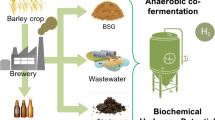
Abstract
Biohydrogen (bioH2) and soluble metabolites products (SMPs) obtention from the co-digestion process of banana processing wastewater (BPW) and synthetic sewage (SS) were investigated. The reactor performance was evaluated by BPW addition with different initial concentrations: 2.0, 5.0, and 9.6 g of total carbohydrate (TC)/L (pure BPW) using SS to complete the working volume. The dark fermentation process was carried out in a 1 L batch reactor operated at 37 °C/52 h and pH 7. The composition of gas and liquid samples (TC, VFAs, alcohols, and pH) were analyzed during reactor operation. The highest bioH2 production yield (210.82 ± 32.07 NmL/g TC) and bioH2 production rate (40.93 ± 7.60 NmL/g TC/h) were obtained at an initial substrate concentration of 2.0 g TC/L. These results indicated that the co-digestion improved carbohydrate utilization and induced a more effective metabolic pathway to bioH2 production. At this condition, the main soluble metabolite products were acetate and butyrate, whereupon Clostridiacae was the main family involved in BPW fermentation. The prediction of functional gene expression evidenced a shift in the mechanisms of SMPs and bio H2 obtention as the initial concentration of substrate changes.
Graphical Abstract

Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Statement of Novelty
Biohydrogen production from wastewater is still a developing area. The knowledge of the ideal fraction of BPW that can be used for energy production, and the approach of the changes in the microbial community dynamics is crucial to optimize systems that the bioH2 is the desired end-product.
Introduction
Hydrogen is a promising fuel source because of its environmentally friendly characteristics. It has a great capacity to reduce pollution, being applicable in various sectors like transport (hydrogen fuel cell), ammonia and methanol synthesis, and petroleum refineries [1, 2]. However, the traditional methods to produce hydrogen are fossil-fuel based, such as natural gas reforming, coal gasification, or partial oxidation of heavy hydrocarbon, which are unfriendly for sustainable development and require high energy input [3]. As an initiative to achieve sustainable production strategies, the nations have focused their interest on reducing the greenhouse effect and other environmental pollutant gas production by using new energy sources instead of fossil-based fuels [2]. In this context, several clean methods that can be used for hydrogen production without harming the environment have been reported in the literature, such as biological, electrical, photonic, and thermal processes [4].
Hydrogen gas obtention via biological systems (i.e. biophotolysis, photofermentation, and dark fermentation) was recently reviewed by Ramprakash et al. [5], and future perspectives and strategies were determined. The authors reported it as a great alternative due to advantages such as specific conversion, simple operations under balmy conditions, carbon neutrality, and a low cost compared to other production methods. Among them, dark fermentation (DF) has been consolidated as an appropriate method to obtain hydrogen because it requires a low external energy input to drive the reaction and the rate of hydrogen production is faster than biophotolysis and photofermentation [5, 6]. In this process, the organic substrates are used as an energy and electron source by microorganisms to produce the commonly known as “biohydrogen”, and other Soluble Metabolite Products (SMPs), such as volatile fatty acids (VFAs), acetone, and alcohols.
Different feedstocks such as agricultural, food, and dairy residues, glycerol, and domestic/industrial wastewaters were recently considered for biohydrogen production. The potential can be identified from the principle that an ideal feedstock must be rich in available carbohydrates [6]. Wastewater is considered a promising source of biohydrogen production due to its abundance and easy access considering the significant domestic and industrial activity. It is a rich source of various organic products, which are majorly soluble and biologically available [7]. According to Qadir et al. [8], the estimated global wastewater production is expected to increase up to 24% by 2030 and 51% by 2050 over the current level, which is around 380 billion m3 annually. Industries - e.g., fruit and food processing, pulp and paper, iron and steel, mining and petrochemical, etc. - are the top contributors to global wastewater generation and require extensive treatment before discharging wastewater safely to the environment. This treatment is motivated by regulations and legislations of the government to protect the environment and marine life from toxic pollutants and pathogens [7, 9]. Recently, studies reported the potential of biohydrogen production through dark fermentation using real wastewater discharged from various industries, such as cheese whey, confectionery wastewater, sugar beet processing wastewater [10], winery wastewater [11], beverage wastewater [12], lactate wastewater [13], cassava starch wastewater [14], among others.
The banana is one of the most important commercial subtropical fruits with global annual production exceeding 120 million tons [15]. Even though it is generally consumed in natura, several banana derivatives, such as candies, chips, dried fruit, jams, and wines, among others, have gained space in the market, which shows the great versatility and appreciation of this fruit and its processed derivatives [16]. Considering its expressive activity nowadays, the industry of banana fruit processing also needs a paradigm shift from wastewater disposal to wastewater utilization. There is a need to promote cleaner and more sustainable processing of fruit waste as well as to contribute to renewable energy insertion in the energetic matrix [17]. Furthermore, containing high content of carbohydrates (glucose, sucrose, and fructose) from the fruit, as well as added sugars in the derivatives elaboration process, BPW should be a potential source for bioH2 production by anaerobic fermentation.
To ensure that the dark fermentation process is viable and a preferred treatment or energy recovery, the evaluation of co-digestion in biohydrogen quantity and quality has been assessed in the open literature. This technique has gained space for promoting using more than one residue while compensating for biomasses’ nutrient limitations and enhancing biohydrogen production. Additionally, the co-digestion technique is useful to enable the dilution of potentially toxic compounds and to improve the synergistic reactions of microorganisms and the buffering capacity of the substrates [14, 18]. The benefits achieved by using at least two different co-substrates were demonstrated by García-Depraect et al. [19] in the co-digestion of vinasse and Nejayote. The results showed a maximum bioH2 production rate of 107 NmL H2/Lr h and bioH2 yield of 115 NmL H2/g TVSadded at a vinasse/Nejayote ratio of 80:20, a result of their mutual complementarity in the concentrations of nitrogen, iron, magnesium, phosphorus, and alkalinity. These co-digestion processes also showed a synergistic effect on bioH2 production for the mixture of fallen leaves and sewage sludge in the investigation conducted by Yang et al. [20]. The authors observed the optimal mixing ratio of leaves/sludge at 80:20, achieving a bioH2 production yield of 37.8 mL/gVSadded, emphasizing the advantages over mono-digestion of sludge (10.3 mL/gVSadded).
Even though different real wastewater treatment using the DF process is well documented in the open literature, as far as is known the potential of biohydrogen and intermediates production from BPW is lacking. Based on this, this study explores the potential and ideal fraction of BPW through co-digestion with Synthetic Sewage (SS) that can be used for energy purposes through bioH2 and soluble metabolites production, involving the microbial community investigation. This study specifically approaches the following questions: Does the initial substrate concentration affect the production yield of bioH2 and soluble metabolites? How does the microbial community change after the fermentation process and what mechanisms are behind the formation of the end products?
