Abstract
In this paper, we present the detailed microfacies study and geochemical characteristics of carbonate exotics (olistoliths) associated with the Eocene Disang group of Assam–Arakan Basin, Northeast India to deduct their relative age, and depositional environment. The studied carbonate rocks contain diverse fauna with the dominance of foraminiferal assemblages (planktonic and benthic). The well-preserved planktonic foraminifera, especially Globotruncana spp. and Heterohelix spp. are recorded in carbonates and age has been assigned as late Cretaceous (Maastrichtian in age). Rare earth element (REE) contents (~ 56 ppm average value) in these carbonates are high compared with the average value of typical marine carbonate (~ 28 ppm). The Post-Archean Australian Shale (PAAS)-normalized REE patterns of these carbonates exhibit seawater-like REE patterns with light rare earth elements (LREE) depletion and relatively heavy rare earth elements (HREE) enrichment with negative Ce anomalies and positive Eu anomalies. The geochemical data along with microfacies suggest that the investigated carbonate rocks might have been formed in low energy environments deposited in neritic to bathyal palaeoenvironment during Campanian–Maastrichtian age. Our study also confirmed that the investigated Disang group carbonates are similar to the adjoining carbonates exposed in the ophiolitic mélange zone of Manipur ophiolites, NE India. It is likely that the carbonate rocks were influenced by diagenetic activities at different tectonic processes during the evolution of MOC and later emplaced with the Disang group flysch-like sediments during subduction and obduction processes of the Indian plate and Myanmar plate collision.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Paleoenvironmental interpretations are made possible by recognizing several varieties of patterns in foraminiferal assemblages. The proportion of planktonic specimens increases from 0 percent in shallow marine environments to more than 90 percent in deep marine environments. Modern planktonic foraminifera generally has globular chambers, often with spines. Some fossil planktonic, however, particularly in the Cretaceous, had a more flattened shape, sometimes with heavy keels running around the shell (Culver 1987). Diversity and distribution of benthic foraminifera in carbonate deposits are good indicators in biostratigraphical, sedimentological and paleoecological analyses of carbonate settings (Geel 2000; Boudagher-Fadel 2008; Flügel 2010). They occur in various environments ranging from terrestrial settings to deeper parts of the sea and from tropical to polar environments and are useful in reconstructions of both ancient and recent environmental conditions (Armstrong and Brasier 2005). Based on their wide distribution, evolution and speciation, most of the benthic foraminiferal species are considered significant tools in different stratigraphy studies.
Conversely, rare earth element (REE) concentrations in carbonate rocks are useful to identify the marine versus non-marine sources of REE (Frimmel 2009; Zhao et al. 2009). REEs are also considered as an indicator to identify the depositional environmental system such as widespread marine anoxia (German and Elderfield 1990; Murray et al. 1991), oceanic palaeo-redox conditions (Elderfield and Pagett 1986; Kato et al. 2002), variations in surface productivity (Toyoda et al. 1990), proximity to source area (Murray et al. 1991), lithology and diagenesis (German and Elderfield 1990; Nath et al. 1992; Armstrong-Altrin et al. 2003, 2011; Madhavaraju et al. 2010) and paleogeography and depositional models (Kemp and Trueman 2003).
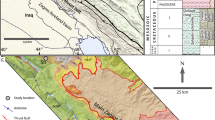
The carbonate rocks from the adjoining mélange zone of Nagaland–Manipur Ophiolites (NMO) of northeast India have been studied (Acharyya et al. 1986; Mitra et al. 1986; Chungkham et al. 1992; Chungkham and Jafar 1998; Tiwari et al. 2011; Singh et al. 2015; Singh et al. 2016), however, the carbonates associated with the Disang group have not been studied in detail till date. Thus, so far there is no consensus on the genesis and depositional environmental system. To fill up this gap, the present study employed microfossils characterization, geochemical features along with field observations and petrographic relations of carbonate exotics (olistoliths) occurring in the Disang group of Assam–Arakan Basin of Northeast India (Fig. 1) to derive a plausible source and to reconstruct the depositional environment of the carbonates rocks.
Generalized geological map of Manipur, Northeast India, showing the studied carbonate exposures within Disang group and adjoining Manipur Ophiolitic Complex (MOC), Indo-Myanmar Orogenic Belt, North East India (modified after Singh et al. 2013)
Geology and field setting
The study area lies in the western parts of the Manipur Ophiolitic Complex (MOC) which is a part of the NMO (Fig. 1). The NMO has an east-dipping thrust contact with the underlying late Cretaceous–late Eocene flysch-like sediments of the Disang and Barail formations exposed in the west, and overthrusted from the east by continental metamorphic rocks of quartz mica–schist, garnet mica–schist, quartzite and granitic gneiss (Brunnschweiler 1966). The boundary thrust between the NMO and the Disang group flysch is marked by brecciation, silicification and the presence of fault gouge and intermixing of litho-units. The Disang group represents enormous thickness (> 3000 m) of fine clastic sediments derived from the hinterland. It is made of shale, slate, phyllite, greywacke, subgraywacke, and calcareous shale with minor limestone. This monotonous, folded and thrusted, argillaceous succession is considered to be a flysch sequence (Mathur and Evans 1964). The Disang group ranges in age from early Cretaceous (Pascoe 1912) to late Eocene. Geologically, the carbonate deposits could be classified into two broad categories: (i) those associated with Disang group and (ii) those associated with oceanic pelagic sediments. In the first category, occur olistostromal limestones, which are exotic in nature. These limestones are usually lensoid in shape and randomly oriented within shales-phyllites of Disang group (Fig. 2). They normally occur along or near the western margin of the main ophiolite belt. On the other hand, oceanic pelagic sediments associated with limestone bodies investigated so far occur in Narum, Lamayang and Kulyang areas of Chandel district. Oceanic pelagic sediments comprise radiolarian chert, shale and sandstone (graywacke). These pelagic limestones are grey to chocolate in colour, fine-grained, and homogenous. Sometimes, they are cherty and occur as small lensoid bodies are of bedded nature, with a few meters in thickness and tens of meters in length. The oceanic pelagic sediments bearing areas are suitable sites for searching limestone bodies. The Disang group can be subdivided into two lithologic formations. They are lower and upper Disang formations. The lower Disang formation extends into eastern Manipur and exhibits argillaceous character. These rocks have been metamorphosed mainly towards the interior i.e. eastern side, where shale has changed to slate, phyllites and schist. They are juxtaposed against the ophiolite belt of Nagaland–Manipur, bordering the easternmost fringe, with a tectonic contact whereas the upper Disang formation is mainly composed of shale with minor sandstone and occasional mudstone.
