Abstract
Objectives
The practice of silence is integral to some meditation traditions. Research is lacking on how silence practice affects brain connectivity. We hypothesized that silent, retreat-based meditation practice would reduce the connection between the language network from core cognitive networks such as the dorsal attention network (DAN) and default mode network (DMN).
Method
In a retrospective study, we analyzed resting state functional MRI (rsfMRI) data in 13 long-term Vipassana meditators (LTM) (~ 11,000 average hours of lifetime meditation experience) and healthy controls (n = 34) with no experience in meditation. We also compared our results with a large-scale dataset—Human Connectome Project (n = 169) (HCP). We compared the within and across functional connectivity among the three networks and correlated meditation experience and days spent in silence with the network connectivities.
Results
We found that the meditators have decoupled functional connectivity strengths (F(2,204) = 10.27, p < 0.01) between the DMN and language network (M = − 0.05, SD = 0.19) as compared to HCP controls (M = 0.14, SD = 0.14). The DAN had a negatively correlated connectivity strength with the language network in meditators (r = − 0.20) as compared to both control groups (r = 0.02) and a strong inverse relation (r = − 0.54) was found between DAN-language connectivity and the number of days spent in silent retreat.
Conclusions
Our study finds a potential role of silence training in changing the connectivities of three cognitive networks, DMN, DAN, and language network, resulting in reduced thoughts during meditation and a deeper experience of meditation.
Preregistration
This study is not preregistered.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Meditation as a means of spiritual growth has been practiced for thousands of years and in recent years has been the focus of many cognitive neuroscience investigations. Numerous studies have documented the effects of meditation practice on the brain, and how these changes in turn impact myriad functions including enhanced cognitive and emotional functioning; reduction of clinical symptoms like anxiety, depression, and PTSD; and increases in compassion (Schuman-Olivier et al., 2020; Seppälä et al. 2014; Sezer et al., 2022; Whitfield et al., 2022; Young et al., 2018).
Meditation is associated with multiple changes in brain functional connectivity (Sezer et al., 2021). Functional MRI paradigms that measure intrinsic or resting state functional connectivity do not examine the organization of brain networks during meditation, but can reveal effective changes in brain connectivity associated with regular/ongoing meditation practice. Meta-analyses have found that meditation is associated with connectivity changes within networks related to self-referential processing, self-awareness, and self-regulation, as well as within networks related to attention, executive function, and memory (Boccia et al., 2015; Rahrig et al., 2022). In particular, the default mode network (DMN) and the dorsal attention network (DAN) are affected by meditation practices. The DMN is associated with aspects of intrinsic processing such as internal mentation, self-projection, autobiographical memory, planning, and mind wandering (Buckner & Carroll, 2007; Buckner & DiNicola, 2019), whereas the DAN is associated with external engagement. Activity in the two networks is negatively correlated (Fox et al., 2005).
A recent study observed that an important indicator of brain health, the strength of negative correlations between DMN and DAN, was greater in long-term Vipassana meditators than in controls (Devaney et al., 2021). A separate longitudinal study found that focused-attention meditation increased the connectivity within DAN and between DMN–DAN, suggesting an ability to quickly switch between an internal mind-wandering state to an external attention state (Zhang et al., 2021). Extensive meditation experience is also associated with altered connectivity within the DMN regions of the posterior cingulate cortex (PCC) and cognitive control network regions of the dorsal anterior cingulate cortex (dACC) and dorsolateral prefrontal cortex (PFC) (Brewer et al., 2011). Additionally, the medial core part of the DMN, PCC, and ventromedial PFC has stronger anterior–posterior connectivity in long-term yoga practitioners (Santaella et al., 2019). Meditators exhibit increased within-network functional connectivity for the DAN and greater across-network connectivity with DMN and salience regions (Froeliger et al., 2012).
The core part of the DMN is more associated with constrained thoughts, whereas the medial temporal lobe part of the DMN, which overlaps with the language network (Lipkin et al., 2022), is related to spontaneous thought. The language network—which spans regions within the temporal, lateral frontal, and medial cortices—is associated with the processing, production, and comprehension of language (Fedorenko et al., 2011). The DMN couples both with the language and control networks (Gordon et al., 2020) and can be associated with hierarchical predictions of prolonged timescales (Heilbron et al., 2022; Margulies et al., 2016).
Ancient meditation traditions had deep insights into the functioning of the mind and its various faculties. Ancient Yogic texts highlight the role of various meditative practices in reducing “oscillations of the mind” which could putatively be interpreted as changes in large-scale cognitive networks like the DMN. Patanjali’s Yoga sutras mention five modulations or oscillations of the mind when it either engages with the world through perception, inference and written knowledge (pramana), or rests during sleep (nidra) or engages in memory (smriti), imagination (vikalpa), or wrong perception (viparyaya) (Shankar, 2022; Tripathi & Bharadwaj, 2021; Vivekananda, 2010). Yoga is defined as the state in which the mind is not engaged in the five modulations. In such a state, if a thought begins to arise, the meditator resists engaging with the thought, which typically leads to it petering out. Thus, there is typically little discursive thought during meditation, particularly during deep meditation states such as jhana or samadhi. Patanjali elucidates imagination as “Shabd artha gyan anupati vastu shunya vikalpa” which translates to “imagination is the knowledge about an inexistent object through comprehension of language” (Shankar, 2022; Vivekananda, 2010). Patanjali explicitly considers imagination as a modulation of the mind that links with language. Pramana, smriti, and viparyaya also involve various large-scale brain networks like the DMN, DAN, and language. Thus, we hypothesized that silence training, which is practiced extensively across spiritual traditions, reduces these oscillations in the mind and can result in lower connectivity between the language network and DAN/DMN.
Here, we focused on brain changes associated with extensive silent meditation practice. Specifically, we hypothesized that the language network will be less connected with the DAN and DMN in individuals who have participated in numerous silent Vipassana meditation retreats, relative to controls without any meditation practice. We compared resting state functional connectivity between the language network and the core subsystem of the DMN and the DAN between long-term meditators who have spent hundreds of days in silence with demographically matched controls with minimal meditation experience, and also with subjects from the Human Connectome Project (HCP).
