Abstract
The relationship between elevation and the natural occurrence of several species of emergent vascular plants was examined in a microtidal deltaic marsh system near Corpus Christi, on the southwestern Texas coast. Vegetation percent cover was estimated quarterly from November 2003 to November 2006 in 374 permanent plots located in 9 sites. Soil elevation was measured in each plot using sub-centimeter real-time kinematic GPS. Cluster analyses determined that there were two significantly (P < 0.05) different sets of co-occurring species, one comprising Borrichia frutescens, Salicornia virginica and Distichlis spicata and the other including Batis maritima, Salicornia bigelovii and Lycium carolinianum. Analysis of species groupings across an elevation gradient using non-metric multi-dimensional scaling and hierarchal cluster analysis indicated that these assemblages represent a low marsh assemblage (25–50 cm NAVD88) and a high marsh assemblage (50–65 cm NAVD88), respectively. The strong relationship between elevation and species distribution suggests that abiotic factors play a key role in determining plant distribution in this ecosystem, and indicate that even apparently small differences in elevation can be important in a microtidal system.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The spatial patterns of salt marsh halophytes are often very complex and may arise from many different abiotic and biotic factors, including competition (Bertness and Ellison 1987; Bertness 1991), nutrient availability (Valiela et al. 1985; Levine et al. 1998), and stressors such as salinity and inundation (Bertness et al. 1992; Pennings and Callaway 1992; Crain et al. 2004; Pennings et al. 2005; Silvestri et al. 2005). Environmental conditions can play a role in determining halophyte distribution and occurrence in both early and late successional stage salt marshes (Erfanzadeh et al. 2010). Elevation above mean sea level (MSL) and tidal patterns determine the frequency and duration of tidal inundation, which in turn influence oxygen and salinity in soil (Adam 1990). Therefore analysis of elevation can provide valuable insights into the physical factors determining the distribution of salt marsh plants.
Impending sea level rise increases the importance of having a thorough understanding of the effects of higher MSL on vegetation patterns and processes. Recent projections of climate change indicate that sea level could rise more than a meter in the coming century (Rahmstorf 2010) and observations indicate that sea level rise is occurring more quickly than estimated by the Intergovernmental Panel on Climate Change (IPCC) in 2001 (Rahmstorf 2007). In microtidal salt marshes, such as in the Gulf of Mexico and Mediterranean, much smaller changes in sea level may have a greater impact on salt marshes (Day et al. 1995). For example, in the Nueces Delta in the western Gulf of Mexico, the frequency of bare space approached 100 % at elevations less than 10 cm above mean sea level, while from 18 to 25 cm, bare space accounted for less than 5 % of sampled areas (Dunton et al. 2001). The effect of such a small difference in elevation may reflect the small tidal amplitude (~15 cm) of the Nueces Delta. The influence of sea level rise on salt marshes with larger tidal amplitudes may be relatively less important. For example, modeled sea level rise scenarios related to global warming in the Tagus Estuary (Portugal) suggested that meso-tidal salt marshes would be impacted minimally by sea level rise (Simas et al. 2001). More recently, Kirwan and Guntenspergen (2010) developed a numerical model that demonstrated that marsh stability, and therefore resilience to sea level rise, is directly related to tidal range. Actual impacts of sea level rise can also be influenced by numerous other factors such as the incidence and severity of storm surges and sedimentation rates (Reed 1990).
In addition to understanding the potential effects of sea level rise, knowledge of the elevations within which species occur is important for restoration efforts. Salt marsh restoration efforts should consider elevations measured in nearby reference marshes because the elevation range of a species can vary among marshes (Zedler et al. 1999). Investigations of the relationship between elevation and species distribution, of the type reported here, provide important baseline data for restoration efforts.