The sedimentary process involved initial deep flysch sedimentation in a tectonic basin, namely the Disang group, which gradually became shallow through sediment filling and tectonic squeezing. There was a gradual shift of sedimentation domain from finer sediments of shaly composition (Disang group) at the bottom through gradual transformation to coarse sand facies towards the top known as the Barail group. This gradual change in marine sedimentation is without any evidence of break-in sedimentation, and as such, there is a likelihood of getting an intervening neritic environment with favourable carbonate deposition (Devi and Duarah 2015). In Ukhrul and Chandel districts of Manipur, the carbonate deposits occur as scattered pockets within the Disang sediments which are overlain conformably by the Barail group of arenaceous with minor shale and silty shale. The sedimentation of the Disang commenced during the late Cretaceous (Maastrichtian) and continued till the end of the Eocene and then began the deposition of Oligocene Barail sediments.
The carbonate rocks occur as exotic blocks in the adjoining melange zone of Disang shales. The carbonate rocks of the Toupokpi area show brown colour and are hard, massive, bedded with conchoidal fractures. The rocks are characterized by the presence of distinct calcite veins and stylolitic structures (Fig. 3T7, T9). The carbonate rocks of the Nungphura occur in conical or pyramidal forms having light to dark grey in colour. The rocks are hard massive, fine-grained with conchoidal to sub-conchoidal fractures. They are characterized by the presence of micro and macro stylolitic structures and calcite veins cut the rocks (Fig. 3NG5, NG9). The carbonate rocks of the New Paoye area show grey, buff and brown colour. The rock is hard, massive and fine-grained. The rock is characterized by the presence of irregular fracture veins and stylolitic structure (Fig. 3P1, P4, P6, P10). The studied carbonates yield diverse microfossil assemblages comprising planktonic and benthic foraminifera.
Materials and methods
About 50 carbonate samples were collected from different localities of the Chandel district viz. Toupokpi (GPS: N-24°14´03.7´´: E-93°54´32.5´´; Fig. 3T7, T9) and Nungphura (GPS: N-24°13´30.4´´: E-93°55´20.01´´; Fig. 3 NG5, NG9) and New Paoye of Ukhrul district (GPS: N-25°2´40´´: E-94°19´41´´; Fig. 3 P1, P4, P6, P10).
Analytical methods
Analysis of eight representative samples (T7, T9, NG5, NG9, P1, P4, P6, P10) was carried out choosing less altered rock for geochemical analysis. Veins and altered external surfaces were removed from the rocks by breaking the rocks into chips before grounding into powder. Rock samples were crushed and then pulverized using an agate carbide ring grinder. Major oxides and selected trace element concentrations present in the rock were measured on powder pellets by X-ray Fluorescence Spectrometer (XRF; Bruker Tiger S-8) in the Wadia Institute of Himalayan Geology (WIHG), Dehradun. XRF technique analytical precision for both major and trace elements lies within ± 2–3% and ± 5–6%, respectively. Loss-on-ignition was determined by heating a separate aliquot (0.5 g rock powder) of each representative sample at 1000 °C for 5 h. Further, the samples were analyzed in the same institute for their REE and few trace elements by Inductively Couple Plasma-Mass Spectrometer (ICP-MS; PerkinElmer SCIEX ELAN DRC-e) using the open system rock digestion method. Rock powders were first thoroughly dissolved in HF and HNO3 in Teflon crucibles and heated over a hot plate for a couple of hours until the sample-precipitate to prepare solutions from which Ta, Hf, Nb (trace elements) and REE abundances were determined. Analytical precision for major elements is well within ± 2–3% and ± 5–6% for trace elements. Accuracy of rare earth elements ranges from 2 to 12% and precision varies from 1 to 8%. Major (wt.%), trace elements (ppm) and rare earth elements (ppm) are presented in Table 1.
