Abstract
The present paper describes two new species of furcocercous cercariae, Cercaria sp. XVIII Malabar n. sp. and Cercaria sp. XIX Malabar n. sp. infecting the freshwater snails, Lymnea luteola and Gyraulus convexiusculus respectively in the Malabar region of Kerala. Cercaria sp. XVIII Malabar n. sp., is a distome, pharyngeate, non-ocellate, longifurcate furcocercous cercaria with two pairs of penetration glands and 16 pairs of flame cells and recovered from L. luteola. Cercaria sp. XIX Malabar n. sp., is also a distome, pharyngeate, longifurcate cercaria with a pair of unpigmented eyespots and 20 pairs of flame cells and recovered from G. convexiusculus. Sporocysts of both the cercariae developed in the digestive glands. The present paper describes the cercariae and compares them with related species to establish their systematic position.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Sewell (1922) in his monograph on cercarial fauna of India described 32 species of cercariae, infecting 10 snail hosts in Kerala. This was followed by numerous studies on larval trematodes in the region. Though, the freshwater snail, Lymnea luteola Lamarck is widely distributed throughout Malabar, the northern region of Kerala, so far only three species of furcocercous cercariae, C. indicae XXX (Sewell 1922), C. indicae XLVII (Sewell 1922), and Cercaria sp. 1 Malabar (Vasandakumar and Janardanan 2005) have been described infecting this snail. The freshwater planorbid snail, Gyraulus convexiusculus (Hutton) is widely distributed throughout the Indian subcontinent and is known to act as intermediate host for several trematodes (Khan 2003). But, so far no cercarial infections have been reported form this snail host in the region. During a screening of freshwater snails in the region, we encountered two species of furcocercous cercariae infecting L. luteola and G. convexiusculus respectively. When compared with previously described forms, both the cercariae were morphologically and morphometrically distinct and hence reported here as new species. Following the earlier pattern, Roman numerals were used to designate the cercariae and this was followed by ‘Malabar’, which indicated the region of collection.
Materials and methods
Lymnea luteola and G. convexiusculus were collected from waterbodies in various localities of Malabar, during May 1989 to October 1990. Infected snails were isolated and maintained for further studies in the laboratory. Emerging cercariae were studied alive using a Carl Zeiss Axioscope—2 plus research microscope with phase-contrast optics. For studying the intra-molluscan developmental stages, infected snails were crushed, stained with neutral red and examined under the microscope. Measurements (n = 30) from 10% formalin-fixed cercariae are given in micrometres (µm) as range followed by mean in parentheses. Structural details from live specimens were added free-hand to camera lucida drawings of the cercariae.
Results and discussion
Cercaria sp. XVIII Malabar n. sp.
-
Host: Lymnea luteola Lamarck.
-
Site of infection: Digestive gland.
-
Type Locality: Kundayithode in Kozhikode district, Kerala
-
Holotype: Deposited in the parasite collections, Parasitology Laboratory, Department of Zoology, University of Calicut (Accession number: CF. 2.3).
-
Etymology: Refers to the region (Malabar) from where the species was first recorded.
-
Period of collection: May, 1989 to October, 1990
-
Prevalence: Eighteen of 1239 snails (1.45%) screened were infected
Cercarial behavior Cercariae emerge during morning hours; positively phototactic; swim actively by lashing movements of their tails; remain suspended in water column with furcal rami spread out and tail stem hanging down during rest.
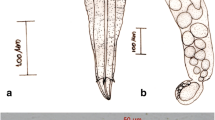
Description (Fig. 1a, b)
Distome, pharyngeate, longifurcate furcocercous cercaria with an elongate body measuring 144.0–180.0 (161.6) × 49.5–67.5 (57.0). Body spination characteristic, with 15–16 anteriorly directed spines overhanging the mouth, followed by a wide band of large, backwardly directed spines. This was followed by an aspinose region occupying the middle region of anterior organ. Ten to 12 rows of medium-sized, backwardly directed spines occupied the region between posterior end of anterior organ and ventral sucker. Tail terminal; tail stem longer than body, devoid of spines; sensory hairs and caudal bodies present; tail stem measured 151.5–192.0 (169.7) × 31.5–40.5 (34.0). Furca exceeded the tail stem in length, not constricted off from stem, devoid of spines and measured 153.0–193.5 (174.8) × 13.5–19.5 (15.3). Caudal excretory canal bifurcates into furcal rami and empties out halfway from furcal origin. Anterior organ elongate and 40.5–52.5 (44.0) × 27.0–37.5 (30.1) in size. Ventral sucker equatorial, slightly wider than long, 25.5–30.0 (27.8) × 25.5–34.5 (31.4) in size; two concentric circles of 90–95 inwardly directed spines present on ventral sucker. Mouth sub-terminal, leading to a short prepharynx; pharynx muscular, measured 10.5–15.0 (13.1) × 7.5–13.5 (11.3). Oesophagus wavy; caeca extend up to the posterior quarter with bulged caecal arms and round tips. Penetration glands two pairs, post-acetabular, enclosed fine, granular contents and prominent nuclei. Gland ducts run forward, enter anterior organ, swell and empty out lateral to mouth. Genital primordia represented by a mass of cells anterior to excretory bladder. Excretory bladder trilobed, anterior lobes broader than long; primary collecting vessels originate from the antero-lateral margins of bladder, run forward and lateral to ventral sucker divide into anterior and posterior collecting vessels. Flame cell formula: 2 [(2 + 1) + (2 + 1 + (2))] = 16.
