Abstract
The aim of the work was to use the hyperspectral imaging as an analytical tool for the classification of flavoured olive oils, according to composition of the starting blends, crop season, flavouring methods, and flavouring agent. For this reason, in the crop season 2013–2014, three mono-cultivar extra virgin olive oils (Coratina, Peranzana, Ogliarola) were used in different proportions to produce three blends, which were successively flavoured by infusion. From each blend, 3 types of flavoured olive oils were produced: basil, chilli pepper, and garlic + chilli pepper. In the crop season 2014–2015, the study was limited to one of the blends used in the previous season and the basil, chilli pepper, and garlic + chilli pepper flavoured olive oils were produced by infusion or by malaxation. Blends and flavoured oils were submitted to hyperspecral imaging in the 400–1000 nm region. To discriminate the oil samples, ANOVA analysis was performed at each wavelength. Wavelengths in the range 400–570 nm and around 695 nm allowed the discrimination of samples.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Flavoured olive oils are typical products of the Mediterranean Countries, obtained by addition of vegetables, spices, herbs, or fruits in one of the steps of the production process with the aim of enhancing the sensory and nutritional properties of the starting oils. The main methods of flavouring comprise infusion and combined cold pressing or malaxation (Caporaso et al. 2013). Infusion consists in putting into contact dried flavouring material and oil, generally at room temperature under frequent stirring (Riva et al. 1993). A successive filtration separates the solid parts from the liquid. The other flavouring method is the addition of herbs, spices, or vegetables during cold pressing or malaxation. Then, the obtained oil is separated from the malaxation mash by pressure or centrifugation.
It is well known that olive oil characteristics are greatly affected by factors such as environment, altitude, climatic differences and seasonal weather conditions (temperature and rainfall), crop year, ripening degree, cultivars and their fruit mixtures or blends (Baiano et al. 2013; Beltrán et al. 2010; Koprivnjak et al. 2012; Lukić et al. 2015). In the case of flavoured oils, variability of physical, chemical, and sensory characteristics is increased by type and amount of flavouring agent, and flavouring method (Baiano et al. 2016; Caponio et al. 2016). This statement highlights the need to apply a lot of physical, chemical, and sensory analyses to authenticate olive oils. In fact, the concept of authentication is not only circumscribed to adulteration but also include aspects as characterisation, geographical origin, processing systems. Few hundreds of chemical compounds need in authentication to identify the unique position that a specific oil sample occupy in an abstract n-dimensional hyperspace and this score can be reached through the so-called chemometrics i.e. the ability to extract chemically relevant information from results of experiments, by means of statistical and mathematical tools. Nevertheless, chemical, physical, and sensory analyses are generally expensive, time-consuming, and destructive, while the modern research in the food science and technology field is aimed to: increase the number of samples analysed in a given time; repeat the same analysis on the sample; obtain real-time information (Zou and Zhao 2015).
Non-destructive techniques, especially image processing, to detect the quality of agricultural products have been recently investigated (Gamal et al. 2007; Patrick et al. 2004; Park et al. 2007). Nevertheless, the application of image processing to oils is rather limited Xie et al. (2014) successfully applied near-infrared hyperspectral imaging and chemometrics algorithms for the identification of different varieties of sesame oil. To identify the most significant wavelengths, authors applied. Competitive adaptive reweighted sampling (CARS), successive projections algorithm (SPA), and x-loading weights (x-LW). Based on the 64 wavelengths suggested by CARS, the classified models namely least squares support vector machine (LS-SVM) and linear discriminant analysis (LDA) gave the highest classification rate (100%) SPA-LS-SVM and SPA-LDA models obtained over 9559% of classification rate with only 7 wavelengths Gila et al. (2012) proposed an online prediction model for acidity, peroxide index, and humidity content of virgin oils, based on an hyperspectral artificial vision system. They developed two methods for the construction of the model: partial least squares regression (PLS), and partial least squares regression applying a genetic algorithm (GA-PLS).
This study explored the possibility using hyperspectral imaging as an analytical tool for the classification of flavoured olive oils, according to blend composition, crop season, flavouring methods, and flavouring agent.