Micropaleontological methods
The micropaleontological separation of foraminifera had been carried out using chemical treatment with the help of a modified version of a maceration technique developed by Zolnaj (1979). The carbonate rock is broken into chips 1 cm and slightly lesser sizes with a hammer. The limestone chips and copper sulphate crystals are mixed and put inside a glass beaker of any size depending on the quantity of the mixture. The quantity of copper sulphate crystals is approximately half of that of limestone chips used. Concentrated acetic acid is poured just up to the level to cover the mixture. This treatment is kept for 3–4 days until the whole mixture turns into a paste. Then the paste is washed and sieved. The undigested chips can be dried and treated again by the same procedure. The basic reaction in this treatment is that the concentrated acetic acid attacks the rocks rapidly but it starts digesting the matrix of microcrystalline calcite (micrite) first and later on only the calcified skeletal grains. If diluted acetic acid of any strength is used, the reaction is much slower and the matrix and the skeletal grains are digested simultaneously. The copper sulphate acts as an anti-reactant to stop the complete digest of the skeletal grain. There is some restriction that the sample does not allow to keep more than 7 days then the whole rock is digested. The crushed and soaked sample is washed under a jet of water over a 230/325 mesh sieve. The washed material is then transported to a hot oven and dried. The dried sample is sieved into different fractions using sieves of different fractions (ASTM 30, 60, 120, 230). The samples thus processed are numbered and stored. The dried residue of the fossil rock mixture obtained as above is ready for sorting. Each fraction is spread in a thin film on a sorting tray. The tray is moved under a stereo zoom microscope (Nikon SMZ 1500) left to right and the microfossils are picked and placed a gummed 254/48 chambered slide. The most commonly used practice of picking microfossils is with a fine hairbrush either 00 or 000 size. Thin sections are also prepared to study the microfossils. The photograph of a thin section is taken in a petrological microscope (Nikon eclipse LV100N POL).
Results
Microfacies types
Four microfacies types viz., (i) MFT1 micrite (ii) MFT2 foraminifer bearing micrite (iii) MFT3 foraminiferal micrite and (iv) MFT4 foraminiferal packstone were identified according to microscopic characteristics of carbonate rocks in the study area (Dunham 1962; Folk 1962; Zeng and Xia 1986).
-
(i) MFT1 micrite (mudstone): This type has mud-supported lithologies with very rare (< 5%) and small allochems scattered in a cryptocrystalline matrix which is found in Nungphura NG9 (Fig. 4). Although the matrix is generally gray. Extremely rare foraminifers are well preserved in this microfacies. The low ratio of grains to the matrix suggests that this MFT was deposited in a low energy facies zone.
-
(ii) MFT2 foraminifer bearing micrite: This type has mud-supported with foraminifers (10–25%) scattered within the cryptocrystalline matrix. The matrix is commonly gray. Allochems are mainly planktonic foraminifers and few benthic foraminifers which are moderate to well preserved and filled with sparite. This type of facies is found in Nungphura NG5. This MFT was deposited in a low energy facies zone. (Fig. 4).
-
(iii) MFT3 foraminiferal micrite: Carbonate rocks of this type, which has a mud-supported structure in the New Paoye P1, P4, P6 and Toupokpi T7, T9 (Fig. 4). The matrix is generally gray with planktonic and benthic foraminifers as allochems, which are mainly filled with sparite. These features are relatively similar to MFT2 but the abundance of grains reaches 25–50%, which is significantly higher than in MFT2. In some samples of the section, the foraminifers are distributed unevenly in thin sections. Oriented foraminifers are concentrated in some areas, indicating a possible origin as gravity flow (Chen et al. 2010).
-
(iv) MFT4 foraminiferal packstone: This microfacies type with a grain-supported structure is identified in the sample of P10 at New Paoye (Fig. 4). The matrix is gray cryptocrystalline and allochems are characterized by moderate to well-preserved planktonic and benthic foraminifers, mainly filled by sparite. The abundance of allochems is higher than 50%.The foraminifers are unevenly distributed in some thin sections of the section and commonly occur in mm-scale bands within a grain-supported structure without mud.
Microfossils
Late Maastrichtian planktonic foraminifer found in the carbonates of the mélange zone of MOC indicates deep water oceanic depositional condition and a slow net rate of sediment accumulation (Chungkham et al.1992). Tewari et al. (2011) suggested that the carbonate rocks from MOC area were deposited under shallow marine depositional environment under humid and warm palaeoclimatic conditions. Later, Singh et al. (2013) have considered a shallow marine depositional environment (neritic to bathyal) for these carbonates based on petrographic and geochemical studies of the carbonate rocks of Ukhrul area. Devi and Duarah (2015) suggest that the carbonate sediments of MOC in Ukhrul area have been deposited in open to protect near shore, marginal neritic environment based on geochemical studies. Benthic foraminifera is useful environmental indicators of oxygen, nutrients and change in water depth (Morkhoven et al. 1986; Murray 1991). In present investigation, foraminferal assemblage has been recovered from the studied areas. Foraminiferal assemblage (Figs. 5, 6) comprises of planktonic (Globotruncana mariei, G. arca, G. conica, G. pettersi, Globotruncanita stuartiformis, G. calcarata, Globigerinelloides volutus, G. prairiehillensis, Heterohelix labellosa, H. globulosa, H. striata, Pseudotextularia nuttalli, Planoglobulina acervulinoides, Gublerina cuvillieri, Pseudoguembelina costulata) and benthic foraminifera (Anomalinoides cenomanica, A. intermdia, A. sp., Bathysiphon brosgei, Bolivina incrassata, Bolivinoides sp., Dentalina hamiltonensis, D. oligostegia, D. praecommunis, D. sp., Ellipsobulimina sp., Eouvigerina aculeata, E. Americana, E. sp. Fissurina sp., Frondicularia inversa, Gavelinella oodnadattensis, Globulina lacrima, Hergottella sp., Hoeglundina sp., Idalina antiqua, Lagena acuticosta, L. gibbera,, L. hispida, L. sulcata, Marginulina bullata, Nodosarella jarvisi, Nodosaria flexocarinata, N. obscura, N. sp., Cibicides sp., Planularia bradyana, Pseudonodosaria humilis, P. sp., Ramulina sp., Stilostomella sp.). All taxa of benthic foraminifera have been assigned as infaunal and epifaunal morphogroups (Table 2) for discussing depositional environment. The epifaunal morphotypes are those that live in the sediment (Collison 1980; Murray 1991). Microfossils characteristics suggest that these carbonates form part of the ophiolitic mélange zone that emplaced during subduction and obduction processes of the Indian plate and Myanmar plate collision.
