Abstract
Anaplastic thyroid carcinoma (ATC) is an aggressive malignancy with dismal outcome especially in metastatic setting. Consensus for ideal treatment of advanced and metastatic ATC remains elusive. This study aimed to analyze the impact of palliative chemotherapy versus supportive care on overall survival in patients with metastatic anaplastic thyroid carcinoma. Patients diagnosed with ATC between the period January 2018 and December 2019 were prospectively followed. The patients opting for palliative chemotherapy received 3 weekly Paclitaxel (175 mg/m2) and Carboplatin (AUC-5). Out of the 31 patients diagnosed with ATC, clinicopathological profile of 29 patients was analyzed (2 patients who underwent upfront surgical resection with curative intent were excluded), out of which 20 patients were included in the survival analysis. The median age of presentation was 55.8 years with male:female ratio 1.9:1. Seventeen out of the total 29 patients presented with anaplastic transformation in long-standing goiter. Nineteen out of 20 (95%) patients presented with distant metastasis with lungs being the most common site. Nodal metastasis was present in all patients. Invasion of the strap muscles (90%) and trachea (80%) was the most common peri-thyroidal tissue invasion followed by invasion of the esophagus (40%), internal jugular vein (30%), and carotid artery (5%). Twelve out of the 20 patients opted for palliative chemotherapy. Overall, median survival from the time of diagnosis was 2.6 months, with median survival in patients receiving chemotherapy 3.1 months and those opting for supportive care 1.6 months (p=0.004). Out of all the factors analyzed, male sex (HR 6.521, 95% CI 1.143–37.206, p value 0.03) and vascular invasion (HR 0.066, 95% CI 0.009–0.499, p value 0.008) were poor prognostic indicators. Palliative chemotherapy showed increased survival benefit in patients with metastatic ATC. Male sex and vascular invasion were found to be significant factors associated with poor outcomes on Cox regression analysis.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Anaplastic thyroid carcinoma (ATC) accounts for less than 1% of thyroid malignancies worldwide but is responsible for 14–39% of mortality attributable to thyroid malignancies [1]. Despite multimodal treatment, the median survival in ATC ranges between 1 and 5 months [2]. Fifteen percent patients present with extensive local disease and distant metastasis is found in 50% patients on further work up [3]. Older age, male gender, bilateral tumors, large primary tumors at presentation, presence of distant metastasis, extensive local invasion, and elevated leucocyte count are unfavorable prognostic factors in patients of ATC [4]. Surgery followed by external beam radiotherapy (EBRT) and chemotherapy are preferred options for resectable tumors. Options for locally advanced and metastatic ATC include radiation with chemotherapy, kinase inhibitors, and immune modulator. Stand-alone chemotherapy is rarely used alone for control of locally advanced disease; but same is recommended by American Thyroid association in a setting of metastatic disease and remains a valid option for metastatic disease due to dearth of RT slots, and also because the metastatic foci may be determinant of overall survival [2]. Doxorubicin, platins, and taxanes have been used as palliative chemotherapy in various combinations [5, 6]. These regimes are affordable compared to newer kinase inhibitors and immunomodulators and remain a valid option in resource constrained setting. Also, clinical trials of potentially effective agents for ATC are hampered by its low incidence and aggressive behavior that limits enrollment and treatment time frame. This study aims to evaluate the impact of palliative combination chemotherapy as a stand-alone therapy on overall survival in patients with inoperable metastatic ATC.
Materials and Methods
Patient Selection
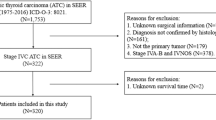
Patients diagnosed with inoperable ATC between January 2018 and December 2019 in the Department of Endocrine Surgery (King George’s Medical University, Lucknow, India) were prospectively followed. The diagnosis of ATC was made by fine needle aspiration cytology (FNA) and core needle biopsy specimens along with immunohistochemistry. Imaging included high-resolution ultrasound (HR USG) of neck, plain radiographs, contrast enhanced computed tomography (CECT) of neck and thorax, and laryngoscopy/upper GI endoscopy for determining the extent of locoregional invasion and staging. USG/CECT abdomen was done to rule out metastasis. FDG PET scan was used selectively. Staging was done according to the AJCC-TNM 8th edition. Patients with operable tumors at the time of presentation (stage IVA), performance status-ECOG (Eastern Cooperative Oncology Group) 3–4, those lost to follow-up, with discordant FNA and core needle biopsy reporting, and incomplete records/lack of imaging were excluded from the survival analysis (Fig. 1).
