Abstract
Cystic echinococcosis is a potential zoonotic helminthic disease affect a broad spectrum of mammals including humans worldwide. The economic impact of the disease interestingly requires updated gathering information on the prevalence in slaughtered animals. Accordingly, in the current study, 573 camels, 4300 sheep, and 1235 pigs were surveyed in four Egyptian municipal abattoirs. Among those, 62 (10.82%) camels, 33 (0.77%) sheep and 3 (0.24%) pigs had cystic echinococcosis in lungs, livers and spleen. The diversity of cysts revealed that positive-cystic echinococcosis animals from all species were detected in El-Basatin abattoir. In El-Monieb abattoir, sheep only were infected. In El-Waraa and Beni-Suef abattoirs, cysts were seen in camels only. Infected animals included both sexes. In camels, lungs were the most affected organs, while in sheep, livers were the most abundant. In pigs, hydatid cysts were detected only in lungs. Moreover, camel cysts were mostly large-sized (diameter > 10 cm) with smaller cysts are also recorded. In sheep, small-, moderately- and large-sized cysts were equally present. Cysts in pigs were large-sized. Camels showed a higher percentage of fertile cysts (46.77%; 29/62) followed by sheep (21.21%; 7/33). Calcified/degenerated cysts were less in camels (38.71%; 24/62) than in sheep (75.76%). Non-viable cysts were the less frequent in both species (14.52% in camels and 3.03% in sheep). All cysts recovered from pigs were fertile. In Egypt, governmental agencies and veterinary authorities are asked to potentially eradicate stray dogs (the main definitive host) and towards the proper hygienic disposal of infected offal in abattoirs to minimize the prevalence of cystic echinococcosis.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
In Egypt, domestic animals including camels and sheep as well as pigs (for some religious communities) are important sources of animal proteins. Parasitism is considered as one of the direct constrains that limit the livestock production. Among those parasitic infections, cystic echinococcosis, caused by unilocular hydatid cysts, is a potentially frequent (Getachew et al. 2012).
Cystic echinococcosis (CE), or hydatidosis, is a potential global zoonotic disease occurs as a result of the infection with the larval stage, hydatid cyst, of the common dwarf dog tapeworm, Echinococcus granulosus (Thompson and McManus 2002; Elmahdi et al. 2004). The parasite has a worldwide distribution, particularly in rural districts where the dog (definitive host)-livestock (intermediate hosts) synanthropic cycle is prevalent causing severe economic losses via condemnation of livers in abattoirs as well as direct effects on the carcasses and serious health problems even in humans (Torgerson and Budke 2003; Thompson 2008). Annually, the economic burden of cystic echinococcosis on the global livestock industry has been estimated to be over $2 billion (Scala et al. 2006). The disease is predominant in endemic regions like the Middle East, Arabic North Africa from Morocco to Egypt, South America and India (Ersahin et al. 1993; Garg et al. 2002; Kouidri et al. 2012). It contributes to the poor overall development and work productivity in the endemic areas (Amer et al. 2015). In Egypt, CE is endemic in both animals and humans (Abdel Aaty et al. 2012). To date, more than one million people suffer from CE, with several individuals exhibiting severe clinical signs which become life threatening if left untreated (Craig et al. 2003, 2007; Abdulhameed et al. 2018).
The cosmopolitan widespread of the parasite might be referred to the broad range of domestic and wild intermediate hosts inside them the parasite metacestodes adapt themselves adequately (Thompson and McManus 2002). Adults Echinococcus granulosus habit in the small intestines of dogs and related wild carnivores and their metacestodes are located in various tissues and organs, like lungs, livers and spleen, of camels, sheep, cattle, pigs, horses and occasionally humans, with lungs and/or livers are the most frequently parasitized organs (Budke et al. 2006; Fikire et al. 2012; Melaku et al. 2012; Faraji et al. 2015; Abdulhameed et al. 2018). In the infected organs, cysts grow slowly, with minimal clinical signs observed in livestock animals, and they are only detected during a routine meat inspection in abattoirs (Abdulhameed et al. 2018; Parkoohi et al. 2018).
