Abstract
Snails are good source of protein and one of the delicacies in majority of the areas in the Philippines. However, they may act as an intermediate host for several parasites such as Angiostrongylus cantonensis. This parasite is commonly found in South East Asia especially in agricultural countries like the Philippines. Hence, the present study aims to determine the prevalence of A. cantonensis in their intermediate host in Village Bagong Sikat, which is situated at the rice granary of the Philippines. A total of 947 snails were randomly collected within 50 m × 50 m transect line through handpicked method. The third larvae (L3) stage from the snails was recovered through artificial tissue digestion. Results showed that 173 out of 947 snails (18.27%) were found positive with A. cantonensis L3 larvae. Three species of snails were recovered having Melanoides tuberculata to have the highest prevalence of A. cantonensis (21.54%) followed by Pomacea canaliculata (17.75%) and Vivipara angularis (17.74%). Statistical analysis further showed that prevalence of A. cantonensis and the length of snails has no significant difference (p > 0.05). The species of snails collected and found infected in the present study are considered part of the delicacies of the locals in Nueva Ecija. Thus, these results highlight the need to raise awareness of the locals regarding the zoonotic potential of A. cantonensis, which are of public health importance.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Rat lungworm, Angiostrongylus cantonensis, is a zoonotic and parasitic nematode thriving in the pulmonary arteries of rats. This was first detected in China and over the past 10 years, its occurrences have been reported that it is endemic to Southeast Asia and the Pacific islands (Pien and Pien 1999; Lv et al. 2009). A. cantonensis’ life cycle involves rodents as a definitive host and different species of gastropods as intermediate hosts (Tunholi-Alves et al. 2012; Stockdale-Walden et al. 2015). However, since humans are not a natural host of A. cantonensis, this may cause eosinophilic meningitis and central nervous system angiostrongyliasis if these are accidentally ingested. Clinical manifestation of this disease includes mild to severe headache, neck stiffness, and meningoencephalitis with permanent neurological injury or even death (Pien and Pien 1999; Luessi et al. 2009; Tujan et al. 2016; Dard et al. 2017).
Occurrence of A. cantonensis was observed in rats and some species of snails in the rice granary of the Philippines, Nueva Ecija, specifically at the town of Muñoz (Tujan et al. 2016; Castillo and Paller 2018). However, occurrence of A. cantonensis in different species of snails were not yet determined in the previous studies, particularly in Village Bagong Sikat, Muñoz, Nueva Ecija. It is significant to present stronger evidence regarding the presence of Angiostrongylus cantonensis in these possible intermediate hosts since most of the residents of the community make these hosts as their food for delicacy. In addition, A. cantonensis was considered as one of the “neglected pathogens” due to the absence of awareness of the society about it (Dalton et al. 2017). Hence, this study was conducted to determine the prevalence of Angiostrongylus cantonensis in every species of snails that were collected in Village Bagong Sikat, Muñoz, Nueva Ecija.
Materials and methods
Ethical considerations
Prior to the conduct of the study, the protocol was approved by the research ethical panel of the Institute with accordance with Administrative Order No. 40 series of 1999 otherwise known as “Rules and Regulations on the Conduct of Scientific Procedures Using Animals” pursuant to Republic Act no. 8485 otherwise known as the “Animal Welfare Act of 1998” and “R.A 9147 or the Wildlife Resource Conservation and Protection Act of 2001”. The survey component of this study was conducted with informed consent from the selected residents of Village Bagong Sikat as respondents, as well as proper consent from the Local Municipal Office. All information obtained during the survey was provided by the respondents voluntarily. Only the information relevant to the study was presented.
Collection of snails
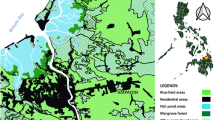
Snails were collected randomly through handpicked method from the surface of the soil and vegetative areas of Village Bagong Sikat Science City of Muñoz, Nueva Ecija along 50 m × 50 m transect lines for 6 weeks (June–July 2018). The collected samples were transported to the laboratory of CAS-Central Luzon State University, Science City of Muñoz for dissection, preservation and processing. Before the snails were processed, the length (cm) of each snails was measured using Vernier caliper and categorized based on their sizes using frequency distribution. Samples from each species were transferred in a vial with 30 ml of 70% ethanol and it was identified through its external morphologies (Igbinosa et al. 2016).