Sporocyst (Fig. 1b)
Were found developing in the digestive gland of infected L. luteola. Body elongate, pale; measured 592.9–3287.9 (1809.5) × 100.1–161.7 (136.0). Two to three developed cercariae, a few developing cercariae and germ balls were observed within the sporocysts.
Remarks
The present cercaria, a longifurcate, pharyngeate, non-ocellate cercaria with two pairs of penetration glands, closely resembles Cercaria indicae I Sewell 1922, Cercaria florensis Probert 1966, Cercaria spiralis Probert 1966 and Cercaria sp. XVI Malabar.
A comparison of characters and measurements of the above four cercariae with that of the present form is given in Table 1. Presence of spinose tail stem and furca; short caeca and nature of body spine distribution differentiate Cercaria indicae I from the present form. Differences in body shape, arrangement of body spines, spinose furca, three circlets of spines on ventral sucker, position/arrangement of penetration glands and absence of dilated gland duct endings distinguishes C. florensis. C. spiralis differs from the present cercaria in body shape, spination, presence of septate caeca, three circlets of spines on ventral sucker and in the position of caudal excretory canal opening. Nature of body spination; post-equatorial ventral sucker; 6–8 overhanging spines at the anterior; shape, position/arrangement of penetration glands and absence of dilated gland duct endings distinguish Cercaria sp. XVI Malabar from the present form. In addition, it differs from all the above compared cercariae in the flame cell formula and snail host. Considering the above differences, the present cercaria is treated as new species and the name Cercaria sp. XVIII Malabar n. sp. is proposed.
Cercaria sp. XIX Malabar n. sp.
-
Host: Gyraulus convexiusculus (Hutton)
-
Site of infection: Digestive gland
-
Locality: Kundayithode in Kozhikode district, Kerala
-
Holotype: Deposited in parasite collections, Parasitology Laboratory, Department of Zoology, University of Calicut (Accession number: CF. 2.4).
-
Etymology: Refers to the region (Malabar) from where the species was first recorded.
-
Period of collection: May, 1989 to February, 1990
-
Prevalence: Four of 142 snails (2.82%) screened were infected
Cercarial behavior Emergence starts during morning hours and continues till noon; swim rapidly toward water surface, rest for a brief period with their furcae spread out parallel to the water surface and then move downward. Cercariae remain active for a few hours and finally settle at the bottom.
Description (Fig. 2a, b)
Belongs to distome, pharyngeate, longifurcate furcocercous group. Body elongate, measured 155.1–191.4 (176.6) × 59.4–79.2 (68.6); body surface armed with small, backwardly directed, uniformly distributed spines; 12–14 large spines found overhanging the mouth; a band of closely packed spines with curved tips found at the anterior region of anterior organ, followed by a region devoid of spines. Two pairs of sensory hairs present near the anterior and posterior extremities. Terminal tail with small, evenly distributed spines, sensory hairs and caudal bodies; tail measured 181.5–283.8 (251.0) × 39.6–52.8 (45.8). Furca without constriction; possesses small, uniformly distributed spines and a pair of sensory hairs on its surface; measured 243.3–277.2 (252.6) × 29.7–36.3 (32.6). Caudal excretory canal bifurcates at the point of furcal origin, enters into each furca and empties on its outer side, roughly one-third from furcal base. Anterior organ terminal, measured 29.7–36.3 (32.8) × 29.7–36.3 (31.2). A pair of unpigmented eyespots in the mid-region of body. Ventral sucker post-equatorial, measured 26.4–29.7 (27.7) × 26.4–29.7 (29.0); armed with a circlet of 60 inwardly directed spines. Mouth sub-terminal; prepharynx short; pharynx pear-shaped, 16.5–23.1 (17.2) × 16.5–19.8 (17.4) in size. Oesophagus long, narrow; caeca extending up to the posterior quarter. Two pairs of pre-acetabular penetration glands with irregular margins, conspicuous nuclei and fine granular contents. Gland ducts run forward as a bunch on either sides and open out independently, lateral to mouth. Genital primordium represented by an irregular mass of cells behind ventral sucker. Excretory bladder, roughly triangular, thin walled; each primary collecting vessel with a bulb-like swelling at its base, arises from the antero-lateral margin of excretory bladder, ascends and lateral to ventral sucker, splits into anterior and posterior collecting vessels. Posteriorly, bladder continues as caudal excretory canal. Flame cell formula: 2 [(2 + 3) + (2 + 1 + (2))] = 20.