Materials and Methods
Substrates and Inoculum
BPW to feed the reactors was collected from a banana processing industry located in Tapiratiba, São Paulo, Brazil, during the process of washing the cooking containers to produce banana candy bars. The BPW was transported and stored in plastic bottles at − 18 °C until its utilization in the anaerobic batch reactors.
The synthetic domestic sewage (SS) to simulate real domestic wastewater was prepared as described by Martín et al. [21] (Supplementary Table 1 of Online Resource 1). The anaerobic consortium of H2-producing bacteria was obtained from a UASB (Upflow Anaerobic Sludge Blanket) reactor used in the treatment of poultry wastes (Tietê, São Paulo, Brazil). The granular sludge was subjected to a thermal pretreatment (100 ºC for 15 min) to inhibit hydrogen-consuming microorganisms and obtain endospore-forming anaerobic bacteria [22]. Table 1 shows the characteristics of the BPW, SS, and the anaerobic sludge pretreated (AS-PT) used in the experiments:
Experimental Set-up
To determine the effect of the initial concentration of wastewater on the DF process, batch fermentation tests were carried out for 52 h in anaerobic batch reactors of 1.0 L (working volume of 0.6 L) containing 20% (v/v) of inoculum (120 mL) and different initial concentrations of BPW, according to Table 2. Endogenous control was measured using only deionized water and inoculum. The initial pH of each reactor was adjusted to 7.0, and sodium bicarbonate (5 g/L) was added as a buffer. Nitrogen gas (99.9%) was fluxioned in each reactor for 5 min to remove the remaining oxygen and promote anaerobic conditions. After that, these were capped with bottle rubber stoppers, wrapped, and incubated without stirring at 37 ºC. The substrate-to-inoculum ratio (S/I) was calculated as the ratio of the volume of BPW to the volume of the inoculum. To ensure the reproducibility of experiments, all assays were done in triplicate.
Analytical Methods
Liquid samples and biogas fractions were collected and analyzed periodically during the reactor’s operation. The amount of gas produced in the reactors was measured by the water displacement method [23]. To liquid samples, TC, pH, and concentration of fermentation products—volatile fatty acids (VFA), acetone, and alcohols—were measured. The determination of TC concentration (measured as glucose) was conducted by the phenol method proposed by Herbert et al. [24]. The pH was determined using a Jenway 3510 pH meter equipped with an electrode. The percentage composition of the biogas (H2, CH4, and CO2) was determined by a gas chromatograph (GC-2014, Shimadzu®) coupled with an using a thermal conductivity detector (TCD) and Carboxen® #1010 PLOT (30 m X 0.53 mm) column, using Argon as a gas carrier. Injector and detector temperatures were set at 220 and 230 °C, respectively. Column temperature ramp was 120 °C (Hold time 1 min), 40 °C/min up to 200 °C (Hold time 3 min), and 50 °C/min up to 230 °C (Hold time 0.5 min) [25]. The concentration of some fermentation products generated in the reactors, such as acetic acid (HAc), butyric acid (HBu), propionic acid (HPr), isobutyric acid (HIsob), isovaleric acid (HIsov), ethanol and methanol, was determined by a gas chromatograph (GC-2030, Shimadzu®), equipped with a flame ionization detector (FID) with an autosampler (AOC 6000 plus), a capillary column HP-INNOWAX (30 m x 0.250 mm x 0.25 μm), and using Nitrogen as the gas carrier [26]. High-Performance Liquid Chromatography (HPLC) was used for the identification and quantification of lactic acid in the pre-filtered samples (0.22 μm pore size filter). The refractive index detector (Waters 2014) was maintained at 40 °C. The analytical column Aminex® HPX-87 H (300 × 7.8 mm) was held at 50 °C with a flow rate of 0.6 mL/min, using sulfuric acid 0.005 mol/L as a mobile phase. The stoichiometric COD factors assumed for the determination of total Soluble Metabolites Products (SMPs) were: 1.24 g COD/glactic_acid, 2.087 g COD/gethanol, 1.066 g COD/gacetic_acid, 1.514 g COD/gpropionic_acid, 1.818 g COD/gbutyric_acid and 2.036 g COD/giso−valeric_acid [27].
The TS, VS, and ashes content in BPW, SS, and AS-PT were determined by gravimetric method, according to APHA standard procedures. COD analysis was conducted on samples collected at the beginning and at the end of the dark fermentation process, on the pure BPW, and on SS [28].
Microbial Community Analysis
The biomass collected before the DF process (AS-PT) and at the end of the assays, separately, was stored at − 80 °C in a 50% glycerin solution, and used for DNA extraction, performed according to Griffiths et al. [29], with adaptations.
The 16 S rRNA amplicon sequencing was performed on Illumina NovaSeq6000 PE 250 platform by ByMyCell (Ribeirão Preto, São Paulo, Brazil). The bacterial 16 S rRNA genes V3–V4 regions were amplified from the microbial genomic DNA using specific primers (515 F/806 R). The taxonomic classification was performed using QIIME (Quantitative insight into microbial ecology) v2022.2.0 [30]. The raw sequencing data had their quality metrics measured at the beginning of the analysis process and reads of unsatisfactory length and chimeras were removed. The curated reads were used in the definition of ASV (Amplicon Sequence Variant) with similarity ≥ 97%, then the α-diversity indexes were calculated. SILVA SSU 138 rRNA Database Project [31] was used for taxonomic assignment.
Metabolism prediction to elucidate the impact of different initial substrate concentrations was done using Phylogenetic Investigation of Communities by Reconstruction of Unobserved States (PICRUSt v2.4.2) based on 16 S rRNA sequencing data. Metagenome predictions were made by corresponding the marker gene data and the reference genomes in the Kyoto Encyclopedia of Genes and Genomes (KEGG) database [32].
The raw sequences were submitted to the NCBI (National Center for Biotechnology Information) SRA database under accession numbers SRR22408487, SRR22408621, and SRR22408622 for the assays using the different concentrations 9.6, 5.0, and 2.0 g TC/L, respectively, and SRR22408407 for the anaerobic granular sludge (BioProject PRJNA643936).
Statistical Analysis and Kinetic Parameters of Hydrogen Production
The experimental data were fitted using the software Statistica 10.0. The average of the hydrogen yield evolution data was adjusted to the modified Gompertz model (Eq. 1) [33].

where P is the BioH2 production potential (NmL/g TC), Rm is the maximum production rate (NmL/g TC/h), t is the incubation time of the reactors (h), ʎ is the phase before BioH2 production starts (h), and e = 2.718. Biohydrogen yields (Y) were calculated by dividing the cumulative hydrogen production potential by the amount of TC and VS added. The experimental data are tested by analysis of variance (ANOVA) and Tukey test (p < 0.05), considering the completely randomized design with three treatments, using the software Microsoft Excel 365 for Windows 11 to assess significant differences among various parameters and expressed as mean values ± SD. Pearson correlation (p < 0.05) analysis was performed to identify significant correlations between DF performance and the microbial community using Jamovi 2.3.18.