Snail processing for artificial tissue digestion
The animal tissue was slowly removed from the shell using needle nose pliers and it was chopped into small pieces for about 1–22 mm. It was subjected to tissue digestion containing pepsin solution (1000 ml of distilled water, 1 ml HCl and 1 g pepsin) with 400 rpm at 37 °C for 30 min to 1 h (Castillo and Paller 2018). The digested tissue was filtered using a strainer and the filtrate was placed in clean test tube. It was vortexed for 2 min then centrifuged for 10 min at 300–400 rpm. The distinct characteristics of A. cantonensis such as filiform body, tapering and transparent smooth rounded head were observed under compound light microscope.
Survey on knowledge and practices of locals on parasites from snails
The knowledge and practices of locals were documented through an interview with the head of each household. Meanwhile, the number of locals interviewed was computed using Slovin’s formula wherein 165 household numbers were obtained.
Statistical analysis
Prevalence of A. cantonensis were computed by
The correlation of the length of the snails with the prevalence of A. cantonensis was determined using Pearson correlation coefficient through SPSS software. In addition, the risk factors associated for the transmission of this zoonotic parasite were determined through frequency distribution.
Results
A total of 947 snail samples were collected from the three sampling sites in Village Bagong Sikat, Science City of Muñoz, Nueva Ecija which corresponds to a total of three (3) species of snails namely: Pomacea canaliculata, Vivipara angularis and Melanoides tuberculata.
Prevalence of Angiostrongylus cantonensis
A total of 173 snails out of 947 (18.27%) were found to be infected with Angiostrongylus cantonensis larva. In addition, distinct characteristics of A. cantonensis were observed (Fig. 1). Meanwhile, it was recorded that Melanoides tuberculata had the highest prevalence of 21.54% followed by Pomacea canaliculata with 17.75% and Vivipara angularis with 17.74% (Table 1). Furthermore, it was noted in the current study that Pomacea canaliculata were obtained in all Zones of Barangay Bagong Sikat. The highest prevalence of A. cantonensis larvae of P. canaliculata was recorded in Zone Matulungin having 30.16%, followed by Zone Masagana (12.90%) and Zone Maligaya (11.33%). On the other hand, V. angularis were only recovered in two zones. The highest prevalence of A. cantonensis in V. angularis was in Zone Maligaya (22.78%) while lower prevalence (11.11%) was recorded on Zone Masagana. M. tuberculata was only obtained from the irrigational canal of Zone Masagana with a prevalence of 21.54% A. cantonensis infection.
Morphology of A. cantonensisa tapering posterior tail (violet arrow) with small protruding (red arrow), b gelatinous body with refractive granules (blue arrow), c male developing copulatory bursa (green arrow) with intestinal wall (gray arrow), d, e female, filiform and cylindrical shape of head f whole larvae (color figure online)
Correlation between the length of the snail and the prevalence of Angiostrongylus cantonensis
Results showed that the prevalence of A. cantonensis is differently correlated in each species. The length of V. angularis which ranges from 0.6 cm–2.4 cm, showed a strong linear correlation (r2 = 0.616486, p > 0.05) to the prevalence of A. cantonensis (Fig. 2); P. canaliculata which ranges from 1.4 to 3.8 cm showed a very weak correlation (r2 = 0.186579, p > 0.05) (Fig. 3); M. tuberculata which ranges from 1.75 to 5.35 cm, showed moderate correlation (r2 = 0.51567, p > 0.05) to the prevalence of A. cantonensis (Fig. 4).
Although the highest prevalence of A. cantonensis in all of the species of snails were observed in the larger group, statistical analysis showed that there was no significant difference between the length of the snails to the prevalence of A. cantonensis (p > 0.05). This implies that there could have been other factors that may have contributed to A. cantonensis infection aside from the size of snails.
Risk factors associated for zoonotic transmission
It was showed that 65.91% of the respondents eat snails due to its availability and since it is easy to collect in the fields. Moreover, it was found that respondents from residential areas do not eat snails, but those families who are living near or within agricultural areas do. The results also showed that males (51.85%) consume snails than women (48.15%). These findings were also supported by the assessment of the different status of household member whereas the manly jobs such as vendor, construction worker and carpenter obtained the highest percentage of eating snails (41.30%) followed by students (26.19%) and housewives (16.17%) (Table 2). It was also noted that they eat mostly M. tuberculata (49.83%) followed by V. angularis (43.05%) and P. canaliculata (7.12%).