The effects of tidal patterns on salt marsh plant community structure are unknown for Gulf of Mexico salt marshes (Pennings and Bertness 2001). The present study examined the relationship between elevation and the distributions of the dominant vascular plants at nine sites within a salt marsh in the western Gulf of Mexico. The objective was to determine the relationship, if any, between species distribution and elevation for these plant species in the Nueces River delta. The specific questions were: (1) What are the common species assemblages in the lower Nueces Marsh? (2) What is the distribution of individual species across the elevation gradient? (3) What are the relationships between species assemblages and elevation?
Methods
Study Site
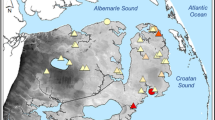
The Nueces Marsh (27° 53’ N, 97° 35’ W) is an estuarine marsh in the western Gulf of Mexico near the Corpus Christi Bay, Texas (United States) (Fig. 1) composed of almost 6,000 ha of salt marsh, tidal flats, and creeks. The climate is semi-arid with an average annual precipitation of 76 cm (Tolan 2007). This region is characterized as micro-tidal, with a daily average tidal amplitude of 15 cm, although tidal surges from tropical storms and winter cold fronts can cause much higher but infrequent flooding throughout the marsh (Ward 1985). Water levels in the Nueces Marsh vary with these irregular events as well as being influenced by tides (Ward 1985). Furthermore, hurricanes are common on the Texas coast, with a hurricane striking any 80 km stretch of the Texas coast once every 6 years (Roth 2010). Two impoundments, approximately 35 km (Lake Corpus Christi) and 80 km (Choke Canyon Reservoir) from the Nueces Marsh have reduced natural flooding (U.S. Bureau of Reclamation 2000).
The two dominant vascular plant species in the lower Nueces Marsh are Borrichia frutescens and Salicornia virginica which are often found growing close to tidal creeks and to Nueces Bay (Rasser 2009). Each of these species often forms mono-specific stands. Sparsely vegetated salt meadows, common at variable distances from tidal creeks, frequently contain Batis maritima and Monanthochloe littoralis. At high elevations more distant from Nueces Bay, the lower salt marsh is bordered by a brackish marsh dominated by Spartina spartinae. S. spartinae as well as Bacopa monnieri, Eleocharis parvula, Iva frutescens, Heliotrope curassavicum, Limonium nashi, Portula oleracea, Pluchea odorata, Aster subulatus, Scirpus maritimus and Typha sp. were observed to be less than 1 % of relative cover in a 10 year study of vegetation in the Nueces Marsh (Forbes and Dunton 2006).
Field Data Collection
Sampling was conducted at a total of 374 permanent plots. Thirty-one to 60 of these plots were located in each of nine sites (Tables 1 and 2) located throughout the lower Nueces Marsh (Figs. 1). Many of the nine sites have been studied for over a decade (Dunton et al. 2001). Previous studies at these sites have examined the response of vegetation at these sites to interannual variation in precipitation (Dunton et al. 2001), freshwater inundation (Alexander and Dunton 2002) and local climate change (Forbes and Dunton 2006).
Plots were located systematically every 2 m along three parallel 20 m transects placed 1 m apart at sites 451, 272, 254, 71, 270, and 450. At sites 1, 2, and 3 transects were 50 m long and plots were spaced 4 m apart between the 24 m and 50 m marks. All transects began at, and ran perpendicular to, the edge of tidal creeks. The cover of each vascular plant species in each plot was measured quarterly from November 2003 through November 2006 using a 0.25 m2 quadrat. The quadrat was divided into 100 5 × 5 cm cells and percent cover was estimated from the number of cells occupied by a species. Cells with no vegetation, covered with water, or containing dead plant material were considered bare (Dunton et al. 2001; Alexander and Dunton 2002). The nine most abundant species, based on percent cover, were included in the analyses.
In February 2007, elevation data were acquired at each of the vegetation plots using real-time kinematic GPS. All measurements were collected at the soil surface and the vertical and horizontal accuracy determined to be within 1 centimeter for all measurements. Elevation was not measured at three plots due to flooding at the time of the survey, and one plot due to surveyor error. All analyses were conducted using the current survey standard, the North American Vertical Datum of 1988 (NAVD88). At the time of analysis, no conversion was available from NAVD88 to mean sea level for the study sites. The MSL of Nueces Bay is estimated to be −17.5 cm and mean high water −6.9 cm (NAVD88) based on preliminary analysis by the Texas Height Modernization program. Actual MSL will be different at the study sites due to the complex hydrology and effects of major wind events influencing the movements of water in and out of the Nueces Marsh.