Sporocyst (Fig. 2b)
Sporocysts recovered from the digestive gland of infected G. convexiusculus. Sporocyst body filamentous, measured 408.1–1293.6 (850.5) × 77.0–154.0 (115.5); harboured two to three cercariae along with a few developing cercariae and germ balls.
Remarks
Based on the morphological features, the present cercaria resembles cercaria of Cotylurus flabelliformis (Faust, 1917) Haitsma, 1931, Cercaria helvetica XXXIV Dubois, 1934, Cercaria tetraglandis Iles, 1959 and Cercaria allotropicalis Nasir and Scorza, 1966.
Comparison of morphology and morphometry of the present cercaria with related forms is given in Table 2. Measurements of body; presence of prepharynx; length and nature of oesophagus and caeca separate the present cercaria from cercaria of C. flabelliformis. It differs from C. helvetica XXXIV in the measurements of body, anterior organ and pharynx; length and nature of oesophagus; shape of penetration glands and position of caudal excretory canal opening. C. tetraglandis distinguishes itself in having a long prepharynx, a septate caeca and the main excretory vessel bifurcating posterior to ventral sucker. Differences in the size of body, tail and furca and in features like position/arrangement of penetration glands; moderately sized oesophagus and caeca extending up to anterior level of genital primordia differentiates C. allotropicalis from the present cercaria. Further, the present cercaria can be discriminated from all the above forms in having a spinose tail, a single circlet of spines on ventral sucker and host species. Based on the above differences, the present cercaria is treated as new species and named Cercaria sp. XIX Malabar n. sp.
References
Dubois G (1934) Contribution a l’etude des cercaires de la region de Neuchatel, suivie d’une note sur les cercaires du Lac Noir (Zermatt). Rev Suisse Zoologie 41(2):73–84
Faust EC (1917) Notes on the Cercariae of the Bitter Root Valley, Montana. J Parasitol 3(3):105–123
Iles C (1959) The larval trematodes of certain freshwater molluscs. I. The furcocercariae. Parasitol 49:478–504
Khan RA (2003) Population dynamics and biology of aquatic gastropod, Gyraulus convexiusculus (Hutton) in a small pond. Rec Zool Surv India 101(Part 3–4):25–37
Nasir P, Scorza JV (1966) Studies on freshwater larval trematodes. Part XIV. A new species of a Strigeid cercaria, Cercaria allotropicalis, from Venezuela, with a key to the related species. Proc Helminth Soc Wash 33(1):87–90
Probert AJ (1966) Studies on larval trematodes infecting the freshwater molluscs of Llangorse Lake, South Wales. III. The furcocercariae. J Helminthol 40:91–114
Sanil NK, Janardanan KP (2017) Two new species of furcocercous cercariae infecting the freshwater snail, Thiara tuberculata (Műller) in Kozhikode and Malappuram districts of Kerala. J Parasit Dis 41(4):1147–1152
Sewell RBS (1922) Cercariae indicae. Indian J Med Res 10:1–370
Vasandakumar MV, Janardanan KP (2005) Two new species of furcocercous cercariae from freshwater snails in Malabar, Kerala. Uttar Pradesh J Zool 25(1):93–96
Acknowledgements
The authors thank the Head, Department of Zoology, University of Calicut for providing necessary facilities. One of the authors (NKS) is grateful to the Council of Scientific and Industrial Research (CSIR) for financial support in the form of a Senior Research Fellowship
Author information
Authors and Affiliations
Contributions
Dr K. P. Janardanan designed and guided the study. Dr N. K. Sanil carried out the survey, collected and studied the cercariae. The manuscript was written by both the authors.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that that there is no conflict of interest between them.
Rights and permissions
About this article
Cite this article
Sanil, N.K., Janardanan, K.P. Furcocercous cercariae infecting freshwater snails in Malabar: two new species from Lymnea luteola Lamarck and Gyraulus convexiusculus (Hutton). J Parasit Dis 42, 220–225 (2018). https://doi.org/10.1007/s12639-018-0987-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12639-018-0987-x