Results and Discussion
Hydrogen Production from BPW
The effects of BPW concentration were evaluated on dark fermentation during 52 h, the results obtained are summarized in Table 3. It’s known that the biohydrogen yield is primarily affected by the content of carbohydrates present in the substrate. Overall, all assays presented a high total carbohydrates removal (83.20–93.66%), resulting in different yields of biohydrogen production, where the highest yield (p < 0.01) of biohydrogen (210.27 ± 32.40 NmL/g TC) from BPW was observed at the lowest initial substrate concentration (2 g TC/L). With the initial total carbohydrate content increase to 5.0 and 9.6 g TC/L, the values decreased to 78.22 ± 7.12 and 56.83 ± 5.32 NmL/g TC, respectively. The same behavior was observed in the lower substrate concentration offered with a significant difference (p < 0.01) in the bioH2 content, in the amount of biohydrogen per gram of VS added, per mol of TC, and per liter of BPW, showing the maximum values of 8.82 ± 0.02%, 155.10 ± 23.90 NmL H2 /g VS, 1.56 ± 0.23 mol H2 /mol TC, and 1538.51 ± 314.42 NmL/L, respectively. No methane gas was detected in fermentation reactors, indicating the effectiveness of the thermal pretreatment of the inoculum (Table 3). In general, although the low substrate concentrations have favored the bioH2 content in the produced biogas, the value obtained in this condition was significantly low. BioH2 contents close to or even superior than 40% have been previously reported in the literature [10, 12, 13]. Many factors might have significant effects on bioH2 production, such as the adjustment of operational parameters (e.g., pH, temperature, S/I, among others), that still need to be studied and were not covered by the scope of the present investigation.
The reactors fed with higher substrate concentration decreased pH (p < 0.01), as seen for the initial concentration of 9.6 g TC/L, resulting in a final pH of 5.8 ± 0.04. In comparison, for concentrations 5.0 and 2.0 g TC/L, final pH of 6.0 ± 0.10 and 6.6 ± 0.02 were observed (Fig. 1). These observations suggest that the accumulation of soluble metabolites in the liquid phase - mainly the acidic products—had the greatest effect on the lowering of pH during DF [13]. Moreover, the highest cumulative biohydrogen of 362.66 ± 29.68 NmL (p < 0.01) was observed at the highest initial substrate concentration (9.6 g TC/L) while lower values of cumulative production were observed as the concentration of BPW decreased to 5.0 and 2.0 g TC/L, obtaining 236.70 ± 21.83 and 158.00 ± 23.36 NmL of biohydrogen, respectively. The pH variation and cumulative bioH2 production are represented in Fig. 1.
The drop in production yields as the initial concentration increases (Table 3) may be related to the accumulation of liquid products with subsequent over-acidifying of bacterial cultures (mostly hydrogen producers). It could be caused by an excessive substrate addiction during the fermentation process [34,35,36,37,38], as seen in the assay 9.6 g TC/L, demonstrating a significant decrease in pH at the end of operational time.
The efficiency of hydrogen production during the fermentation of wastewater is strongly influenced by the waste concentration and by the nature of the substrate, which some studies evidence high yields of biohydrogen production from wastewater at lower substrate concentrations (lower than 40 g COD/L) [39]. Fangkum & Reungsang [34] also tested the effect of initial total carbohydrate (5–40 g TC/L) in DF of sugarcane bagasse hydrolysate. The study demonstrated that the hydrogen production yield gradually declined at concentrations greater than 10 g TC/L, achieving the maximum H2 production yield of 0.84 mol H2/mol of TCconsumed at the initial concentration of 10 g TC/L. In the work of Sivaramakrishna et al. [35], the hydrogen yield from probiotic wastewater (PW) under mesophilic conditions strongly depended on the initial substrate concentration. Production yield, similar to the one obtained in the present study (1.8 mol bioH2/mol TC) was achieved at a substrate concentration of 5 g/L and in the range of 2–8 gPW/L using mixed anaerobic consortia.
The performance of the DF process also can be affected by the S/I ratio. According to the work of Litti et al. (2022) [10], the optimal S/I in terms of H2 yield and H2 production from the fermentation of cheese whey in anaerobic batch reactors were obtained at the range of 0.5–1.0. This result explains the representative yield of biohydrogen production at the lower offered concentration, corresponding to an S/I of 0.8 (Table 3).
A low degree of COD removal after DF of BPW was also verified (4.66–18.94%). The result could be related to the primary organic matter being converted mostly into organic acids and alcohols due to partial anaerobic digestion. Consequently, hydrogen makes up only a small part of the COD balance of end products of dark fermentation [40].
The modified Gompertz model was used to describe the progress of the batch fermentative hydrogen production from BPW. The fitted model in comparison with the experimental data was plotted in Fig. 2, while Table 4 lists the fitted parameters. The correlation coefficients were 0.99 for all assays, indicating the reliability of the values obtained. The maximum hydrogen production yield (210.82 ± 32.07 NmL/gTC) and the maximum production rate (40.93 ± 7.60 NmL/gTC/h) were achieved using the lower concentration of BPW (2.0 g TC/L), at a significance level of p < 0.01, indicating the efficiency of the co-digestion with SS. A lower value of lag phase (ʎ) was observed in the assay 5.0 g TC/L (p < 0.001) indicating that this kinetic parameter can be affected by changes in different initial concentrations.
Soluble Metabolites Production
Biohydrogen production is accompanied by the formation of different SMPs during the fermentative process. The SMPs concentration and distribution are useful indicators for monitoring the gas production by predicting the metabolic pathways assumed by the anaerobic consortium involved. It is possible to mainly observe the presence of the metabolites lactic acid (HLa), acetic acid (HAc), butyric acid (HBu), ethanol, and methanol at the beginning of the reactor operation. This is probably attributed to the natural fermentation of the residue. The presence of indigenous microorganisms can be originally present in the substrate or proliferate during storage and transportation [41]. It was previously correlated to high amounts of lactate and acetate in the substrates by different studies, e.g. cassava starch wastewater [42] and sugarcane molasses [43]. Only propionic, isobutyric, isovaleric, and caproic acid traces were detected after each run (Fig. 3).