Proposed protocol for patient selection for palliative chemotherapy. Abbreviations: ATC anaplastic thyroid carcinoma, HRUSG high-resolution ultrasonography, CECT contrast-enhanced computed tomography, FNAC fine needle aspiration cytology, IHC immunohistochemistry, MRI magnetic resonance imaging, PET-CT positron emission tomography, PS performance status, ECOG Eastern Cooperative Oncology Group
In view of limited life expectancy, locally advanced metastatic disease, and restricted resource setting, systemic control of disease with chemotherapy was planned alternatively to radiotherapy for locoregional control. Patients with ECOG 0–2 were given option of palliative chemotherapy versus supportive care, after due discussion with the patients and their caregivers. Patients opting for chemotherapy received 3 weekly Paclitaxel (175 mg/m2) and Carboplatin (AUC-5). Patients not opting for chemotherapy received only supportive care and symptomatic treatment. As patients were metastatic at the time of presentation, none of the patients received locoregional radiotherapy. The records of all patients were retrospectively analyzed (Fig. 2).
Statistical Analysis
Data of all 29 patients were used for evaluation of demographic and clinical presentation. Nine patients were excluded from radiological and survival analysis. Patient characteristics were summarized using counts and percentages for categorical variables, and medians and interquartile range for continuous variables. Cox regression models were used to identify risk factors contributing to overall survival (OS). Survival rates were estimated using the Kaplan-Meier method, and the significance of comparisons was calculated by the log-rank test. All statistical analyses were conducted with IBM SPSS version 24 (Chicago, IL, USA). A two-sided P value < 0.05 was considered to be statistically significant.
Results
Of the 29 patients, 19 (65.51%) were males and 10 (34.49%) were females. The median age of presentation was 55.8 years (range 32–78 years), with 10 patients (34.49%) less than 55 years of age. Seventeen (58.62%) out of the total 29 patients had history of long-standing goiter (Table 1).
The median duration between onset of symptoms and diagnosis was 3.5 months. This duration was calculated from the point of acute onset of symptoms and excluded the duration of long-standing goiter. Most common clinical presentation was rapid progression of neck mass seen in 28 (96.55%) out of 20 patients, followed by dyspnea (75.86%), voice change (62.06%), neck and shoulder pain (55.17%), dysphagia (41.32%), and stridor (27.58%). Laboratory investigations revealed leukocytosis in 25 (86.20%) patients with mean leucocyte count of 17846.6/mm3. Two (6.89%) patients had suppressed serum TSH levels with elevated serum T4 levels at the time of presentation (Table 2).
Radiologically (n=20), both lobes were involved in 4 (20%) patients, right lobe in 10 (50%), and left lobe in 6 (30%) patients. Infiltration was most commonly seen in the strap muscles (90%), followed by the trachea (80%), esophagus (40%), recurrent laryngeal nerve (40%), internal jugular (30%), and least in carotid artery (5%). Nodal metastasis was seen in all patients, and the lung was the most common site of distant metastasis (Table 3).
Out of the 20 patients included for survival analysis, 12 (60%) patients opted for palliative chemotherapy and rest received supportive care. Seven male patients and 5 female patients opted for palliative chemotherapy (OR 0.467, 95% CI 0.65–3.34, p-value 0.48). Out of the 12 patients that received palliative chemotherapy, only 2 patients completed all 6 cycles of 3 weekly palliative chemotherapy regimen. Overall, the median time of survival was 2.8 months after diagnosis. For the patients who received chemotherapy, the median survival was 3.46 months as compared to those who received only supportive care whose median survival was 1.64 months (p-value 0.004, significant) (Fig. 3). As far as the chemotherapy-related toxicities were concerned, 3 (15%) patients developed grade I neutropenia and 1 (5%) patient had significant chemotherapy-induced nausea and vomiting (CINV), which responded to neurokinin1 inhibitors. The 3 weekly taxane- and platin-based chemotherapy regimen was otherwise well tolerated. Partial response, in terms of reduction in size of swelling, dysphagia, and dyspnea, was seen in 7 out of the total 12 patients who received chemotherapy, after 2 cycles. However, these patients subsequently progressed and succumbed to the disease. Down-staging was not seen in any of the patients.
