Abstract
Benzo(a)pyrene (BaP) is a widespread environmental contaminant that has been associated with neurotoxicity in mammals. It has strong toxic effects on the developing central nervous system. Cerebellum is associated with locomotor activity and anxiety behavior, but there is very little research about the toxic effects of BaP in cerebellum. The present study aims to investigate the global influence of BaP subacute exposure on the metabolome of rat cerebellum. Male neonatal rats (postnatal day 5) were divided into two groups: control group and BaP-treated group (2 mg/kg/day for 7 weeks). Open field test and transmission electron microscopy were performed to analyze neurobehavior and ultramicrostructure alteration. Gas chromatography-mass spectrometry (GC-MS) was used to analyze metabolites of the cerebellum in both groups. The results revealed that postnatal exposure to BaP promoted pathological changes in the cerebellum and increased locomotor and anxiety activities in early adulthood. Twenty differential significant metabolites were identified by multivariate statistical analysis. Further metabolic pathway impact analysis and network analysis suggested that the primary metabolic pathways affected included pathway involved in energy metabolism, methionine and cysteine metabolism, and glutathione metabolism. These findings suggest that BaP-induced cerebellum injury may be correlated with metabolic changes and provide an area to target to reduce the negative effects of BaP.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Benzo(a)pyrene (BaP) is a typical polycyclic aromatic hydrocarbon (PAH): uptake occurs mostly by inhalation and ingestion of contaminated food and water and polluted air (Knuckles et al. 2001; Archibong et al. 2002). BaP and its metabolites can cause central nervous system (CNS) impairments (Chepelev et al. 2015). It has also been reported that the fetus in utero and young children is more susceptible to the toxicant and that postnatal BaP exposure may result in neurobehavioral impairment in adulthood (Brown et al. 2007; Chen et al. 2012). In fact, the developing CNS is much more sensitive to injury from toxicants than the adult CNS and can interfere with any of the processes involved in its development (Rodier 1995).
Every development of an organism has a critical period for its normal maturation. In the development of the brain, it is referred to as the “brain growth spurt” (Eriksson 1997). During this period, the glial cells start to develop, axons grow, dendrites branch, and synapses are formed (Gale et al. 2003). Also in this critical period, animals can develop new motor and sensory abilities and spontaneous motor peaks (Viberg et al. 2008). This is the key period that the brain’s physiological structure and function mature from the infant period. Studies have shown that early postnatal toxicant exposure has profound effects on adult neural behavior (Tchekalarova et al. 2005; Viberg et al. 2008; Gassowska et al. 2016). It is worth noting that infants and children are unavoidably exposed to BaP, which is ubiquitous and consistently present in the environment, and is a possible causal factor for abnormal adult neurobehavioral (Chen et al. 2012).
The cerebellum has an important role in gait, reflex adaptation, and motor function and learning (Glickstein and Doron 2008). Recent work has revealed that the cerebellum intervenes in the regulation of some neocortical cognitive function and emotional activities (Habas 2001; Gillig and Sanders 2010). BaP exposure can adversely affect both adult and children’s neurobehavior related to the function of the cerebellum, such as mood state, digital span and symbol, visual retention, manual dexterity, anxiety, depression, and attention (Niu et al. 2010; Perera et al. 2012; Qiu et al. 2013; Zhang et al. 2013). A handful of studies of BaP neurotoxicity have mentioned that BaP could disturb gene expression in cerebellum and may correlate with anxiety behavior and motor activity change (Saunders et al. 2002; Grova et al. 2008; Chepelev et al. 2016), but the underlying mechanisms are not clear.
In recent years, metabolomics has been used to study the molecular mechanisms of chemical toxicity (Liu et al. 2015; Xu et al. 2015; Ba et al. 2016) and some diseases (Constantinou et al. 2011; Kimball et al. 2016; Zhou et al. 2016). Metabolomics depicts the metabolic profile in complex systems (Naz et al. 2014), through the systematic identification and quantification of metabolites (Jager et al. 2016). Metabolism is the endpoint of the enzyme (protein) action and the final consequence of biological function, so it can directly indicate aberrant physiological status (Huang et al. 2016). A few reports have shown that BaP disrupts the metabolism of the Manila clam, oyster pinctada, rat serum, murine hepatocytes, and human endothelia (Zhang et al. 2011; Kalkhof et al. 2015; Wang et al. 2015; Chen et al. 2016). An untargeted metabolomic approach has not yet been applied to analyze rat cerebellum following BaP treatment (Naz et al. 2014). In the present study, untargeted metabolomics was employed using GC-MS to explore the effect of postnatal exposure to BaP on rat cerebellum.
Materials and Methods
Chemical
Benzo(a)pyrene (> 96% pure), the derivatization reagent methyl chloroformate (MCF), the internal standard D4-L-alanine, and pyridine were purchased from Sigma-Aldrich (St. Louis, USA). Methanol, chloroform, sodium hydroxide, and sodium bicarbonate were obtained from Merck (Darmastadt, Germany). Anhydrous sodium sulfate was purchased from Fluka (Steinheim, Germany). All the reagents in this research were of analytical grade.
