Abstract
Cancers of the appendix are rare. Most of them are found accidentally on appendectomies performed for appendicitis. When reviewed, majority of the tumors were carcinoid, adenoma, and lymphoma. Adenocarcinomas of the appendix are only 0.08% of all cancers, and the treatment remains controversial. It can present as appendicitis or lump or abscess. Gastrointestinal tuberculosis most commonly involves the ileocecal region. Disruption of the integrity of mucosal barriers and impairment in cell-mediated immunity associated with cancerous growth are known to predispose to bacterial infection. The role of antituberculosis therapy and chemotherapy is unclear due to lacking randomized trials but seems to be accepted if there is lymph node involvement or peritoneal seeding. Here, we are reporting a 69-year-old lady presented with symptoms of abdominal wall abscess with tuberculosis infection which was then diagnosed with mucinous adenocarcinoma of the appendix. The patient was treated with incision and drainage followed by the right hemicolectomy. Up to date, she remains asymptomatic and continuing with chemotherapy.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Vermiform appendix is a vestigial organ in human body; a questionable immunological significance, but very rarely, the appendix may become cancerous. Cancer of the appendix may cause appendicitis or rupture of the appendix. Most of the appendicular malignancy present as appendicitis. The majority of appendiceal tumors are carcinoids 80–90%, while the remaining are mucinous cystadenocarcinoma, adenocarcinoma, lymphosarcoma, paraganglioma, and granular cell tumors. Most common symptoms include acute pain in the right lower quadrant with or without lump and other symptoms of inflammation like fever, leukocytosis, and so forth. The appendix may perforate leading to appendicular lump formation or very rarely an abscess formation. Gastrointestinal tuberculosis most commonly involves the ileocecal region. Ulcerative lesions of tuberculosis may be precursors of carcinomas, derived from a chronic inflammatory process with repetition of erosion, ulcer, and consequent regeneration. Disruption of the integrity of mucosal barriers and impairment in cell-mediated immunity associated with cancerous growth are known to predispose to bacterial infection. Clinical features of both tuberculosis and cancer may overlap, e.g., weight loss, constipation, occult blood in stool etc., and the radiologic picture of an extensive ileocecal region and ascending colon tuberculosis may be so dramatic as to mask a coexisting malignant process if not contemplated. Careful endoscopy and biopsy of such “typical” looking colonic lesions are essential to rule out a coexisting carcinoma. Antituberculosis therapy (ATT) is of doubtful significance without surgery, but a course of ATT is still advocated. Appendectomy is performed as clinically indicated. If a mass in the appendix is encountered incidentally during the course of abdominal surgery, an appendectomy is performed with frozen-section analysis of the mass. Most masses are benign mucocele or very small carcinoids. They do not require any further management. However, if lymphoma or larger carcinoid was identified, chemotherapy or more extensive surgery will be required. When the mass is adenocarcinoma, the treatment algorithm is less defined as the data remain controversial.
Case Report
Here, we are reporting an interesting case of a 69-year-old lady who presented to us with pain in the right iliac fossa associated with progressive swelling in the right iliac fossa with fever with chills since 10 days. There was no associated history of vomiting, bowel/bladder habits abnormalities, trauma, and itching/insect bite.
On the examination, she had tachycardia with tense shining swelling over the right iliac fossa with pointing sign suggestive of abscess. No other lump or abnormalities were detected on the examination.
On further evaluation, the total count was very high 45,700/cumm with neutrophilia and toxic granules were seen. ESR was raised to 120 mm. We did an MRI of spine with abdominal screening which revealed an ill-defined collection involving the subcutaneous plane along the right posterolateral aspect lateral to paraspinal muscles with air pockets with retroperitoneal extension extending to ileocecal junction.
In view of raised counts with pointing abscess, we decided to go ahead with extraperitoneal drainage of the abscess. Incision and drainage of abscess was done. Around 1000 ml of frank pus drained which was sent for culture and sensitivity and the granulation tissue from abscess wall scrapped and sent for histopathology. Thorough wash was given and dressing was done.
Pus culture showed growth of Escherichia coli which was a multidrug sensitive, gene expert detected Mycobacterium tuberculosis, and AFB culture showed no growth. Histopathology of abscess wall scrapings showed pyogenic inflammation with a well-formed epithelioid granulomas but no caseous necrosis.
The patient managed with sensitive antibiotics and started on antituberculous therapy. The patient was improving over time and wound healed over time.