Meanwhile, 61 (7.58%) household members eat rodent meat, which is a definitive host of A. cantonensis and 5 households (3.03%) eat raw or fresh “kinilaw” snails. Household members also prefer to eat undercooked vegetables (63.03%) and the major sources of drinking water on three Zones are mineral water (47.30%) and steam pump (41.22%). For the practices of locals, 132 households were eating using with their hands (80%); most locals used their hands three (3) to five (5) times per week (48.48%). As for the awareness of inhabitants in different zoonotic parasites, it was revealed that there are no household members who are aware of these parasites however, 10 (6.06%) out of 165 households stated that snails can be a possible source of bacteria such as Escherichia coli (Table 2).
Discussions
Prevalence of Angiostrongylus cantonensis
The obtained intermediate hosts which thrive in the agricultural areas of Village Bagong Sikat were positive for the infection of A. cantonensis. The definitive host, rats, which are dominant in rice farm villages may pose as key drivers for the transmission of A. cantonensis to its intermediate hosts (Tujan et al. 2016; Castillo and Paller 2018).
Egg masses of P. canaliculata attached to the vegetation were highly observed on Zone Matulungin (Site 2) and they have high feeding rate during their reproductive stage (Brito and Joshi 2016). In line with this, P. canaliculata are more prone for the infection of A. cantonensis which may accidentally harbored through scraping. Meanwhile, no egg masses were observed in Zone Maligaya (Site 1) and Masagana (Site 3) and it also have cultivated farm which are not a perfect habitat for the definitive hosts because of lack of food resources. Castillo and Paller (2018) stated that several definitive hosts such as Rattus norvegicus (house rat) were not commonly found on rural areas due to cultivation, where mean intensity is much higher compared to Rattus tanezumi (field rat); species commonly found in agricultural areas..
Evidently, high prevalence of V. angularis was recorded on Zone Maligaya (Site 1) due to the ambient temperature in the creek. This result is supported by the study of Dias and Lima (2011) and Mozzer et al. (2014), wherein they stated that the third stage larvae of Angiostrongylus sp. have higher survival rate and better developmental time in lower temperature resulting on increasing of potential for the transmission of larvae to its hosts. Moreover, Schug et al. (2018) recorded several cases of Angiostrongylus sp. during autumn and winter months in Germany.
On other hand, high temperature in the creek of Zone Masagana (Site 3) was observed. Viability of Metastongyloidea larvae such as A. cantonensis greatly reduced as the temperature increases because its metabolic rate was greatly affected (Dias and Lima 2011). Unlike on Zone Maligaya (Site 1), competition was observed which results to less susceptibility for parasitic infection due to the limitation in food resources. Tujan et al. (2016) found that the collected V. angularis Muñoz, Nueva Ecija was negative A. cantonensis larvae. Hence, this is the first report of A. cantonensis larvae infection in V. angularis in the Philippines.
M. tuberculata are the dominant species in the irrigational canal of Zone Masagana (Site 3) resulting for the greater resources than other species of snails. M. tuberculata is considered as competitively superior k-adapted species and takes over a wider area than in native species of snails through interspecific competition (Hamalainen and Jarvinenen 2012; Raw et al. 2016). For this reason, greater resources may increase the chances of infection.
Correlation between the length of the snail and the prevalence of Angiostrongylus cantonensis
Results showed that the prevalence of A. cantonensis in all species of each snails tend to be higher in a larger sizes. Ibrahim (2007) and Martin and Cabrera (2018) stressed that larger hosts provide more space and resources and may accumulate infection on their young stage, thus increasing larger populations of parasite. But then again, larger-older snails may have been exposed to more larvae. This was supported by the study of Minchella (1985) in which large snails is older within a given population. Parasite infections were directly proportional to increase with age since they have a greater chance of infection because of their greater contact with parasites during their lifetime (Graham 2003; Ibrahim 2007; Castillo and Paller 2018).
Statistical analysis, on the other hand, showed that there was no significant difference between the lengths of the snails to the prevalence of A. cantonensis (p > 0.05) which suggest that there could have been other factors such as weight and age that may have contributed to A. cantonensis infection aside from the size of snails. In coherence with the study of Oliveira et al. (2015), they state that there was no significant correlation between the snail size and prevalence of A. cantonensis.
Risk factors associated for zoonotic transmission
Results showed that males obtained a higher percentage in terms of snail consumption since they occasionally eat this with alcoholic drinks. Meanwhile, most of locals prefer to eat snails (raw or cooked) and undercooked vegetables. One of the major causes of Angiostrongyliasis all over the world was due from ingesting of uncooked hosts such as shrimps, crabs and frogs and under- or poorly cooked contaminated vegetables and water (Simoes et al. 2016; Nguyen et al. 2017). On the other hand, eating rodent meat is a huge threat for the possible transmission of A. cantonensis to human since in the study of Castillo and Paller (2018) on the neighbouring Villages of Bagong Sikat, 100% prevalence was observed on different definitive host of A. cantonensis. The most vital pathways of infections are those definitive and intermediate hosts which might carry large parasite loads (Cowie 2013). Moreover, almost half of the respondents use steam pump as their source of drinking water. Since they live in agricultural areas, a possible transmission of A. cantonensis from the water of steam pump to human is a serious risk factor especially if they do not regularly clean this underground water because A. cantonensis can survive in water for 72 h (Cowie 2013).