Overall, it was observed that as the offered substrate concentration increased from 2.0 to 9.6 g TC/L, there was an increased formation of HBu. In contrast, for decreasing concentration, there was an increased formation of HAc. Table 5 summarizes the final concentration of the main SMPs. In the first 10 h, HLa concentration in the assays 9.6 and 5.0 g TC/L increased considerably, achieving concentrations of 4.84 and 1.2 g COD/L, which corresponds to a portion of 62% and 42% of total SMPs, respectively (Supplementary Fig. 1 of Online Resource 1). For the highest concentration of 9.6 g TC/L (Fig. 3a), the HBu had the higher abundance at the end of the fermentation, corresponding to 43% of the total SMPs, followed by ethanol (25%), and HAc (24%). The amount of HBu, ethanol, and HAc produced was 2.09 ± 0.93, 1.22 ± 0.56 g COD/L, and 1.15 ± 0.79 g COD/L, respectively at the end of fermentation. Interestingly, mainly the HBu level increased continuously with a reduction of HLa at the rapid phase ranging from 10 to 52 h, coinciding with the exponential phase of hydrogen production (Fig. 2), suggesting that HBu and bioH2 were generated from lactate consumption during this stage.
As demonstrated by Matsumoto & Nishimura [44], several species of microorganisms from the Clostridium genus have been associated with the production of HBu and BioH2 from HLa, for example, Clostridium butyricum, C. diolisJPCC H3, and C. beijerinckii. Furthermore, Kim et al. [45] reported the positive effect of HLa in a bioH2-producing system. The authors reported that HBu made up the greatest portion of all metabolites produced when the initial concentration was 8 g HLa/L, which also promoted the highest hydrogen yield. Considering the significant production of HBu feeding the reactor 9.6 g TC/L, the predominant pathway assumed was the butyric acid route. In the butyrate-type fermentation, only 2 mols of hydrogen are produced when butyrate is the main fermentation product.
Several researchers[34,35,36,37,38] suggest that excessive addition of substrate concentration results in the generation of too high partial pressure of hydrogen in the headspace. This fact can inhibit further hydrogen formation and decrease the specific yield of hydrogen due to the occurrence of a solvent production pathway, resulting in enhanced alcohol production. In the present study, it was verified the maximum final concentration of ethanol (p < 0.05) in the assay fed with 9.6 g TC/L (1.22 ± 0.56 g COD/L), confirms the solvent pathway.
In assay 2.0 g TC/L there was a marked formation of acetic acid during the operation time, which at the end of the reaction corresponded to a portion of 61% of the total soluble metabolites formed, achieving the final concentration of 1.63 ± 0.27 g COD/L (Fig. 3c). In this assay, the strong predominance of acetic acid formation explains the high hydrogen yield and content in biogas since the acetic acid pathway is more favorable to hydrogen production. Theoretically, 4 mols of hydrogen are produced from 1 mol of glucose in acetate-type fermentation.
As the substrate concentration offered decreased to 5.0 and 2.0 g TC/L, it was verified that, in the interval from 12 to 52 h of operation, a decrease in the concentration of ethanol and an increase in HAc and BioH2 production occurred, achieving final concentration of 0.37 ± 0.25 and 0.04 ± 0.01 g COD/L, respectively. These results may characterize a metabolic strategy used by bacteria to obtain energy at low carbohydrate availability conditions using ethanol fermentation as an alternative pathway [46]. Similar results were obtained by Villa Montoya et al. [47] in the co-digestion of coffee waste (coffee pulp, husk, and processing wastewater) at similar conditions to this study (neutral pH and mesophilic conditions). The study reported an increase in HAc generation and a decrease in the concentration of other organic acids and alcohols (mainly ethanol) at the end of fermentation. These results suggested that the formation of ethanol without bioH2 production occurred during the first hours of fermentation, a thermodynamically favorable reaction in neutral pHs, with the subsequent consumption of ethanol into HAc and bioH2 by the microorganisms through acetogenesis, considering the high concentrations of this volatile fatty acid.
Taxonomic Profile and Prediction of Functional Genes Expression
The microbial diversity in an evaluated sample is commonly estimated by the Alpha diversity indexes. The Shannon index demonstrates the measure of diversity within the sample, then a higher value of this index means higher microbial diversity in a sample [20]. Microbial diversity data was shown in Table 6. Higher diversity and richness of the microbial communities were verified in the granular sludge used as inoculum (7.96), followed by the reactor fed with the initial substrate concentration of 2 g TC/L (7.29). Microbial diversity tended to decrease as substrate concentration increased to 9.6 g TC/L (6.67).
Figure 4 illustrates the taxonomic profile of microbial communities for the samples from the assays at different initial concentrations of BPW after 52 h of operation and the inoculum
At the phylum level (Fig. 4a), the microbial community of the inoculum sample was relatively diverse with an abundance of 20 phyla higher than 1%, including mainly the phyla Bacteroidota (27.38%), Patescibacteria (10.87%), Firmicutes (10.55%), Cloacimonadota (10.18%), Desulfobacterota (6.60%), and Chloroflexi (5.63%). After 52 h of fermentation, all assays were dominated by the phylum Firmicutes (with relative abundance ranging from 58.43 to 63.12%). The prevalence of the phylum Firmicutes might be related to its ability to form endospores to resistant extreme environmental conditions, which promotes a better ability to recover their activity after the heat-shock pretreatment compared with other remaining phyla [48]. Furthermore, the phylum Firmicutes includes the most hydrogen-producing microbes, which can utilize a wide range of substrates for their growth, even including carbohydrates and proteins [49]. This explains the considerable growth with the addition of BPW.
At the genus level, it can be seen from Fig. 4b that there were 19 genera with relative abundance higher than 1% in the inoculum. The most dominant genus was Bacteroidetes_vadinHA17 (9.29%), among others such as Lentimicrobium (6.86%), SAR324 (4.03%), and Christensenellacae R7 group (3.24%). After 52 h of fermentation in each reactor fed with different initial substrate concentrations, changes in microbial structures and dominant species in the fermentation systems were observed as a result of a decrease in pH due to the soluble metabolites formation and the acclimation of some indigenous microorganisms, mainly from the Clostridium genus, which may be associated with the consumption of the total carbohydrates contained in the BPW and the HLa generated at the beginning of DF process of each reactor.
To assay 2.0 g TC/L, the microbial community demonstrated high diversity, indicating a synergistic activity of various microorganisms on the DF process. The most dominant genus was Clostridium sensu stricto 2 (5.51%) followed by Lactococcus (5.58%), Clostridium sensu stricto 1 (5.36%), Clostridium sensu stricto 10 (4.07%), and Clostridium sensu stricto 13 (4.01%). The occurrence of lactate-producing bacteria (e.g. Lactococcus, Lactobacillus, Streptococcus, among others) in DF systems has been widely discussed in an attempt to understand the effects of these microorganisms on bioH2 production. In some studies it has shown a detrimental effect, while in others it was observed that lactate improves bioH2 production [12]. The main reason underlying this discrepancy still remains unknown. However, the presence of these microorganisms associated with higher bioH2 production yield suggests that hydrogen-producing bacteria and lactate-producing bacteria were able to cohabit under specific conditions without apparently leading to negative impacts on hydrogen production. In addition, studies have demonstrated that lactate-based DF processes have been favored under carbohydrate-limiting conditions [50], which justifies the high yield of bioH2 production to the tested condition of lower initial substrate concentration. Furthermore, this successful hydrogen production via lactate-type fermentation represents an opportunity to achieve practical DF processes without the need for prior substrate sterilization and pretreatment of inoculum and/or substrate [41].