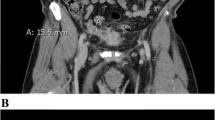
After 3 months, the patient returned with complaints of fever since 4 days with raised WBC counts (20,000/cumm). Contrast enhanced computed tomography (CECT) of abdomen and pelvis showed resolution of previous abscess with persistent soft tissue thickening of posterior wall of cecum with residual extension to retroperitoneal disease. Residual disease with partially necrotic right iliac lymph nodes (Fig. 1). Serum CEA levels were normal.
Colonoscopy was done which showed features of tuberculosis with fistulous opening in cecum (Fig. 2).
The patient underwent right hemicolectomy for same and managed accordingly. Postoperative period was uneventful, and patient recovered well.
Histopathology report showed a 3 × 3 × 3.5 cm growth arising from the appendix involving adjacent cecum with multiple tiny 0.5 to 1 cm gray white nodules over the serosa in the region of ileocecal junction. Margins were clear (Fig. 3).
Microscopy was suggestive of low-grade mucinous adenocarcinoma with perineural invasion. A total of 15 lymph nodes were identified from the specimen. All lymph nodes were negative for metastasis (pathological stage—pT3N0Mx).
The patient was referred to an oncologist who started her on 5-fluorouracil regimen-based chemotherapy. Currently, she is doing well, tolerating orals, passing normal stools, and urine. She has received six cycles of chemotherapy till now. Review CT scan was done after four cycles showed no evidence of residual disease.
Discussion
Appendix cancer is rare and most commonly found incidentally in an appendectomy specimen (incidence < 1%) (1), which was obtained for an unrelated condition. The main histologic types are carcinoids, adenocarcinomas, adenocarcinoids, cystadenomas, and cystadenocarcinomas.
In contrast to other appendiceal tumors, adenocarcinomas are more often present with a clinical picture of acute appendicitis. Standard treatment is a right colectomy.
Obstruction of the appendiceal lumen has been recognized as the major etiologic factor of appendicitis. Neoplasms of the appendix or cecum may obstruct the appendiceal lumen, incite an inflammatory reaction subsequent to obstruction of the organ, and then progress to rupture of the appendix with abscess formation. Abscess may form in the periappendiceal region, in the right paracolic gutter, in pelvis, or may track onto the abdominal wall (2).
Pathophysiologically, the relations between appendicular cancer and appendicular abscess may be the following:
-
1.
Appendicular cancer and appendicular abscess may be two coexisting and independent affections.
-
2.
Appendicular neoplastic lesion may cause appendicitis by mechanical obstruction at the orifice of the vermiform appendix.
-
3.
Adenocarcinoma of the appendix may present clinically as appendiceal abscess due to transmural invasion with perforation.
Patients with perforated cancer are at risk of diffusion of cancer cells within the abdomen and pelvis and consequently of peritoneal carcinomatosis (3). However, different clinical reports show that the presence of perforation does not necessarily predispose a poor prognosis and long-term survivals depending on tumor stages without a significant difference between perforated and uncomplicated cancers (4,5,6).
Gastrointestinal tuberculosis most commonly involves the ileocecal region followed by jejunum and colon. Multiple sites of involvement are common. Tanaka et al. (7) have reviewed 27 cases of coexistent tuberculosis colon cancer (CTCC). The site of occurrence of such carcinoma matches well with that favored by tuberculous colitis. Another possibility, although less likely, is that carcinoma predisposes to tuberculous infection. Disruption of the integrity of mucosal barriers and impairment in cell-mediated immunity associated with cancerous growth are known to predispose to bacterial infection (8). However, the evidence of TB in the colon at a site distant from carcinoma in at least two cases of Jain et al. contradicts the hypothesis of a secondary infection in a preexisting tumor. Moruta et al. (9) have postulated that ulcerative lesions of tuberculosis may be precursors of carcinomas, derived from a chronic inflammatory process with repetition of erosion, ulcer, and consequent regeneration. The role of bacteria in the causation of colorectal carcinoma has only been discussed in association with toxic metabolite production through sterols secreted in the bile (10). Whether mycobacteria have any significant bearing in this context is a matter of debate.
There have also been recent reports of decreased natural killer (NK) cell activity (11) and increased incidence of chromosomal aberrations (12) in association with tuberculosis. Specific relationship, of both accelerating and controlling growth, exist between fibroblasts and epithelial cells (13) and excessive fibroblastic activity is a constant finding in a long-standing tuberculous process.