Planktonic and benthic foraminifera present in the studied carbonate rocks 1. Stilostomella sp. (Benthic foraminifera) 2. Globigerinelloides volutus 3. Globigerinelloides prairiehillensis 4. Globotruncana calcarata 5. Globotruncana stuartiformis 6. Heterohelix striata 7. Globotruncana mariei 8. Pseudoguembelina costulata 9. Globigerinelloides prairiehillensis 10. Stilostomella sp. (Benthic foraminifera) 11. Heterohelix striata 12. Pseudoguembelina costulata
Geochemistry proxies
Major oxide geochemistry
The major element data of the analyzed rocks have been used to understand the chemical variations, commensurate with their petrographic typification, diagnostic chemical signatures, and to decipher relevant parameters of petrogenetic significance. The interpretation of such data in the light of modern concepts helps in evaluating the physio-chemical environments of deposition of these carbonates which have been a sequel. The major oxides variations of carbonates are shown in Table 3. The SiO2 ranges from 13.53 to 24.36 (wt.%) with an average value of 17.83 (wt.%). In some samples, the amount of SiO2% is very high (i.e. 24.36 in NG9), which is due to the presence of silicate minerals in the form of chert and detrital quartz. The SiO2 is the second major oxide present in the carbonate rocks of the study area. The total iron of the carbonate rocks of the study area ranges from 5.54 to 2.15 (wt. %) with an average value of 3.17 (wt.%). In some samples, the content of iron is high due to the presence of iron oxide like ilmenite, chamosite, glauconite, siderite, etc. The Al2O3 ranges from 11.29 to 3.84(wt.%) with an average of 6.50 (wt.%). MgO concentration ranges from 0.54 to 1.7 to (wt.%) with an average of 0.885 (wt.%). Geochemically, manganese is a strong lithophile metal, redox-sensitive and high content probably indicates reduction (Wallace 1990). Many workers have demonstrated that the Mn content and the varying amount in trace elements can be useful in differentiating shallow water and deep water limestones (Flugel 1982; Flugel and Stanley 1984). The concentration of manganese oxide content ranges from 0.057 to 0.141(wt.%) with an average of 0.08 (wt.%). The P2O5 content in carbonate rock is an indicator of reducing the environment with organic matter or the presence of phosphate minerals. Genetically, P2O5 content is related to the organic matter viz., fossils. The distribution of P2O5 content ranges from 0.282 to 0.114 (wt. %) with an average of 0.16(wt.%). A relatively higher percentage of P2O5 in the carbonate rock may be due to protected climatic condition and/or higher salinity in the study areas.
Trace element characteristics
Trace element concentrations in the carbonate rocks are given in (Table 3). These data have been used to differentiate the shallow and deep water limestones (Wedepohl 1970). The majority of the trace elements known in carbonate rocks are bounded to the detrital silica oxide fraction of the limestone. Mg, Sr and Mn are linked in specific ways with the carbonate phase depending on the similarity of the crystal chemistry of main components of more abundant carbonate mineral (Sr2+ and Ca2+ having the similar size of the same ionic charge while Mn2+ and Ca2+ have the same charge but slightly different size) and on the relatively high concentration in the ocean. The distribution of the abundances of trace elements (Table 3) of the study area in ppm is Sr (270–856), Rb (35–69), Ba (49–199), Zn (48–166), V (110–177), Co (10–21). The strontium (Sr) concentration of the Toupokpi carbonate ranges from 332 to 816 ppm with an average of 574.7 ppm, whereas the concentration of strontium ranges from 535 to 578 ppm with an average of 556.9 ppm in Nungphura and the concentration of strontium ranges from 270 to 856 ppm with an average of 596.2 ppm in New Paoye carbonate (Flugel and Wedepohl 1967), Sr content for shallow marine environment ranges from 100 to 400 ppm and for the deep marine environment it ranges from 500 to 3000 ppm. The variations of average Sr concentration in carbonates suggest a relatively deeper environment of deposition in New Paoye, Nungphura and Toupokpi, respectively. Anderson (1974) explained the effect of low water salinity on the depletion of strontium, so precipitations under a high saline environment result in a high concentration of strontium. Hence, the high percentage of strontium in New Paoye, Nungphura and Toupokpi carbonates might also indicate the formation of the carbonate under a relatively higher saline environment. The stable calcite becomes progressively enriched in magnesium and the stable dolomite becomes progressively enriched in calcium with increasing temperature (Morse and Mackenzie 1990). Many organisms secrete shells high in Mg, rather than low magnesian calcite. The Sr concentration of these carbonate rocks varies considerably but is generally about a factor or two higher than that of low magnesian biogenic calcite. The concentration of barium (Ba) ranges from 69 to 116 ppm in Toupokpi carbonate with an average of 92.53 ppm whereas the concentration of strontium ranges from 119 to 199 ppm with an average of 159.6 ppm in Nungphura, and the concentration of barium ranges from 41 to 134 ppm with an average of 99.53 ppm. In New Paoye limestone. As compared with the above table the study area has an average trace element (ppm) i.e. Co-16, Ba-112 and Ni-34 which are close to deep-sea carbonates (Table 4).