Among risk factors that increase the possibility of the occurrence of dogs-livestock life cycle, the improper disposal of dead carcasses, unauthorized slaughtering, the breeding and grazing of domestic animals in such way that allows a close contact with stray dogs, the almost lack of awareness of breeders and farmers to the regular use of anthelmintics, the ease access of stray dogs to the offal of slaughtered animals and the carelessness of some authorities to the periodic elimination of stray dogs (Christodoulopoulos et al. 2008; Ibrahim 2010; Singh et al. 2014).
In terms of assessing CE, questionnaires of abattoirs in all seasons, particularly in endemic areas, are recommended for obtaining data on diseases, interestingly subclinical infections, of the livestock because it is considered the most available way to detect CE in domestic animals (Craig 1997; Njoroge et al. 2002; Ibrahim 2010). The current study was conducted to evaluate the recent data on the prevalence and diversity of cystic echinococcosis among Egyptian slaughtered animals (camels, sheep and pigs) posing on the public health significance to minimize the possible hazards and economic losses of such disease.
Materials and methods
Study area
The survey included four abattoirs in three Egyptian provinces; El-Monieb and El-Waraa abattoirs in Giza (30°01′N 31°13′E) province, El-Basatin abattoir in Cairo (30°2′N 31°14′E) province and Beni-Suef abattoir in Beni-Suef (29°04′N 31°05′E) province. A lot of farmers live in the study area economically depend on cultivation of crops and animals breeding. The livestock bred in those areas included large numbers of camels, sheep and pigs as well as small groups of cattle.
Sampling
A total of 4300 sheep, 573 camels and 1235 pigs examined for the presence of hydatid cyst during meat inspection in abattoirs during the period from May 2018 to December 2018.Sheep were slaughtered in El-Monieb and El-Basatin abattoirs, camels were slaughtered in El-Basatin, El-Waraa and Beni-Suef abattoirs, and pigs were slaughtered in El-Basatin abattoir only. In the later, all species were slaughtered. During the ante-mortem examination, each animal was given an identification number. The age (based on dentition and owner’s information), sex, breed and origin of animals were recorded. All examined species included both sexes. Concerning the age of slaughtered species, camels aged 4–8 years, sheep aged 6 months-1 year (females up to 5 years) and pigs aged 5–18 months. Hydatid cysts were collected from infected livers, lungs and spleen of slaughtered animals. Specimens were carefully transported in an ice box to the laboratory Parasitology of Faculty of Veterinary Medicine in Beni-Suef University for further investigations.
Routine parasitological examination
Macroscopic examination
Each organ was examined macroscopically by visual inspection, palpation and systematic incision. Where needed, one/more incisions were made to detect small-sized hydatid cysts (Kebede et al. 2011; Getachew et al. 2012). The infected organs from each CE-positive animal were collected. The total number of hydatid cysts were counted per infected organ and recorded. Cysts were grossly examined for the detection of being either fully formed or degenerated/calcified. The size, fertility and viability were encountered. The diameter of cysts was measured (the average diameter of 5 cysts was considered) and they were categorized as large (diameter > 10 cm), medium (diameter 5–10 cm) and small (diameter < 5 cm) (Oostburg et al. 2000; Kebede et al. 2011). The weight of cysts (n = 24 in camels, n = 15 in sheep and one cyst in pigs) was measured by an electric balance (Njoroge et al. 2002).
Microscopic examination
The cyst viability (fertility) was determined according to Daryani et al. (2006). The presence of protoscolices as white dots on the inner germinal layer as well as in the hydatid fluid indicates a fertile cyst (Kebede et al. 2009). Accordingly, cysts may be fertile (viable), calcified (non-viable) (with dead protoscolices) or sterile (no protoscolices). Briefly, collected cysts (from each organ) were carefully incised and examined. The cyst wall was penetrated using a sterile needle and cut with a scalpel and scissors. The contents were collected into a sterile container. A drop of the cyst fluid was added to a microscopic glass slide covered with a slip. The peristaltic movement of viable protoscolices looks like the motility of flame cells. When it becomes doubtful for motility, a drop of 0.1% aqueous eosin solution was added. Alive protoscolices could not be stained (Getachew et al. 2012).