Materials and methods
Olives and oil-making
The starting extra-virgin olive oils were supplied by Olearia Clemente (Manfredonia, Italy). In the 2013–2014 e 2014–2015 crop seasons, 3 mono-cultivar extra-virgin oils (Ogliarola, Peranzana, and Coratina) were produced by Olearia Clemente (Manfredonia, Italy) by a continuous process including olive grinding through a hammer crusher, malaxation for 30 min at 26 °C, centrifugation through a two phase decanter. Afterwards, in the 2013–2014 crop season, the following 3 blends were produced: B1 (40% Peranzana, 40% Coratina, 20% Ogliarola); B2 (50% Ogliarola, 30% Coratina, 20% Peranzana); B3 (70% Coratina, 20% Ogliarola, 10% Peranzana) and the flavoured olive oils were produced by infusion as in Baiano et al. (2016). In particular, 3 types of flavoured olive oils were produced from each blend: basil flavoured olive oil (B1-b-I-2013, B2-b-I-2013, and B3-b-I-2013—dry basil 5% w/w, daily stirring for 15 days at 15–18 °C); chilli pepper flavoured olive oil (B1-cp-I-2013, B2-cp-I-2013, and B3-cp-I-2013—dry chilli pepper 20% w/w, daily stirring for 7 days at 15–18 °C); garlic & chilli pepper flavoured olive oil (B1-cp&g-I-2013, B2-cp&g-I-2013, and B3-cp&g-I-2013—dry chilli pepper 20% w/w, dry garlic 10% w/w, daily stirring for 7 days at 15–18 °C). After infusion, the oils were filtered through cardboard filters in order to remove the solid fraction.
In the 2014–2015, the experimental design included only the blend B2. In particular, the flavoured olive oils were produced by infusion (B2-b-I-2014, B2-cp-I-2014, and B2-cp&g-I-2014) as described above, and by combined malaxation of olive paste and spices for 30 min at 26 °C in presence of basil 5% w/w or dry chilli pepper 20% w/w or dry chilli pepper 20% w/w, dry garlic 10% w/w (B2-b-M-2014, B2-cp-M-2014, and B2-cp&g-M-2014).
Hyperspectral image system, oil samples acquisition, and hyperspectral image’s pre-processing
Transmittance hyperspectral images of oil samples were acquired by a hyperspectral imaging system (DV srl, Padova, Italy), composed by a 12 bit CCD camera (Pile F-100B, Allied Vision Technologies gmbh, Stadtroda, Germany), coupled to a spectrograph (ImSpector V10, Specim Ltd, Haarlem, The Netherlands) equipped by a standard C-mount f/16 mm lens. The spectral range of this system was 400–1000 nm, having resolution of 5 nm. The light source consisted of a 150 W halogen lamp (EKE 21V150 W, Japan) and of an optic fibre, which transfer the radiation to a flat light diffuser placed on a flat bed plane under the field of view (FOV) of the camera. The flat bed plane was equipped by a step-motor to move the sample through the camera FOV, and scan it by lines. The sample was placed in a Petri dish at a distance of 360 mm from the camera. The camera settings were the same of a previous work conducted by authors (Baiano et al. 2012). The acquisition was controlled through a PC, by the Spectral Scanner software (DV srl, Padova, Italy).
The hyperspectral images were acquired in transmittance mode. Then, the oil samples were contained in Petri’s dishes and placed over the flat light diffuser. After each acquisition session, the hyperspectral image system was calibrating to set the 0–100% transmittance. In particular, scanning an empty Petri’s dish the 100% value was set. The hyperspectral images were stored as a three dimensional form (x, y, λ), where x an y represents the spatial resolution (1000 × 1000 pixels) and λ is the wavelength (nm). The transmittance spectra were obtained using the Spectral Scanner software, by selecting a circular region of interest (ROI), covering the 80% of Petri’s dish. In this manner the edge interferences was avoided. Operating in this way, the transmittance spectra obtained was the mean of the spectra of each pixel falling in the ROI 10 replicates of each oil samples were acquired.
Statistical analysis
To discriminate the oil samples, ANOVA analysis was performed, using Kruskal–Wallis non-parametric test as function of mean separation test (p < 005). For each wavelength, the ANOVA test was performed for 10 replicates of each oil samples arranged in the different combinations considered. The statistical analysis was performed through the software Matlab R2015b (Mathworks inc, Natick, MA, USA).
