes from enzymes, which are no longer associated with viable cells. So, enzyme activity does not necessarily correlate with soil microbial biomass. In fact, it is the cumulative effect of long-term microbial activity and activity of the viable population at the time of sampling (http://soilquality.org/home.html). However, an exception is the dehydrogenase enzyme activity, which in theory can only occur in viable cells and therefore indicates the activity of viable cells (Salazar et al. 2011).
In the present study, the effect of seasonal variations and Yamuna River water pollution on soil health is studied through various physicochemical properties and soil enzymes activities. The activity of four soil enzymes dehydrogenase, urease, arginine deaminase and nitrate reductase was studied. Dehydrogenase enzyme takes part in the electron transport system of microbial respiration, while urease and arginine deaminase enzymes are responsible for nitrogen metabolism. Nitrate reductase, on the other hand, converts nitrate and nitrite into atmospheric nitrogen and thus completes the nitrogen cycle. Heavy metal content of soil as well river water was also analyzed to observe their contamination level. Therefore, this study helps us in better understanding the complex relationship between microbes and its surrounding environment.
Materials and methods
Soil sampling and sampling sites
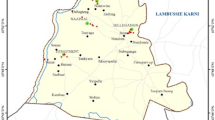
To analyze the impact of seasonal variation and heavy metal concentration on soil enzyme activity, sampling was performed from ten different points along the river course in three different seasons (summer, rainy and winter) for three consecutive years (2010–2011, 2011–2012, 2012–2013) between 28°48′–28°31′ N and 077°12′–077°20′ E (Fig. 1). These points were divided into three different streams, i.e., upstream (least polluted), middle stream (less polluted) and lower stream (most polluted). Soil sampling was done randomly from 1 to 15 cm depth using the rectangular sampler (5 × 5 × 10). At each sampling site, two soil samples were collected, one from river bank and another from nearby fields which are some 100 m away from the river. In order to determine the water pollution status, water sample was also collected from the above-mentioned sampling points. These collected soil samples were stored at 4 °C until further analyzed.
Physicochemical properties of soil and water
Physicochemical analysis except moisture content was performed with air-dried and sieved (< 2 mm) soil samples according to their respective standard techniques. Soil texture was determined through sieving separation method (Gee and Bauder 1986) and analyzed using soil texture triangle (Toogood 1958). Soil moisture content and soil respiration were determined using R. Ohlinger method (Ohlinger 1996). The CHNS content was analyzed using C.H.N.S.O analyzer (Elementar Analysesysteme GmbH Vario EL V3.00) from the University Science Instrument Center, Delhi University. Soil pH was measured by using two different buffers having pH 4.0 and pH 7.0 using Corning pH meter. The metal content of seventeen heavy metals, i.e., iron (Fe), zinc (Zn), cadmium (Cd), lead (Pb), nickel (Ni), arsenic (Ar), cobalt (Co), chromium (Cr), magnesium (Mg), lithium (Li), sodium (Na), potassium (K), calcium (Ca), aluminum (Al), tin (Sn), vanadium (V) and silver (Ag) was determined using AAS (Sensa AAS Dual, GBC, Australia) by EPA’s acid digestion procedure (EPA, 3050B). Total organic carbon of all the soil samples was determined using modified Walkley–Black method (Walkley and Black 1934). Water sample was also subjected to analyze COD, BOD, DO and pH using their respective protocols as described in Methods in Applied Soil Microbiology and Biochemistry by Alef and Nannipieri (1995).
Enzyme activity assays
Enzyme activity assays were performed using the freshly sampled sieved (< 2 mm) soils. The activity of four soil enzymes viz. dehydrogenase, urease, arginine deaminase and nitrate reductase was determined using their respective protocols as described in Methods in Applied Soil Microbiology and Biochemistry by Alef and Nannipieri (1995).