Method
Participants
The Vipassana Meditation Cohort (VMC) dataset included 16 experienced meditators (11 males, mean age = 34.33 years) out of which 13 (9 males) had resting state data. Subjects were recruited using word of mouth from a local Vipassana center in Massachusetts. The subjects had on average 7 years of meditation experience, with an average estimated 8311 hr of meditation (range = 1300–50,000 hr) along with 291 ± 86 days (range = 25–319) spent on silent retreats. The average weekly time dedicated to meditation was 843 ± 230 (range 540–1560) min spread over 14 ± 4 sessions (range 9–26) ranging between 55 and 65 min. The data were collected for a prior study (Devaney et al., 2021) investigating differences in attentional processes between meditators and non-meditators. Age, gender, and expertise-matched controls were recruited (16 total, 11 males) from which eight subjects (five males) had resting state data that was used in the current manuscript. Since prior research has shown that recruitment based on expertise can result in differences in task performance (Boot et al., 2011), the expertise-matched controls were included on the basis of a “sham expertise” to control for meditators who were explicitly recruited for their meditation expertise. Potential participants initially were asked to self-rate their abilities on a broad range of tasks including athletic abilities, swimming, driving, etc., and subsequently were told that they were being invited to participate because of their specific “expertise” in a field for which they highly rated their own abilities (for details see Devaney et al., 2021). We included an additional 26 (11 male, mean age = 29.21 years) control subjects (CNT dataset) from the Boston University community to overcome the attrition in the sham expertise control group and improve statistical robustness. Overall, we had a total of 34 controls (16 males, mean age = 31.54 years). All subjects in the VMC and CNT groups were right-handed. The study was conducted in accordance with the Declaration of Helsinki. The Institutional Review Board of the University approved the study and informed consent was obtained from all subjects who were financially compensated ($75/hr).
We compared the resting state connectivity metrics with subjects from the Human Connectome Project (HCP) dataset (Van Essen et al., 2013, db.humanconnectome.org) which consisted of 169 healthy subjects (104 females) in the age range 22–35 (exact ages were restricted in the HCP dataset) who had data collected for resting state and seven cognitive tasks as detailed in Barch et al. (2013) and Van Essen et al. (2012). The handedness was also restricted for the HCP subjects but about 10% of the subjects were left-handed as mentioned elsewhere (Ruck & Schoenemann, 2021).
Procedure
VMC and CNT dataset was acquired on a 3-T Siemens Tim Trio using a 32-channel Siemens head coil. T1-weighted (MPRAGE) high-resolution data (TR = 6.6 ms, TE = 2.9 ms, flip angle = 8°, voxel size = 1.0 × 1.0 × 1.3 mm) were acquired for each participant along with gradient echo EPI sequences (TR = 2600 ms, TE = 30 ms, flip angle = 90°, voxel size = 3.0 × 3.0 × 3.1 mm, 42 slices, whole brain coverage) for the resting state data that consisted of two runs of 6 min each (278 time points). The resting state data were collected with a fixation cross on the screen and the subjects were asked to fixate on the cross and not do anything in particular, including explicit instructions not to meditate.
The Human Connectome Project (HCP) resting state data were acquired on a 3-T Siemens Connectom scanner with high-resolution T1 (voxel size = 1.0 × 1.0 × 1.3 mm) and T2 data. Resting state data were acquired using gradient echo EPI sequences (TR = 720 ms, multiband factor = 3, voxel size = 2 mm isotropic, 72 slices, total 1200 TRs per run) collected across two fMRI scanning days which were interspersed with other MRI data. Across the four runs, data were normalized and concatenated resulting in 4800 total volumes.
We used Freesurfer (http://surfer.nmr.mgh.harvard.edu/) for cortical reconstruction and volumetric segmentation of the T1 data. More details about the technical methods can be found in prior publications (Dale et al., 1999; Fischl & Dale, 2000; Fischl et al., 1999, 2002, 2004; Jovicich et al., 2006; Power et al., 2012; Ségonne et al., 2004). For the resting state data, we performed slice time correction, motion correction, and volumetric spatial smoothing with an FWHM of 1.5 mm followed by intensity normalization and boundary-based registration to the subject’s own high-resolution anatomical data. We then used six motion parameters (reduced to three singular value decomposition–based eigenvectors) as nuisance regressors for motion correction of the resting state data which was followed by ventricular and white matter regression. Time points with excessive motion above a threshold of 0.5 mm were removed and replaced using linear interpolation. We then performed bandpass filtering between 0.01 and 0.08 Hz, followed by smoothing global mean gray matter signal regression.
The HCP dataset was preprocessed using the minimal preprocessing pipeline (Glasser et al., 2013) that incorporated Freesurfer-based surface registration to the CIFTI surface (32 k grayordinate format). The preprocessing incorporated artifact correction, gradient non-linearity correction, motion correction and EPI distortion correction, temporal denoising, and bandpass filtering between 0.01 and 0.08 Hz. Structural and functional images were registered from the native subject space to the MNI space which was followed by cortical segmentation in the native surface mesh using the Freesurfer pipeline. The data were registered from the native to 168 k to 32 k vertices surface CIFTI format. We finally applied a spatial smoothing of 2-mm FWHM at the surface level.
Measures
We extracted the language network from the Schaefer ROIs using the definitions of the language network as defined by Federenko and colleagues (Lipkin et al., 2022). We limited our analysis to the left lateral representation of the language network because most of the subjects in the analyses were right-handed and the extent of the network in the language atlas for the right hemisphere was limited. We took the core subsystem of the default mode network (Christoff et al., 2016) without the dorsal inferior parietal lobule to avoid overlap with some parts of the language network. We extracted the dorsal attention network using the Schaefer 200 parcel cortical atlas (Schaefer et al., 2017) and the 17-network Yeo parcellation definition (Yeo et al., 2011). We computed the seed-to-seed resting state functional connectivity (rsFC) analysis by taking an average of all the vertices within a seed across time and then taking Pearson correlations across the seeds (Finn et al., 2015; Rosenberg et al., 2015).
Data Analyses
To compare the seed-seed rsFC measure, both within and across networks, between the meditator (VMC) and non-meditator groups (CNT and HCP), we Fisher z-transformed the correlation coefficients between the networks within a participant and then used the two-way ANOVA method followed by Tukey’s HSD post hoc analysis to statistically analyze the correlation strengths across groups. We used the implementation of ANOVA from the statsmodels toolbox (Seabold & Perktold, 2010) which utilizes the Scipy Python toolbox (Virtanen et al., 2020). We also reported adjusted p-values for the post-hoc analyses.