On preforming Cox regression analysis to identify factors affecting overall survival, it was found that only male sex (HR 6.521 95% CI 1.143–37.206 p value 0.03) and vascular invasion (HR 0.066 95% CI 0.009–0.499 p value 0.008) were significantly associated with reduced overall survival. Other factors like age >55 years, long-standing goiter, infiltration of trachea and esophagus, distant metastasis, and leukocytosis did not significantly affect the outcome in patients with advanced ATC (Table 4) (Fig. 4).
Discussion
ATC is one of the most aggressive solid malignancies in humans, with extremely poor prognosis. There is no consensus on the appropriate management in advanced and metastatic ATC, with focus on palliation and quality of life. There are no randomized trials to conclusively demonstrate the efficacy of one regime over another among the various chemotherapy agents used in advanced ATC. Moreover, response to chemotherapy cannot be assessed on the conventional basis using RECIST criteria, making results of the of various trials difficult to interpret [7, 8]. Some studies have shown better survival benefit when chemotherapy is combined with radiotherapy as compared to chemotherapy alone [1]. This study assessed the effect of palliative chemotherapy on overall survival in patients with advanced and metastatic ATC and factors assessing the overall survival.
The reported mean age of presentation is seventh decade, with less than 10% patients less than 50 years of age. Age has been found to be an independent prognostic factor in patients with ATC [7]. This is in contrast to the present study, where 34.49% of patients were less than 55 years of age. As far as gender is concerned, 60–70% of all tumors occur in women [8]. However, in the present study, more than half of the study population are males. In relation to the Indian subcontinent, the distribution of ATC is almost equal between males and females as demonstrated by Pradhan et al. with male:female ratio 1:1.2 [9]. The median time elapsed between onset of symptoms to diagnosis was 3.5 months, which was more than the international average of 1.5 months [10]. Such a delay may be the cause of late presentation with advanced inoperable disease and metastasis. The delay may be due to the higher percentage of rural dwellers (75.86%), with difficult access to tertiary health care facilities and thyroid surgeons. Pre-existing goiters have been identified as a risk factor for malignant transformation [11], which were present in more than half the population in the present study. The most common presentation was a rapidly enlarging neck mass which was present in 96.55% of the whole study group. The same results were shown by Nagaiah et al. [12] and Giuffrida et al. [13]. Overall, distant metastasis at the time of presentation are seen in 15–50% patients and nodal metastasis in 40% patients [8, 10]. This is in contrast the present study where distant metastasis was seen in 95% of the study population (most common site being the lungs) and nodal metastasis in all patients.
Radiologically, infiltration into the strap muscles was most commonly seen (90%) followed by the trachea. The involvement of the recurrent laryngeal nerve was seen in 40% patients. Both FNAC and core needle biopsy were performed in all patients for confirmation of diagnosis, as FNA alone may be inconclusive due to extensive necrosis. Histopathological diagnosis was supplemented by immunohistochemistry markers.