Clinical features of both tuberculosis and cancer may overlap, e.g., weight loss, constipation, occult blood in stool etc., and the radiologic picture of an extensive ileocecal region and ascending colon tuberculosis may be so dramatic as to mask a coexisting malignant process if not contemplated. This is true especially in an endemic region, where the bias is predominantly in favor of tuberculosis. Careful endoscopy and biopsy of such “typical” looking colonic lesions is essential to rule out a coexisting carcinoma. The diagnostic possibility may also be considered on a barium enema showing overt malignancy of colon entailing an unduly long segment, particularly in the proximal colon.
The treatment of appendiceal abscesses is still debatable, and many different approaches are nowadays adopted. Expectant management, consisting of intravenous antibiotics, percutaneous drainage, and an interval appendectomy at a later date, is gaining a wide consent as it seems associated with less morbidity and shorter overall hospital stay (14, 15). Intraoperative abscess drainage and resection should be performed in selected cases to avoid the risk of delaying the diagnosis and unrecognizing malignant lesion as well as to achieve a safe and adequate treatment (16). In the suspect of neoplastic lesion of cecum or appendix, the right hemicolectomy is the procedure of choice to make a correct diagnosis and to avoid the risk of leaving residual tissue (17, 18).
The role of adjuvant chemotherapy for adenocarcinoma of the appendix is unknown. Despite the lack of available data, many oncologists recommend adjuvant 5-FU-based chemotherapy particularly for patients with node-positive intestinal type adenocarcinoma, extrapolating from data on adjuvant chemotherapy for node-positive colon cancer. Optimal treatment of patients with intraperitoneal dissemination of appendiceal adenocarcinoma (mucinous peritoneal carcinomatosis as distinguished from DPAM) is unclear. Selected patients treated with aggressive surgical cytoreduction and IPHC may do well in long term, but patient selection and the experience of the treating team are critical. In uncontrolled series from experienced institutions, long-term survival rates in highly selected patients range from 28 to 72% at 3 to 10 years. The benefit of systemic chemotherapy for advanced disease is unknown. Although mucinous appendiceal adenocarcinomas have been thought to be relatively chemotherapy refractory, case reports suggest some level of benefit for therapy in individual patients (19,20,21,22).
For appendiceal carcinoids, reoperation and right colectomy is recommended for tumors larger than 2 cm and for smaller tumors with mesoappendiceal invasion. Most patients have localized disease, and the prognosis is excellent (23).
The spectrum of epithelial tumors of the appendix ranges from the benign mucocele to an aggressive adenocarcinoma. Simple appendectomy, taking care not to rupture the tumor intraoperatively, is a sufficient therapy for benign appendiceal mucocele, cystadenomas, and some cystadenocarcinomas. A right colectomy is indicated for cystadenocarcinomas with mesenteric or adjacent organ involvement and complicated mucocele with involvement of the terminal ileum or cecum cystadenocarcinomas (1, 23).
Pseudomyxoma peritonei (PMP) is a unique condition characterized by diffuse collections of gelatinous material in the abdomen and pelvis, associated with mucinous implants on the peritoneal surfaces. The term should be reserved for the clinical situation in which a ruptured cystadenoma seeds the peritoneal cavity with mucus-producing epithelial cells, termed diffuse peritoneal adenomucinosis (DPAM) (24). The natural history is one of indolent but progressive growth, and if left untreated, this is a fatal condition. Standard treatment for PMP is repeated surgical debulking for symptomatic disease. This treatment is not curative but aims to limit the buildup of mucus and its pressure effect. A more aggressive approach using radical surgical cytoreduction of all intraabdominal and pelvic disease and intraperitoneal heated chemotherapy (IPHC) has been adopted by some clinicians, aiming for cure (19, 24, 25). Although randomized trials have not been conducted, five-year survival rates of 70 to 86% have been reported for highly selected patients.
The cancers of the appendix could represent a challenge for diagnosis and management. No standard of care is established due to rare frequency of occurrence; our case and the literature review are a summary of the appendix tumor, and the management based more on personal opinion of the oncologists who deal with this rare type of tumor, hopefully, would help in the future to create clinical trials to establish more evidence-based medicine help in the management.
Conclusion
Appendiceal cancers are rare. Most present with symptoms of appendicitis, appendicular abscess, or lump. Rarely present as abdominal wall abscess. Most require incision and drainage followed by the right hemicolectomy with further chemotherapy. Prognosis for earlier stages is good but poor in wide spread cases.