As the initial substrate concentration offered increased, the microbial community was dominated by the representative of the genus Clostridium sensu stricto 1. When the initial concentration of substrate increased to 5.0 g TC/L, the most dominant genus was Clostridium sensu stricto 1 (15.67%), followed by Terrisporobacter (9.10%), Clostridium sensu stricto 13 (7.62%), and Clostridium sensu stricto 2 (5.85%). At an initial concentration of 9.6 g TC/L, Clostridium sensu stricto 1 (27.65%) became the most dominant genus, followed by Paraclostridium (5.43%), Terrisporobacter (5.10%), Clostridium sensu stricto 2 (3.56%).
The genera Clostridium sensu stricto 1, Clostridium sensu stricto 13, Clostridium sensu stricto 2, Clostridium sensu stricto 7, Clostridium sensu stricto 10, and Lactococcus were not found in the inoculum. These indigenous microorganisms, probably coming from the non-sterile BPW, were favored to grow in the environmental conditions imposed such as pH, temperature, and substrate availability.
To better understand the functions of individual microbial groups in dark fermentation of BPW, possible relationships between main characteristic genera and kinetic parameters of bioH2 production (final pH, H2 in biogas, yield, S/I, ethanol, HAc, HBu, and metabolites production) were evaluated by the Pearson correlation analysis. Only two correlations exhibited significatively data: the relative abundance of Clostridium sensu stricto 1 was correlated negatively with the percentage of H2 in biogas (r = − 0.99, p < 0.05), meanwhile, the relative abundance of the Clostridium sensu stricto 13 genus was correlated positively with the acetate production via fermentation (r = 0.99, p < 0.05) Fig. 5.
Clostridium sensu stricto 1 is a widely reported hydrogen-producing genus, which is capable of using different feedstocks (e.g. sewage sludge, food waste, lignocellulosic biomass, sucrose, starch, hemicellulose, glucose, and cellulose) for bioH2 production, and the typical species of this genus is Clostridium butyricum [50]. Although a positive correlation between biohydrogen productivity and the genus Clostridium sensu stricto has been reported [50], in the present work the abundance of this genus resulted in a decrease of the bioH2 presence in the biogas, accompanied by a low yield of bioH2 and production of mostly HBu (Table 3; Figs. 2 and 3). This behavior may be explained by a change in Clostridial-type hydrogen producing fermentation mechanism, as described by Hallenbeck study: these microorganisms are potentially capable, under the right conditions, of producing additional hydrogen by reoxidizing the NADH generated during glycolysis. Since two moles of NADH are produced during glycolysis, up to a maximum of two additional molecules of H2 could potentially be generated by this pathway. Thus, these organisms appear to be capable in theory of producing up to 4H2/glucose. However, proton reduction with NADH is unfavorable since the redox potential of hydrogen (∼450 mV at neutral pH, STP) is appreciably lower than that of the NADH/NAD couple (∼320 mV). Thus, the production of H2 with electrons derived from NADH is only possible at greatly reduced hydrogen partial pressure. At moderate to high hydrogen partial pressures (favored at high substrate concentrations), the inability to reoxidize NADH leads to its oxidation by pathways that produce reduced organic compounds at the expense of hydrogen, commonly butyric acid [51].
The prediction of encoding genes related to key enzymes involved in the BPW fermentation might suggest the presence of particular pathways helping to elucidate the process. PICRUSt analysis was further performed to reveal the metabolism prediction and assess the impact of the different initial concentrations of BPW on the expression of functional genes encoding the enzymes closely related to the formation of bioH2 and SMPs. Thus, KEGG database-based PICRUSt analysis was adopted in this study to give more comprehensive information concerning the microbial functional gene expressions. Figure 6 describes 19 key enzymes encoded by correlated genes involved in bioH2 production and direct and indirect SMPs formation during BPW fermentation at different initial concentrations, according to functional orthologs from KEGG orthology (K)).
In the inoculum is possible to observe the predominance of 6-phosphofructokinase (73.79%) and phosphotransbutyrylase (26.20%), which was drastically reduced at the assays since they are conducted after the inoculum pretreatment, whereupon most microbes are inhibited due de unfavorable conditions. The changes in the mechanisms of VFAs and bioH2 production after the pretreatment might be related to the fermentation conditions that selectively enriched the microbes favorable for the environment, directly influencing the tolerance of different genes to pretreatment, determining which one will be recovered [52].
Regarding the assay 2.0 g TC/L, Hac—the major soluble metabolite produced—proceeds through Acetate-CoA ligase (4.73%), responsible for the conversion of Acetyl-CoA into acetate. Moreover, the acetyl-CoA produced can be converted to acetate with concomitant ATP synthesis from the acetyl-phosphate intermediate, generated by Phosphotransacetylase (5.51%). Butyrogenesis proceeds through butyryl-CoA generation from Acetyl-CoA via the intermediates acetoacetyl-CoA, hydroxybutyryl-CoA and crotonyl-CoA, both converted by acetyl-CoA-acetyltranferase (10.36%), 3-hydroxybutyryl-CoA dehydrogenase (5.56%) and 3-hydroxybutyryl-CoA dehydratase (3.70%), respectively. The presence of phosphotransbutyrylase (33.70%) and butyrate kinase (5.03%) completes the conversion into butyrate. Alcohol dehydrogenase was also observed at the abundance of 11.80% and might be related to the consumption of ethanol at the end of the assay. A proportion of glucokinase (9.72%) and pyruvate kinase (9.84%) played a relatively major role in bioH2 production.
The fermentation process at higher initial concentrations demonstrates some changes in the mechanism of the obtainment of the DF products. For the assay 5.0 g TC/L, the bioH2 production was mainly related to the activity of ferredoxin hydrogenase (6.58%), which drives the hydrogen evolution through the acceptance of reduced ferredoxin produced during the conversion of pyruvate into acetyl-CoA and CO2. It’s possible to observe the butyrate production proceeding directly via butyrate kinase (13.38%), starting from de precursors acetyl-CoA-acetyltransferase (20.75%), 3-hydroxybutyryl-CoA dehydratase (14.06%), butyryl-CoA dehydrogenase (8.53%) and butyrate kinase (13.38%), and also infer the indirect conversion of butyrate by butyryl-CoA:acetate-CoA transferase, which converts external acetate present in the medium into butyrate. The relevant presence of alcohol dehydrogenase (26.61%) might be related to ethanol consumption during the last hours of the fermentation process.