Statistical analysis
Level of significance due to seasonal variations and waste water contamination in all the above-studied parameters were assessed using two-way analysis of variance (ANOVA) followed by the Tukey’s test (p < 0.001, Sigma plot version 12.5) except heavy metal concentration. The statistical significant difference in the heavy metal concentrations was analyzed by three-way ANOVA followed by the Tukey’s test (p < 0.001, SigmaPlot version 12.5).
Results and discussion
Physicochemical properties of soil and water
Soil texture directly affects the physicochemical properties of soil and hence the nutrient availability within the soil (Bowman and Hutka 2002). All the soil samples collected from river bank and fields belong to silty loam class. Moisture content of river bank soil was slightly higher (22.49% in summer to 29.43% in Rainy) than that of field soil (13.69% in summer to 25.54% in Rainy). Carbon and nitrogen are the two extremely important elements, especially in relation with each other, i.e., C/N ratio (Jiana et al. 2016). The ratio of the mass of carbon to the mass of nitrogen in the soil, i.e., C/N ratio is an indicator for nitrogen limitation of plants and other organisms in the soil. The average C/N ratio of the river bank soil samples ranged between 2.1% mg−1 (up stream) and 2.4% mg−1 (lower stream) while in case of field soil, it ranged between 2.0% mg−1 (Up stream) and 2.4% mg−1 (lower stream) and did not show any significant seasonal variations during the entire study period. pH of Yamuna River soil and field soil remained slightly alkaline throughout the study period ranging from 7.2 (up stream) to 8.1 (lower stream). The slightly higher alkaline pH in the lower stream might be due to the presence of organic matter contaminations and high salt concentration in soil. Yamuna River water and field soil showed higher Na and Ca content which may make a buffer for soil pH. The presence of K also helps to stabilize pH between 7 and 8 which is suitable for most enzymatic reaction (http://www.superiorminerals.co.nz). Additions of pesticides, industrial wastes, including various pollutants and heavy metals in the soil and river water, are also responsible for increase in the soil pH (Ying et al. 2013). Physicochemical properties of water samples were also observed (Fig. 2). Increase in BOD while decrease in COD and DO levels signifies the increase in pollution level from up stream to lower stream. COD is a chemical oxygen demand; as the concentration of organic/inorganic chemicals increases in river water, concentration of dissolved oxygen (mg/l) decreases and biological oxygen demand increases (Said and Hussain 2019).
Heavy metal analysis
Heavy metal plays important roles in the activity of soil enzymes. They are generally required only in trace amounts and excess level of any metal have a toxic effect on soil enzyme activities (Bansal 2018). Since most of the agricultural practices in Delhi–NCR depend on Yamuna for irrigation, the field soil is directly exposed to the heavy metals contaminated river water. These heavy metals enter our body through food chains.
The concentrations of heavy metals from Yamuna River water and field soil were analyzed (Fig. 3). The result was discussed and compared with standard value recommended by WHO (Table 1) (http://www.who.int/water_sanitation_health/bathing/srwe1/en/). The mean square value of heavy metal content was found to be higher in the lower stream followed by middle stream and up stream. Sewage water and wastewater are the major source of heavy metal contamination. The heavy metal concentration varied with the seasonal variation, i.e., temperature, rainfall, etc., as summer season showed significantly higher (p < 0.001) concentration followed by winter and rainy seasons. Decrease in the river water level during the summer season lead to increase in concentration of heavy metals in water and sediments (Shanbehzadeh et al. 2014), whereas heavy rainfall and increase in river water level during the rainy season dilutes the heavy metal concentration (Ahmad et al. 2010; Kaur and Mehra 2013).
The concentration of heavy metal in field soil also corresponds to that of river water with lower stream showing significantly higher concentration followed by middle stream and up stream (Fig. 3). However, the concentration level of these heavy metals in field soil was found to be lower than WHO recommended safety limits. Seasonally field soil also showed significantly higher (p < 0.001) mean square value of heavy metal content in the summer season than winter and rainy seasons.