We computed the difference between the edge connectivity (Faskowitz et al., 2020) of language and DMN, DMN and DAN, and DAN and language edges across the meditator and control groups to analyze how the effect of silence and meditation training impacts the networks and the relationship among them.
The HCP dataset also included a language task where subjects listened to 20–25-s stories and responded to yes/no questions after the story block ended. We correlated the accuracy and reaction time in the language task with the connectivities between DMN, DAN, and language regions.
Results
We analyzed the seed-based connectivity between the core DMN (cDMN) and language network (Fig. 1). We computed the correlation strength between the networks within an individual subject, Fisher z-transformed them, and used a two-way ANOVA to compare the network strengths across the groups and found that cDMN-language connectivity strength differed (F(2,204) = 12.28, p < 0.01) across the groups. Tukey’s post hoc analysis found differences between CNT (M = 0.06, SD = 0.13) and HCP (M = 0.14, SD = 0.14) groups (adjusted p = 0.03) and also between the HCP and VMC (M = –0.05, SD = 0.19) groups (adjusted p = 0.001). The HCP group had stronger rsFC correlation strengths as compared to the two other groups.
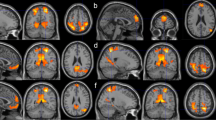
Network definitions and cDMN-language connectivity analysis. a) We extracted the averaged time series from eyes-open resting state data in the subjects for the lateral part of the left hemispheric language network as defined in Lipkin et al. (2022) and correlated it to the medial part of the core DMN. We performed Pearson correlation to estimate the functional connectivity of the two networks. b) We plotted the cDMN-language network connectivity for the three datasets (Vipassana meditation cohort, VMC; controls, CNT; Human Connectome Project controls, HCP). *Statistical significance with p < 0.05
Analyzing the cDMN and DAN network connectivity (Fig. 2), we found that the three groups differed (F(2,204) = 69.63, p < 0.01) with post hoc differences between CNT (M = –0.08, SD = 0.2) and HCP (M = –0.37, SD = 0.1) groups (adjusted p = 0.001) and HCP and VMC (M = –0.15, SD = 0.10) groups (adjusted p = 0.001) with the HCP group having stronger inverse correlation strengths as compared to VMC and CNT. Here, inverse correlations signify an opposing activity in the networks wherein when the BOLD activity in one network increases, it decreases in the other network.
Functional connectivity among the networks across groups. Analyzing the functional connectivity in core DMN, DAN, and language network for the three groups (Vipassana meditation cohort, VMC; controls, CNT; Human Connectome Project controls, HCP). The cDMN-DAN connectivity was low for the meditators compared to HCP. The control groups had near zero DAN and language network whereas they were negatively correlated for the meditators. *Statistical significance with p < 0.05
The resting state functional connectivity between the DAN and language network was negatively correlated for the VMC (M = –0.2, SD = 0.15) group but not for the HCP (M = 0.02, SD = 0.13) and CNT (M = 0.03, SD = 0.18) groups. Group differences were statistically significant (F(2,204) = 10.27, p < 0.01) with post hoc differences between VMC and HCP groups (adjusted p < 0.001) and CNT and VMC groups (adjusted p = 0.001). We correlated the time spent in silent retreats with the connectivity changes. Prior to computing statistics, we removed the outliers (two standard deviations away from the mean time period spent in silence; one subject with the most amount of silence practice at 843 days was removed). DAN-language connectivity (Fig. 3) showed a trending negative correlation with the number of days spent in silent retreat (r(11) = –0.54, p = 0.06), which did not reach statistical significance. Weaker correlations with meditation experience were found for cDMN connectivity with other networks: cDMN-DAN (r(11) = 0.17, p = 0.57) and cDMN-language (r(11) = 0.27, p = 0.39), which were not statistically significant.
Relationship between network connectivity and meditation experience. We found a negatively correlated relationship between days spent in silent retreat with connectivity between DAN and language network connectivity (r(11) = –0.54, p = 0.06). We excluded the most experienced meditator with 895 days in silent retreat. The more experienced participants had a tendency toward stronger negatively correlated language network and DAN
The HCP dataset included a language task and we correlated the accuracy of the task with the functional connectivity among the various networks to determine if there are behavioral differences with change in network connectivity measures in the healthy subjects. We did not find significant correlations (p > 0.11) between responses to language tasks (accuracy, reaction time) and DMN-language connectivity. Since, we did not collect language-based behavioral measures for the meditator and control datasets, we cannot determine if silence practice can result in discernable behavioral changes in language processing. Further research would be required to find behavioral measures that can change with the practice of silence and meditation. We summarize network connectivity differences between the two groups in Fig. 4; controls exhibit positive correlation between DAN and language networks, no correlation between DAN and language networks, and inverse correlations between the DAN and DMN, whereas in meditators the language network is decoupled from the DMN and more negatively correlated with the DAN.
Summarizing network effects. Possible effects of silence and meditation training on resting state functional connectivity on meditators. The green arrow indicates a positive relationship, whereas the red arrow represents an inversely correlated relationship between the networks. The cDMN and DAN are negatively correlated in the two groups. The cDMN and language network are positively correlated in controls and uncorrelated in meditators, whereas the DAN and language network are inversely correlated in meditators and decoupled in the control groups
Discussion
We found that in long-term meditators who have extensive experience with silent meditation retreats, the language network decouples from the core DMN and is negatively correlated with the DAN. Our study was retrospective, so we could not investigate a causal link between silence training and the decoupling of the language network. However, these correlational findings are noteworthy and deserve future investigation. The behavioral effects of language network decoupling on the efficacy of meditation practice are another point for future investigations. Specifically, the reduced influence of the language network on the core DMN and DAN may assist a meditation practitioner in attaining and/or maintaining deeper states of meditation.