Animals and Treatment
Thirty-four healthy male neonatal Sprague-Dawley (SD) rats aged postnatal day (PND) 4 (the day of parturition was considered PND 0) and their maternal rats were purchased from the animal center of Chongqing Medical University (Chongqing, China) [certificate SCXK (YU) 2007-0001]. All animals were kept under standard laboratory conditions: 18–28 °C temperature and 40–80% humidity with a light/dark cycle of 12 h. All animals had free access to water and food. Each neonatal rat was marked an individual number on their feet and body by a nontoxic ink. All neonatal rats were randomly divided into two groups: control group and BaP-treated group. Neonatal rats were weaned on PND 21 and housed in the same treatment group. All animal care and experimental procedures were in accordance with the National Institutes of Health Guidelines for Animal Research (Clark et al. 1997). This study was approved by the Ethics Committee of Chongqing Medical University.
Owing to its lipophilicity, BaP was dissolved in corn oil. The BaP-treated group of rats was administered via oral gavage each day with a dose of 2 mg/kg. This dose corresponded to human environmental exposure levels (ATSDR 1995). This dose has also been studied in our previous studies (Chengzhi et al. 2011; Chen et al. 2012; Yang et al. 2017). The control group rats received an equivalent volume of corn oil.
In rats, the brain growth spurt period corresponds to the third human gestational trimester and continues throughout the first years of life, spanning the first 3–4 weeks of neonatal life and reaching its peak around PND 10 (Viberg et al. 2008). In this study, the treatment began at PND 5. To simulate the infant and children continuously being exposed to BaP before maturation, the treatment lasted for 7 weeks.
After 7-week treatment, 18 rats (n = 9 each group) were directly sacrificed by rapid decapitation; the cerebellum was quickly acquired for metabolomic analysis (n = 6 each group) and electron microscopy (n = 3 each group). Sixteen rats (n = 8 each group) performed behavioral tests using the open field test.
Open Field Test
Rats were placed in the testing room for a 30-min period to acclimatize. The open field test apparatus consists of a black square cage (100 × 100 × 40 cm) with a floor arena divided into 25 × 25 cm2 equal squares. Each rat was placed in the center of the cage. After 30 s of adapting, all behaviors were recorded for 5 min using a camera positioned 200 cm above the arena. The cage was thoroughly cleaned with ethanol swabs between each test. The rats were then returned to their home cage after the test, and then to a hold room once all animals were tested. The locomotor activity (total distance and time) and the number of central activity and rearing behaviors (defined as an upright posture sustained on hind paws) were clearly visible on the video. The test took place in a soundproof room between 9:00 a.m. and 17:00 p.m.
Sample Preparation for Electron Microscopy
The rats were perfused with normal saline solution followed by 4% paraformaldehyde, and the brain was quickly acquired and stored at 4% paraformaldehyde for 12 h. The sample was then sliced into sections of approximately 1 mm2 and kept in the 2.5% glutaraldehyde for 24 h at 4 °C. Samples were post-fixed in 1% osmium tetroxide for 2 h, dehydrated in a series of alcohol solutions, and embedded in epoxy resin. Ultra-thin (1 μm) sections were placed on grids and stained with uranyl acetate and lead citrate before examination with a transmission electron microscope.
Sample Preparation for GC-MS Analysis
The cerebellum (m = 50 ± 0.5 mg) was homogenized after adding 20 μL internal standard and 1.5 mL of prechilled methanol/water (1:1, v/v) in a 2-mL centrifuge tube. The mixture was centrifuged at 16,000×g for 10 min at 4 °C, and the supernatant was transferred into a new 2-mL Eppendorf tube. Then, 1.6 mL of prechilled dichloromethane/methanol (3:1, v/v) was added to precipitate. The mixture was again centrifuged under the same conditions, to obtain the supernatant. The two supernatants were combined and dried in a vacuum concentrator. Subsequently, 200 μL of sodium hydroxide was added to each of the dried samples before they were transferred to a silanized glass tube. One-hundred sixty-seven microliters of methanol and 34 μL of pyridine were added to the solution, followed by the addition of 20 μL of MCF with vigorous mixing for 30s, and then, a further 20 μL of MCF was added, accompanied by vigorous mixing for 30 s. Four hundred microliters of chloroform and 400 μL of 50 mM sodium bicarbonate were added, followed by 10 s of vigorous mixing to separate the MCF derivatives. After centrifuging at 1500 rpm for 10 min, the aqueous layer was discarded and the remaining sample was dehydrated with anhydrous sodium sulfate before being transferred to GC vials for analysis (Smart et al. 2010; Dai et al. 2016).
GC-MS Analysis
GC-MS analysis was performed on an Agilent 7890A gas chromatography system coupled with an Agilent 5975C mass spectrometer (Agilent, Santa Clara, CA, USA). The system utilized a fused silica Zebron ZB-1701 capillary column (30 × 250 × 0.15 μm) coated with 86% dimethylpolysiloxane and 14% cyanopropylphenyl (Phenomenex, Torrance, CA, USA). The GC-MS parameters were based on Thomas et al. (2015). A 1.0 μL amount of the sample was injected into the GC-MS under pulsed splitless mode. The inlet temperature was set to 290 °C. Carrier gas was instrument grade helium (99.99%, BOC); the front inlet purge flow was set to 25 mL/min 1 min after injection. The GC oven initial temperature was kept at 45 °C for 2 min, raised to 180 °C at rate of 9 °C/min, held 5 min; increased 220 °C at rate of 40 °C/min, held 5 min; increased 240 °C at rate of 40 °C/min, held 11.5 min; finally increased 280 °C at rate of 40 °C/min, held 2 min. The transfer line, source, and quadrupole mass analyzer temperatures were 250, 230, and 150 °C, respectively. The detector was run in positive ionization mode with electron energy of 70 eV and turned on 5.5 min into the run. The mass spectrometry data was acquired in scan mode with the range from 38 m/z to 550 m/z with scan time 0.1 s, scan speed 1562 μ/s, and 100 ion counts as the detection threshold. A solvent blank was run after every six sample injections to check for instrument carryover.