Data Analysis
For each of the 374 plots, the cover of each species was averaged over all sample dates (n = 13), producing an array of average cover values for 9 species. Similarly, the proportion of bare ground was averaged over all dates, separately for each plot. Preliminary data analysis determined that it was not possible to examine the interaction among sites and elevation as there was insufficient data to compare sites or to maintain site as a variable in the analyses. The 4 plots without elevation measurements were excluded from all elevation analyses.
The Primer 6 package (Plymouth Marine Lab, UK) was used for all multivariate analyses. A resemblance matrix of the Bray-Curtis similarity index (Bray and Curtis 1957) was calculated from the cover data array (including bare ground) and used in all similarity analyses. To identify groups of spatially-associated species, a hierarchical cluster analysis using only the species cover data (including bare ground, but not elevation data) was calculated. The Primer 6 routine SIMPROF (Clarke and Warwick 2001) was used to identify significantly different clusters in this analysis.
Elevations were classified into bins by 5 cm intervals except for the highest and lowest elevation values. All elevations < 25 cm were grouped together as were values > 65 cm. A cluster analysis of these bins, based on average species cover per elevation bin, was calculated. SIMPROF was used to identify significantly different clusters. A non-metric multi-dimensional scaling (MDS) ordination of the square-root-transformed percent cover data (Clarke and Warwick 2001) was computed to visualize and compare elevation bins.
Results
Site Characterization
The nine sites sampled were 600 m to 9,500 m inland from Nueces Bay (Fig. 1). Mean site elevation varied from 36.2 cm (Site 271) to 52.2 cm (Site 1). In all sites elevation was lowest next to the tidal creek (Fig. 2). Most transects had a relatively high-elevation portion close to the tidal creek (Fig. 2). Nine species (Table 3) accounted for almost all plant cover and were included in the analyses. Each of these nine species was present in at least four of the nine sites, but only two species (B. frutescens and L. carolinianum)were present in all nine sites. B. frutescens (24.6 %), S. virginica (10.0 %), B. maritima (7.0 %), and D. spicata (6.2 %) were the most abundant species. S. alterniflora, L. carolinianum, S. bigelovii, M. littoralis and S. linearis each represented less than 3 % percent cover (Table 3). Other plant species, such as Limonium nashii and Heliotropium curassavicum, each accounted for less than 0.5 % of the total cover sampled and were therefore not included in any of the multivariate analyses. Bare ground was 39.9 % of the total area.
Cluster Analysis of Plant Species
A cluster analysis of plant cover alone (Fig. 3) identified two significantly different (P ≤0.05, SIMPROF) clusters, involving six species among the nine plant species examined. Cluster A was B. frutescens, S. virginica, and Distichlis spicata and cluster B included Salicornia bigelovii, Lycium carolinianum and Batis maritima.
Species Distributions in Relation to Elevation
The three species in cluster A were most abundant at elevations close to 35 cm (Fig 4). Among cluster B species, Salicornia bigelovii and Batis maritima were most abundant near 60 cm, while Lycium carolinianum was more like the unclustered Monanthochloe littoralis and Suaeda linearis in being most abundant near the highest elevation (70 cm). Bare area was most common at the lowest elevations and around 55 cm. Two species of cluster A, D. spicata and B. frutescens, were also relatively abundant at the highest elevations. Spartina alterniflora was common at the lowest elevations (< 25 cm). In the multivariate analyses, elevations 25–50 cm clustered together and elevations 50–65 cm clustered together (Fig. 5). The first of these reflects the dominance of group A species at 25–50 cm elevations; the second reflects the presence of all the species (except S. alterniflora and S. virginica) at higher elevations. The 10–25 cm elevation did not cluster with any other elevation interval, reflecting its dominance by S. alterniflora.