Long-term meditators experience changes in their DMN connectivity (Boccia et al., 2015; Brewer et al., 2011) and our findings add to that literature. The DMN is active in self-projection scenarios like thinking about the past or the future or thinking about others. The DMN integrates these processes into a cohesive narrative (Buckner & Carroll, 2007) which spans long timescales (Heilbron et al., 2022; Margulies et al., 2016), whereas recent studies of the language network have shown that it is limited to short-timescale information processing such as next word or sentence prediction (Caucheteux & King, 2022; Schrimpf et al., 2021). As time spent in silence changes language activity, its connectivity with DMN would decouple resulting in a reduction of spontaneous imaginative thoughts and more stability in meditation. There could be a possible reduction in the hierarchical predictive processing engaged by these large-scale networks as some studies have suggested (Kirk et al., 2019; Lutz et al., 2019; Pagnoni, 2019) and can disengage anticipatory processing and the modulations of the mind, thus allowing the practitioner to be more present and mindful in the now (Laukkonen & Slagter, 2021). These effects could be beneficial to allow the practitioner to go into deeper states of meditation like samadhi (Tripathi & Bharadwaj, 2021) and jhana (Dennison, 2019; Hagerty et al., 2013). Silence and meditation may offer the advantage of diminishing samskaras, as suggested by Srinivasan (2013). In Buddhist and Vedic traditions, samskaras are considered to be the accumulated imprints of memories and emotions on the body, which shape our physical and mental habits. These habitual patterns could be correlated with the predictive patterns of the brain. Through regular periods of silence and meditation, a practitioner deliberately withdraws from external stimuli, potentially allowing the brain to reconfigure these established patterns and reduce the various modulations of the mind. This process could effectively lessen the influence of previous experiences on current thoughts and behaviors, granting the individual greater autonomy and volition in the present, unencumbered by historical imprints. This hypothesis is supported by evidence suggesting that meditation can alleviate the impact of traumatic memories, as seen in studies on post-traumatic stress disorder (PTSD) (Seppälä et al. 2014). Srinivasan (2013) posits that liberation from samskaras correlates with an increased sense of freedom, relaxation, and presence in the moment.
The DAN is associated with external attention, and engaging and demanding tasks (Fox et al., 2005). DAN dysfunction is associated with ADHD and attention deficits (McCarthy et al., 2013). DMN and DAN have a dynamic relationship which is usually anti-correlated such that demanding tasks drive the DAN and suppress the DMN (Fox et al., 2005; Greicius & Menon, 2004) but can vary throughout the day based on cognitive state, mood, and arousal levels (Dixon et al., 2017). We found a relationship between the number of days spent in silent retreats and the degree of DAN-language inverse connectivity. The negative association of the DAN with the language network in long-term meditators in the current study could suggest a reduction in spontaneous thoughts which shifts the external attention to internal stimuli (e.g., interoceptive processes, consciousness itself). Multiple studies have shown that meditation changes functional connectivity within the DAN and across DAN-DMN (Fialoke et al., 2023; Devaney et al., 2021; Froeliger et al., 2012). The mental training in ignoring distractions associated with meditation helps strengthen the DAN in practitioners (Zhang et al., 2021) and improves performance in attention tasks (Kozasa et al., 2012). The degree to which the long-term meditators are able to suppress spontaneous thoughts, the better they are at maintaining attention on their chosen object of attention. Many spiritual practices involve attention to the breath or some external point (focused attention) that could benefit from having a negatively correlated DAN-language network, allowing for better sustained attention which could result in a more effective meditation without a large degree of internal distractions.
We observed that the more days spent in silence, the more negatively correlated the DAN-language regions (although the test did not cross statistical significance possibly due to the lower number of long-term meditators in the study). The DAN-language correlation for the most experienced meditator in our cohort with 895 days spent in silence resembled that of the least experienced subjects which is in line with earlier studies which show an inverted U-shaped curve between brain activation and experience, possibly due to long-term synaptic plasticity reducing the amount of energy required to sustain a mental process (Brefczynski-Lewis et al., 2007). With a larger number of long-term meditators with 500–900 days of silent retreat experience, we may have been able to see a cleaner U-shaped curve between network connectivity and experience. We also found weak correlations between meditation experience and cDMN-DAN and cDMN-language connectivity strengths, which again would benefit from more participants in the study.
We found an inverse correlation between the core DMN-DAN connectivity in both meditators and controls. Our group previously demonstrated that when considering the entire DMN, the inverse correlation with the DAN is stronger in meditators than in controls (Devaney et al., 2021). It is important to note that in that study, the full DMN was defined as one of seven cortical networks (Yeo et al., 2011), which encompass not only the core medial DMN regions analyzed here (defined from the 17-network parcellation of Yeo et al. (2011)) but also key portions of the language network. This early parcellation map included the language network into the DMN. Our present analysis more finely dissects the DMN and indicates that decoupling of the language network with both DAN and core DMN is a key driver of the effects earlier reported with the coarser network definitions.
The language network is juxtaposed alongside the higher-order association networks across the brain and its deactivation during the periods of silence could help decrease activity in the closely connected DMN regions (Du et al., 2023). The language network is differentiated into two distinct pathways: production and comprehension streams. In the current study, we did not have sufficient data to separate out the finer details of the language network but the practice of silence can have divergent influences on language production and comprehension. Further research would be needed to determine if silence practice can result in any language deficits or improvements.
The key insight behind the paper was inspired by Patanjali’s Yoga Sutras’ definition of the various modulations of the mind and how the process of imagination (vikalpa) could be linked to the activity and the connectivity of the language region and DMN. The intrinsic processing-centered DMN gets decoupled with the language network, thus pointing to a reduction in spontaneous imaginative thoughts or vikalpa. Smriti (memory) can be associated with the hippocampus-centered episodic projection memory subsystem which is connected with a subnetwork of DMN (Zheng et al., 2021; Du et al., 2023) and the practice of silence can reduce the time spent on past memories allowing the practitioner to be more in the present. Recent research on the self-projection parts of the DMN suggests that the same network is involved in both thinking about the past and the future (DiNicola et al., 2020), what the authors now call the scene construction network. The language network and the DMN are tightly juxtaposed and can function together in some of these mental modulations. The pramana (proof) modulation could engage the language network along with the DAN and get more negatively correlated in long-term meditators with extensive silence practice, and probably be linked to reduced mind oscillations. The modulations of the mind elucidated by Patanjali describe an interesting model of mental states and can help us quantify the progress related to various meditative practices based on time spent on these mental modulations. We would need further research to find functional activity and connectivity-based measures associated with the Patanjalian mental model.