In connection, there could be a high chance for the transmission of zoonotic parasites of snails such as A. cantonensis because majority of household member use bear hands in eating. The biggest threat for the transmission of this parasite is through hand to mouth after handling infected hosts (Cowie 2013) and other outdoor activities (Kramer et al. 2018) related to farming. Lack of awareness regarding A. cantonensis was also highly observed. Hence, human awareness is essential to establish in order to control the transmission of this zoonotic parasite since some antihelminthic drugs such as albendazole for eosinophilic mengitis is still debated (Gederen 2010).
Summary and conclusion
In conclusion, the result of the current study has public health significance since survey revealed that the species of intermediate hosts, which were found positive with Angiostrongylus cantonensis infection, are part of the delicacy of the locals. Thus, authorities such as the local municipal health officers may help in spreading the knowledge about this zoonotic parasites and proper cooking of snails to minimize the chance of infection of this parasite. In addition, suitable handling of different intermediate host and vegetables that may get contaminated should be practiced.
Recommendation
The following recommendations are given based from the findings: (1) determine the prevalence of A. cantonensis during wet and dry season. (2) Take the locals under serological examination with the help of licensed health officers to know if there is any case of Angiostrongyliasis
References
Brito F, Joshi R (2016) The golden apple snail Pomacea canaliculata: a review on invasion, dispersion and control. Research Information LTD, Burnham
Castillo DS, Paller V (2018) Occurrence of Angiostrongylus cantonensis in rodents from the rice granary of the philippines and associated risk factors for zoonotic transmission. J Parasit Dis. https://doi.org/10.1007/s12639-018-1005-z
Cowie RH (2013) Pathways for transmission of angiostrongyliasis and the risk of disease associated with them. Hawai’i J Med Public Health 72(6 Supplement 2):70–74
Dalton M, Fenton H, Cleveland C, Elsmo E, Yabsley M (2017) Eosinophilic meningoencephalitis associated with rat lungworm (Angiostrongylus cantonensis) migration in two nine-banded armadillos (Dasypus novemcinctus) and an opossum (Didelphis virginiana) in the southeastern United States. Int J Parasitol Parasites Wildl. https://doi.org/10.1016/j.ijppaw.2017.05.004
Dard C, Piloquet JE, Qvarnstrom Y, Fox L, Mkada H, Hebert JC, Mattera D, Harrois D (2017) First evidence of Angiostrongyliasis caused by Angiostrongylus cantonensis in Guadeloupe, Lesser Antilles. Am Soc Trop Med Hyg 96(3):692–697
Dias S, Lima W (2011) Effect of temperature on activity of third-stage larvae of Angiostrongylus vasorum. Parasitol Res. https://doi.org/10.1007/s00436-011-2624-9
Gederen P (2010) Antihenlminthic drugs. Side effects of drugs annual
Graham A (2003) Effects of snail size and age on the prevalence and intensity of avian schistosome infection: relating laboratory to field studies. J Parasitol 89(3):458–463
Hamalainen E, Jarvinenen S (2012) Snails: biology, ecology and conservation. Animal science, issues and prefessions. Nova Science Publisher, New York
Ibrahim M (2007) Prevalence and intensity of angiostrongylus cantonensis in freshwater snails in relation to some ecological and biological factors. Parasite 14:61–70. https://doi.org/10.1051/parasite/200714106
Igbinosa B, Isaac C, Adamui O, Adeleke G (2016) Parasites of edible land snails in Edo State, Nigeria. Helminthologia 53:331–335. https://doi.org/10.1515/helmin-2016-0031
Kramer K, Posner J, Gosnell W (2018) The role of gastropod mucus in the transmission of Angiostrongylus cantonensis, a potentially serious neurological infection. ACS Chem Neurosci. https://doi.org/10.1021/acschemneuro.7b00491
Luessi F, Sollors J, Torzewski M, Muller H, Siegel E, Blum J, Sommer C, Vogt T, Thomke F (2009) Eosinophilic Meningitis due to Angiostrongylus cantonensis in Germany. International Society of Travel Medicine, 1195-1982. J Travel Med 16(4):292–294