Only 12 out of the total 20 patients accepted palliative chemotherapy. The rest consented for supportive care. Based on the ATA recommendations for palliative therapy in ATC [2], locally available agents, and patient affordability, patients received 3 weekly platin- and taxane-based regimen. Chemotherapy as a single modality of treatment for advanced ATC has been sparsely studied across literature. This may be due to the variable response of ATC to chemotherapeutic agents due to overexpression of multidrug resistance-associated protein, under expression of multiple drug resistance (mdr1) mRNA and P-glycoprotein and high volume of tumor-associated macrophages (TAMs) occupying about 50% of tumor [14]. Doxorubicin and taxane are commonly used chemotherapeutic agents with response rates of up to 22% and 54%, respectively [15, 16]. Platin-based therapies have been shown to have a poor response rate as compared to doxorubicin and paclitaxel [17]. In the present study, partial response, in terms of reduction in size of swelling, dysphagia, and dyspnea, was seen in 7 out of 12 patients who received chemotherapy, after 2 cycles (Fig. 5). For the patients who received chemotherapy, the median survival was 3.46 months as compared to those who received only supportive care whose median survival was 1.64 months (p-value 0.004, significant). Various studies have shown conflicting evidence regarding use of chemotherapy in patients with ATC, and most of these studies have enrolled operable patients of ATC with focus on the post-operative outcomes with chemotherapy. In meta-analysis by Xin et al. [17], the overall survival was unaltered by chemotherapy alone. Sosa et al. [18], Koussis et al. [19], and Busnardo et al. [20] showed improved overall survival with the use of chemotherapy when compared to patients who did not receive any systemic therapy. As more data becomes available regarding the molecular pathogenesis of ATC, newer agents like tyrosine kinase inhibitors, BRAF inhibitors (Dabrafenib/Trametinib), fusion inhibitors (ALK fusion-Crizotinib/Ceritinib, RET fusion - Pralsetinib/Selpercatinib, NTRK fusion-Larotrectinib/Entrectinib), anti-EGFR agents, agents targeting NF-kB pathway, farnesyl-transferase, and PPAR have shown promising results [2]. Average cost of locally available targeted therapy with agents like dabrafenib, which have shown increased progression free survival, range from INR 6lacs-10lacs (8000–13600 USD) per cycle versus INR 10000–14000 (135–190 USD) per chemotherapy cycle (Fig. 6). Hence, due to high cost, restricted patient affordability, and limited local availability, conventional chemotherapy continues to remain a feasible option compared to the newer agents in a resource-limited setting.
Prognostication of patients with thyroid malignancy depends upon age, sex, size, extrathyroidal extension, and metastasis among other factors. Sugitani et al. [8] and Smallridge et al. [21] found that presence of acute symptoms, large tumor (>5cm), distant metastasis, and leukocytosis > 10,000/cumm (collectively called prognostic index), along with age, sex, extent of surgery, higher-dose radiotherapy, coexistence of differentiated thyroid cancer, and multi-modality therapy were significant factors affecting prognosis; and were useful for selecting patients for aggressive multimodal therapy. In the present series, only male sex and vascular invasion were associated with poor prognosis on Cox regression model.
Although patients who received chemotherapy showed improvement in symptoms and overall survival, the present study had certain limitations. Retrospective nature of the study comes with its own set of biases. As anaplastic thyroid carcinoma is less common, the number of patients in the study is limited. Histological subclassification of ATC was lacking, which could have impacted the prognostication of patients. Patients who received systemic therapy did not undergo radiation which may have added benefit on locoregional disease and symptom control. The response to palliative chemotherapy was subjective in terms of symptom improvement and size reduction. Lastly, quality of life assessment for patients on chemotherapy versus those on supportive care is warranted, as chemotherapy in the setting of advanced ATC is palliative. The study of newer agents in the setting of a metastatic disease and its impact on overall survival is also essential.
Conclusion
Although the prognosis of patients with ATC, whether operable or advanced, continues to be poor, palliative treatment with single modality like chemotherapy can bring symptom relief along with improvement in overall survival of patients with metastatic disease. Further prospective studies are required to determine appropriate palliative treatment protocols for advanced ATC.