Limitations and Future Research
While the present findings are intriguing, it is important to acknowledge the key limitations of this work. Our study lacked behavioral or imaging data on language tasks to assess language functionality. Also, as noted above, the retrospective nature of this study did not permit us to investigate the causal influence of silence practice on language network decoupling. We had a limited number of expert meditators in the study due to recruitment challenges which resulted in non-statistically significant correlations between network connectivity and meditation experience. An expanded study with a larger number of expert meditators could help overcome this issue. One of the major limitations of the study was a gender imbalance among the participants. We used a snowball sampling approach for recruitment and it resulted in more male experienced meditators as a part of the study. In future studies, we will carefully balance across genders during recruitment and also control for handedness as hand dominance affects the lateralization of the language region (Knecht et al., 2000).
Another line of future research would be to determine if the decoupling of the language network is a continuous or sudden process or does it take at least some number of silent retreats before connectivity changes. One could collect language-based imaging data on meditators and analyze the change in the word prediction response with the practice of meditation. One line of research could involve neuromodulating the language network using tDCS, tACS, or TMS during meditation and observing responses in the various signatures of meditation with E/MEG. We could also see changes in DMN-based connectivity with neuromodulation methods possibly at the alpha and beta rhythms as earlier studies (Cheng et al., 2022; Marino et al., 2019; Tripathi & Somers, 2023) have suggested a relationship between the two modalities.
Data Availability
The data presented in this study are available on request from the corresponding author. The data are not publicly available due to participant privacy. The Human Connectome Project (HCP) dataset is publicly available on db.humanconnectome.org.
References
Barch, D. M., Burgess, G. C., Harms, M. P., Petersen, S. E., Schlaggar, B. L., Corbetta, M., Glasser, M. F., Curtiss, S., Dixit, S., Feldt, C., Nolan, D., Bryant, E., Hartley, T., Footer, O., Bjork, J. M., Poldrack, R., Smith, S., Johansen-Berg, H., Snyder, A. Z., & Essen, D. C. V. (2013). Function in the Human Connectome: Task-fMRI and individual differences in behavior. NeuroImage, 80, 169. https://doi.org/10.1016/j.neuroimage.2013.05.033
Boccia, M., Piccardi, L., & Guariglia, P. (2015). The meditative mind: A comprehensive meta-analysis of MRI studies. BioMed Research International, 2015, 419808. https://doi.org/10.1155/2015/419808
Boot, W. R., Blakely, D. P., & Simons, D. J. (2011). Do action video games improve perception and cognition? Frontiers in Psychology, 2, 226. https://doi.org/10.3389/fpsyg.2011.00226
Brefczynski-Lewis, J. A., Lutz, A., Schaefer, H. S., Levinson, D. B., & Davidson, R. J. (2007). Neural correlates of attentional expertise in long-term meditation practitioners. Proceedings of the National Academy of Sciences, 104(27), 11483–11488. https://doi.org/10.1073/pnas.0606552104
Brewer, J. A., Worhunsky, P. D., Gray, J. R., Tang, Y.-Y., Weber, J., & Kober, H. (2011). Meditation experience is associated with differences in default mode network activity and connectivity. Proceedings of the National Academy of Sciences, 108(50), 20254–20259. https://doi.org/10.1073/pnas.1112029108
Buckner, R. L., & Carroll, D. C. (2007). Self-projection and the brain. Trends in Cognitive Sciences, 11(2), 49–57. https://doi.org/10.1016/j.tics.2006.11.004
Buckner, R. L., & DiNicola, L. M. (2019). The brain’s default network: Updated anatomy, physiology and evolving insights. Nature Reviews Neuroscience, 20(10), 593–608. https://doi.org/10.1038/s41583-019-0212-7
Caucheteux, C., & King, J.-R. (2022). Brains and algorithms partially converge in natural language processing. Communications Biology, 5, 134. https://doi.org/10.1038/s42003-022-03036-1
Cheng, P., Grover, S., Wen, W., Sankaranarayanan, S., Davies, S., Fragetta, J., Soto, D., & Reinhart, R. M. G. (2022). Dissociable rhythmic mechanisms enhance memory for conscious and nonconscious perceptual contents. Proceedings of the National Academy of Sciences, 119(44), e2211147119. https://doi.org/10.1073/pnas.2211147119
Christoff, K., Irving, Z. C., Fox, K. C. R., Spreng, R. N., & Andrews-Hanna, J. R. (2016). Mind-wandering as spontaneous thought: A dynamic framework. Nature Reviews Neuroscience, 17(11), 718–731. https://doi.org/10.1038/nrn.2016.113
Dale, A. M., Fischl, B., & Sereno, M. I. (1999). Cortical surface-based analysis: I. Segmentation and Surface Reconstruction. Neuroimage, 9(2), 179–194. https://doi.org/10.1006/nimg.1998.0395
Dennison, P. (2019). The human default consciousness and its disruption: Insights from an EEG study of Buddhist Jhāna meditation. Frontiers in Human Neuroscience, 13, 178. https://doi.org/10.3389/fnhum.2019.00178
Devaney, K. J., Levin, E. J., Tripathi, V., Higgins, J. P., Lazar, S. W., & Somers, D. C. (2021). Attention and default mode network assessments of meditation experience during active cognition and rest. Brain Sciences, 11(5), 566. https://doi.org/10.3390/brainsci11050566
DiNicola, L. M., Braga, R. M., & Buckner, R. L. (2020). Parallel distributed networks dissociate episodic and social functions within the individual. Journal of Neurophysiology, 123(3), 1144–1179. https://doi.org/10.1152/jn.00529.2019