Average percent cover of vascular plant species and bare area with respect to soil elevation for all sampling sites in the Nueces Marsh. Based on average percent cover within plots categorized in elevation bins (see Table 3) for 13 sampling events (n 39–1235). Error bars are standard error
NMDS plot of Bray-Curtis similarity of species percent cover for elevation category data. Data grouped from all sites. Elevation categories are superimposed on plot (see Table 2). Lines around groupings represent clusters that are at least 60 % similar to each other
Discussion
Analyses of data from 374 plots in nine sites in the lower Nueces Marsh of Texas revealed consistent relationships between elevation and plant community composition. At the lowest elevations (< 25 cm), S. alterniflora dominates (Fig. 6). At somewhat higher elevations (25–50 cm), the vegetation is dominated by B. frutescens, D. spicata, and S. virginica. The other five common plant species (B. maritima, L. carolinianum, M. littoralis, S. bigelovii, and S. linearis) are each most abundant at elevations above 55 cm. Bare, vegetation-free areas are most common below 30 cm elevation and between 45 and 60 cm.
A similar strong association between species composition and elevation has been documented in other salt marshes. For example, Sadro et al. (2007) used remotely sensed data, including light detection and ranging (LIDAR), to determine three dominant plant classifications that occurred within a 17 cm elevation range in a southern California salt marsh. Similarly, Silvestri et al. (2005) found a strong dependence of species frequency based on elevation range of 0–50 cm above MSL in the Venice Lagoon (Italy). The authors were unable to link the distribution of salt marsh plants to any single factor such as salinity.
The strong association between species distributions and elevation suggests that abiotic factors are important in determining the spatial distribution of plant species in the Nueces Marsh. Small differences in inundation frequency, duration and timing may account for differences in plant distributions (Bornman et al. 2008). For example Sadro et al. (2007) found differences in plant classes that experienced only a 7 % difference in period of tidal inundation.
The higher proportion of bare area below 30 cm elevation may be due to the physiological stress of more frequent inundation that occurs near MSL. Dunton et al. (2001) examined three of the same sites (450, 252 and 254; noted as 50, 52, and 54 respectively in this earlier study) and found that there was nearly a hundred percent bare area from 0–10 cm MSL and that bare area decreased significantly from 10–25 cm above MSL. Although only relative elevation was measured by Dunton et al. (2001) it appears that plants are generally absent between mean sea level and mean high water (MHW) which corresponds to approximately 10 cm above MSL in Nueces Bay.
Spartina alterniflora occurred at the lowest elevations measured and has been measured to occur approximately 20 cm above mean sea level in the Nueces Marsh (Dunton et al. 2001). The dominance of S. alterniflora at lower elevation supports studies that have found that this species is very tolerant of inundation (Gleason and Zieman 1981; Naidoo et al. 1992) and subjective to competitive displacement (Bertness and Ellison 1987; Bertness 1988, 1991). It is likely that this species has a better ability to tolerate flooding than other species in the Nueces Marsh. The upper limit of S. alterniflora is likely set by competition in the Nueces Marsh with B. frutescens. There is experimental evidence that the upper border of S. alterniflora is set by competition with B. frutescens in a salt marsh in Georgia (Pennings and Moore 2001). However, the relative unpredictability of water levels may make competitive exclusion less common on the Texas Coast (Kunza and Pennings 2008). The limited distribution and abundance of S. alterniflora throughout the Nueces Marsh is likely due to the small tidal amplitude of the Nueces Marsh, which limits the elevation range in which frequent exposure to flooding occurs. This same positive relationship between S. alterniflora abundance and tidal amplitude and has been demonstrated to occur at numerous salt marshes (McKee and Patrick 1988).