References
Sun XS, Sun SR, Guevara N et al (2013) Chemo radiation in anaplastic thyroid carcinomas. Crit Rev Oncol/ Haematol 86(3):290–301
Bible KC, Kebebew E, Brierley J, Brito JP, Cabanillas ME, Clark TJ Jr, Di Cristofano A, Foote R, Giordano T, Kasperbauer J, Newbold K, Nikiforov YE, Randolph G, Rosenthal MS, Sawka AM, Shah M, Shaha A, Smallridge R, Wong-Clark CK (2021 Mar) 2021 American Thyroid Association guidelines for management of patients with anaplastic thyroid cancer. Thyroid. 31(3):337–386
Chen J, Tward JD, Shrieve DC, Hitchcock YJ (2008) Surgery and radiotherapy improves survival in patients with anaplastic thyroid carcinoma. Am J Clin Oncol 31(5):460–464
Sharifi A, Shojaeifard A, Soroush A, Jafari M, Abdehgah AG, Mahmoudzade H (2016) Predictors of regional lymph node recurrence after initial thyroidectomy in patients with thyroid cancer. J Thyroid Rese 2016: 4127278
Shimaoka K, Schoenfeld DA, DeWys WD, Creech RH, DeConti R (1985) A randomized trial of doxorubicin versus doxorubicin plus cisplatin in patients with advanced thyroid carcinoma. Cancer 56(9):2155–2160
Higashiyama T, Ito Y, Hirokawa M et al (2010) Induction chemotherapy with weekly paclitaxel administration for anaplastic thyroid carcinoma. Thyroid 20(1):7–14
Sugitani I, Kasai N, Fujimoto Y, Yanagisawa A (2001) Prognostic factors and therapeutic strategy for anaplastic carcinoma of the thyroid. World J Surg. 25:617–22
Kebebew E, Greenspan FS, Clark OH, Woeber KA (2005) McMillan A Anaplastic thyroid carcinoma. Treatment outcome and prognostic factors. . Cancer. 103(7):1330
Pradhan R, Agarwal A, Lal P et al (2018) Clinico-pathological profile of anaplastic thyroid carcinoma in an endemic goiter area. Indian J Endocrinol Metab. 22(6):793–797. https://doi.org/10.4103/ijem.IJEM_264_18
Tennvall J, Lundell G, Wahlberg P, Bergenfelz A, Grimelius L, Akerman M et al (2002) Anaplastic thyroid carcinoma: three protocols combining doxorubicin, hyperfractionated radiotherapy and surgery. Br J Cancer. 86:1848–53
Hundahl SA, Cady B, Cunningham MP, Mazzaferri E, McKee RF, Rosai J et al (2000) Initial results from a prospective cohort study of 5583 cases of thyroid carcinoma treated in the united states during 1996. U.S. and German Thyroid Cancer Study Group. An American College of Surgeons Commission on Cancer Patient Care Evaluation study. Cancer. 89:202–17
Nagaiah G, Hossain A, Mooney CJ, Parmentier J, Remick SC (2011) Anaplastic thyroid cancer: a review of epidemiology, pathogenesis, and treatment. J Oncol. 2011:542358
Giuffrida D, Gharib H (2000) Anaplastic thyroid carcinoma: current diagnosis and treatment. Ann Oncol. 11:1083–9
Lehnert M (1996) Clinical multidrug resistance in cancer: a multifactorial problem. Eur J Cancer. 32(6):912–920
Ahuja S, Ernst H (1987) Chemotherapy of thyroid carcinoma. J Endocrinol Invest. 10(3):303–310
Ain KB, Egorin MJ, DeSimone PA (2000) Treatment of anaplastic thyroid carcinoma with paclitaxel: phase 2 trial using ninety-six-hour infusion. Collaborative Anaplastic Thyroid Cancer Health Intervention Trials (CATCHIT) Group. Thyroid. 10(7):587–594
Xia Q, Wang W, Xu J, Chen X, Zhong Z, Sun C (2018) Evidence from an updated meta-analysis of the prognostic impacts of postoperative radiotherapy and chemotherapy in patients with anaplastic thyroid carcinoma. Onco Targets Ther. 11:2251–2257
Sosa JA, Elisei R, Jarzab B, et al (2010) Randomized phase 2/3 trial of a tunor vascular disrupting agent, fosbertubulin (CA4P), with carboplatin(C), and paclitaxel(P) in anaplastic thyroid cancer(ATC): interim safety and efficacy results of the FACT trial. Milan, Italy
Koussis H, Scola A, Tonello S et al (2006) Multimodality therapeutic approach in anaplastic thyroid cancer (ATC): study of 56 patients. J Clin Oncol. 24(18S, supplement) ASCO Annual Meeting Proceedings Part I. Vol. 24
Busnardo B, Daniele O, Pelizzo MR et al (2000) A multimodality therapeutic approach in anaplastic thyroid carcinoma: study on 39 patients. J Endocrinol Investig. 23(11):755–761
Smallridge RC, Copland JA (2010) Anaplastic thyroid carcinoma: pathogenesis and emerging therapies. Clin Oncol 22:486–497
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Garg, S., Mouli, S., Singh, K.R. et al. Revisiting Combination Chemotherapy as a Single Modality Palliative Therapy for Advanced Anaplastic Thyroid Carcinoma—a Single Institution Experience. Indian J Surg Oncol 13, 208–215 (2022). https://doi.org/10.1007/s13193-021-01401-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13193-021-01401-w