Results and discussion
Transmittance spectra of unflavoured and flavoured olive oils
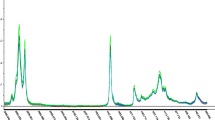
Figures 1 and 2 show the average relative transmittance (amount of light transmitted) spectra of the unflavoured and flavoured oils produced in the crop seasons 2013–2014 e 2014–2015, in the 400–1000 nm range. The statistical analysis was done to select the suitable wavelengths for estimation of difference among samples. In fact, although general trends of spectral curves of the samples were similar, the results of the statistical treatment highlighted significant differences in the visible region, in particular in the range 400–570 nm and around 695 nm. Variation in form of the spectral-transmittance curves for the various oils is proof of the presence of various pigments. In fact, a and b chlorophylls and their derivatives (pheophytins), carotenoids, and flavonoids have distinctive absorption bands in the visible region. In particular, 500 nm is the reflection wavelength of the chlorophyll pigments, while the range 660–700 nm is the reflection band of the anthocyanin pigments (Baiano et al. 2012). Thus, taking into account that transmittance is inversely proportional to absorption, visible absorption or transmittance spectra can be considered a fingerprint of the olive oil and they could be tentatively used in the classifications of samples based on blend composition, crop season, flavouring methods, and flavouring agent.
Average transmittance spectra in a range of 400–1000 nm of the unflavoured and flavoured oil blends produced in the crop season 2013–2014. B1 40% cv Peranzana, 40% cv Coratina, 20% cv Ogliarola; B2 50% cv Ogliarola, 30% cv Coratina, 20% cv Peranzana; B3 70% cv Coratina, 20% cv Ogliarola, 10% cv Peranzana; b basil; cp chilli pepper; cp&g chilli pepper and garlic; I infusion; 2013 crop season 2013–2014; 2014
Average transmittance spectra in a range of 400–1000 nm of the unflavoured and flavoured oil blends produced in the crop season 2014–2015. B2 50% cv Ogliarola, 30% cv Coratina, 20% cv Peranzana; b: basil; cp chilli pepper; cp&g chilli pepper and garlic; I infusion; 2014 M Malaxation; 2014 crop season 2014–2015
The colour of olive oil as it is depends on 2 types of natural pigments, chlorophylls and carotenoids, these account for greenness and yellowness respectively. In olive oils, the following chlorophylls can be found: the bluish-green chlorophyll a and the yellowish–green chloprophyll b [whose ratio oscillates between 6 and 8 (Criado et al. 2007)], pheophytins, chlorophyllides, pheophorbides, pyropheophytins, chlorines, rhodins, and purpurins (Mínguez-Mosquera 1997). Chlorophyll a, which is the main chlorophyll pigment in olive drupes has 2 main absorption maxima at around 429 and 659 nm, while, for example, chlorophyll b has the 2 main absorprtion peak at 455 and 642 nm, and pheophytin a has its absorption maxima at 407 and 665 nm. Chlorophylls also adsorb at 550 nm. Carotenoids including β-carotene, lutein, luteoxanthin, auroxanthin, mutatoxanthin, neoxanthin, violaxanthin, antheraxanthin, and neochrome were reported in olive oil (Moyano et al. 2010). Carotenoids have a significant role in the absorption at 520 nm as demonstrated by the significant correlation of reflectance near 520 nm with carotenoids content (Zur et al. 2000). Carotenoids and chlorophyll have overlapping absorption bands in the blue range, making it difficult to distinguish between them (Zur et al. 2000).
The colour of flavoured olive oils also depends on the flavouring agent. Basil mainly contains chlorophylls and carotenoids (Kopsell et al. 2005) that, added to those of the same pigments in the olive oils, give absorption and transmittance spectra similar to those of unflavoured olive oils for shape but different for intensity. Concerning garlic, blue–green pigments have the ability to form in crushed cloves following a rapid series of enzymatic and non-enzymatic reactions (Lee et al. 2006). According to some researchers, the formed blue–green pigments are thought to consist of tri-, tetra-, or polypyrroles, compounds structurally similar to chlorophyll (Block 2010), while other studies stated that the pigments formed are new “nitrogenous water-soluble compounds differing significantly from all previously reported green pigments in plants” (Lee et al. 2007). These compounds showed absorption at 440 nm and 590 nm (Cho et al. 2009). Chilli pepper pigments include chlorophylls a and b, yellow–orange carotenoids such as α- and β-carotene, zeaxanthin, lutein, and β-cryptoxanthin, and the red carotenoids capsanthin, capsorubin, and capsanthin-5,6-epoxide (Gómez-García and Ochoa-Alejo 2013). In the present research, the use of red chilli peppers strongly modify the absorption and transmittance spectra of the corresponding flavoured olive oils.