At slightly higher elevations than S. alterniflora, the species assemblage consists of D. spicata, B. frutescens, and S. virginica. It seems likely that B. frutescens, the most abundant plant sampled, is largely limited by abiotic conditions. Previous research in the Nueces River Delta has noted the vegetative expansion of B. frutescens following large precipitation events (Weilhoefer 1998) and the introduction of treated wastewater that have reduced porewater salinity (Forbes et al. 2008). Similarly, during periods of drought, S. virginica abundance has increased (Forbes and Dunton 2006) suggesting that this species is relatively more tolerant of salinity. More recently, Rasser (2009) experimentally demonstrated that B. frutescens growth is impaired at salinity of 30‰ whereas S. virginica is not. D. spicata is common to disturbed marsh habitats (Bertness and Ellison 1987; Allison 1996; Levine et al. 1998) and has been experimentally shown to be susceptible to competitive displacement (Crain et al. 2004). Competitive displacement by B. frutscens and S. virginica, both clonal plants, may limit the distribution of D. spicata.
The occurrence of bare space (45–60 cm) is likely related to physiological constraints of very high porewater salinity. Intermediate elevations in salt marsh can contain the highest porewater salinity as these areas are low enough to be flooded during periods of high water but high enough to not significantly benefit from freshwater inflow or more frequent tidal events (Pennings and Bertness 2001). In addition, relatively low areas along the elevation profile (Fig. 2) may accumulate very high porewater salinity from repeated flooding and evaporation events.
The higher elevation assemblage of B. maritima, S. bigelovii and L. carolinianum (Fig. 6) as well as M. litoralis that are more abundant at higher elevation (Fig. 4) are generally believed to have a higher salinity tolerance. Tiner (1999) lists all of these species as indicators of a hyperhaline salt marsh (> 40 ‰) as compared to all of the lower elevation species S. virginica (euhaline 30–40 ‰) and S. alterniflora, B. frutescens, and D. spicata (euhaline/polyhaline (18–40 ‰). This suggests that these species may have a higher salinity tolerance. The abundance of the annual S. bigelovii at elevations near the maximum of bare area (Fig. 4) may reflect its ability to take advantage of lower level of salinity following freshwater input (Alexander and Dunton 2002). Forbes and Dunton (2006) hypothesized that this species is capable of producing a viable seed bank that persists through periods of hypersalinity. In contrast, perennial plants such as B. maritima may not be able to persist in bare areas because of intermittent periods of much higher salinity. More spatially and temporally detailed studies would be needed to test such hypotheses.
The patterns of distribution related to elevation are likely driven by both changes in abiotic conditions such as salinity and flooding as well as competition. Nevertheless, some plant species can still be found across a wide range of elevations. For example, B. frutescens was common at both higher and lower elevations (Fig. 4) perhaps a reflection of this plant being able to competitively exclude other plants at the edge of tidal creeks, which frequently have a higher relative elevation (Fig. 2). Examination of factors such as inundation and frequency may provide further insight into the variables influencing salt marshes rather than just elevation alone (Bockelman et al. 2002). This understanding is crucial for natural resources managers to understand the potential impacts of anthropogenic impacts such as reduced freshwater inflow, climate change, and sea level rise might impact salt marsh function.
We have documented that distribution of individual species as well as species assemblages in the Nueces Marsh are influenced by very small changes in elevation. These patterns were observed despite tidal amplitude of only 15 cm. If sea level model predictions of 0.5 m to 1.0 m prove true, the entire plant community in the Nueces Marsh will be influenced by higher water levels. In the near future, smaller changes in sea level of 10–20 cm may have the ability to alter vegetative community structure in micro-tidal salt marshes such as the Nueces Marsh. It is possible that other microtidal salt marshes throughout the Gulf of Mexico or even other regions of the world such as the Mediterranean Sea, may be similarly impacted.