Metabolomic Data Analysis
The raw data obtained from the GC-MS was converted into common data format (CDF) by the Automated Mass Deconvolution and Identification System (AMDIS—http://www.amdis.net/). Different metabolites of the cerebellum were identified by an in-house mass spectral library and the NIST0.5 library (Thomas et al. 2015). The results include sample information, retention time, and peak intensities. Compounds with a match factor > 70% to one of the libraries were included in the analysis (Thomas et al. 2015). We used the relative quantification of peak intensities to estimate the level of the metabolites. The data was normalized using an internal standard and weight of the brain tissue. Principal component analysis (PCA) was used to observe the distribution of all samples, and partial least-squares discriminate analysis (PLS-DA) was used to differentiate the control group from the BaP-treated group by SIMCA-P 11.5 (Umetrics, Umea, Sweden). Significantly changed compounds were selected based on their variable of importance in the project (VIP) value. To ensure that the selected metabolites significantly changed among groups, a Student’s t test was conducted in SPSS 17.0 (SPSS, Inc., Chicago, IL) with the significance level set at VIP > 1 and P < 0.05. In addition, metabolic pathway analysis was performed in Metaboanalyst (http://www.metaboanalyst.ca/), followed by network analysis performed in Metscape (a plug-in of Cytoscape 3.5.0) to select the main affected pathway.
Statistical Analysis
The data obtained in the open field test was analyzed using a Student’s t test. Data was reported as mean ± SD, and P < 0.05 was considered to be statistically significant in the open field test. All statistical analyses were performed in SPSS v. 17.0.
Results
Open Field Test
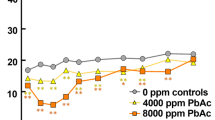
Locomotor activity and mood changes were evaluated in the open field test. Compared with the control group, the rats in the BaP-treated group had significantly higher total distance locomotor activity (Fig. 1a, t = 6.039, P < 0.000), but there was no statistically significant difference in the time of the locomotor activity (Fig. 1b, t = 0.077, P = 0.940). Moreover, the number of central activities (Fig. 1c, t = 2.263, P = 0.040) and rearing activities performed by the BaP-treated group was significantly increased (Fig. 1d, t = 2.426, P = 0.029).
Ultrastructural Feature in Cerebellum
Compared with the control group (Fig. 2a, c, e), the BaP-treated group showed ultrastructural features of typical cellular swelling. The characterization of cellular swelling in the BaP-treated group was local swelling which was recognized by a pale and watery cytoplasm under electron microscopic analysis (Fig. 2b) and swollen organelles, including mitochondria deformation (Fig. 2d) and endoplasmic reticulum expansion (Fig. 2f), which were obvious in the cell body and the neuropil.
Ultrastructural changes in cerebellum of control and BaP-treated rats. a Unaltered structure of neural cell and neuropil. b Neural cells and neuropil swelling. c Ultrastructurally unchanged mitochondria. d Mitochondria swollen and deformation. e Ultrastructurally unchanged endoplasmic reticulum. f Endoplasmic reticulum expansion. Pictures are representative for each of three control and BaP-treated group rats
Analysis of Brain Metabolomics
The GC-MS metabolomic profiling of the cerebellum samples produced a total of 120 compounds across the two groups, which were used in the subsequent multivariate analysis. The PCA plot showed that the BaP-treated group could be distinguished from the control group (R 2 X = 0.68, Q 2 = 0.32; Fig. 3a), and the PLS-DA plot showed an obvious distinction between the two groups (R 2 X = 0.416, R 2 Y = 0.992, Q 2 = 0.915, Fig. 3b). Based on the PLS-DA (VIP > 1) and t test (P < 0.05), 20 differential metabolites were identified in the cerebellum between the control group and BaP-treated group (Table 1). In order to visualize the metabolomic results, a heat map (Fig. 4) was generated to show the level of the 20 significant metabolites. Pathway analysis, performed by inputting metabolites into Metaboanalyst (http://www.metaboanalyst.ca/), suggested that BaP significantly impacted a number of metabolic pathways (Fig. 5, Table 2). Further analysis was conducted based on the 20 significant metabolites, to further study the associations and interaction patterns between metabolites. There were only six metabolites that were highly linked and were considered as a hub of a network (Fig. 6). This network hub included pathways related to energy metabolism, methionine cycle, and glutathione metabolism. Figure 7 visually summarizes the potential mechanisms of BaP in rat cerebellum through the metabolomic changes observed.