Results
The current study revealed that among slaughtered species, 62 (10.82%) camels, 33 (0.77%) sheep and 3 (0.24%) pigs had cystic echinococcosis in some of the internal organs. It has been found that the highest infection was recorded in camels, while the lowest infection was found in pigs (Table 1). All slaughtered animals were apparently symptomless. Despite all slaughtered species include both sexes, the prevalence of CE relative to sex could not be detected, as the routine daily procedures, by co-workers and assistants, in Egyptian abattoirs make it is too difficult to identify the infected organ belonging to its corresponding animal sex. The detection of cysts in slaughtered animals based on the abattoir inspection with careful determination of them in their predilection organs (lungs, livers and spleen) as well as laboratory identification using the microscopic detection of protoscolices.
The diversity of CE in the surveyed abattoirs revealed that positive-CE animals from camels, sheep and pigs were detected in El-Basatin abattoir. In El-Monieb, infected animals were only sheep. In El-Waraa and Beni-Suef abattoirs, CE was seen in camels only (Table 2).
Three organs were found to be infected; lungs, livers and spleen. In camels, the most infected organ was lungs followed by livers and spleen (Figs. 1, 2), while in sheep, livers was the most predominant infected organ followed by lungs. Spleen of examined sheep had no infection. Surprisingly, one sheep showed a mixed infection of hydatid cyst and the common bladder worm, Cysticercus tenuicollis. Among slaughtered pigs, only three individuals revealed hydatid cysts in their lungs (Table 3 and Fig. 3).
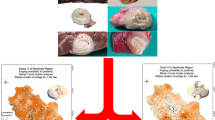
Hydatid cysts recovered from various organs of slaughtered camels. a A large-sized cyst revealed from a lung of a camel. b A heavy weight of a camel-lung cyst indicating a large quantity of the hydatid fluid. Note that it approximately exceeds 790 g. c Multiple hydatid cyst recovered from one lung. d An exposed germinal epithelium of an incised cyst. e Caseous materials from a degenerated lung cyst. f A large-sized cyst from a liver lobe
Hydatid cysts recovered from various organs of slaughtered sheep and pigs. a Multiple large-sized cysts from a liver of sheep. b Multiple small sized-cysts from a liver of sheep. c A cavity left after extirpation of a large-sized sheep liver-hydatid cyst. d A large-sized cyst recovered from a lung of sheep. e A mixed infection of moderate-sized hydatid cyst and the bladder worm, Cysticercus tenuicollis, recovered from an infected sheep liver. f A moderate-sized cyst recovered from a lung of a slaughtered pig
In camels, the majority (48.39%) of cysts were uniquely large-sized containing large quantities of hydatid fluid associated with a less frequency of smaller-sized cysts. Moreover, multiple infections with variable sizes were observed. In sheep, all cyst sizes are found in more or less equal percentages. Swine cysts were large-sized (Table 4).
Concerning cyst viability, it has been found that camels showed a higher percentage of fertile cysts (46.77%; 29/62) followed by sheep (21.21%; 7/33). Calcified/degenerated cysts were less in camels (38.71%; 24/62) than in sheep (75.76%). Sterile (non-viable) cysts were the less frequent in both species (14.52% in camels and 3.03% in sheep). All cysts recovered from pigs were fertile (Table 5).
In camels, the recovered cysts weighed 13.37–790.56 g (average 84.88 g; all from lungs except one cyst from spleen). In sheep, cysts weighed 10.71–233.99 g (average 39.46 g; all from livers). Only one cyst from a lung of a pig could be weighed (29.23 g).
Discussion
Cystic echinococcosis (hydatidosis) is an important veterinary and medical constrain in the underdeveloped countries and the Middle East. The disease is now categorized as a neglected tropical disease, and it is included as a part of the strategic plan of the World Health Organization (WHO) (da Silva 2010; Abbas et al. 2016). The induction of the disease always needs stray dogs as a definitive host and various domestic animals, like camels, sheep and pigs, as well as humans are considered as potential intermediate hosts (Daryani et al. 2007; Ibrahim 2010). The occurrence of the life cycle of the adult tapeworm, Echinococcus granulosus, as well as the wide diversity of the disease recommend that importation of infected animals might be synchronized with the high risk of the disease (Fathi et al. 2011).