Dixon, M. L., Andrews-Hanna, J. R., Spreng, R. N., Irving, Z. C., Mills, C., Girn, M., & Christoff, K. (2017). Interactions between the default network and dorsal attention network vary across default subsystems, time, and cognitive states. NeuroImage, 147, 632–649. https://doi.org/10.1016/j.neuroimage.2016.12.073
Du, J., DiNicola, L. M., Angeli, P. A., Saadon-Grosman, N., Sun, W., Kaiser, S., Ladopoulou, J., Xue, A., Yeo, B. T. T., Eldaief, M. C., & Buckner, R. L. (2023). Within-individual organization of the human cerebral cortex: Networks, global topography, and function. bioRxiv. https://doi.org/10.1101/2023.08.08.552437
Faskowitz, J., Esfahlani, F. Z., Jo, Y., Sporns, O., & Betzel, R. F. (2020). Edge-centric functional network representations of human cerebral cortex reveal overlapping system-level architecture. Nature Neuroscience, 23, 1644–1654. https://doi.org/10.1038/s41593-020-00719-y
Fedorenko, E., Behr, M. K., & Kanwisher, N. (2011). Functional specificity for high-level linguistic processing in the human brain. Proceedings of the National Academy of Sciences, 108(39), 16428–16433. https://doi.org/10.1073/pnas.1112937108
Fialoke, S., Tripathi, V., Thakral, S., Dhawan, A., Majahan, V., & Garg, R. (2023). Brain connectivity changes in meditators and novices during yoga nidra: A novel fMRI study. bioRxiv. https://doi.org/10.1101/2023.09.15.557655
Finn, E. S., Shen, X., Scheinost, D., Rosenberg, M. D., Huang, J., Chun, M. M., Papademetris, X., & Constable, R. T. (2015). Functional connectome fingerprinting: Identifying individuals using patterns of brain connectivity. Nature Neuroscience, 18, 1664–1671. https://doi.org/10.1038/nn.4135
Fischl, B., & Dale, A. M. (2000). Measuring the thickness of the human cerebral cortex from magnetic resonance images. Proceedings of the National Academy of Sciences, 97(20), 11050–11055. https://doi.org/10.1073/pnas.200033797
Fischl, B., Sereno, M. I., & Dale, A. M. (1999). Cortical surface-based analysis II Inflation, flattening, and a surface-based coordinate system. NeuroImage, 9(2), 195–207. https://doi.org/10.1006/nimg.1998.0396
Fischl, B., Salat, D. H., Busa, E., Albert, M., Dieterich, M., Haselgrove, C., van der Kouwe, A., Killiany, R., Kennedy, D., Klaveness, S., Montillo, A., Makris, N., Rosen, B., & Dale, A. M. (2002). Whole brain segmentation: Automated labeling of neuroanatomical structures in the human brain. Neuron, 33(3), 341–355. https://doi.org/10.1016/S0896-6273(02)00569-X
Fischl, B., Salat, D. H., van der Kouwe, A. J. W., Makris, N., Ségonne, F., Quinn, B. T., & Dale, A. M. (2004). Sequence-independent segmentation of magnetic resonance images. NeuroImage, 23, S69–S84. https://doi.org/10.1016/j.neuroimage.2004.07.016
Fox, M. D., Snyder, A. Z., Vincent, J. L., Corbetta, M., Van Essen, D. C., & Raichle, M. E. (2005). The human brain is intrinsically organized into dynamic, anticorrelated functional networks. Proceedings of the National Academy of Sciences, 102(27), 9673–9678. https://doi.org/10.1002/poc.610010207
Froeliger, B., Garland, E. L., Kozink, R. V., Modlin, L. A., Chen, N.-K., McClernon, F. J., Greeson, J. M., & Sobin, P. (2012). Meditation-state functional connectivity (msFC): Strengthening of the dorsal attention network and beyond. Evidence-Based Complementary and Alternative Medicine, 2012, 680407. https://doi.org/10.1155/2012/680407
Glasser, M. F., Sotiropoulos, S. N., Wilson, J. A., Coalson, T. S., Fischl, B., Andersson, J. L., Xu, J., Jbabdi, S., Webster, M., Polimeni, J. R., Van Essen, D. C., & Jenkinson, M. (2013). The minimal preprocessing pipelines for the Human Connectome Project. NeuroImage, 80, 105–124. https://doi.org/10.1016/j.neuroimage.2013.04.127
Gordon, E. M., Laumann, T. O., Marek, S., Raut, R. V., Gratton, C., Newbold, D. J., Greene, D. J., Coalson, R. S., Snyder, A. Z., Schlaggar, B. L., Petersen, S. E., Dosenbach, N. U. F., & Nelson, S. M. (2020). Default-mode network streams for coupling to language and control systems. Proceedings of the National Academy of Sciences of the United States of America, 117(29), 17308–17319. https://doi.org/10.1073/pnas.2005238117
Greicius, M., & Menon, V. (2004). Default-mode activity during a passive sensory task: Uncoupled from deactivation but impacting activation. Journal of Cognitive Neuroscience, 16(9), 1484–1492. https://doi.org/10.1162/0898929042568532
Hagerty, M. R., Isaacs, J., Brasington, L., Shupe, L., Fetz, E. E., & Cramer, S. C. (2013). Case study of ecstatic meditation: FMRI and EEG evidence of self-stimulating a reward system. Neural Plasticity, 2013, 653572. https://doi.org/10.1155/2013/653572
Heilbron, M., Armeni, K., Schoffelen, J.-M., Hagoort, P., & de Lange, F. P. (2022). A hierarchy of linguistic predictions during natural language comprehension. Proceedings of the National Academy of Sciences, 119(32), e2201968119. https://doi.org/10.1073/pnas.2201968119
Jovicich, J., Czanner, S., Greve, D., Haley, E., van der Kouwe, A., Gollub, R., Kennedy, D., Schmitt, F., Brown, G., MacFall, J., Fischl, B., & Dale, A. (2006). Reliability in multi-site structural MRI studies: Effects of gradient non-linearity correction on phantom and human data. NeuroImage, 30(2), 436–443. https://doi.org/10.1016/j.neuroimage.2005.09.046
Kirk, U., Pagnoni, G., Hétu, S., & Montague, R. (2019). Short-term mindfulness practice attenuates reward prediction errors signals in the brain. Scientific Reports, 9(1), 6964. https://doi.org/10.1038/s41598-019-43474-2
Knecht, S., Dräger, B., Deppe, M., Bobe, L., Lohmann, H., Flöel, A., Ringelstein, E.-B., & Henningsen, H. (2000). Handedness and hemispheric language dominance in healthy humans. Brain, 123(12), 2512–2518. https://doi.org/10.1093/brain/123.12.2512