The pathway impact of BaP on rat cerebellum metabolites. All matched pathway according to P values from pathway enrichment analysis and pathway impact value from pathway topology analysis by Metaboanalyst. Enrich metabolic pathways were ranked by FDR values. The significant pathways for BaP-treated group compared to control group were represented by bigger/red dots and by those dots with higher log P value. And the pathway impact is calculated as the sum of the importance measures of the matched metabolites normalized by the sum of the importance measures of all metabolites in each pathway. Specifics are shown in Table 2
Integrated network combining the major signaling pathway associated with the differential metabolites between control group and BaP-treated group. The network analysis of differential metabolites was constructed using Metscape (a plug-in Cytoscape 3.5.0). Nodes in red represent the hub of network. The scale of red node represents the size of impact. There are six metabolites highly linked that were considered as a hub of network and involved three metabolisms including energy metabolism, methionine cycle, and glutathione metabolism
The summary overview of the metabolite pathway of BaP affected neural function in rat cerebellum. Molecular upregulation (pink). Molecular downregulation (green). Potential mechanism of toxic effects (green). Cr/PCr/CK creatine/phosphocreatine/creatine kinase system, TCA tricarboxylic acid cycle, GSSG oxidized glutathione, GSH glutathione, GPx glutathione peroxidase, GST glutathione-S-transferases, CAT reactivating H2O2-inactiated catalase, TrxR thioredoxin reductase, HCys homocysteine, AdoHcy S-adenosylhomocysteine, AdoHmet S-adenosylmethionine, SAH hydrolase S-adenosylhomocysteine hydrolase, BHM betaine-homocysteine methyltransferase, CBS cystathionine-beta-synthase, TCA tricarboxylic acid
Discussion
The open field test was used to comprehensively assess locomotor activity and anxiety-like and exploratory behaviors (Prut and Belzung 2003; Tatem et al. 2014). In this study, compared with the control group, the rats in the BaP-treated group had significantly increased locomotor activity, rearing activity, and central activity. These results suggest that early life exposure to BaP can affect locomotor activity and anxiety-like behavior in adulthood. These findings are in accordance with previous research (Chen et al. 2012; Perera et al. 2012; Patel et al. 2016). Moreover, electron microscope analysis revealed that BaP-treated rats had obvious neural cell and neuropil swelling; mitochondria deformation including elongation, swelling, or shrinking; and endoplasmic reticulum expansion. Therefore, subacute postnatal BaP oral exposure may have subsequent effects on the development of rat cerebellum.
The primary metabolism of the SD rat cerebellum exposed to BaP after 7 weeks by oral gavage was studied, using GC-MS metabolomics for the acquisition of the metabolic profiles. Metabolomic analysis enabled the identification of 20 metabolites that were significantly different between the two groups, and they were involved in major metabolic pathways and functions. Our findings demonstrated that the rat cerebellum is metabolically responsive to BaP. Thus, the metabolic changes observed in mammalian cerebellum indicate that BaP may affect brain metabolism.
The metabolomic observations indicate that insufficient energy was being supplied in the cerebellum. There was no significant change in some metabolites that represent glycolysis and the tricarboxylic acid (TCA) cycle, but the level of phosphoenolpyruvic (PEP) increased, and the levels of some amino acids, including asparagine, proline, valine, leucine, and serine, decreased. The creatine/phosphocreatine/creatine kinase (Cr/PCr/CK) system was interrupted, indicating that there was shortage of energy supply in cerebellum. PEP is a high-energy intermediate product of glycolysis, which can be converted into pyruvate (Saiki et al. 1997). This step is essentially irreversible in the whole process of glycolytic and oxidative phosphorylation (Saiki et al. 1997). The accumulation of PEP suggests that glycolysis was abnormal. While the TCA cycle is the main pathway of oxidative phosphorylation in mitochondria (Fernie et al. 2004), and is the major common pathway for oxidation, some amino acids are metabolized to obtain energy needed to maintain CNS function (Rodrigues and Cerdán 2007). The levels of asparagine, proline, valine, and leucine were decreased which may have indicated that amino acid oxidation was occurring to compensate for the shortage of energy (Reitzer 2004; Fernstrom 2005; Wyse and Netto 2011). The Cr/PCr/CK system, a high-energy phosphate shuttle from mitochondria to cytoplasmic sites and an intracellular buffer for ATP, is another high-energy reservoir for CNS (Beard and Braissant 2010; Lage et al. 2013). CK activity is important for brain energy metabolism homeostasis and especially ATP transfer and buffering and could be inhibited because of the increased levels of lysine (Tonin et al. 2009). The decrease of creatinine, which is final product of creatine, was further evidence that the Cr/PCr/CK system was disrupted. The shortage of energy can impair the main functions of CNS, including generation, processing, and transmission of impulses. Because action potentials generated and transmitted rely on Na+, K+, and Ca+ to maintain electrochemical disequilibrium across the neuronal plasma membrane, the CNS is in need of a constant supply of energy (Erecinska and Silver 1989). Sodium-potassium adenosine triphosphatase (Na+-K+-ATPase), a highly expressed membrane protein that hydrolyzes ATP to transduce Na+ and K+, can modulate the resting transmembrane potential, some postsynaptic activities, and neurotransmitter turnover (Villa et al. 2013; Kurauchi et al. 2016). Calcium adenosine triphosphatase (Ca+-ATPase) can hydrolyze ATP to maintain the homeostasis of intracellular Ca+ (Kurauchi et al. 2016). The rates of ion transport and ATPase activity require a large supply of energy (Erecinska and Silver 1989). If this equilibria was broken, it could lead to nerve ending depolarization with Ca+ entry into the cell, followed by neurotransmitter release and neuronal swelling (de Lores Arnaiz and Ordieres 2014). Our previous studies found that BaP exposure can lead to decreased ATPase activity as well as Ca+ overload (Duan et al. 2013; Dong et al. 2015; Yang et al. 2017). Furthermore, the upregulation of locomotor activity and anxiety aggravated the consumption of energy and accelerated amino acid metabolism—particularly valine and leucine metabolism.