Rare earth element (REE) distributions
The total REE contents of the studied carbonates range from 22 to 169 ppm, with an average of 84 ppm (Table 3). Significant variations in REE content are noticed in the different types of carbonates. The carbonate samples from Nungphura have the highest concentration (35–169 ppm) of REE whereas the carbonates from Toupokpi have the lowest REE content (0.02–9 ppm). The low REE in Toupokpi carbonate is probably due to marine carbonate phases, which generally contain significantly lower REE content than detrital clays and heavy minerals (Palmer 1985). Post-Archean Australian Shale (PAAS)-normalized REE patterns of these carbonates are shown in (Fig. 7a). These carbonates exhibit a seawater-like REE pattern with LREE depletion [average (Nd/Yb)SN (shale normalized) = 0.98 ± 1.5] and consistent negative CeSN and positive LaSN anomalies (Singh et al. 2015). The carbonate minerals precipitated in equilibrium with seawater show distinct negative Ce anomalies and this may also be reflected in the bulk REE pattern (Palmer 1985). The (Dy/Yb)SN ratio in the MOC carbonates varies from 1.24 to 1.53 (average 1.3 ± 1.5), which is similar to the modern seawater (*0.8–1.1). The high (Dy/Yb)SN ratios in Manipur carbonates show enrichment in HREE rather than LREE, similar to modern seawater. LaSN and CeSN anomalies were calculated using the techniques (Bau and Dulski 1996; Webb and Kamber 2000). In the PAAS-normalized Pr/Pr* [Pr/(0.5Ce + 0.5Nd)]SN vs. Ce/Ce* [3Ce/(2La + Nd)SN plot (Fig. 7b)] all carbonate samples cluster in the field of negative Ce and positive La anomalies in agreement with modern open oceanic surface water (Fig. 7b). The effects of LREE/HREE fractionation in modern and ancient marine systems can be represented by the Er/Nd ratio (German and Elderfield 1989). The high Er/Nd ratio of carbonates effectively reveals the seawater signature retained by the marine carbonate. Er/Nd ratio in normal seawater is about 0.27 (De Baar et al. 1988). The addition of detrital material or diagenesis can reduce the Er/Nd value to 0.1 due to the preferential concentration of Nd relative to Er (German and Elderfield 1989; Bellanca et al. 1997). The Er/Nd ratio of the MOC carbonates is ranging from 1.2 to 0.9 which indicates the influence of detrital materials in these carbonates (Table 3). The shale normalized positive Eu anomalies (Eu/Eu*) are found either in waters affected by eolian input (Elderfield 1988) via the river, hydrothermal solutions, and the sediments resulting from high T-basalt alteration along with mid-ocean ridges, back-arc spreading center (German et al. 1993; Siby et al. 2008), and diagenesis (Murray et al. 1991) or variations in plagioclase content (Nath et al. 1992; Madhavaraju and Lee 2009). Positive Eu anomalies are uncommon in seawater, which generally results by input from hydrothermal anomalies which is unusual in seawater probably resulted by the diagenetic alteration in the carbonate (Brand and Veizer 1980); a slight increase in the primary or detrital feldspar component (Madhavaraju et al. 2010) and an increased oceanic input of hydrothermally originated fluids at mid-oceanic ridges (German et al. 1999). The ratio of REE is shown in Table 3.
a Post-Archean Australian Shale (PAAS) normalized REE patterns of carbonates from Manipur within Disang shale, Northeast India. b Plot of PAAS-normalized Pr/Pr* [Pr/(0.5Ce ? 0.5Nd)]SN versus Ce/Ce* (3Ce/(2La ? Nd) SN (after Bau and Dulski 1996; Webb and Kamber 2000). The plot discriminates between La and Ce anomalies, both may be present in seawater. All carbonate samples cluster in the field of negative Ce and positive La anomalies in perfect agreement with modern open oceanic surface water. The field of modern seawater is shown for comparison (after Nagarajan et al. 2011). Nungphura (NG5 and NG9), New Paoye (P1, P4, P6 and P10) and Toupokpi (T7 and T9)
Eu is the only REE showing a changing valency in the near-surface environment, where Eu3+ was reduced to Eu2+ under extremely reducing conditions. The redox potential of Eu/Eu* in aqueous solutions depends mainly on temperature and to a lesser extent on pressure, Ph, and REE speciation (Bau 1996), which explains that the positive Eu anomalies are typically found in acidic, reducing hydrothermal fluids. In this study, the samples display a large variations in Eu/Eu*, ranging from 0.9 to 1.1(Normalised PASS values) Positive Eu anomalies are uncommon in seawater, which is generally resulted by these actors: (1) an increased oceanic input of hydrothermally originated fluids at mid-oceanic ridges (German et al. 1999); (2) a slight increase in the primary detrital feldspar component (Madhavaraju et al. 2010) and (3) the digenetic alteration in the carbonate/limestone (Brand 1980).
Weak or no correlations between Ce/Ce* and Eu/Eu*, and between Ce/Ce* and REE (Fig. 8a, b) also suggest that the affection of the diagenetic process on REE concentrations is limited. Hence, we considered that the positive Eu anomalies are produced by the hydrothermal activity. Ce anomaly is mainly observed in the seawater (Elderfield 1982), and the values of Ce anomalies in marine carbonate provide important aspects of the geological record, i.e., terrigenous input and redox condition at the time and place of deposition (Madhavaraju and Lee 2009; Madhavaraju et al. 2010). The light negative Ce anomalies suggest that these carbonate rocks were deposited in an oxygenated environment, for the terrigenous input is limited.
Discussions
Depositional environment and age of carbonate
Benthic foraminifer is a useful environmental indicator of oxygen, nutrients, and change in water depth (Morkhoven et al. 1986; Murray 1991). The occurrence of taxa Globotruncana prefers warmer water bodies or low latitude whereas Heterohelix characterizes the oxygen minimum zone of a water column (Haq and Boersma 1998). All taxa of benthic foraminifera have been assigned as infaunal and epifaunal morphogroups (Table 2) for discussing depositional environment. The epifaunal morphotypes are those that live in the sediment (Collison 1980; Murray 1991). The planktonic benthic ratio is high (> 95%) in all the samples. Shallow bathyal environment (about 200–600 m depth) is indicated by the presence of benthic foraminifera taxa Bolivinoides, Lagena, Anomalinoides, Eouvigerina and Bathysiphon. Also, the presence of planktonic foraminifera taxa Globotuncana and benthic foraminifera assemblages suggest that the carbonate rocks were deposited under warmer or tropical and subtropical climate in deep marine conditions.