The estimation of significant differences among the relative transmittances of samples gave good discrimination at the wavelengths reported in Table 1, as described in the following paragraph.
Discrimination among blends having different composition, crop seasons, flavouring methods, and flavouring agents
In order to discriminate among blends having different composition, the samples B1, B2, and B3 produced in the crop season 2013–2014 were compared. The results of Table 1 show that the relative transmittance spectrum of B1 blend significantly differed from those of the other two blends at 625 nm, the wavelength at which all the chlorophylls absorb although it doesn’t correspond to a main absorption peak. The blends B2 and B3 are indistinguishable from each other at each wavelength in the range 400–1000 nm. Instead, according to a previous study (Baiano et al. 2016), phenolic profile (in particular compounds such as hydroxytyrosol, tyrosol, tyrosol acetate, the dialdehydic form of elenolic acid linked to hydroxytyrosol and tyrosol, pinoresinol, and oleuropein aglycone) and volatile composition (volatile acids, esters, and terpenes) allowed a better discrimination among blends.
The hyperspectral imaging appeared more suitable to discriminate among infusion and malaxation, the two flavouring methods used in the present work. With reference to the 2014–2015 crop season, the flavoured oils obtained by malaxation always showed higher relative transmittance than those produced by infusion at 695 (basil), 505 (chilli pepper), and 495 and 545 (chilli pepper and garlic) nm (Table 1) 695 nm corresponds to the wavelength of maximum absorption of the light-harvesting complex proteins 2 (Lhc2) associated with both photosystem I and II (Ganeteg et al. 2001). Lhc proteins bind chlorophylls a and b and carotenoids with weak, non-covalent bonds. Concerning the differences between the two flavouring methods in chilli peppers and chilli peppers and garlic flavoured oils, they are referred to wavelengths comprised in the maximum absorption range of carotenoids (460–550 nm) or coinciding with those at which the blue–green pigments synthetized in crushed garlic cloves absorb. A previous work of Caponio et al. (2016) demonstrated that the flavouring method significantly affected the phenolic profiles (concentrations of hydroxytyrosol, the dialdehydic form of elenolic acid linked to hydroxytyrosol and tyrosol, tyrosol acetate, 1-acetoxypinoresinol, and pinoresinol), the concentration of sulphur compounds, and sensory properties such as colour, bitter, and pungent intensity.
The presence and type of flavouring agents can be properly discriminate by hyperspectral imaging, which highlighted significant differences among relative transmittances of unflavoured and basil, chilli pepper, and chilli pepper and garlic flavoured oils at 510 or 515 nm, independently on the starting blend (Table 1). These wavelengths just correspond to the main absorption bands of the xanthophyll lutein and of other carotenoids (Croce et al. 2007; Govindjee 1999). In fact, the mean concentrations of lutein and β-carotene is in the following decreasing order: basil (6.64 and 6.01 mg/100 g fresh weight); chilli pepper (< 0.3 and < 1.5 mg/100 g fresh weight); garlic (0.05 mg and not determined in 100 g of dry weight) (Kopsell et al. 2005; Aruna and Baskaran 2010; Pugliese et al. 2013).
Hyperspectral imaging was also suitable to discriminate the crop season of the olive oils independently on the presence of a flavouring agent. In fact, unflavoured and flavoured oils of the 2014–2015 crop season showed higher relative transmittance that those of the 2013–2014 crop season (Table 1). The differences between the unflavoured oils of the two crop seasons were highlighted at 425 and 500 nm (absorption of chlorophyll a and carotenoids, respectively). The differences between the basil flavoured oils were observed in the range 400–695 nm (absorption of chlorophylls and carotenoids). The differences between the chilli pepper flavoured oils were observed in the already discussed 490–695 nm and in the range 725–855 nm. This latter range is interesting since a previous work (Koehne et al. 1999) correlated the ability of adapting of the green alga Ostreobium to low-light conditions in which wavelengths > 700 nm prevail to a red form of chlorophyll a-containing light-harvesting complex, namely Lhca1 and possibly also Lhca4. The differences between the chilli pepper and garlic flavoured oils were observed at the already discussed 695 nm and in the range 570–715 nm, which includes absorption of chlorophylls and carotenoids. These results allow to hypothesize that the differences highlighted are related to the different concentrations in chlorophyll and carotenoids of the starting blends within the 2 considered crop years. In this regard, it is well known that seasonal aspects (including climatic conditions such as temperature and rainfall) influence the physiology of the plant and thus the quality of the olive oil (including content of minor components such as chlorophyll and carotenoids) (Panelli et al. 1994). In particular, in the considered location, (http://www.ilrestodelgargano.it/attualita/2014/11/news/anno-nero-per-le-olive-crolla-la-produzione-in-puglia-prezzi-in-rialzo-4457.html/) the crop year 2014/2015 was characterized by unfavourable weather conditions, the massive presence of the olive fruit fly, and the strong winds that determined a consistent fall of olives. As results, the Italian production of olive oil was reduced by 35% over the previous crop year together with a lower olive oil quality.