The upregulation of NADP/NADPH and the downregulation of glutathione (GSH) and dehydroascorbic acid (DRA) suggest that the antioxidant system is disordered in the cerebellum. NADP/NADPH and GSH are known as classic molecules involved in energy metabolism, reductive biosynthesis, and antioxidation (Wu et al. 2004; Ying 2008). They are important components in the intracellular antioxidant system and play an essential role in cell defense against reactive oxygen species (ROS) (Wu et al. 2004; Ying 2008). NADPH mediates cellular antioxidation mainly through its effects on GSH regeneration: the regeneration of GSH from GSSH, through the action of glutathione reductase, requires NADPH (Hayes and McLellan 1999; Ying 2008). DRA, an oxidized form of ascorbic acid (AA), could inhibit mitochondrial damage and cell death following oxidative injury and can also complement the lack of AA at the expense of GSH (Kim et al. 2008). The antioxidant system is responsible for balancing the production of ROS that is generated as part of normal cellular metabolism in the brain (Liu et al. 2015). The antioxidant system breakdown following BaP treatment suggests that there was a ROS generation overload, resulting in neuronal oxidative stress. Also, the upregulated level of intracellular Ca+ can trigger the mitochondria to produce more ROS (Bakthavachalam and Shanmugam 2017). These findings are in accordance with our previous studies which found that BaP could induce oxidative stress in the rat brain (Duan et al. 2013; Yang et al. 2017). Excessive ROS induced and enhanced neurobehavioral impairment and neural cell damage, mainly through altered neuronal signaling, neuroinflammation, and activation of cell death mechanisms (Popa-Wagner et al. 2013).
S-adenosylhomocysteine (AdoHcy), an important intermediate product in the methionine cycle, can influence cellular methylation function through regulation of the levels of S-adenosylmethionine (AdoHmet) and homocysteine (HCys) (Miller 2003). AdoHcy is generated from AdoHmet after donation of a methyl group (Miller 2003). The level of AdoHcy and AdoHmet can modulate cellular methylation; thus, the ratio of AdoHcy/AdoHmet is often used as an index of methylation capacity (Choumenkovitch et al. 2002). AdoHcy acts as a potent methyltransferase inhibitor that could cause global cellular hypomethylation (Esse et al. 2014). AdoHcy hydrolase cleaves AdoHcy to adenosine and Hcys in the methionine cycle (Miller 2003). The increase of AdoHcy could induce the pathophysiology of cystathionine-beta-synthase (CBS) deficiency (Choumenkovitch et al. 2002; Esse et al. 2014). HCys is converted to cystathionine by CBS and is associated with the incidence of hyperhomocysteinemia that could cause or promote CNS dysfunction (Gerritsen and Waisman 1964; Robert et al. 2003; Troen 2005). In this study, we found that cystathionine downregulated HCys metabolism which suggests that CBS deficiency plays a critical role in metabolism. In addition, CBS deficiency could also induce the increase of HCys and AdoHcy (Choumenkovitch et al. 2002; Esse et al. 2014). The accumulation of HCys in neurons could not act on the extracellular transport of HCys. High brain concentrations of homocysteine or its oxidized derivatives might indirectly alter neurotransmission and induce excitotoxicity in neurons, or induce AdoHcy metabolism and cerebrovascular function damage, leading to secondary neuronal dysfunction and degeneration (Troen 2005). Methionine and HCys may also modulate ROS generated (Gomez et al. 2011), which suggests that the concentration of AdoHcy may be associated with ROS generation.
This is the first study to apply a metabolomic approach to investigate the effect of BaP on rat cerebellum metabolism. Administration of BaP caused anxiety behavior and increased locomotor activity and neural cell swelling. Our findings suggest that postnatal exposure to BaP induced toxic effects on the brain. Untargeted GC-MS-based metabolomics revealed that the cerebellum neurodevelopment was predominantly affected in three key areas: energy metabolism, the antioxidant system, and the methionine cycle.
-
1.
Energy metabolism plays an important role in maintaining CNS function. Insufficient energy supply is linked with neurobehavioral impairment and cell swelling and could also promote ROS production and disrupt the methionine cycle.
-
2.
ROS were modulated by GSH (glutathione) and NADP-NADPH by glutathione peroxidase (GPx), glutathione-S-transferases (GST), reactivating H2O2-inactiated catalase (CAT), and thioredoxin reductase (TrxR)-mediated regeneration of thioredoxin. Methionine and homocysteine (HCys) may modulate ROS generation in the brain.
-
3.
S-adenosylhomocysteine (AdoHcy), a potent methyltransferase inhibitor, could induce global cellular hypomethylation. The accumulation of AdoHcy and the absence of cystathionine may lead to BHM enzyme inactivity and CBS deficiency, which are correlated with hyperhomocysteinemia that can cause CNS dysfunction.
In this study, the untargeted GC-MS metabolomic approach was used to investigate the effects of BaP on cerebellar development. More detailed mechanisms should be explored in future studies.