Differences in ∑REE content among the individual samples are mainly due to variations in the amount of terrigenous sediment included in these carbonates. The average REE concentration of the MOC carbonates (∑REE = 169 ± 22 ppm) is more or less comparable with the shallow marine carbonates of Maastrichtian limestones (∑REE = 73 ± 20). However, they are higher than the typical marine carbonate value of *28 ppm (Bellanca et al. 1997). The value of ∑REE = 3.36 ± 2.55 ppm indicates the shallow marine continental platform carbonates during late Neo Proterozoic (Mazumdar et al. 2003). REEs (La to Lu) show positive inter-elemental relationships, indicating their coherent nature, which reveals that they are highly linked with seawater during the REE fractionation. The extent of Ce depletion reflects the oxygenation state of the water (Komiya et al. 2008; Zhao et al. 2009). The negative anomalies of Ce in these carbonates suggest that they were deposited under an oxygenated environment. In addition, there is no remarkable variation in Ce anomalies in these carbonates which suggests that there was not much fluctuation in the bottom-water oxygen level.
Most of the planktonic foraminiferal assemblages recovered from pelagic limestones in the melange zone of Ukhrul, Manipur supported early Campanian age for the oldest pelagic limestone and terminal late Maastrichtian age for the youngest and supports the age described by Chungkham et al. (1992), Chungkham and Jafar (1998) and Singh et al. (2016). Foraminifera of late Cretaceous to Eocene age have been identified (Devi and Duarah 2018). In the present study age of the carbonates in the study area is found to be late Cretaceous (late Maastrichtian in age) which is confirmed by the presence of index fossils of planktonic foraminifera viz. Globigerinelloides volutus, Globotruncana calcarata, Globotruncana stuartiformis, Heterohelix striata, Globotruncana mariei, Pseudoguembelina costulata, Globigerinelloides prairiehillensis, Pseudoguembelina costulata. Thus microfossils evidences and geochemical characteristics suggest that the present investigated carbonates are similar to the carbonates of the mélange zone of MOC. Considering of these investigated carbonates, it is likely that the carbonate rocks were influenced by diagenetic activities at different tectonic processes during the evolution of MOC that emplaced during subduction and obduction processes of the Indian plate and Myanmar plate collision.
Conclusions
Based on microfacies evidences and geochemical characteristics of the studied carbonate rocks, the following main conclusions are summarized:
-
1. The investigated Disang group carbonates of Assam–Arakan Basin, Northeast India contain diverse fauna with the dominance of foraminiferal assemblages (planktonic and benthic). As a whole, sparse biomicrite or biomicrite is the dominant microfacies in which the fossil allochems are embedded in the groundmass of calcite matrix or the fossil allochems are cemented by calcite matrix which is indicative of low energy environment.
-
2. Age of the carbonates in the study area has been assigned as late Cretaceous (Maastrichtian in age) based on the well-preserved planktonic foraminifera, especially Globotruncana spp. and Heterohelix spp.
-
3. These carbonates show variable contents of REE range from 22 to 169 ppm and an average of 84 ppm which is higher than the average value of typical marine carbonate (*28 ppm). They also exhibit a seawater-like REE pattern with LREE depletion [average (Nd/Yb)SN (shale normalized) = 1 ± 0.9] and consistent negative CeSN and positive LaSN anomalies.
-
4. Similarity in microfossils assemblages preserved and geochemical characteristics, it is suggested that the present investigated carbonates are genetically related with the carbonates of the mélange zone of MOC. It is also concluded that carbonate rocks were influenced by diagenetic activities at different tectonic processes during the evolution of MOC and later emplaced within the Disang group flysch-like sediments during tectonic processes of the Indian plate and Myanmar plate collision.