In Egypt, the disease is endemic, with variable infection rates were recorded in cattle, goats, sheep and camels (Omar et al. 2013). This is due to being that the environmental conditions are more or less appropriate for the dogs-livestock transmission; the existence of large populations of Echinococcus granulosus-infected stray dogs (used for guarding purposes) as well as the ease access of dogs to abattoirs ingesting the condemned infected offal. In the current study, regional distribution patterns of CE in slaughtered camels, sheep and pigs were assessed in order to re-describe the map of CE in Egypt.
The present study revealed that 10.82% (62/573) of camels, 0.77% (33/4300) of sheep and 0.24% (3/1235) of pigs were CE-positive. These findings were lower than those reported by Rahman et al. (1992) who detected percentages of 31.0, 1.33, 4.62%, respectively in the same species in Egypt. Moreover, Haridy et al. (1998) stated that CE in camels was 5.5–8.2% in the period of 1992–1995. A similar low percentage (0.33%) in slaughtered sheep in municipal abattoirs was reported by Haridy et al. (2006) during the period of 1995–1999. Dyab et al. (2005) demonstrated more or less a lower percentage (7.67%) of CE in slaughtered camels. In the authors’ opinion, the variation in prevalences, particularly in camels which may be inbreed or imported, might be attributed to fluctuating strategy of eradication of stray dogs among various decades, the lack of the understanding of herdsmen to the life cycle of a such parasite as well as the personal behavior of workers about the elimination of infected offal in abattoirs.
A considerable higher percentage (6.4%) of CE in sheep was revealed in the Delta district (in the north), Egypt by Sabri et al. (2005). It is worthy to mention that, the Delta district includes have larger animal population (the Northern Egypt is uniquely possesses higher green areas), so, these areas have a greater risk of CE than the Southeast areas. In other related African countries, previous reports indicated higher percentages in both camels and sheep. In the former, they were 80.0% in Morocco (Pandey et al. 1986), 53.7% in Mauritania (Pangui and Ould 1991), 48.0% in Libya (Ibrahem and Craig 1998) and 59.0% in Sudan (Omer et al. 2010). In sheep, prevalences were 10.58% in Morocco (Azlaf and Dakkak 2006); 10.6% in Ethiopia (Kebede et al. 2009); 11.0% in Sudan (Omer et al. 2010) and 7.7% in Ethiopia (Getachew et al. 2012). In the authors’ opinion, higher infection rates in those countries, in the past, might be related to the discrepancies of the climatic conditions as well as the raising techniques of animals, particularly in Sudan and Ethiopia which have huge populations of the livestock and several large-scaled grazing areas. In such concern, together with the lack of veterinary care general awareness, the opportunity of dogs-livestock transmission was highly possible. Supporting this hypothesis, the current study recorded larger populations of examined and infected animals in El-Basatin and El-Monieb abattoirs due to the agricultural nature of those areas, on the contrast to El-Waraa and Beni-Suef abattoirs. Currently, the lower prevalence (0.24%) of CE in pigs might be attributed to the limitation of breeding of pigs to farms and avoiding breeding by the individuals, and thus, minimizing the possibility of hydatidosis. In Ethiopia, during a survey of CE among domestic animals, Fromsa and Jobre (2011) revealed pigs free from the disease.