Regarding the assay 9.6 g TC/L, is possible to infer the direct production of HBu via the precursors Acetyl-CoA-acetyltransferase (10.33%), 3-hydroxybutyryl-CoA dehydratase (13.99%), which converts 3-hydroxybutyryl-CoA into Crotonyl-CoA, that enables the formation of Butyryl-CoA, used by Phosphotransbutyrylase (4.45%) to generates Butyrate-phosphate, the main component used to butyrate formation via Butyrate kinase (even expressed at low abundance). At the same time, the direct conversion of acetate by the key enzymes involved in acetate production and consumption, Phosphotransacetylase (5.81%) and Acetate kinase (6.16%), through the intermediate Acetyl-phosphate, was assumed. Alcohol dehydrogenase was also identified (11.98%), and this enzyme has the ability to convert Acetyl-CoA into acetaldehyde, which will be converted into ethanol while reoxidizing NADH to provide the NAD+ needed by the glycolytic pathway for further substrate utilization [51]. Because of the presence of Glucokinase (10.88%), Pyruvate kinase (9.59%), 6-phosphofructokinase (13.72%), it can be inferred that the abundance of these enzymes related to the breakdown of carbohydrates was associated with the initial concentration of substrate offered, indicating the adaption of microorganisms to good performance on carbohydrate utilization, also being probably the main pathway to produce bioH2. These enzymes are implicated in the formation of pyruvate, strongly involved in hydrogen production, especially in clostridial-type fermentation, and this is in accordance with the predominance of the family Clostridiacae in the assay 9.6 g TC/L [53].
Both lactate and ethanol-forming processes are accompanied by the conversion of NADH to NAD+. As hydrogen can be produced from NADH by hydrogenases, NADH-consuming metabolic pathways are considered restrictive for hydrogen production [52]. In this work, key enzymes related to hydrogen-consuming pathways such as D-lactate dehydrogenase and L-lactate dehydrogenase weren’t observed at considerable relative abundances. In contrast, the significant formation of ethanol associated with the strong relative abundance of alcohol dehydrogenase in the assay 9.6 g TC/L makes clear the limited bioH2 yield.
Conclusions
This study demonstrates that the initial substrate concentration effectively affected bioH2 and SMPs production. The highest bioH2 production yield (210.82 ± 32.07 NmL/g TC) and bioH2 production rate (40.93 ± 7.60 NmL/g TC/h) were obtained at the initial substrate concentration of 2.0 g TC/L. The main SMPs produced in the assays were acetate, butyrate, and ethanol. However, a shift in the metabolic pathway was noted as the initial substrate concentrations changed from 2.0 g TC/L to 9.6 g TC/L, whereupon the acetate pathway was followed at low BPW concentration. In contrast, the butyrate pathway was followed at a higher BPW concentration. Although the data obtained in the present study are promising for bioH2 production from BPW, low percentages of hydrogen were observed in biogas produced. Thus, further investigation on other variables that influence the fermentation process is still needed aiming to optimize the system. Moreover, future research directions should take into account assessing the continuous operation of the system.
The predictive functional profiling of microbial communities revealed the changes in the mechanisms of DF products formation and the dynamic of dominant genera as the initial substrate concentration changes, making the microbial analysis more comprehensive and elucidating the reasons for the changed metabolites formation. Synergistic activity of various microorganisms was observed at low initial concentrations, such as Clostridium sensu stricto 2, Lactococcus, and Clostridium sensu stricto 1, whereas the last one became dominant at high substrate concentrations. To the initial concentration of substrate with the best performance on DF, genes related to the breakdown of carbohydrates, and production of bioH2 and acetate, such as Glucokinase, Puyruvate kinase, and Phosphotransacetylase, respectively, were identified. The described process proved to be a sustainable way to treat unexplored wastewater towards a circular economy, recovering energy and contributing to the reduction of greenhouse gas emissions, boosting the development of the banana production and processing chain.
Data Availability
The data that support this paper are available from the corresponding author upon request from readers.
References
Tak, S.S., Shetye, O., Muley, O., Jaiswal, H., Malik, S.N.: Emerging technologies for hydrogen production from wastewater. Int. J. Hydrogen Energy (2022). https://doi.org/10.1016/J.IJHYDENE.2022.06.225
Qyyum, M.A., Ihsanullah, I., Ahmad, R., Ismail, S., Khan, A., Nizami, A.S., Tawfik, A.: Biohydrogen production from real industrial wastewater: Potential bioreactors, challenges in commercialization and future directions. Int. J. Hydrogen Energy (2022). https://doi.org/10.1016/J.IJHYDENE.2022.01.195
Mei, D., Qiu, X., Liu, H., Wu, Q., Yu, S., Xu, L., Zuo, T., Wang, Y.: Progress on methanol reforming technologies for highly efficient hydrogen production and applications. Int. J. Hydrogen Energy (2022). https://doi.org/10.1016/J.IJHYDENE.2022.08.134
Acar, C., Dincer, I.: Review and evaluation of hydrogen production options for better environment. J. Clean Prod. 218, 835–849 (2019). https://doi.org/10.1016/J.JCLEPRO.2019.02.046
Ramprakash, B., Lindblad, P., Eaton-Rye, J.J., Incharoensakdi, A.: Current strategies and future perspectives in biological hydrogen production: a review. Renew. Sustain. Energy Rev. 168, 112773 (2022). https://doi.org/10.1016/J.RSER.2022.112773
Sivaramakrishnan, R., Shanmugam, S., Sekar, M., Mathimani, T., Incharoensakdi, A., Kim, S.H., Parthiban, A., Edwin Geo, V., Brindhadevi, K., Pugazhendhi, A.: Insights on biological hydrogen production routes and potential microorganisms for high hydrogen yield. Fuel 291, 120136 (2021). https://doi.org/10.1016/J.FUEL.2021.120136
Aydin, M.I., Karaca, A.E., Qureshy, A.M.M.I., Dincer, I.: A comparative review on clean hydrogen production from wastewaters. J. Environ. Manag. 279, 111793 (2021). https://doi.org/10.1016/J.JENVMAN.2020.111793