References
Archibong AE, Inyang F, Ramesh A, Greenwood M, Nayyar T, Kopsombut P, Hood DB, Nyanda AM (2002) Alteration of pregnancy related hormones and fetal survival in F-344 rats exposed by inhalation to benzo(a)pyrene. Reprod Toxicol 16:801–808
ATSDR (1995) Toxicological profile for polycyclic aromatic hydrocarbons(PAHs). U.S. Department of Health and Human Services, Atlanta
Ba Q, Huang C, Fu Y, Li J, Li J, Chu R, Jia X, Wang H (2016) Cumulative metabolic effects of low-dose benzo(a)pyrene exposure on human cells. Toxicol Res 5(1):107–115
Bakthavachalam P, Shanmugam PS (2017) Mitochondrial dysfunction—silent killer in cerebral ischemia. J Neurol Sci 375:417–423
Beard E, Braissant O (2010) Synthesis and transport of creatine in the CNS: importance for cerebral functions. J Neurochem 115(2):297–313
Brown LA, Khousbouei H, Goodwin JS, Irvin-Wilson CV, Ramesh A, Sheng L, McCallister MM, Jiang GC, Aschner M, Hood DB (2007) Down-regulation of early ionotrophic glutamate receptor subunit developmental expression as a mechanism for observed plasticity deficits following gestational exposure to benzo(a)pyrene. Neurotoxicology 28(5):965–978
Chen C, Tang Y, Jiang X, Qi Y, Cheng S, Qiu C, Peng B, Tu B (2012) Early postnatal benzo(a)pyrene exposure in Sprague-Dawley rats causes persistent neurobehavioral impairments that emerge postnatally and continue into adolescence and adulthood. Toxicol Sci 125(1):248–261
Chen H, Song Q, Diao X, Zhou H (2016) Proteomic and metabolomic analysis on the toxicological effects of benzo[a]pyrene in pearl oyster Pinctada martensii. Aquat Toxicol 175:81–89
Chengzhi C, Yan T, Shuqun C, Xuejun J, Youbin Q, Yinyin X, Qian T, Baijie T (2011) New candidate proteins for benzo(a)pyrene-induced spatial learning and memory deficits. J Toxicol Sci 36(2):163–171
Chepelev NL, Moffat ID, Bowers WJ, Yauk CL (2015) Neurotoxicity may be an overlooked consequence of benzo[a]pyrene exposure that is relevant to human health risk assessment. Mutat Res Rev Mutat Res 764:64–89
Chepelev NL, Long AS, Bowers WJ, Gagne R, Williams A, Kuo B, Phillips DH, Arlt VM, White PA, Yauk CL (2016) Transcriptional profiling of the mouse hippocampus supports an NMDAR-mediated neurotoxic mode of action for benzo[a]pyrene. Environ Mol Mutagen 57(5):350–363
Choumenkovitch SF, Selhub J, Bagley PJ, Maeda N, Nadeau MR, Smith DE, Choi SW (2002) In the cystathionine beta-synthase knockout mouse, elevations in total plasma homocysteine increase tissue S-adenosylhomocysteine, but responses of S-adenosylmethionine and DNA methylation are tissue specific. J Nutr 132(8):2157–2160
Clark JD, Gebhart GF, Gonder JC, Keeling ME, Kohn DF (1997) The 1996 guide for the care and use of laboratoty animals. ILAR J 38:41–48
Constantinou C, Chrysanthopoulos PK, Margarity M, Klapa MI (2011) GC-MS metabolomic analysis reveals significant alterations in cerebellar metabolic physiology in a mouse model of adult onset hypothyroidism. J Proteome Res 10(2):869–879
Dai H, Xia YY, Ting-Li Han TH, Philip NBPN, Tang X, Zhang RY, Du H, Cai TJ, Cheng SQ (2016) Effect of chronic arsenic exposure on mouse brain tissue and serum metabolomics. Nan Fang Yi Ke Da Xue Xue Bao 36(9):1192–1197
de Lores Arnaiz GR, Ordieres MGL (2014) Brain Na+, K+−ATPase activity in aging and disease. Int J Biomed Sci 10(2):85–102
Dong T, Ni J, Wei K, Liang X, Qin Q, Tu B (2015) Effects of benzo(a)pyrene exposure on the ATPase activity and content of Ca(2)(+) in the hippocampus of neonatal SD rats. Zhong Nan Da Xue Xue Bao Yi Xue Ban 40(4):356–361
Duan L, Tang Y, Chen CZ, Pen B, Qiu CY, Qi YB, Tu BJ (2013) Effects of benzo(a)pyrene exposure on oxidative stress and ATPase in the hippocampus of rats. Zhonghua Lao Dong Wei Sheng Zhi Ye Bing Za Zhi 31(7):500–503
Erecinska M, Silver IA (1989) ATP and brain function. J Cereb Blood Flow Metab 9(1):2–19
Eriksson P (1997) Developmental neurotoxicity of environmental agents in the neonate. Neurotoxicology 18(3):719–726
Esse R, Imbard A, Florindo C, Gupta S, Quinlivan EP, Davids M, Teerlink T, Tavares de Almeida I, Kruger WD, Blom HJ, Castro R (2014) Protein arginine hypomethylation in a mouse model of cystathionine beta-synthase deficiency. FASEB J 28(6):2686–2695
Fernie AR, Carrari F, Sweetlove LJ (2004) Respiratory metabolism: glycolysis, the TCA cycle and mitochondrial electron transport. Curr Opin Plant Biol 7(3):254–261
Fernstrom JD (2005) Branched-chain amino acids and brain function. J Nutr 135(6 Suppl):1539S–1546S
Gale CR, Walton S, Martyn CN (2003) Foetal and postnatal head growth and risk of cognitive decline in old age. Brain 126(Pt 10):2273–2278
Gassowska M, Baranowska-Bosiacka I, Moczydlowska J, Frontczak-Baniewicz M, Gewartowska M, Struzynska L, Gutowska I, Chlubek D, Adamczyk A (2016) Perinatal exposure to lead (Pb) induces ultrastructural and molecular alterations in synapses of rat offspring. Toxicology 373:13–29