References
Adam P (1990) Saltmarsh ecology. Cambridge University Press, Cambridge
Alexander HD, Dunton KH (2002) Freshwater inundation effects on emergent vegetation of a hypersaline salt marsh. Estuaries and Coast 25:1559–2731
Allison SK (1996) Recruitment and establishment of salt marsh plants following disturbance by flooding. American Midland Naturalist 136:232–247
Bertness MD (1988) Peat accumulation and the success of salt marsh plants. Ecology 69:703–713
Bertness MD (1991) Interspecific interactions among high marsh perennials in a New England salt marsh. Ecology 72:125–137
Bertness MD, Ellison AM (1987) Determinants of pattern in a New England salt marsh plant community. Ecological Monographs 57:129–147
Bertness MD, Gough L, Shumway SW (1992) Salt tolerance and the distribution of fugitive salt marsh plants. Ecology 72:1842–1851
Bockelman AC, Bakker JP, Neuhaus R, Lage J (2002) The relation between vegetation zonation, elevation, and inundation frequency. Aquatic Botany 73:211–221
Bornman TG, Adams JB, Bate GC (2008) Environmental factors controlling the vegetation zonation patterns and distribution of vegetation types in the Olifants Estuary, South Africa. South African Journal of Botany 74:685–695
Bray JR, Curtis JT (1957) An ordination of the upland forest communities of Southern Wisconsin. Ecological Monographs 27:325–349
Clarke KR, Warwick RM (2001) Change in marine communities: an approach to statistical analysis and interpretation, 2nd edn. PRIMER-E, Plymouth
Crain CM, Silliman BR, Bertness SL, Bertness MD (2004) Physical and biotic drivers of plant distribution across estuarine salinity gradients. Ecology 85:2539–2549
Day JW, Pont D, Hensel PF, Ibañez C (1995) Impacts of sea-level rise on deltas in the Gulf of Mexico and the Mediterranean: the importance of pulsing events to sustainability. Estuaries 18:636–647
Dunton KH, Hardgree B, Whitledge TE (2001) Response of estuarine marsh Vegetation to interannual variations in precipitation. Estuaries 24:851–861
Erfanzadeh R, Petillon J, Maelfait JP, Hoffmann M (2010) Environmental determinism versus stochasticity in the appearance of plant species in salt-marsh succession. Plant Ecology and Evolution 143:43–50
Forbes MG, Dunton KH (2006) Response of a subtropical estuarine marsh to local climate change in the southwestern Gulf of Mexico. Estuaries and Coasts 29:1242–1254
Forbes MG, Alexander HD, Dunton KH (2008) Effects of pulsed riverine versus non-pulsed wastewater inputs of freshwater on plant community structure in a semi-arid salt marsh. Wetlands 28:984–989
Gleason ML, Zieman JC (1981) Influence of tidal inundation on internal oxygen supply of Spartina alterniflora and spartina patens. Estuarine, Coastal and Shelf Science 13:47–57
Kirwan ML, Guntenspergen GR (2010) Influence of tidal range on the stability of coastal marshland. Journal of Geophysical Research 115:FO2009. doi:10.1029/2009JF001400
Kunza AE, Pennings SC (2008) Patterns of plant diversity in Georgia and Texas salt marshes. Estuaries and Coasts 31:673–681
Levine JM, Brewer MS, Bertness MD (1998) Nutrients, competition, and plant zonation in a New England salt marsh. Journal of Ecology 86:285–292
McKee KL, Patrick WH (1988) The relationship of smooth cordgrass (Spartina alterniflora) to tidal datums: a review. Estuaries 11:143–151
Naidoo G, McKee KL, Mendelssohn IA (1992) Anatomical and Metabolic Responses to Waterlogging and Salinity in Spartina alterniflora and S. patens (Poaceae). American Journal of Botany 79:765–770
Pennings SC, Bertness MD (2001) Salt marsh communities. In: Bertness MD, Gaines SD, Hay ME (eds) Marine community ecology. Sinaeur Associates, Sunderland Massachusetts, pp 289–316
Pennings SC, Callaway RM (1992) Salt marsh plant zonation: the relative importance of competition and physical factors. Ecology 73:681–690