Total organic carbon
The total organic carbon (TOC) was significantly higher (p < 0.001) in lower stream than up and middle stream (Fig. 4A). Total organic carbon includes all the elements (hydrogen, oxygen, nitrogen, etc.) that are components of organic compounds, not just carbon. Organic compounds are the main source of nutrients for soil microbes. TOC is generally used as a nonspecific indicator of water quality and hence indicates soil pollution level (EIA 2013). Sewage and municipal waste from various drains dumping into the Yamuna River in Delhi–NCR are rich source of organic compound and other pollutants (Pascual et al. 2000). Due to higher concentration of pollutants, lower stream might show higher TOC value. However, no significant seasonal variation was observed.
Mean value of total organic carbon (A), soil respiration (B), and soil enzymes activities, i.e., dehydrogenase enzyme (C), urease enzyme (D), arginine deaminase enzyme (E), and nitrate reductase enzyme (F) of Yamuna River soil and field soil during the three years (2010–2013). Values with the same letter are not significantly different from each other. *a, b, c = significant among the three streams at p < 0.001 according to Tukey’s test. *A, B, C = significant among the three seasons at p < 0.001 according to Tukey’s test
Soil respiration
The soil respiration was significantly (p < 0.001) higher in lower stream during the summer season (Fig. 4B). It is the amount of total CO2 released from soil and includes respiration of plant roots, the rhizosphere, microbes and fauna. Various anthropogenic activities led to increase in the global CO2 level, which in turn increases the soil respiration rate (Yu et al. 2011). It increases exponentially with temperature and shows a negative relationship with soil moisture content (Lai et al. 2012). Higher temperature during the summer season and maximum pollution in the lower stream might be the reason for increase in soil respiration.
Enzyme activity assays
Soil enzymes are either intracellular or extracellular in nature. They play a key role in maintaining the nutrient availability within the soil. Any type of fluctuation in the environment and microbial community structure cause variations in their activity (Saxena and Singh 2013).
Dehydrogenase enzyme Dehydrogenase enzyme showed significantly (p < 0.001) higher activity in lower stream followed by middle stream and upstream (Fig. 4C). It is an intracellular enzyme which takes part in the oxidative phosphorylation mechanism of soil microorganisms; thus, it displays strong fluctuation in the activity of soil microorganisms. It plays a key role in cellular respiration and also helps to maintain the carbon cycle in the environment; therefore, its activity varies with the increase in pollution concentration (Moeskops et al. 2010). Sewage sludge is composed of highly oxidizable organic substrate and contains a huge amount of biomass. An increase in the amount of organic pollutants leads to increase in nutrient availability to soil microbes which results in increase the dehydrogenase enzyme activity in soil treated with polluted water (Pascual et al. 2000; Olaniran et al. 2017). These drains are the rich source of sewage sludge and municipal waste which favors the growth of microbial population. Lower stream is the most polluted stretch of Yamuna River because of the various drains which directly or indirectly merge with the Yamuna River. One of the largest drain in Delhi–NCR is the Shahdara drain which discharges its waste at the Noida Bridge in the lower part of the Yamuna River.
Dehydrogenase enzyme activity was maximum in winter season followed by rainy and summer seasons (Fig. 4C). During winter season, low temperature increases the moisture content and decreases the availability of oxygen in the soil therefore favors the anaerobic conditions. Microbes responsible for dehydrogenase activity are obligate anaerobes, and hence, activity increases with high moisture content (Wolińska and Stępniewska 2012). Low moisture content and high heavy metal concentration during the summer season reduce the intracellular water potential, thereby reducing hydration and dehydrogenase enzyme activity. Also frequent drying and rewetting cycles of soil during the rainy season decreases the cellular respiration rate resulting in lower dehydrogenase enzyme activity (Leitner et al. 2015).