Lv S, Zhang Y, Liu H, Hu L, Yang K, Steinmann P, Chen Z, Wang L, Utzinger J, Zhou X (2009) Invasive snails and an emerging infectious disease: results from the first national survey on Angiostrongylus cantonensis in China. www.plosntds.org
Martin GL, Cabrera EC (2018) Morphological characterization of emerging cercariae among lymnaeid snails from Baranggay Cawongan, Padre Garcia, Batangas, Philippines. J Parasitol Res. https://doi.org/10.1155/2018/5241217
Minchella D (1985) Host life-history variation in response to parasitism. Parasitology. https://doi.org/10.1017/S0031182000049143
Mozzer LR, Coaglio A, Dracz R, Ribeiro V, Lima W (2014) The development of Angiostrongylus vasorum (Baillet, 1866) in the freshwater snail Pomacea canaliculata (Lamarck, 1822). J Helminthol 89:755–759. https://doi.org/10.1017/s0022149x14000856
Nguyen Y, Rossi B, Argy N, Baker C, Nickel B, Marti H, Zarrouk V, Houzé S, Fantin B, Lefort A (2017) Autochthonous case of eosinophilic meningitis caused by Angiostrongylus cantonensis, France, 2016. Emerg Infect Dis 23(6):1045–1046
Oliveira A, Gentile R, Maldonado A Jr, Torres EJL, Thiengo SC (2015) Angiostrongylus cantonensis infection in Molluscs in the Municipality of São Gonçalo, a Metropolitan Area of Rio de Janeiro, Brazil: role of the invasive species Achatina fulica in parasite transmission dynamics. Mem Inst Oswaldo Cruz 110(6):739–744. https://doi.org/10.1590/0074-02760150106
Pien F, Pien B (1999) Angiostrongylus cantonensis: Eosinophilic Meningitis. Straub Clinic and Hospital, University of Hawaii John A. Burns School of Medicine, Honolulu
Raw J, Perissinotto R, Miranda N, Peer N (2016) Feeding Dynamics of Melanoides tuberculata (Muller, 1774). J Molluscan Stud. https://doi.org/10.1093/mollus/eyv070
Schug K, Kramer F, Scaper R, Hirzmann J, Failing K, Hermosilla C, Taubert A (2018) Prevalence Survey on Lungworm Angiostrongylus vasorum, Crenosoma vulpis, Eucoleus aerophilus) Infections of Wild Red Foxes (Vulpes vulpes) in Central Germany. Parasites and Vectors 11:85
Simoes R, Monte T, Boia M (2016) Junior A. Encephalitis caused by Helminths. Laboratorio de Biologia e Parasitologia de Mamiferos Silvestres Reservatorios, Fundacao Oswaldo Cruz, Brazil. SMGroup
Stockdale-Walden H, Slapcinsky J, Qvarnstrom Y, McIntosh A, Bishop H, Rosseland B (2015) Angiostrongylus cantonensis in introduced gastropods in Southern Florida. J Parasitol 101(2):156–159
Tujan MA, Fontanilla IA, Paller VG (2016) Vectors and spatial pattern of Angiostrongylus cantonensis in selected rice-farming villages of Muñoz, Nueva Ecija, Philippines. J Parasitol Res 2016:3085639
Tunholi-Alves V, Tunholi V, Pinheiro J, Thiengo S (2012) Effects of infection by larvae of Angiostrongulus cantonensis (Nematoda, Metastongylidae) on the metabolism of the experimental intermediate host Biomphalaria glabrata. Exp Parasitol 131:143–147
Acknowledgements
We would like to express our deepest appreciation and gratitude to the locals of Village Bagong Sikat, Munoz, Nueva Ecija, who have the great participation for the success of our study and CLSU-Environmental Science students, faculty and laboratory assistants, who helped, guide and provided us facilities and equipment.
Author information
Authors and Affiliations
Contributions
Conceived and designed the experiments: JRC, CJTQ, DSCC, KOP; Performed the experiment, data analysis: JRC, CJTQ; all authors participated in writing the final paper.
Corresponding author
Ethics declarations
Conflict of interest
None declared.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Cawas, J.R., Quisao, C.J.T., Castillo, D.S.C. et al. Prevalence of Angiostrongylus cantonensis among different species of snails in the village of Bagong Sikat Muñoz, Nueva Ecija, Philippines and its associated risk factors for zoonotic transmission. J Parasit Dis 44, 388–394 (2020). https://doi.org/10.1007/s12639-020-01200-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12639-020-01200-0