The present study demonstrated that CE was predominant in camel lungs and sheep liver, otherwise, both organs in the two species harboured hydatid cysts. In camels, spleen also was infected. The size of cysts varies according the time of formation and establishment inside the intermediate host (Omar et al. 2013). It has been found that lungs and livers are more susceptible to CE. This may be due to the fact that these organs have unique blood circulation characteristics. Lungs have intensive capillary networks and the liver is abundantly supplied with blood from both portal veins and hepatic arteries, therefore, higher prevalences are recorded in those organs (Qingling et al. 2014). The organ involved might depend upon the host species and on both anatomical and physiological factors (Fromsa and Jobre 2011; Omar et al. 2013). Herein, the distribution of cysts in the invaded organs coincided with those detected in previous literature (Rahman et al. 1992; Haridy et al. 2006; Fromsa and Jobre 2011; Omar et al. 2013). Currently, one sheep had a mixed infection with the bladder worm, Cysticercus tenuicollis. This finding argues the fact that stray dogs, the definitive host of the adult tapeworm Taenia hydatigena, were in a close contact with the infected sheep.
Regarding the cysts viability, the present work detected that camels had a higher percentage of fertile cysts (46.77%) followed by sheep (21.21%). Calcified/degenerated cysts were less in camels (38.71%) than in sheep (75.76%). Sterile cysts were the less frequent in both species (14.52% in camels and 3.03% in sheep). Furthermore, cysts recovered from pigs were fertile. In Egypt, a similar finding was reported by Haridy et al. (1998) who revealed the fertility rate of camel cysts is 49.0%. Meanwhile, Omar et al. (2013) detected 66.6% fertility rate in camel cysts and 15.51% in sheep. On the other hand, in Libya, Ibrahem and Craig (1998) reported that 91.6% of examined cysts were fertile.
Conclusion
The present study highlighted on the CE in slaughtered camels, sheep and pigs in four municipal abattoirs in three Egyptian provinces. Topographical variations, grazing system, behaviour of humans as well as the application of strict hygienic and control measures are major incorporated factors. The authorized agencies are recommended to apply proper hygienic instructions in abattoirs towards the elimination of condemned infected offal safely. Moreover, the eradication of stray dogs is a major task. Regular surveillances of CE in Egypt is urgently needed to follow up the updated information of the disease in both animals and humans.
References
Abbas IEA, Al-Kappany YM, Al-Araby MA (2016) Prevalence and molecular characterization of hydatid cyst isolates from cattle in Egypt. Asian J Anim Vet Adv 11(12):794–804
Abdel Aaty H, Abdel-Hameed D, Alam-Eldin Y, El-Shennawy S, Aminou H, Makled SS, Darweesh SK (2012) Molecular genotyping of Echinococcus granulosus in animal and human isolates from Egypt. Acta Trop 121:125–128. https://doi.org/10.1016/j.actatropica.2011.10.014PMID: 22062047
Abdulhameed MF, Habib I, Al-Azizz SA, Robertson I (2018) Cystic echinococcosis in marketed offal of sheep in Basrah, Iraq: abattoir-based survey and a probabilistic model estimation of the direct economic losses due to hydatid cyst. Parasite Epidemiol Control 3:43–51
Amer S, Helal IB, Kamau E, Feng Y, Xiao L (2015) Molecular characterization of Echinococcus granulosus sensu lato from farm animals in Egypt. PLoS ONE 10(3):e0118509. https://doi.org/10.1371/journal.pone.0118509