Urease and arginine deaminase enzymes Both play significant role in the bio-geochemical cycle of nitrogen. Urea is a most common nitrogen source for bacteria and plants. Chemical hydrolysis of urea within the soil is a very slow process; therefore, hydrolysis of urea in soil is mainly catalyzed by the action of urease enzyme (Dharmakeerthi and Theenabadhu 1996). Arginine, on the other hand, can serve as a source of carbon, nitrogen as well as energy through various metabolic pathways (Lu 2006). Therefore, ammonification by arginine deaminase is generally studied to measure the microbial biomass and their activity in the soil (Alef and Kleiner 1987).
Urease enzyme activity is highly sensitive toward the salinity of soil. Its activity decreases with an increase in the salt concentration of the soil (Adetunji et al. 2017). This might be the reason why the soil urease enzyme activity in the present study was significantly higher in the up stream where the river water is least polluted (Fig. 4D). Moreover, the chemical fertilizers used in various agricultural practices in the Delhi–Haryana border might also be contributed to increase the urease enzyme activity in this region. On the other hand, the activity of arginine deaminase enzyme was significantly higher in the lower stream followed by middle and up stream respectively (Fig. 4E). The presence of excess amounts of nitrogenous waste from various drains, dumping sewage and municipal waste is responsible for the increased arginine deaminase activity in the lower stream. Besides, the arginine deaminase enzyme activity also facilitates the acid-resistant mechanism of various microbes in soil (Colon and Marquis 1988). The urease and arginine deaminase activity was significantly higher in the summer season (Fig. 4D, E). High temperature during the summer season favors both these enzymes activities, as the optimal temperature for urease enzyme activity is reported in the range of 50–60 °C (Bremner and Jantua 1975).
Nitrate reductase enzyme It catalyzes the process of denitrification, the conversion of nitrate and nitrites into atmospheric nitrogen. Denitrifying microbes require very low oxygen concentration of less than 10% therefore; prefer anaerobic conditions (Guimarães et al. 2017). Nitrate reductase enzyme activity was found to be significantly higher (p < 0.001) in lower stream than up and middle stream (Fig. 4F). Various organic pollutants in the lower stream resulted in partial anaerobic conditions within the soil which might enhance the activity of the nitrate reductase enzyme (Pascual et al. 2000). The activity of the nitrate reductase enzyme do not show any significant seasonal variation.
There were a significant correlation between total heavy metal content and enzyme activity, reflecting the common geochemical origin (Table S1) (Nabulo et al. 2010). Although the total soil concentration of heavy metal commonly used to indicate degree of contamination, however, inhibition of soil enzyme activity by heavy metal contamination is well documented (Hagmann et al. 2015; Dindar et al. 2015).
Conclusion
Increase in the heavy metal concentration and overall water pollution level in the Yamuna River led to increase in the activity of dehydrogenase, arginine deaminase and nitrate reductase enzyme while inhibiting the urease enzyme activity. Higher temperature during the summer season increased the arginine deaminase and urease enzyme activity while higher soil moisture content during winter season favored dehydrogenase enzyme activity. Soil enzymes are highly subtle toward any changes in the environment; therefore, they are often used as biomarkers for soil ecosystem. Regular use of polluted river water for irrigation results in degradation of soil health and therefore these soils enzymes can be used as the indicator of soil health.