Conclusion
Unflavoured and flavoured olive oils were analysed by means of hyperspectral imaging in the 400–1000 nm range. The transmittance spectra provided a sort of fingerprint of the oils. In particular, the processing of the spectral data the range 400–570 nm and around 695 nm was appropriate for the classification of flavoured olive oils, according to crop season, flavouring methods, and flavouring agent. The obtained data are preliminary results but they encourage the use of hyperspectral imaging as non-destructive tools in replacement of analytical instruments for the quality control of unflavoured and flavoured olive oils.
References
Aruna G, Baskaran V (2010) Comparative study on the levels of carotenoids lutein, zeaxanthin and b-carotene in Indian spices of nutritional and medicinal importance. Food Chem 123:404–409
Baiano A, Terracone C, Peri G, Romaniello R (2012) Application of hyperspectral imaging for prediction of physico-chemical and sensory characteristics of table grapes. Comput Electron Agric 87:142–151
Baiano A, Terracone C, Viggiani I, Del Nobile MA (2013) Effects of cultivars and location on quality indices, phenolic content and antioxidant activity of extra-virgin olive oils. J Am Oil Chem Soc 90:103–111
Baiano A, Previtali MA, Viggiani I, Varva G, Squeo G, Paradiso VM, Summo C, Gomes T, Caponio F (2016) As oil blending affects physical, chemical, and sensory characteristics of flavoured olive oils. Eur Food Res Technol. https://doi.org/10.1007/s00217-016-2669-1
Beltrán G, Jiménez A, del Rio C, Sánchez S, Martínez L, Uceda M, Aguilera MP (2010) Variability of vitamin E in virgin olive oil by agronomical and genetic factors. J Food Comp Anal 23:633–639
Block E (2010) Garlic and other alliums: the lore and the science. Royal Society of Chemistry, Cambridge
Caponio F, Durante V, Varva G, Silletti R, Previtali MA, Viggiani I, Squeo G, Summo C, Pasqualone A, Gomes T, Baiano A (2016) Effect of infusion of spices into the oil vs combined malaxation of olive paste and spices on quality of naturally flavoured virgin olive oils. Food Chem 202:221–228
Caporaso N, Padano A, Nicoletti G, Sacchi R (2013) Capsaicinoids, antioxidant activity, and volatile compounds in olive oil flavoured with dried chilli pepper (Capsicum annuum). Eur J Lipid Sci Technol 115:1434–1442
Cho J, Lee E, Yoo K, Lee S, Patil B (2009) Identification of candidate amino acids involved in the formation of blue pigments in crushed garlic cloves (Allium sativum L). J Food Sci 74:C11–C16
Criado MN, Romero MP, Motilva MJ (2007) Effect of the technological and agronomical factors on pigment transfer during olive oil extraction. J Agric Food Chem 55:5681–5688
Croce R, Mozzo M, Morosinotto T, Romeo A, Hienerwadel R, Bassi R (2007) Singlet and triplet state transitions of carotenoids in the antenna complexes of higher-plant photosystem I. Biochem 46:3846–3855
del Rocío Gómez-García M, Ochoa-Alejo N (2013) Biochemistry and molecular biology of carotenoid biosynthesis in chili peppers (Capsicum spp). Int J Mol Sci 14:19025–19053
Gamal E, Ning W, Adel E, Michael N (2007) Hyperspectral imaging for nondestructive determination of some quality attributes for strawberry. J Food Eng 81:98–107
Ganeteg U, Strand Å, Gustafsson P, Jansson S (2001) The properties of the chlorophyll a/b-binding proteins Lhca2 and Lhca3 studied in vivo using antisense inhibition. Plant Physiol 127:150–158
Gila DM, Cano Marchal P, Gámez Garcia J, Gómez Ortega J (2012) Hyperspectral imaging for determination of some quality parameters for olive oil. In Proceedings of 18th international conference on automation and computing (ICAC), Loughborough 7–8 September 2012, pp 1–6