Gerritsen T, Waisman HA (1964) Homocystinuria: absence of cystathionine in the brain. Science 145(3632):588
Gillig PM, Sanders RD (2010) Psychiatry, neurology, and the role of the cerebellum. Psychiatry (Edgmont) 7(9):38–43
Glickstein M, Doron K (2008) Cerebellum: connections and functions. Cerebellum 7(4):589–594
Gomez J, Sanchez-Roman I, Gomez A, Sanchez C, Suarez H, Lopez-Torres M, Barja G (2011) Methionine and homocysteine modulate the rate of ROS generation of isolated mitochondria in vitro. J Bioenerg Biomembr 43(4):377–386
Grova N, Schroeder H, Farinelle S, Prodhomme E, Valley A, Muller CP (2008) Sub-acute administration of benzo[a]pyrene (B[a]P) reduces anxiety-related behaviour in adult mice and modulates regional expression of N-methyl-D-aspartate (NMDA) receptors genes in relevant brain regions. Chemosphere 73(1 Suppl):S295–S302
Habas C (2001) The cerebellum: from motor coordination to cognitive function. Rev Neurol (Paris) 157(12):1471–1497
Hayes JD, McLellan LI (1999) Glutathione and glutathione-dependent enzymes represent a co-ordinately regulated defence against oxidative stress. Free Radic Res 31(4):273–300
Huang Q, Luo L, Alamdar A, Zhang J, Liu L, Tian M, Eqani SA, Shen H (2016) Integrated proteomics and metabolomics analysis of rat testis: mechanism of arsenic-induced male reproductive toxicity. Sci Rep 6:32518
Jager C, Hiller K, Buttini M (2016) Metabolic profiling and quantification of neurotransmitters in mouse brain by gas chromatography-mass spectrometry. Curr Protoc Mouse Biol 6(3):333–342
Kalkhof S, Dautel F, Loguercio S, Baumann S, Trump S, Jungnickel H, Otto W, Rudzok S, Potratz S, Luch A, Lehmann I, Beyer A, von Bergen M (2015) Pathway and time-resolved benzo[a]pyrene toxicity on Hepa1c1c7 cells at toxic and subtoxic exposure. J Proteome Res 14(1):164–182
Kim EJ, Won R, Sohn JH, Chung MA, Nam TS, Lee HJ, Lee BH (2008) Anti-oxidant effect of ascorbic and dehydroascorbic acids in hippocampal slice culture. Biochem Biophys Res Commun 366(1):8–14
Kimball BA, Wilson DA, Wesson DW (2016) Alterations of the volatile metabolome in mouse models of Alzheimer’s disease. Sci Rep 6:19495
Knuckles ME, Inyang F, Ramesh A (2001) Acute and subchronic oral toxicities of benzo[a]pyrene in F-344 rats. Toxicol Sci 60:382–388
Kurauchi Y, Hisatsune A, Seki T, Katsuki H (2016) Na(+), K(+)-ATPase dysfunction causes cerebrovascular endothelial cell degeneration in rat prefrontal cortex slice cultures. Brain Res 1644:249–257
Lage S, Andrade F, Prieto JA, Asla I, Rodriguez A, Ruiz N, Echeverria J, Luz Couce M, Sanjurjo P, Aldamiz-Echevarria L (2013) Arginine-guanidinoacetate-creatine pathway in preterm newborns: creatine biosynthesis in newborns. J Pediatr Endocrinol Metab 26(1–2):53–60
Liu B, Gu Y, Xiao H, Lei X, Liang W, Zhang J (2015) Altered metabolomic profiles may be associated with sevoflurane-induced neurotoxicity in neonatal rats. Neurochem Res 40(4):788–799
Miller AL (2003) The methionine-homocysteine cycle and its effects on cognitive diseases. Altern Med Rev 8(1):7–19
Naz S, dos Santos DCM, García A, Barbas C (2014) Analytical protocols based on LC–MS, GC–MS and CE–MS for nontargeted metabolomics of biological tissues.pdf. Bioanalysis 1757–6180:1657–1677
Niu Q, Zhang H, Li X, Li M (2010) Benzo[a]pyrene-induced neurobehavioral function and neurotransmitter alterations in coke oven workers. Occup Environ Med 67(7):444–448
Patel B, Das SK, Patri M (2016) Neonatal benzo[a]pyrene exposure induces oxidative stress and DNA damage causing Neurobehavioural changes during the early adolescence period in rats. Dev Neurosci 38(2):150–162
Perera FP, Tang D, Wang S, Vishnevetsky J, Zhang B, Diaz D, Camann D, Rauh V (2012) Prenatal polycyclic aromatic hydrocarbon (PAH) exposure and child behavior at age 6-7 years. Environ Health Perspect 120(6):921–926
Popa-Wagner A, Mitran S, Sivanesan S, Chang E, Buga AM (2013) ROS and brain diseases: the good, the bad, and the ugly. Oxidative Med Cell Longev 2013:963520
Prut L, Belzung C (2003) The open field as a paradigm to measure the effects of drugs on anxiety-like behaviors: a review. Eur J Pharmacol 463(1–3):3–33
Qiu C, Peng B, Cheng S, Xia Y, Tu B (2013) The effect of occupational exposure to benzo[a]pyrene on neurobehavioral function in coke oven workers. Am J Ind Med 56(3):347–355
Reitzer L (2004) Biosynthesis of glutamate, aspartate, asparagine, L-alanine, and D-alanine. EcoSal Plus 1(1)
Robert K, Vialard F, Thiery E, Toyama K, Sinet PM, Janel N, London J (2003) Expression of the cystathionine beta synthase (CBS) gene during mouse development and immunolocalization in adult brain. J Histochem Cytochem 51(3):363–371