References
McCusker ME, Coté TR, Clegg LX, Sobin LH (2002) Primary malignant neoplasms of the appendix: a population-based study from the surveillance, epidemiology and end-results program, 1973-1998. Cancer 94(12):3307–3312
Okita A, Kubo Y, Tanada M, Kurita A, Takashima S (2007) Unusual abscesses associated with colon cancer: report of three cases. Acta Med Okayama 61(2):107–113
Sugarbaker PH (1999) Successful management of microscopic residual disease in large bowel cancer. Cancer Chemother Pharmacol 43(Suppl):S15–S25
Cerame MA (1988) A 25-year review of adenocarcinoma of the appendix. A frequently perforating carcinoma Dis Colon Rectum 31(2):145–150
Kriwanek S, Armbruster C, Dittrich K, Beckerhinn P (1996) Perforated colorectal cancer. Dis Colon Rectum 39(12):1409–1414
Alcobendas F, Jorba R, Poves I, Busquets J, Engel A, Jaurrieta E (2000) Perforated colonic cancer. Evolution and prognosis Rev Esp Enferm Dig 92(5):326–333
Tanaka K, Kondo S, Hattori F, Yamashita Y, Matsuda M, Itoh K et al (1987) A case of colonic carcinoma associated with intestinal tuberculosis, and an analysis of 26 cases reported in Japan. Gan No Rinsho. 33(9):1117–1123
Jain BK, Chandra SS, Narasimhan R, Ananthakrishnan N, Mehta RB (1991) Coexisting tuberculosis and carcinoma of the colon. Aust N Z J Surg 61(11):828–831
Maruta K, Tanaka S, Nakamura T, Hagio S, Sato E, Nishi M (1983) Carcinoma of the colon coexisting with tuberculosis--report of 2 cases. Gan No Rinsho 29(5):448–453
Bresalier R.S. KYS. Malignant neoplasms of the large and small intestine. 4th ed. Sleisenger FJS, editor. Philadelphia: W.B. Saunders; 1989
Restrepo LM, Barrera LF, Garcia LF (1990) Natural killer cell activity in patients with pulmonary tuberculosis and in healthy controls. Tubercle 71(2):95–102
Rao VV, Gupta EV, Thomas IM (1990) Chromosome damage in untreated tuberculosis patients. Tubercle. 71(3):169–172
Potter JD (1992) Reconciling the epidemiology, physiology, and molecular biology of colon cancer. JAMA 268(12):1573–1577
Brown CV, Abrishami M, Muller M, Velmahos GC (2003) Appendiceal abscess: immediate operation or percutaneous drainage? Am Surg 69(10):829–832
Lasson A, Lundagårds J, Lorén I, Nilsson PE (2002) Appendiceal abscesses: primary percutaneous drainage and selective interval appendicectomy. Eur J Surg 168(5):264–269
Zarba Meli E, Mazzocchi P, Lepiane P, Dalsasso G, Giacovazzo F, Salvio A, et al. [The role of surgery in the treatment of appendicular abscesses]. Minerva Chir 1997;52(5):577–581
Poon RT, Chu KW (1999) Inflammatory cecal masses in patients presenting with appendicitis. World J Surg 23(7):713–716 discussion 6
Cirocco WC (2005) Right hemicolectomy remains the treatment of choice for adenocarcinoma of the appendix. Gastrointest Endosc 61(6):791 author reply 2
Nitecki SS, Wolff BG, Schlinkert R, Sarr MG (1994) The natural history of surgically treated primary adenocarcinoma of the appendix. Ann Surg 219(1):51–57
Pickhardt PJ, Levy AD, Rohrmann CA, Kende AI (2003) Primary neoplasms of the appendix: radiologic spectrum of disease with pathologic correlation. Radiographics 23(3):645–662
Hartley JE, Drew PJ, Qureshi A, MacDonald A, Monson JR (1996) Primary adenocarcinoma of the appendix. J R Soc Med 89(2):111P–113P
Ferro M, Anthony PP (1985) Adenocarcinoma of the appendix. Dis Colon Rectum 28(6):457–459
Modlin IM, Sandor A (1997) An analysis of 8305 cases of carcinoid tumors. Cancer 79(4):813–829
Sugarbaker PH, Kern K, Lack E (1987) Malignant pseudomyxoma peritonei of colonic origin. Natural history and presentation of a curative approach to treatment Dis Colon Rectum 30(10):772–779
Pahlavan PS, Kanthan R (2005) Goblet cell carcinoid of the appendix. World J Surg Oncol 3:36
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Patel, R.R., Nandu, V.V. Primary Appendicular Malignancy Presenting as Abdominal Wall Abscess with Secondary Tuberculosis Infection: an Interesting Case Report. Indian J Surg Oncol 9, 613–617 (2018). https://doi.org/10.1007/s13193-018-0804-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13193-018-0804-0