Govindjee (1999) Carotenoids in photosynthesis: an historical perspective. In: Frank HA, Young AJ, Britton G, Cogdell RJ (eds) The photochemistry of carotenoids. Kluwer Academic Publishers, Dordrecht, pp 1–19
Koehne B, Elli G, Jennings RC, Wilhelm C, Trissl HW (1999) Spectroscopic and molecular characterization of a long wavelength absorbing antenna of Ostreobium sp. BBA Bioenerg 1412:94–107
Koprivnjak O, Majetić V, Brkić Bubola K, Kosić U (2012) Variability of phenolic and volatile compounds in virgin olive oil from Leccino and Istarska Bjelica cultivars in relation to their fruit mixtures. Food Technol Biotechnol 50:216–221
Kopsell DA, Kopsell DE, Curran-Celentano J (2005) Carotenoid and chlorophyll pigments in sweet basil grown in the field and greenhouse. HortSci 40:1230–1233
Lee EJ, Cho J, Lee S (2006) The chemical basis of green pigment formation (“greening”) in crushed garlic (Allium sativum L.) cloves. Food Sci Biotechnol 15:838–843
Lee EJ, Cho J, Kim J, Lee S (2007) Green pigment in crushed garlic (Allium sativum L) cloves: purification and partial characterization. Food Chem 101:1677–1718
Lukić M, Lukić I, Sladonja B, Piližota V (2015) Variability of 4-monomethylsterols and 4,4′-dimethylsterols in olive oil and their use as indicators of olive variety, ripening degree, and oil storage temperature. J Agric Food Chem 63:499–5508
Mínguez-Mosquera MI (1997) Clorofilas y carotenoides en tecnología de alimentos. Sevilla Secretariado de publicaciones de la Universidad de Sevilla, Spain, p 189
Moyano MJ, Heredia FJ, Meléndez-Martínez AJ (2010) The color of olive oils: the pigments and their likely health benefits and visual and instrumental methods of analysis. Compr Rev Food Sci Food Saf 9:278–291
Panelli G, Servili M, Selvaggini R, Baldioli M, Montedoro GF (1994) Effect of agronomic and seasonal factors on olive (Olea europaea L.) production and on the qualitative characteristics of the oil. Acta Hortic 356:239–244
Park B, Windham WR, Lawrenc KC, Smith DP (2007) Contaminant classification of poultry hyperspectral imagery using a spectral angle mapper algorithm. Biosyst Eng 96:323–333
Patrick MM, Yud-Ren C, Moon SK, Diane EC (2004) Development of hyperspectral image technique for the detection of apple surface defects and contaminations. J Food Eng 61:67–81
Pugliese A, Loizzo MR, Tundis R, O’Callaghan Y, Galvin K, Menichini F, O’Brien N (2013) The effect of domestic processing on the content and bioaccessibility of carotenoids from chili peppers (Capsicum species). Food Chem 141:2606–2613
Riva M, Di Cesare LF, Schiraldi A (1993) Microwave and traditional technology to prepare garlic aromatized olive oil. In: Charalambous G (ed) Food flavours, ingredients and composition. Elsevier, Holland
Xie C, Wang Q, He Y (2014) Identification of different varieties of sesame oil using near-infrared hyperspectral imaging and chemometrics algorithms. PLoS ONE 9:e98522
Zou X, Zhao J (2015) Nondestructive measurement in food and agro-products. Science Press, Beijing
Zur Y, Gitelson AA, Chivkunova OB, Merzlyak MN (2000) The spectral contribution of carotenoids to light absorption and reflectance in green leaves. In: Proceedings of the 2nd international conference geospatial information in agriculture and forestry, Buena Vista, FL, January 10–12, 2000, v 2
Acknowledgements
Research supported with the contribute of a project co-financed by EU “PO Puglia FESR 2007/2013, Asse I, Linea 12, Azione 124, Bando—Aiuti a sostegno dei partenariati regionali per l’innovazione Investiamo nel vostro futuro”.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Romaniello, R., Baiano, A. Discrimination of flavoured olive oil based on hyperspectral imaging. J Food Sci Technol 55, 2429–2435 (2018). https://doi.org/10.1007/s13197-018-3160-8
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13197-018-3160-8