References
Adetunji AT, Lewu FB, Mulidzi R, Ncube B (2017) The biological activities of β-glucosidase, phosphatase and urease as soil quality indicators: a review. J Soil Sci Plant Nutr 17:794–807
Ahmad MK, Islam S, Rahman S, Haque MR, Islam MM (2010) Heavy metals in water, sediment and some fishes of Buriganga river, Bangladesh. Int J Environ Res 4:321–332
Alef K, Kleiner D (1987) Applicability of arginine ammonification as indicator of microbial activity in different soils. Biol Fert Soils 5:148–151
Alef K, Nannipieri P (1995) Methods in applied soil microbiology and biochemistry, 3rd edn. Academic Press, London
Bansal OP (2018) The influence of potentially toxic elements on soil biological and chemical properties, metals in soil—contamination and remediation. IntechOpen, Rijeka. https://doi.org/10.5772/intechopen.81348
Bowman GM, Hutka J (2002) Particle size analysis. In: McKenzie N, Coughlan K, Cresswell H (eds) Soil physical measurement and interpretation for land evaluation. CSIRO Publishing, Collingwood, pp 224–239
Bremner JM, Jantua MI (1975) Enzyme activity at subzero temperature. Soil Biol Biochem 7:383–387
Colon AC, Marquis RE (1988) Role of the arginine deiminase system in protecting oral bacteria and an enzymatic basis for acid tolerance. App Environ Microbiol 54:1318–1324
Datt N, Singh D (2019) Enzymes in relation to soil biological properties and sustainability. In: Meena R, Kumar S, Bohra J, Jat M (eds) Sustainable management of soil and environment. Springer, Singapore, pp 383–406
Dharmakeerthi RS, Theenabadhu MW (1996) Urease activity in soil: a review. J Natn Sci Coun Srilanks 24:159–195
Dhillon MK, George MP, Mishra S (2013) Water quality of River Yamuna—Delhi stretch. Int J Environ Sci 3:1416–1423
Dindar E, Şağban FOT, Başkaya HS (2015) Variations of soil enzyme activities in petroleum-hydrocarbon contaminated soil. Int Biodeter Biodegr 105:268–275
Gee GW, Bauder JW (1986) Particle-size analysis. In: Klute A (ed) Methods of soil analysis, part 1. Physical and mineralogical methods. Agronomy monograph No. 9. American Society of Agronomy/Soil Science Society of America, Madison, WI, pp 383–411
Guidelines for safe recreational water environments website, http://www.who.int/water_sanitation_health/bathing/srwe1/en/. Last accessed on 30.08.2014
Guimarães LB, Mezzari MP, Daudt GC, Costa RHR (2017) Microbial pathways of nitrogen removal in aerobic granular sludge treating domestic wastewater. J Chem Technol Biotechnol 92:1756–1765
Hagmann DF, Goodey NM, Mathieua C, Evans J (2015) Effect of metal contamination on microbial enzymatic activity in soil. Soil Biol Biochem 91:291–297
Jiana S, Lia J, Chen J, Wang G, Mayesd MA, Dzantora KE, Huie D, Luo Y (2016) Soil extracellular enzyme activities, soil carbon and nitrogen storage under nitrogen fertilization: a meta-analysis. Soil Biolog Biochem 101:32–43
Kaur S, Mehra P (2013) Assessment of heavy metals in summer & winter seasons in river Yamuna segment flowing through Delhi, India. J Environ Ecol 3:149–165
Lai L, Zhao X, Jiang L, Wang Y, Luo L (2012) Soil respiration in different agricultural and natural ecosystems in an arid region. PLoS ONE 7:48011–48019
Leitner S, Saronjic N, Kobler J, Holtermann C, Zechmeister-Boltenstern S, Zimmermann M (2015) Impact of repeated dry-wet cycles on soil CO2 efflux in a beech forest. Geophys Res Abstr 17:EGU2015-15130
Lu CD (2006) Pathways and regulation of bacterial arginine metabolism and perspectives for obtaining arginine overproducing strains. Appl Microbiol Biotechnol 70:261–272
Moeskops B, Sukristiyonubo Bucha D, Sleutel S, Herawat L, Husen E et al (2010) Soil microbial communities and activities under intensive organic and conventional vegetable farming in West Java, Indonesia. Appl Soil Ecol 45:112–120