Azlaf R, Dakkak A (2006) Epidemiological study of the cystic echinococcosis in Morocco. Vet Parasitol 137:83–93
Budke CM, Deplaxes P, Torgerson PR (2006) Global socio-economic impact of CE. Emerg Infect Dis 12(2):296–303
Christodoulopoulos G, Theodoropoulos G, Petrakos G (2008) Epidemiological survey of cestode-larva disease in Greek sheep flocks. Vet Parasitol 153(3–4):368–573
Craig PS (1997) Immunodiagnosis of Echinococcus granulosus and a comparison of techniques for diagnosis of canine echinococcosis. In: Andersen FL, Ouhelli H, Kachani M (eds) Compendium on cystic echinococcosis in Africa and in Middle Eastern countries with special reference to Morocco. Brigham Young University, Provo, UT, pp 85–118
Craig PS, Rogan MT, Campos-Ponce M (2003) Echinococcosis: disease detection and transmission. Parasitology 127:5–20
Craig PS, McManus DP, Lightowlers MW, Chabalgoity JA, Garcia HH, Gavidia CM, Gilman RH, Gonzalez AE, Lorca M, Naquira C, Nieto A, Schantz PM (2007) Prevention and control of cystic echinococcosis. Lancet Infect Dis 7:385–394
da Silva AM (2010) Human echinococcosis: a neglected disease. Gastroenterol Res Pract, 1409872 . https://doi.org/10.1155/2010/583297
Daryani A, Aiaei R, Arab R, Sharif M (2006) Prevalence of hydatid cyst in slaughtered animals in Northwest Iran. J Anim Vet Adv 5:330–334
Daryani A, Alaei R, Arab R, Sharif M, Dehghan MH, Ziaei H (2007) The prevalence, intensity and viability of hydatid cysts in slaughtered animals in the Ardabil province of Northwest Iran. J Helminthol 81:13–17
Dyab KA, Hassanein R, Hussein AA, Metwally SE, Gaad HM (2005) Hydatidosis among man and animals in Assiut and Aswan Governorates. J Egypt Soc Parasitol 35:157–166
Elmahdi IE, Ali QM, Magzoub MM, Ibrahim AM, Saad MB, Romig T (2004) Cystic echinococcosis of livestock and humans in central Sudan. Ann Trop Med Parasitol 98:473–479
Ersahin Y, Mutuler S, Guzelbag E (1993) Intracranial hydatid cysts in children. Neurosurgery 33(2):219–225
Faraji R, Javadi GR, Barshahi PM, Ahmadian F, Sarebanhassanabadi M, Firoozabadi AD, Moloudi AR (2015) Prevalence of hydatid cyst in slaughtered livestock in Kermanshah (West Iran). Adv Microbiol 5:252–257. https://doi.org/10.4236/aim.2015.54023
Fathi S, Dehaghi MM, Radfar MH (2011) Fertility and viability rates of hydatid cysts in camels slaughtered in Kerman region, the southeast of Iran. Sci Parasitol 12(2):77–83
Fikire Z, Tolosa T, Nigussie Z, Macias C, Kebede N (2012) Prevalence and characterization of hydatidosis in animals slaughtered at Addis Ababa abattoir, Ethiopia. J Parasitol Vector Biol 4(1):1–6. https://doi.org/10.5897/JPVB11.020
Fromsa A, Jobre Y (2011) Infection prevalence of hydatidosis (Echinococcus granulosus, Batsch, 1786) in domestic animals in Ethiopia: a synthesis report of previous surveys. Ethiop Vet J 15:11–33
Garg M, Gupta RK, Prasad KN, Sikora SS, Pal L, Chawla S, Kumar R, Husain M, Saxena S, Husain N, Roy R (2002) Fertility assessment of hydatid cyst by Proton MR spectroscopy. J Surg Res 106:196–201. https://doi.org/10.1006/jsre.2002.6397
Getachew D, Almaw G, Terefe G (2012) Occurrence and fertility rates of hydatid cysts in sheep and goats slaughtered at Modjo Luna Export Slaughter House, Ethiopia. Ethiop Vet J 16(1):83–91. https://doi.org/10.4314/evj.v16i1.7
Haridy FM, Ibrahim BB, Morsy TA (1998) Studies on hydatidosis in slaughtered camels in Egypt. J Egypt Soc Parasitol 28:673–681
Haridy FM, Ibrahim BM, Elshazly AM, Awad SE, Sultan DM, El-Sherbini G, Morsy TA (2006) Hydatidosis granulosus in Egyptian slaughtered animals in the years 2000–2005. J Egypt Soc Parasitol 36:1087–1100