Kozasa, E. H., Sato, J. R., Lacerda, S. S., Barreiros, M. A. M., Radvany, J., Russell, T. A., Sanches, L. G., Mello, L. E. A. M., & Amaro, E. (2012). Meditation training increases brain efficiency in an attention task. NeuroImage, 59(1), 745–749. https://doi.org/10.1016/j.neuroimage.2011.06.088
Laukkonen, R. E., & Slagter, H. A. (2021). From many to (n)one: Meditation and the plasticity of the predictive mind. Neuroscience & Biobehavioral Reviews, 128, 199–217. https://doi.org/10.1016/j.neubiorev.2021.06.021
Lipkin, B., Tuckute, G., Affourtit, J., Small, H., Mineroff, Z., Kean, H., Jouravlev, O., Rakocevic, L., Pritchett, B., Siegelman, M., Hoeflin, C., Pongos, A., Blank, I. A., Struhl, M. K., Ivanova, A., Shannon, S., Sathe, A., Hoffmann, M., Nieto-Castañón, A., & Fedorenko, E. (2022). Probabilistic atlas for the language network based on precision fMRI data from >800 individuals. Scientific Data, 9, 529. https://doi.org/10.1038/s41597-022-01645-3
Lutz, A., Mattout, J., & Pagnoni, G. (2019). The epistemic and pragmatic value of non-action a predictive coding perspective on meditation. Current Opinion in Psychology, 28, 166–171. https://doi.org/10.1016/j.copsyc.2018.12.019
Margulies, D. S., Ghosh, S. S., Goulas, A., Falkiewicz, M., Huntenburg, J. M., Langs, G., Bezgin, G., Eickhoff, S. B., Castellanos, F. X., Petrides, M., Jefferies, E., & Smallwood, J. (2016). Situating the default-mode network along a principal gradient of macroscale cortical organization. Proceedings of the National Academy of Sciences of the United States of America, 113(44), 12574–12579. https://doi.org/10.1073/pnas.1608282113
Marino, M., Arcara, G., Porcaro, C., & Mantini, D. (2019). Hemodynamic correlates of electrophysiological activity in the default mode network. Frontiers in Neuroscience, 13, 1–12. https://doi.org/10.3389/fnins.2019.01060
McCarthy, H., Skokauskas, N., Mulligan, A., Donohoe, G., Mullins, D., Kelly, J., Johnson, K., Fagan, A., Gill, M., Meaney, J., & Frodl, T. (2013). Attention network hypoconnectivity with default and affective network hyperconnectivity in adults diagnosed with attention-deficit/hyperactivity disorder in childhood. JAMA Psychiatry, 70(12), 1329–1337. https://doi.org/10.1001/jamapsychiatry.2013.2174
Pagnoni, G. (2019). The contemplative exercise through the lenses of predictive processing: A promising approach. In Progress in Brain Research (Vol. 244, pp. 299–322). Elsevier. https://doi.org/10.1016/bs.pbr.2018.10.022
Power, J. D., Barnes, K. A., Snyder, A. Z., Schlaggar, B. L., & Petersen, S. E. (2012). Spurious but systematic correlations in functional connectivity MRI networks arise from subject motion. NeuroImage, 59(3), 2142–2154. https://doi.org/10.1016/j.neuroimage.2011.10.018
Rahrig, H., Vago, D. R., Passarelli, M. A., Auten, A., Lynn, N. A., & Brown, K. W. (2022). Meta-analytic evidence that mindfulness training alters resting state default mode network connectivity. Scientific Reports, 12(1), 12260. https://doi.org/10.1038/s41598-022-15195-6
Rosenberg, M. D., Finn, E. S., Scheinost, D., Papademetris, X., Shen, X., Constable, R. T., & Chun, M. M. (2015). A neuromarker of sustained attention from whole-brain functional connectivity. Nature Neuroscience, 19(1), 165–171. https://doi.org/10.1038/nn.4179
Ruck, L., & Schoenemann, P. T. (2021). Handedness measures for the Human Connectome Project: Implications for data analysis. Laterality, 26(5), 584–606. https://doi.org/10.1080/1357650X.2020.1866001
Santaella, D. F., Balardin, J. B., Afonso, R. F., Giorjiani, G. M., Sato, J. R., Lacerda, S. S., Amaro, E., Lazar, S., & Kozasa, E. H. (2019). Greater anteroposterior default mode network functional connectivity in long-term elderly yoga practitioners. Frontiers in Aging Neuroscience, 10, 1–7. https://doi.org/10.3389/fnagi.2019.00158
Schaefer, A., Kong, R., Gordon, E. M., Laumann, T. O., Zuo, X.-N., Holmes, A. J., Eickhoff, S. B., & Yeo, B. T. T. (2017). Local-global parcellation of the human cerebral cortex from intrinsic functional connectivity MRI. Cerebral Cortex, 28(9), 3095–3114. https://doi.org/10.1093/cercor/bhx179
Schrimpf, M., Blank, I. A., Tuckute, G., Kauf, C., Hosseini, E. A., Kanwisher, N., Tenenbaum, J. B., & Fedorenko, E. (2021). The neural architecture of language: Integrative modeling converges on predictive processing. Proceedings of the National Academy of Sciences, 118(45), e2105646118. https://doi.org/10.1073/pnas.2105646118
Schuman-Olivier, Z., Trombka, M., Lovas, D. A., Brewer, J. A., Vago, D. R., Gawande, R., Dunne, J. P., Lazar, S. W., Loucks, E. B., & Fulwiler, C. (2020). Mindfulness and behavior change. Harvard Review of Psychiatry, 28(6), 371–394. https://doi.org/10.1097/HRP.0000000000000277
Ségonne, F., Dale, A. M., Busa, E., Glessner, M., Salat, D., Hahn, H. K., & Fischl, B. (2004). A hybrid approach to the skull stripping problem in MRI. NeuroImage, 22(3), 1060–1075. https://doi.org/10.1016/j.neuroimage.2004.03.032
Seppälä, EM, Nitschke, JB, Tudorascu, DL, Hayes, A, Goldstein, MR, Nguyen, DT, Perlman, D, & Davidson, RJ (2014) Breathing-based meditation decreases posttraumatic stress disorder symptoms in U.S. military veterans A randomized controlled longitudinal study Journal of Traumatic Stress, 27(4), 397–405
Seabold, S., & Perktold, J. (2010). Statsmodels: Econometric and statistical modeling with python. In Proceedings of the 9th Python in Science Conference, SciPy 2010. https://doi.org/10.25080/majora-92bf1922-011
Sezer, I., Pizzagalli, D. A., & Sacchet, M. D. (2022). Resting-state fMRI functional connectivity and mindfulness in clinical and non-clinical contexts: A review and synthesis. Neuroscience & Biobehavioral Reviews, 135, 104583. https://doi.org/10.1016/j.neubiorev.2022.104583
Shankar, R. (2022). Patanjali Yoga Sutras: The heart of yoga (Vol. 1). Sri Sri Publications Trust.