Pennings SC, Moore DJ (2001) Zonation of shrubs in western Atlantic salt marshes. Oecologia 126:587–594
Pennings SC, Grant M, Bertness MD (2005) Plant zonation in a low-latitude salt marsh: disentangling the roles of flooding, salinity, and competition. Journal of Ecology 93:159–167
Rahmstorf S (2010) A new view on sea level rise. Nature Reports Climate Change. doi:10.1038/climate.2010.20
Rahmstorf S, Cazenave A, Church JA, Hansen JE, Keeling RF et al (2007) Recent climate observations compared to projections. Science 316:709
Rasser MK (2009) Ph.D. Dissertation . The role of biotic and abiotic processes in the zonation of salt marsh plants in the Nueces River Delta, Texas. The University of Texas at Austin, Austin Texas
Reed JD (1990) The impact of sea-level rise on coastal salt marshes. Progress in Physical Geography 14(4):465–481
Roth D (2010) Texas hurricane history. National Weather Service, Camp Springs
Sadro S, Gastil-Buhl M, Melack J (2007) Characterizing patterns of plant distribution in a southern California salt marsh using remotely sensed topographic and hyperspectral data and local tidal fluctuations. Remote Sensing of Environment 110:226–239
Silvestri S, Defina A, Marani M (2005) Tidal regime, salinity and salt marsh plant zonation. Estuarine, Coastal and Shelf Science 62:109–130
Simas T, Nunes JP, Ferreira JG (2001) Effects of global climate change on coastal marshes. Ecological Modeling 139:1–15
Tiner R (1999) Wetland Indicators: A guide to wetland identification, delineation, classification, and mapping. Lewis Publishers, Boca Raton
Tolan JM (2007) El Niño-Southern Oscillation impacts translated to the watershed scale: estuarine salinity patterns along the Texas Gulf Coast, 1982 to 2004. Estuarine, Coastal and Shelf Science 72:247–260
U.S. Bureau of Reclamation (2000) In: Harris RG (ed) Concluding report: Rincon Bayou demonstration project. volume II. findings. U.S. Department of the Interior, Bureau of Reclamation, Austin
Valiela I, Cosgwell C, Ahrtman J, Allen S, Van Etten R, Goehringer D (1985) Some long–term consequences of sewage contamination in salt-marsh ecosystems. In: Godfrey PJ, Kaynor ER, Pelezarski S, Benforado J (eds) Ecological considerations in wetlands treatment of municipal wastewaters. Van Nostrand Reinhold Co, New York, pp 301–316
Ward GH (1985) Evaluation of marsh enhancement by freshwater diversion. Journal of Water Resources Planning and Management, ASCE 111:1–23
Weilhoefer LC (1998) Effects of freshwater inflow, salinity, and nutrients on salt marsh vegetation in south Texas. M.S. Thesis, Austin, Texas: The University of Texas at Austin
Zedler JB, Callaway JC, Desmond JS, Vivian-Smith G, Williams GD, Sullivan, Brewster AE, Bradshaw BK (1999) California salt-marsh vegetation: an improved model of spatial pattern. Ecosystems 2:19–35
Acknowledgments
This research was supported by the City of Corpus Christi, Project Number 3007 and would not have been possible without considerable field and laboratory assistance from Kim Jackson, Tami Beyer, Craig Aumack, Troy Mutchler, Summer Martin, Susan Schonberg, Maggie Forbes, Lauren Hutchinson, Faye Berens, Kelly Fellows, and others. Greg Hauger provided information and clarification regarding tidal information from the Texas Height Modernization program. Chris Wilson and several anonymous peer reviewers provided feedback that improved the clarity and quality of the manuscript. Connie Murphy provided technical editing and Nancy Buskey artwork that contributed to the manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Rasser, M.K., Fowler, N.L. & Dunton, K.H. Elevation and Plant Community Distribution in a Microtidal Salt Marsh of the Western Gulf of Mexico. Wetlands 33, 575–583 (2013). https://doi.org/10.1007/s13157-013-0398-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13157-013-0398-9