Nabulo G, Young SD, Black CR (2010) Assessing risk to human health from tropical leafy vegetables grown on contaminated urban soils. Sci Total Environ. 2408:5338–5351
Ohlinger R (1996) soil respiration by titration. In: Schinner F, Öhlinger R, Kandeler E, Margesin R (eds) Methods in soil biology. Springer, Berlin, pp 95–98
Olaniran AO, Balgobind A, Kumar A, Pillay B (2017) Treatment additives reduced arsenic and cadmium bioavailability and increased 1,2-dichloroethane biodegradation and microbial enzyme activities in co-contaminated soil. J Soils Sediments 17:2019
Pascual JA, Garcia C, Hernandez T, Moreno JL, Ros M (2000) Soil microbial activity as a biomarker of degradation and remediation processes. Soil Biol Biochem 32:1877–1883
Pollution in Yamuna River Website, http://www.ndtvmi.com/b8/Dopesheets/chetanyash.pdf. Accessed on 1 June 2014
Said S, Hussain A (2019) Pollution mapping of Yamuna River segment passing through Delhi using high-resolution GeoEye-2 imagery. Appl Water Sci 9:46
Salazar S, Sánchez EL, Alvarez J, Valverde A, Galindo P, Lgual JM et al (2011) Correlation among soil enzyme activities under different forest system management practices. Ecol Eng 37:1123–1131
Saxena N, Singh K (2013) Seasonal variation in dehydrogenase activity and biomass carbon in forest ecosystem. Int J Adv Biotecnol Res 4:241–252
Shanbehzadeh S, Dastjerdi MV, Hassanzadeh A, Kiyanizadeh T (2014) Heavy metals in water and sediment: a case study of Tembi river. J Environ Public Health 1–5
Singer MJ, Ewing S (2000) Soil quality. In: Huang PM, Li YC, Sumner ME (eds) Handbook of soil science. CRC, Boca Raton, pp 271–298
Singh R, Gautam N, Mishra A, Gupta R (2011) Heavy metals and living systems: an overview. Indian J Pharmacol 43:246–253
Soil Quality Website, http://www.soilquality.org.au. Accessed on 1 June 2014
Superior minerals Website, http://www.superiorminerals.co.nz. Last accessed on 5 July 2014
Technically Recoverable Shale Oil and Shale Gas Resources: An Assessment of 137 Shale Formations in 41 countries outside the United States. U.S. Energy Information Administration (EIA) 2013. http://www.eia.gov/analysis/studies/worldshalegas/pdf/fullreport.pdf. Accessed on 30 July 2014
Toogood JA (1958) A simplified textural classification diagram. Can J Soil Sci 38:54–55
USEPA (1996) Method 3050B Acid digestion of sediments, sluges and soils, revision 2, EPA, Washington, USA, 1996, pp 3–5
Walkley A, Black IA (1934) An examination of the detjare method for determining soil organic matter and a proposed modification of the chromic acid titration. J Soil Sci 37:29–36
Wolińska A, Stępniewska Z (2012) Dehydrogenase activity in the soil environment, vol 8. IntechOpen, Rijeka, pp 183–210
Ying W, Jiang F, Qianxin L, Xianguo L, Xiaoyu W, Guoping W (2013) Effects of crude oil contamination on soil physical and chemical properties in Momoge Wetland of China. Chin Geogr Sci 23:708–715
Yu X, Zha T, Pang Z, Wu B, Wang X (2011) Response of soil respiration to soil temperature and moisture in a 50-year-old oriental arborvitae plantation in china. PLoS ONE 6:e28397
Acknowledgements
I implore my profound gratitude to DU-DST Purse program of the University of Delhi, for providing all the necessary research facilities.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Sharma, R., Singh, N.S. & Singh, D.K. Impact of heavy metal contamination and seasonal variations on enzyme’s activity of Yamuna river soil in Delhi and NCR. Appl Water Sci 10, 83 (2020). https://doi.org/10.1007/s13201-020-1166-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13201-020-1166-7