References
Acharyya SK, Mitra ND, Nandy DR (1986) Regional geology and tectonic frame. In: Geology of Nagaland ophiolite. Memoirs of the Geological Survey of India, vol 119, pp 6–12
Anderson JW (1974) Influence of salinity and temperature on the O2 consumption of Neanthes arenaceodentata Moore. Amer Soc Zool 14:1260
Armstrong HA, Brasier MD (2005) Microfossil, 2nd edn. Blackwell publishing, p 287
Armstrong-Altrin JS, Verma SP, Madhavaraju J, Lee YI, Ramasamy S (2003) Geochemistry of late Miocene Kudankulam limestones, South India. Int Geol Rev 45:16–26
Armstrong-Altrin JS, Madhavaraju J, Sial AN, Kasper-Zubillaga JJ, Nagarajan R, Flores-Castro K, Rodrigues JF (2011) Petrography and stable isotope geochemistry of the Cretaceous EI Abra Limestone(Actopan), Mexico: implication on diagenesis. J Geol Soc Ind 77:349–359
Bau M, Dulski P (1996) Anthropogenic origin of positive gadolinium anomalies in river waters. Earth Planet Sci Lett 143:245–255
Bellanca A, Masetti D, Neri R (1997) Rare earth elements in limestone/marlstone couplets from the Albian-Cenomanian Cismon section (Venetian region, northern Italy): assessing REE sensitivity to environmental changes. Chem Geol 141:141–152
Boudagher-Fadel MK (2008) Evolution and geological significance of larger benthic forminifera. In: Wignall PB (ed) Developments in paleontology and stratigraphy, vol 21. Elsevier Publication, p 540
Brand U, Veizer J (1980) Chemical diagenesis of a multicomponent carbonate system. 1 trace element. J Sediment Res 50:1219–1236
Brunnschweiler RO (1966) On the geology of Indo-Burma range. Geol Soc Aust 13:137–194
Chen X, Wang C, Kuhnt W, Holbourn A, Huang Y, Ma C (2010) Lithofacies, microfacies and depositional environments of Upper Cretaceous Oceanic red beds (Chuangde Formation) in southern Tibet. Sediment Geol 235:100–110
Chungkham P, Jafar SA (1998) Late Cretaceous (Santonian-Maastrichtian) integrated coccolith-globotruncanid biostratigraphy of pelagic limestone from the accretionary prism of Manipur, Northeastern India. Micropaleontology 44:68–83
Chungkham P, Mishra PK, Sahni A (1992) Late and terminal Cretaceous foraminifera assemblages from Ukhrul mélange zone, Manipur. Curr Sci 62(6):478–481
Collison P (1980) Vertical distribution of foraminifera off the coast of Northumberland, England. J Foraminifer Res 10(1):716–719
Culver SJ (1987) Foraminifera. In: Broadhead TW (ed) Fossil prokaryotes and protists. Notes for a short course. University of Tennessee, Department of Geological Sciences, Studies in Geology, pp 169–212
De Baar HJW, German CR, Elderfield H, Van Gaans P (1988) Rare earth element distributions in anoxic waters of the Cariaco Trench. Geochim Cosmochim Acta 52:1203–1219
Devi KR, Duarah BP (2015) Geochemistry of Ukhrul limestone of Assam-Arakan subduction basin, Manipur, Northeast India. J Geol Soc India 85:367–376
Devi KR, Duarah BP (2018) Microfacies analysis and depositional environment of Maastrichtian-Eocene limestone of the Ukhrul district, Manipur, Northeast India. J Indian Assoc Sedimentol 35(1):99–106
Dunham RJ (1962) Classification of carbonate rocks according to depositional texture. In: Ham WE (ed) Classification of carbonate rocks, vol 1. Memoir of American Association of Petroleum Geologists, Tulsa, pp 108–121
Elderfield H (1988) The oceanic chemistry of the rare-earth elements. Philos Trans Royal Soc London 325:105–126
Elderfield H, Greaves MJ (1982) The rare earth elements in seawater. Nature 296:214–219
Elderfield H, Pagett R (1986) Rare earth elements in icthyoliths: variations with redox conditions and depositional environments. Sci Total Environ 49:175–197
Flugel E (1982) Microfacies analysis of limestones. Springer-Verlag, Heidelberg, p 634
Flugel E (2010) Microfacies of carbonate rocks, analysis interpretation and application. Springer-Verlag, Berlin, p 662
Flugel E, Stanley GD (1984) Reorganisation, development and evolution of post-Permian reef organisms. Amer J Palaeontographica 54:177–186
Flugel HW, Wedepohl KH (1967) Die-vertielung des strontiums in oberjurassicchen karbonatgeserien Der Nordlichen Kalkalpen. Contrib Mineral Petrol 14(3):249–299
Folk RL (1962) Spectral subdivision of limestone types. In: Ham WE (ed) Classification of carbonate rocks. Memoir of American Association of Petroleum Geologists, Tulsa, pp 62–84
Frimmel HE (2009) Trace element distribution in Neoproterozoic carbonates as palaeoenvironmental indicator. Chem Geol 258:338–353
Geel T (2000) Recognition of stratigraphic sequence in carbonate platform and slope deposits: empirical models based on microfacies analysis of Palaeogene deposits in southeastren Spain. Palaeogeogr Palaeoclimatol Palaeoecol 155:211–238
German CR, Elderfield H (1989) Rare earth elements in Saanich Inlet, British Columbia, a seasonally anoxic basin. Geochim Cosmochim Acta 53:2561–2571
German CR, Elderfield H (1990) Application of the Ce anomaly as a paleoredox indicator: the ground rules. Paleoceanography 5:823–833
German CR, Higgs NC, Thomson J, Mills R, Elderfield H, Blusztajn J, Fleer AP, Bacon AP (1993) A geochemical study of metalliferous sediment from the TAG hydrothermal mound, 26°08ʹN, Mid-Atlantic Ridge. J Geophys Res 98:9683–9692
German CR, Hergt J, Palmer MR, Edmond JM (1999) Geochemistry of a hydrothermal sediment core from the OBS vent-field, 21°N East Pacific Rise. Chem Geol 155:65–75
Haq BU, Boersma A (1998) Introduction to marine micropaleontology. Elsevier, Amsterdam
Kato Y, Nakao K, Isozaki Y (2002) Geochemistry of Late Permian Triassic pelagic cherts from southwest Japan: implications for an oceanic redox change. Chem Geol 182:15–34
Kemp RA, Trueman CN (2003) Rare earth elements in Solnhofen biogenic apatite: geochemical clues to the palaeoenvironment. Sediment Geol 155:109–127