Srinivasan, T. M. (2013). From meditation to dhyana. International Journal of Yoga, 6(1), 1–3. https://doi.org/10.4103/0973-6131.105934
Tripathi, V., & Bharadwaj, P. (2021). Neuroscience of the yogic theory of consciousness. Neuroscience of Consciousness, 7(2), 1–15.
Tripathi, V., & Somers, D. C. (2023). Default mode and dorsal attention network functional connectivity associated with alpha and beta peak frequency in individuals. biorXiv. https://doi.org/10.1101/2023.02.19.529136
Van Essen, D. C., Ugurbil, K., Auerbach, E., Barch, D., Behrens, T. E. J., Bucholz, R., Chang, A., Chen, L., Corbetta, M., Curtiss, S. W., Della Penna, S., Feinberg, D., Glasser, M. F., Harel, N., Heath, A. C., Larson-Prior, L., Marcus, D., Michalareas, G., Moeller, S., … Yacoub, E. (2012). The Human Connectome Project: A data acquisition perspective. NeuroImage, 62(4), 2222–2231. https://doi.org/10.1016/j.neuroimage.2012.02.018
Van Essen, D. C., Smith, S. M., Barch, D. M., Behrens, T. E. J., Yacoub, E., & Ugurbil, K. (2013). The WU-Minn Human Connectome Project: An overview. NeuroImage. https://doi.org/10.1016/j.neuroimage.2013.05.041
Virtanen, P., Gommers, R., Oliphant, T. E., Haberland, M., Reddy, T., Cournapeau, D., Burovski, E., Peterson, P., Weckesser, W., Bright, J., van der Walt, S. J., Brett, M., Wilson, J., Millman, K. J., Mayorov, N., Nelson, A. R. J., Jones, E., Kern, R., Larson, E., … Vázquez-Baeza, Y. (2020). SciPy 1.0: Fundamental algorithms for scientific computing in Python. Nature Methods, 17(3), 261–272. https://doi.org/10.1038/s41592-019-0686-2
Vivekananda, S. (2010). Patanjali Yoga Sutras. https://archive.org/details/PatanjaliYogaSutraBySwamiVivekananda/
Whitfield, T., Barnhofer, T., Acabchuk, R., Cohen, A., Lee, M., Schlosser, M., Arenaza-Urquijo, E. M., Böttcher, A., Britton, W., Coll-Padros, N., Collette, F., Chételat, G., Dautricourt, S., Demnitz-King, H., Dumais, T., Klimecki, O., Meiberth, D., Moulinet, I., Müller, T., & Marchant, N. L. (2022). The effect of mindfulness-based programs on cognitive function in adults: A systematic review and meta-analysis. Neuropsychology Review, 32(3), 677–702. https://doi.org/10.1007/s11065-021-09519-y
Yeo, B. T. T., Krienen, F. M., Sepulcre, J., Sabuncu, M. R., Lashkari, D., Hollinshead, M., Roffman, J. L., Smoller, J. W., Zöllei, L., Polimeni, J. R., Fischl, B., Liu, H., & Buckner, R. L. (2011). The organization of the human cerebral cortex estimated by intrinsic functional connectivity. Journal of Neurophysiology, 106(3), 1125–1165. https://doi.org/10.1152/jn.00338.2011
Young, K. S., Van Der Velden, A. M., Craske, M. G., Pallesen, K. J., Fjorback, L., Roepstorff, A., & Parsons, C. E. (2018). The impact of mindfulness-based interventions on brain activity: A systematic review of functional magnetic resonance imaging studies. Neuroscience & Biobehavioral Reviews, 84, 424–433. https://doi.org/10.1016/j.neubiorev.2017.08.003
Zhang, Z., Luh, W. M., Duan, W., Zhou, G. D., Weinschenk, G., Anderson, A. K., & Dai, W. (2021). Longitudinal effects of meditation on brain resting-state functional connectivity. Scientific Reports, 11, 11361. https://doi.org/10.1038/s41598-021-90729-y
Zheng, A., Montez, D. F., Marek, S., Gilmore, A. W., Newbold, D. J., Laumann, T. O., Kay, B. P., Seider, N. A., Van, A. N., Hampton, J. M., Alexopoulos, D., Schlaggar, B. L., Sylvester, C. M., Greene, D. J., Shimony, J. S., Nelson, S. M., Wig, G. S., Gratton, C., McDermott, K. B., & Dosenbach, N. U. F. (2021). Parallel hippocampal-parietal circuits for self- and goal-oriented processing. Proceedings of the National Academy of Sciences, 118(34), e2101743118. https://doi.org/10.1073/pnas.2101743118
Funding
This work was funded by National Science Foundation Graduate Research Fellowship DGE-1247312 and Ad Astra Chandaria Foundation (K.J.D.), and National Science Foundation Grant SMA-0835976 and National Institutes of Health grant R01-EY022229 (D.C.S.). No funding was received to assist with the preparation of this manuscript.
Author information
Authors and Affiliations
Contributions
Vaibhav Tripathi: conceptualization, methodology, software, formal analysis, investigation, writing—original draft, visualization, writing—review and editing; Kathryn J. Devaney: methodology, investigation, project administration, data curation, writing—review and editing; Sara W. Lazar: supervision, writing—review and editing; David C Somers: investigation, resources, writing—review and editing, supervision, funding acquisition.
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare no competing interests.
Ethics Approval
The study was approved by the Institutional Review Board of Boston University (1040E, 2734E) and conducted in accordance with the Declaration of Helsinki.
Informed Consent
Written informed consent was obtained from all subjects involved in the study.
Use of Artificial Intelligence
ChatGPT-4 was used to refactor a few paragraphs of the discussion with the prompt “Can you suggest alternative ways of writing ‘X’ ?” The authors reviewed and edited the content as needed and take full responsibility for the content of the publication.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Tripathi, V., Devaney, K.J., Lazar, S.W. et al. Silence Practice Modulates the Resting State Functional Connectivity of Language Network with Default Mode and Dorsal Attention Networks in Long-Term Meditators. Mindfulness 15, 665–674 (2024). https://doi.org/10.1007/s12671-024-02316-7
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12671-024-02316-7