Qadir, M., Drechsel, P., Jiménez Cisneros, B., Kim, Y., Pramanik, A., Mehta, P., Olaniyan, O.: Global and regional potential of wastewater as a water, nutrient and energy source. Nat. Resour. Forum. 44, 40–51 (2020). https://doi.org/10.1111/1477-8947.12187
Caligan, C.J.A., Garcia, M.M.S., Mitra, J.L., San Juan, J.L.G.: Multi-objective optimization for a wastewater treatment plant and sludge-to-energy network. J. Clean Prod. 368, 133047 (2022). https://doi.org/10.1016/J.JCLEPRO.2022.133047
Litti, Y.V., Potekhina, M.A., Zhuravleva, E.A., Vishnyakova, A.V., Gruzdev, D.S., Kovalev, A.A., Kovalev, D.A., Katraeva, I.V., Parshina, S.N.: Dark fermentative hydrogen production from simple sugars and various wastewaters by a newly isolated thermoanaerobacterium thermosaccharolyticum SP-H2. Int. J. Hydrogen Energy (2022). https://doi.org/10.1016/J.IJHYDENE.2022.05.235
Policastro, G., Carraturo, F., Compagnone, M., Guida, M., Fabbricino, M.: Enhancing hydrogen production from winery wastewater through fermentative microbial culture selection. Bioresour. Technol. Rep. (2022). https://doi.org/10.1016/j.biteb.2022.101196
Alvarez, A.J., Fuentes, K.L., Arias, A., Chaparro, C.: Production of hydrogen from beverage wastewater by dark fermentation in an internal circulation reactor: effect on pH and hydraulic retention time. Energy Convers. Manag. X. (2022). https://doi.org/10.1016/J.ECMX.2022.100232
Ziara, R.M.M., Miller, D.N., Subbiah, J., Dvorak, B.I.: Lactate wastewater dark fermentation: the effect of temperature and initial pH on biohydrogen production and microbial community. Int. J. Hydrogen Energy 44, 661–673 (2019). https://doi.org/10.1016/j.ijhydene.2018.11.045
Wadjeam, P., Reungsang, A., Imai, T., Plangklang, P.: Co-digestion of cassava starch wastewater with buffalo dung for bio-hydrogen production. Int. J. Hydrogen Energy 44, 14694–14706 (2019). https://doi.org/10.1016/j.ijhydene.2019.04.138
Manhongo, T.T., Chimphango, A.F.A., Thornley, P., Röder, M.: Current status and opportunities for fruit processing waste biorefineries. Renew. Sustain. Energy Rev. (2022). https://doi.org/10.1016/j.rser.2021.111823
Aurore, G., Parfait, B., Fahrasmane, L.: Bananas, raw materials for making processed food products. Trends Food Sci. Technol. 20, 78–91 (2009). https://doi.org/10.1016/J.TIFS.2008.10.003
Pereira, B.S., de Freitas, C., Vieira, R.M., Brienzo, M.: Brazilian banana, guava, and orange fruit and waste production as a potential biorefinery feedstock. J. Mater. Cycles Waste Manag. 24, 2126–2140 (2022). https://doi.org/10.1007/S10163-022-01495-6/TABLES/3
Rasmeni, Z.Z., Madyira, D.M., Matheri, A.N.: Optimum loading ratio for co-digested wastewater sludge and brewery spent yeast. Energy Rep. 8, 1141–1149 (2022). https://doi.org/10.1016/J.EGYR.2022.06.082
García-Depraect, O., Gómez-Romero, J., León-Becerril, E., López-López, A.: A novel biohydrogen production process: Co-digestion of vinasse and nejayote as complex raw substrates using a robust inoculum. Int. J. Hydrogen Energy 42, 5820–5831 (2017). https://doi.org/10.1016/j.ijhydene.2016.11.204
Yang, G., Hu, Y., Wang, J.: Biohydrogen production from co-fermentation of fallen leaves and sewage sludge. Bioresour. Technol. 285, 121342 (2019). https://doi.org/10.1016/j.biortech.2019.121342
Martín, M.A., de la Rubia, M.A., Martín, A., Borja, R., Montalvo, S., Sánchez, E.: Kinetic evaluation of the psychrophylic anaerobic digestion of synthetic domestic sewage using an upflow filter. Bioresour. Technol. 101, 131–137 (2010). https://doi.org/10.1016/j.biortech.2009.08.010
Wang, J., Wan, W.: Comparison of different pretreatment methods for enriching hydrogen-producing bacteria from digested sludge. Int. J. Hydrogen Energy 33, 2934–2941 (2008). https://doi.org/10.1016/j.ijhydene.2008.03.048
Aquino, S.F., Chernicharo, C.A.L., De, M., Florêncio, L., Santos, D.: Methodologies for determining the specific methanogenic activity (SMA) in anaerobic sludges. San. Environ. Eng. (2007). https://doi.org/10.1590/S1413-41522007000200010
Herbert, D., Phipps, P.J., Strange, R.E.: Chapter III chemical analysis of microbial cells. Methods Microbiol. 5, 209–344 (1971). https://doi.org/10.1016/S0580-9517(08)70641-X
Maintinguer, S.I., Fernandes, B.S., Duarte, I.C.S., Saavedra, N.K., Adorno, M.A.T., Varesche, M.B.: Fermentative hydrogen production by microbial consortium. Int. J. Hydrogen Energy 33, 4309–4317 (2008). https://doi.org/10.1016/j.ijhydene.2008.06.053
Angela, M., Adorno, T., Hirasawa, J.S., Bernadete, M., Varesche, A.: Development and validation of two methods to quantify volatile acids (C2–C6) by GC/FID: headspace (automatic and manual) and liquid-liquid extraction (LLE). Am. J. Analyt Chem. 05, 406–414 (2014). https://doi.org/10.4236/AJAC.2014.57049
Perimenis, A., Nicolay, T., Leclercq, M., Gerin, P.A.: Comparison of the acidogenic and methanogenic potential of agroindustrial residues. Waste Manage 72, 178–185 (2018). https://doi.org/10.1016/J.WASMAN.2017.11.033
APHA: Standard methods for the examination of water and wastewater. American Public Health Association, Washington, D.C. (2005)
Griffiths, R.I., Whiteley, A.S., O’Donnell, A.G., Bailey, M.J.: Rapid method for coextraction of DNA and RNA from natural environments for analysis of ribosomal DNA- and rRNA-based microbial community composition. Appl. Environ. Microbiol. 66, 5488–5491 (2000). https://doi.org/10.1128/AEM.66.12.5488-5491.2000
Callahan, B.J., McMurdie, P.J., Rosen, M.J., Han, A.W., Johnson, A.J.A., Holmes, S.P.: DADA2: high-resolution sample inference from illumina amplicon data. Nat. Methods 13, 7 (2016). https://doi.org/10.1038/nmeth.3869
McLaren, M.R., Callahan, B.J.: Silva 138.1 prokaryotic SSU taxonomic training data formatted for DADA2. Zenodo Database (2021). https://doi.org/10.5281/ZENODO.458795
Kanehisa, M., Goto, S.: KEGG: Kyoto Encyclopedia of genes and genomes. Nucleic Acids Res. 28, 27–30 (2000)
Wang, J., Wan, W.: Kinetic models for fermentative hydrogen production: a review. Green Energy Technol. (2009). https://doi.org/10.1007/978-981-10-4675-9_6