Rodier PM (1995) Developing brain as a target of toxicity. Environ Health Perspect 103(6):73–76
Rodrigues, T. B. and S. Cerdán (2007). 1.4 the cerebral tricarboxylic acid cycles. 63–91
Saiki S, Yamaguchi K, Chijiiwa K, Shimizu S, Hamasaki N, Tanaka M (1997) Phosphoenolpyruvate prevents the decline in hepatic ATP and energy charge after ischemia and reperfusion injury in rats. J Surg Res 73(1):59–65
Saunders CR, Ramesh A, Shockley DC (2002) Modulation of neurotoxic behavior in F-344 rats by temporal disposition of benzo(a)pyrene. Toxicol Lett 129:33–45
Smart KF, Aggio RB, Van Houtte JR, Villas-Boas SG (2010) Analytical platform for metabolome analysis of microbial cells using methyl chloroformate derivatization followed by gas chromatography-mass spectrometry. Nat Protoc 5(10):1709–1729
Tatem KS, Quinn JL, Phadke A, Yu Q, Gordish-Dressman H, Nagaraju K (2014) Behavioral and locomotor measurements using an open field activity monitoring system for skeletal muscle diseases. J Vis Exp 91:51785
Tchekalarova J, Kubova H, Mares P (2005) Postnatal caffeine exposure: effects on motor skills and locomotor activity during ontogenesis. Behav Brain Res 160(1):99–106
Thomas MM, Sulek K, McKenzie EJ, Jones B, Han TL, Villas-Boas SG, Kenny LC, McCowan LM, Baker PN (2015) Metabolite profile of cervicovaginal fluids from early pregnancy is not predictive of spontaneous preterm birth. Int J Mol Sci 16(11):27741–27748
Tonin AM, Ferreira GC, Schuck PF, Viegas CM, Zanatta A, Leipnitz G, Seminotti B, Duvall Wannmacher CM, Wajner M (2009) Inhibition of creatine kinase activity by lysine in rat cerebral cortex. Metab Brain Dis 24(2):349–360
Troen AM (2005) The central nervous system in animal models of hyperhomocysteinemia. Prog Neuro-Psychopharmacol Biol Psychiatry 29(7):1140–1151
Viberg H, Mundy W, Eriksson P (2008) Neonatal exposure to decabrominated diphenyl ether (PBDE 209) results in changes in BDNF, CaMKII and GAP-43, biochemical substrates of neuronal survival, growth, and synaptogenesis. Neurotoxicology 29(1):152–159
Villa RF, Ferrari F, Gorini A (2013) ATP-ases of synaptic plasma membranes in striatum: enzymatic systems for synapses functionality by in vivo administration of L-acetylcarnitine in relation to Parkinson’s disease. Neuroscience 248:414–426
Wang X, Zhang J, Huang Q, Alamdar A, Tian M, Liu L, Shen H (2015) Serum metabolomics analysis reveals impaired lipid metabolism in rats after oral exposure to benzo(a)pyrene. Mol BioSyst 11(3):753–759
Wu G, Fang YZ, Yang S, Lupton JR, Turner ND (2004) Glutathione metabolism and its implications for health. J Nutr 134(3):489–492
Wyse AT, Netto CA (2011) Behavioral and neurochemical effects of proline. Metab Brain Dis 26(3):159–172
Xu MY, Sun YJ, Wang P, Xu HY, Chen LP, Zhu L, Wu YJ (2015) Metabolomics analysis and biomarker identification for brains of rats exposed subchronically to the mixtures of low-dose cadmium and chlorpyrifos. Chem Res Toxicol 28(6):1216–1223
Yang K, Chen C, Cheng S, Cao X, Tu B (2017) Effects of benzo(a)pyrene exposure on the ATPase activity and calcium concentration in the hippocampus of neonatal rats. Int J Occup Med Environ Health 30(2):203–211
Ying W (2008) NAD+/NADH and NADP+/NADPH in cellular functions and cell death: regulation and biological consequences. Antioxid Redox Signal 10(2):179–206
Zhang H, Nie J, Li X, Niu Q (2013) Association of aryl hydrocarbon receptor gene polymorphism with the neurobehavioral function and autonomic nervous system function changes induced by benzo[a]pyrene exposure in coke oven workers. J Occup Environ Med 55(3):265–271
Zhang L, Liu X, You L, Zhou D, Wang Q, Li F, Cong M, Li L, Zhao J, Liu D, Yu J, Wu H (2011) Benzo(a)pyrene-induced metabolic responses in Manila clam Ruditapes philippinarum by proton nuclear magnetic resonance ((1)H NMR) based metabolomics. Environ Toxicol Pharmacol 32(2):218–225
Zhou X, Liu L, Zhang Y, Pu J, Yang L, Zhou C, Yuan S, Zhang H, Xie P (2016) Metabolomics identifies perturbations in amino acid metabolism in the prefrontal cortex of the learned helplessness rat model of depression. Neuroscience 343:1–9
Funding
This report was financially supported by National Natural Science Foundation of China (Grant No. 81502777 and Grant No. 81372957).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest in the submission of this manuscript.
Rights and permissions
About this article
Cite this article
Li, C., Wang, J., Su, Q. et al. Postnatal Subacute Benzo(a)Pyrene Exposure Caused Neurobehavioral Impairment and Metabolomic Changes of Cerebellum in the Early Adulthood Period of Sprague-Dawley Rats. Neurotox Res 33, 812–823 (2018). https://doi.org/10.1007/s12640-017-9832-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12640-017-9832-8