Komiya T, Hirata T, Kitajima K, Yamamoto S, Shibuya T, Sawaki Y, Ishikawa T, Shu D, Li Y, Han J (2008) Evolution of the composition of seawater through geologic time, and its influence on the evolution of life. Gondwana Res 14:159–174
Madhavaraju J, Lee YI (2009) Geochemistry of the Dalmiapuram formation of the Uttatur group (Early Cretaceous), Cauvery basin, southeastern India: implications on provenance and paleo-redox conditions. Revista Mexicana De Ciencias Geológicas 26:380–394
Madhavaraju J, González-León CM, Lee YI, Armstrong-Altrin JS, Reyes-Campero LM (2010) Geochemistry of the mural formation (Aptian–Albian) of the Bisbee group, Northern Sonora, Mexico. Cretac Res 31:400–414
Mathur LP, Evans P (1964) Oil in India. In: International geological congress, 22nd session, pp 1–85
Mazumdar A, Tanaka K, Takahashi T, Kawabe I (2003) Characteristics of rare earth element abundance in shallow marine continental platform carbonates of Late Neoproterozoic successions from India. Gechem J 37:277–289
Mitra ND, Vidyadharan KT, Gour MP, Singh SK, Mishra UK, Khan IK, Ghosh S (1986) A note on the olistostrome deposits of Manipur. Rec Geol Surv India 114(4):61–76
Morkhoven FPCM, van Berggren WA, Edwards AS, Oertli HJ (1986) Cenozoic cosmopolitan deep-water benthic foraminifera. Bulletin des centres de recherches Exploration-production Elf-Aquitaine Mémoire 11:1–421
Morse JW, Mackenzie FT (1990) Geochemistry of sedimentary carbonates. Elsevier Scientific Publ. Co., New York, p 696
Murray JWL (1991) Ecology and palaeoecology of benthic foraminifera, vol 397. Longman, Harlow
Murray RW, Buchholtz Brink MR, Brink MR, Gerlach DC, Russ GP III, Jones DL (1991) Rare earth, major and trace elements in chert from the Franciscan complex and Monterey group, California: assessing REE sources to fine grained marine sediments. Geochim Cosmochim Acta 55:1875–1895
Nagarajan R, Madhavaraju J, Armstrong-Altrin JS, Nagendra R (2011) Geochemistry of Neoproterozoic limestones of the Shahabad formation, Bhima Basin, Karnataka, southern India. Geosci J 15(1):9–25
Nath BN, Roelandts I, Sudhakar M, Plueger WL (1992) Rare earth element patterns of the Central Indian Basin sediments related to their lithology. Geophys Res Lett 19:1197–1200
Palmer MR (1985) Rare earth elements in foraminifera tests. Earth Planet Sci Lett 73:285–298
Pascoe EH (1912) A traverse across the Naga hills of Assam from the Dimapur to the neighbourhood of Saramati peak. Rec Geol Surv India 42(4):261
Siby K, Nath BN, Ramaswamy V, Naman D, Gnaneshwar Rao T, Kamesh Raju KA, Selvaraj K, Chen CTA (2008) Possible detrital, diagenetic and hydrothermal sources for Holocene sediments of the Andaman backarc basin. Mar Geol 247:178–193
Singh AK, Devi LD, Singh NI, Subramanyam KSV, Singh RKB, Satyanarayanan M (2013) Platinum-group elements and gold distributions inperidotites and associated podiform chromitites of the Manipur Ophiolitic Complex Indo-Myanmar Orogenic Belt, Northeast India. Chem Erde 73:147–161
Singh AK, Tewari VC, Sial AN, Khanna PP, Singh NI (2015) Rare earth elements and stable isotope geochemistry of carbonates from the mélange zone of Manipur ophiolitic complex, Indo-Myanmar orogenic belt, Northeast India. Carbonate Evaporite. https://doi.org/10.1007/s13146-015-0249-2
Singh YR, Singh BP, Thanglemmoi MS, Naorem R (2016) Planktonic and benthic foraminifera of the Ophiolite associated limestones, around Ukhrul area of Manipur India: age and palaeoenvironment. In: Srivastava SK (ed) Recent trends in earth science research with special reference to NE India. Today and Tomorrow’s Printers and Publishers, New Delhi, pp 145–155
Tewari VC, Singh AK, Sial AN, Singh NI (2011) Stable isotope geochemistry of carbonate rocks from ophiolitic melange zone in Manipur, Northeast India. J Indian Geol Congr 3(2):17–27
Toyoda K, Nakamura Y, Masuda A (1990) Rare earth elements of Pacific pelagic sediments. Geochim Cosmochim Acta 54:1093–1103
Turekian KK, Wedepohl KH (1961) Distribution of the elements in some major units of the Earth’s crust. GSA Bull 72(2):175–192
Wallace MW (1990) Origin of dolomitization of the Barbowire, Canning Basin, Western Australia. J Ind Assoc Sedimentologist 37(1):105–122
Webb GE, Kamber BS (2000) Rare earth elements in Holocene reefal microbialites: a new shallow seawater proxy. Geochim Cosmochim Acta 64:1557–1565
Wedepohl KH (1970) Geochemistry data on sedimentary carbonates and carbonate rocks and their facies and petrogenic evaluation. Verh Geol Bundesanst 4:692–705
Zeng YF, Xia WJ (1986) Sedimentary petrology. Geological Publishing House, Beijing
Zhao YY, Zheng YF, Chen F (2009) Trace element and strontium isotope constraints on sedimentary environment of Ediacaran carbonates in southern. J Chemical Geol 265:345–362
Zolnaj S (1979) Carbonate microfossils separation from hard limestone. VII international symposium on Ostracod, Beogradd, pp 246–269
Acknowledgements
The authors are grateful to the Director, Wadia Institute of Himalayan Geology (WIHG), Dehradun, India for providing geochemical analytical facilities. The authors (YRS) are grateful to the Science and Research Board (SERB), Government of India (Grant No. EEQ/2016/000062) for funding of this work.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Guruaribam, V., Singh, Y.R. & Singh, A.K. Age and depositional environment of carbonate exotics associated with the Disang group of Assam–Arakan Basin, Northeast India: constraints from microfossils and geochemistry. Carbonates Evaporites 36, 46 (2021). https://doi.org/10.1007/s13146-021-00715-8
Accepted:
Published:
DOI: https://doi.org/10.1007/s13146-021-00715-8