Fangkum, A., Reungsang, A.: Biohydrogen production from sugarcane bagasse hydrolysate by elephant dung: effects of initial pH and substrate concentration. Int. J. Hydrogen Energy 36, 8687–8696 (2011). https://doi.org/10.1016/J.IJHYDENE.2010.05.119
Sivaramakrishna, D., Sreekanth, D., Himabindu, V., Anjaneyulu, Y.: Biological hydrogen production from probiotic wastewater as substrate by selectively enriched anaerobic mixed microflora. Renew. Energy 34, 937–940 (2009). https://doi.org/10.1016/J.RENENE.2008.04.016
Fan, Y.T., Zhang, Y.H., Zhang, S.F., Hou, H.W., Ren, B.Z.: Efficient conversion of wheat straw wastes into biohydrogen gas by cow dung compost. Bioresour. Technol. 97, 500–505 (2006). https://doi.org/10.1016/J.BIORTECH.2005.02.049
Wicher, E., Seifert, K., Zagrodnik, R., Pietrzyk, B., Laniecki, M.: Hydrogen gas production from distillery wastewater by dark fermentation. Int. J. Hydrogen Energy 38, 7767–7773 (2013). https://doi.org/10.1016/j.ijhydene.2013.04.008
Sá, L.R., Cammarota, M.C., Ferreira-Leitão, V.S.: Produção de hidrogênio via fermentação anaeróbia-aspectos gerais e possibilidade de utilização de resíduos agroindustriais brasileiros. Quím. Nova. 37(5), 857–867 (2014). https://doi.org/10.5935/0100-4042.20140138
Lin, C.Y., Lay, C.H., Sen, B., Chu, C.Y., Kumar, G., Chen, C.C., Chang, J.S.: Fermentative hydrogen production from wastewaters: a review and prognosis. Int. J. Hydrogen Energy 37, 15632–15642 (2012). https://doi.org/10.1016/J.IJHYDENE.2012.02.072
Fernandes, B.S., Peixoto, G., Albrecht, F.R., del Saavedra, N.K., Zaiat, M.: Potential to produce biohydrogen from various wastewaters. Energy Sustain. Dev. 14, 143–148 (2010). https://doi.org/10.1016/J.ESD.2010.03.004
García-Depraect, O., Castro-Muñoz, R., Muñoz, R., Rene, E.R., León-Becerril, E., Valdez-Vazquez, I., Kumar, G., Reyes-Alvarado, L.C., Martínez-Mendoza, L.J., Carrillo-Reyes, J., Buitrón, G.: A review on the factors influencing biohydrogen production from lactate: the key to unlocking enhanced dark fermentative processes. Bioresour. Technol. 324, 124595 (2021). https://doi.org/10.1016/J.BIORTECH.2020.124595
Andreani, C.L., Tonello, T.U., Mari, A.G., Leite, L.C.C., Campaña, H.D., Lopes, D.D., Rodrigues, J.A.D., Gomes, S.D.: Impact of operational conditions on development of the hydrogen-producing microbial consortium in an AnSBBR from cassava wastewater rich in lactic acid. Int. J. Hydrogen Energy 44, 1474–1482 (2019). https://doi.org/10.1016/J.IJHYDENE.2018.11.155
Baima Ferreira Freitas, I., Aparecida de Menezes, C., Luiz Silva, E.: An alternative for value aggregation to the sugarcane chain: biohydrogen and volatile fatty acids production from sugarcane molasses in mesophilic expanded granular sludge bed reactors. Fuel 260, 116419 (2020). https://doi.org/10.1016/J.FUEL.2019.116419
Matsumoto, M., Nishimura, Y.: Hydrogen production by fermentation using acetic acid and lactic acid. J. Biosci. Bioeng. 103, 236–241 (2007). https://doi.org/10.1263/JBB.103.236
Kim, T.H., Lee, Y., Chang, K.H., Hwang, S.J.: Effects of initial lactic acid concentration, HRTs, and OLRs on bio-hydrogen production from lactate-type fermentation. Bioresour. Technol. 103, 136–141 (2012). https://doi.org/10.1016/J.BIORTECH.2011.09.093
Fuess, L.T., Ferraz, A.D.N., Machado, C.B., Zaiat, M.: Temporal dynamics and metabolic correlation between lactate-producing and hydrogen-producing bacteria in sugarcane vinasse dark fermentation: the key role of lactate. Bioresour. Technol. 247, 426–433 (2018). https://doi.org/10.1016/J.BIORTECH.2017.09.121
Villa Montoya, A.C., Cristina da Silva Mazareli, R., Delforno, T.P., Centurion, V.B., Sakamoto, I.K., Maia de Oliveira, V., Silva, E.L., Amâncio Varesche, M.B.: Hydrogen, alcohols and volatile fatty acids from the co-digestion of coffee waste (coffee pulp, husk, and processing wastewater) by applying autochthonous microorganisms. Int. J. Hydrogen Energy 44, 21434–21450 (2019). https://doi.org/10.1016/J.IJHYDENE.2019.06.115
Xu, R., Yang, Z.H., Zheng, Y., Zhang, H.B., Liu, J.B., Xiong, W.P., Zhang, Y.R., Ahmad, K.: Depth-resolved microbial community analyses in the anaerobic co-digester of dewatered sewage sludge with food waste. Bioresour. Technol. 244, 824–835 (2017). https://doi.org/10.1016/J.BIORTECH.2017.07.056
Yang, G., Wang, J.: Changes in microbial community structure during dark fermentative hydrogen production. Int. J. Hydrogen Energy 44, 25542–25550 (2019). https://doi.org/10.1016/J.IJHYDENE.2019.08.039
Yang, G., Wang, J.: Biohydrogen production by co-fermentation of antibiotic fermentation residue and fallen leaves: insights into the microbial community and functional genes. Bioresour. Technol. 337, 125380 (2021). https://doi.org/10.1016/J.BIORTECH.2021.125380
Hallenbeck, P.C.: Fermentative hydrogen production: principles, progress, and prognosis. Int. J. Hydrogen Energy 34, 7379–7389 (2009). https://doi.org/10.1016/j.ijhydene.2008.12.080
Yin, Y., Wang, J.: Predictive functional profiling of microbial communities in fermentative hydrogen production system using PICRUSt. Int. J. Hydrogen Energy 46, 3716–3725 (2021). https://doi.org/10.1016/J.IJHYDENE.2020.10.246
Hallenbeck, P.C.: Fundamentals of the fermentative production of hydrogen. Water Sci. Technol. 52, 21–29 (2005)
Funding
The financial assistance was provided by São Paulo Research Foundation (FAPESP), Grant No. 2020/15155-3.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by CCCF, as well as the written of the first draft of the manuscript. CF performed the High-Performance Liquid Chromatography analysis. DFCM contributed to the experimental outline and discussion. SIM and MB made funding acquisition, advising and discussion. All authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
da Costa Freire, C.C., Marin, D.F.C., da Silva Mazareli, R.C. et al. Unravelling the Biohydrogen Production Potential from a Co-Digestion Process of Banana Processing Wastewater and Synthetic Sewage by Anaerobic Fermentation: Performance Evaluation and Microbial Community Analysis. Waste Biomass Valor 15, 1587–1601 (2024). https://doi.org/10.1007/s12649-023-02258-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12649-023-02258-6