Ibrahem MM, Craig PS (1998) Prevalence of cystic echinococcosis in camels (Camelus dromedarius) in Libya. J Helminthol 72:27–31
Ibrahim M (2010) Study of cystic echinococcosis in slaughtered animals in Al Baha region, Saudi Arabia: interaction between some biotic and abiotic factors. Acta Trop 113:26–33
Kebede N, Mitiku A, Tilahun G (2009) Hydatidosis of slaughtered animals in Bahir Dar abattoir, Northwestern Ethiopia. Trop Anim Health Prod 41:43–50
Kebede N, Gebre-Egziabher Z, Tilahun G, Wossene A (2011) Prevalence and financial effects of hydatidosis in cattle slaughtered in Birre-Sheleko and Dangila abattoirs, Northwestern Ethiopia. Zoonoses Public Health 58:41–46
Kouidri M, Khoudja FB, Boulkaboul A, Selles M (2012) Prevalence, fertility and viability of cystic echinococcosis in sheep and cattle of Algeria. Bulg J Vet Med 15(3):191–197
Melaku A, Lukas B, Bogale B (2012) Cyst viability, organ distribution and financial losses due to hydatidosis in cattle slaughtered at Dessie municipal abattoir, North-eastern Ethiopia. Vet World 5(4):213–218
Njoroge EM, Mbithi PMF, Gathuma JM, Wachira TM, Gathura PB, Magambo JK, Zeyhle E (2002) A study of cystic echinococcosis in slaughter animals in three selected areas of northern Turkana, Kenya. Vet Parasitol 104:85–91
Omar M, Sultan K, Haridy M, Omran A (2013) Prevalence of cystic echinococcosis in slaughtered ruminants in different abattoirs, Upper Egypt. Am J Anim Vet Sci 8(3):117–121
Omer RA, Dinkel A, Romig T, Mackenstedt U, Elnahas AA, Aradaib IE, Ahmed ME, Elmalik KH, Adam A (2010) A molecular survey of cystic echinococcosis in Sudan. Vet Parasitol 169:340–346
Oostburg BFJ, Vrede MA, Bergen AE (2000) The occurrence of polycystic echinococcosis in Suriname. Ann Trop Med Parasitol 94:247–252
Pandey VS, Ouhelli H, Ouchton M (1986) Hydatidosis in sheep, goats and dromedaries in Morocco. Ann Trop Med Parasitol 80:525–529
Pangui LJ, Ould AE (1991) Incidence of camel hydatidosis in Mauritania. Bull Anim Health Prod Afr 39:25–26
Parkoohi PI, Jahani M, Hosseinzadeh F, Taghian S, Rostami F, Mousavi A, Rezai M (2018) Epidemiology and clinical features of hydatid cyst in Northern Iran from 2005 to 2015. Iran J Parasitol 13(2):310–316
Qingling M, Guanglei W, Jun Q, Xinquan Z, Tianli L, Xuemei S, Jinsheng Z, Huisheng W, Kuojun C, Chuangfu C (2014) Prevalence of hydatid cysts in livestock animals in Xinjiang, China. Korean J Parasitol 52(3):331–334
Rahman MS, Sokar SM, Dahab S (1992) Comparative studies on hydatidosis in farm animals in Egypt. Deutsch Tierärztl Wochenschr 99:438–440
Sabri JH, Hassan MA, Ramadan MY, Khalifa NO (2005) Hydatidosis in sheep, goat and human contacts. Benha Vet Med J 16:2–2
Scala A, Garippa G, Varcasia A, Tranquillo VM, Genchi C (2006) Cystic echinococcosis in slaughtered sheep in Sardinia (Italy). Vet Parasitol 135(1):33–38
Singh BB, Dhand NK, Ghatak S, Gill JPS (2014) Economic losses due to cystic echinococcosis in India: needfor urgent action to control the disease. Prev Vet Med 113:1–12
Thompson RC (2008) The taxonomy, phylogeny and transmission of Echinococcus. Exp Parasitol 119:439–446
Thompson RC, McManus DP (2002) Towards a taxonomic revision of the genus Echinococcus. Trends Parasitol 18:452–457
Torgerson PR, Budke C (2003) Echinococcosis: an international public health challenge. Res Vet Sci 74:191–202
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
El-Dakhly, K.M., Arafa, W.M., El-Nahass, ES.N. et al. The current prevalence and diversity of cystic echinococcosis in slaughtered animals in Egypt. J Parasit Dis 43, 711–717 (2019). https://doi.org/10.1007/s12639-019-01151-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12639-019-01151-1