Abstract
Purpose
Deep brain stimulation (DBS) can be an effective treatment option for patients with essential tremor and Parkinson’s disease. This review provides an overview on the functioning of neurostimulators and recent advances in this technology and presents an updated guide on the anesthetic management of patients with an implanted neurostimulator undergoing surgery or medical intervention.
Source
A search was conducted on MEDLINE®, EMBASE™, and Cochrane Database of Systematic Reviews databases to identify studies published in English from 1974 to December 2015. Our search also included relevant and available incident reports from the manufacturers, Health Canada, the United States Food and Drug Administration, and the European Medicines Agency. Thirty of 232 articles identified were found to be relevant to this review.
Principal findings
Deep brain stimulation systems now offer a range of options, including pulse generators with dual-channel capabilities, rechargeable batteries, and current-control modes. Preoperatively, the anesthesiologist should ascertain the indications for DBS therapy, identify the type of device implanted, and consult a DBS specialist for specific precautions and device management. The major perioperative concern is the potential for interactions with the medical device resulting in patient morbidity. Neurostimulators should be turned off intraoperatively to minimize electromagnetic interference, and precautions should be taken when using electrosurgical equipment. Following surgery, the device should be turned on and checked by a DBS specialist.
Conclusion
The anesthesiologist plays an important role to ensure a safe operating environment for patients with an implanted DBS device. Pertinent issues include identifying the type of device, involving a DBS-trained physician, turning off the device intraoperatively, implementing precautions when using electrosurgical equipment, and checking the device postoperatively.
Résumé
Objectif
La stimulation cérébrale profonde (SCP) peut constituer une option thérapeutique efficace pour les patients atteints de tremblement essentiel ou de la maladie de Parkinson. Ce compte rendu propose un aperçu du fonctionnement des neurostimulateurs et des progrès récents de cette technologie et présente un guide mis à jour de la prise en charge anesthésique des patients ayant un neurostimulateur implanté et devant subir une chirurgie ou une intervention médicale.
Source
Nous avons réalisé des recherches dans les bases de données MEDLINE®, EMBASE™ et la base de données Cochrane de comptes rendus méthodiques (Cochrane Database of Systematic Reviews) afin d’identifier les études publiées en anglais entre 1974 et décembre 2015. Nos recherches ont également inclus les rapports d’incident pertinents et disponibles des fabricants, de Santé Canada, de la FDA (Food and Drug Administration) américaine, et de l’Agence européenne des médicaments. Au total, trente des 232 articles identifiés ont été jugés pertinents à ce compte rendu.
Constatations principales
Les dispositifs de stimulation cérébrale profonde proposent aujourd’hui une vaste gamme d’options, notamment des générateurs d’impulsions capables d’utiliser deux canaux, des piles rechargeables, et des modes de contrôle du courant. Avant l’opération, l’anesthésiologiste devrait vérifier les indications thérapeutiques d’une SCP, identifier le type de dispositif implanté, et consulter un spécialiste de la SCP pour connaître les précautions spécifiques et la prise en charge du système. La préoccupation périopératoire majeure est le potentiel d’interactions avec le dispositif médical, ce qui pourrait provoquer une morbidité pour le patient. Les neurostimulateurs devraient être éteints pendant l’opération afin de minimiser les interférences électromagnétiques, et des précautions doivent être prises lors de l’utilisation de matériel électrochirurgical. Après la chirurgie, le dispositif devrait être rallumé et vérifié par un spécialiste de la SCP.
Conclusion
L’anesthésiologiste joue un rôle important pour garantir un environnement opératoire sécuritaire pour les patients munis d’un dispositif de SCP implanté. Les questions pertinentes sont l’identification du type de dispositif, la consultation d’un médecin formé en SCP, la mise hors tension du dispositif pendant l’opération, la mise en œuvre de précautions lors du recours à du matériel électrochirurgical, et la vérification postopératoire du dispositif.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Deep brain stimulation (DBS) is an effective treatment option for patients with a movement disorder, and an increasing number of patients with essential tremor or Parkinson’s disease are being treated with this technology.1 Therefore, the anesthesiologist will increasingly encounter patients with an implanted DBS system in the operating room and interventional radiology suites. Most anesthesiologists are familiar with the anesthetic management of patients with a cardiac implantable electronic device, and there are established practice guidelines from various organizations.2,3 Nevertheless, there is a paucity of information on the anesthetic management of patients with an implanted neurostimulator, and most data on patient management derive from isolated case reports and available manufacturer information sheets. The purpose of this review is to provide a brief overview on DBS systems and present an up-to-date guide on the anesthetic management of patients with an implanted DBS device.
Deep brain stimulation—a primer for anesthesiologists
Indications
The use of DBS was first reported in 1987 in patients with tremor-predominant Parkinson’s disease.4 This technology revolutionized the treatment of patients with a movement disorder such as essential tremor, dystonia, and Parkinson’s disease. The advantages of DBS over ablative surgeries (e.g., thalamotomy and pallidotomy) include its reversibility, adjustability, and safety profile. To date, the use of DBS has been approved by the United States Food and Drug Administration for treatment of patients with Parkinson’s disease or essential tremor as well as patients under a humanitarian device exemption for dystonia or obsessive compulsive disorder.5,6 Ongoing research supports the potential benefit of DBS in a range of disorders, including epilepsy, chronic pain, major depression, anorexia nervosa, and Alzheimer’s disease.5-7
Deep brain stimulation system
Current DBS systems consist of one or more cylindrical electrodes (each housing multiple contacts) implanted at pre-planned targets within the brain parenchyma, an implantable pulse generator (IPG) inserted most commonly below the clavicle, and extension wires that connect the electrodes to the IPG. This system delivers stimulation to the electrodes (unilaterally or bilaterally) at a set amplitude, pulse width, and frequency. Patients with a bilateral electrode insertion may have two implanted IPGs or a single IPG with dual-channel capabilities, allowing independent control of both electrodes from a single generator. After insertion, the treating physician can program the system wirelessly with a handheld device. The clinician and patient have separate programming devices. Depending on the manufacturer, the patient’s programmer allows the patient to perform basic functions, such as checking the lifespan of the neurostimulator and programmer battery, turning the neurostimulator on or off, and adjusting therapy settings within the limits set by the physician. In addition to these functions, the physician’s programmer enables the clinician to enter and check the patient’s profile and system information, program stimulation parameters, perform electrode impedance measurements, and set patient control limits. Older neurostimulator models, such as Medtronic Kinetra® Model 7428 and Soletra® Model 7426 (Medtronic, Minneapolis, MN, USA), have magnetically controlled switches that can be turned on or off with an external magnet, but newer models from Medtronic Inc. and other manufacturers do not turn off with an external magnet. Medtronic Inc. was previously the sole manufacturer of neurostimulators, but in recent years, two other companies, St Jude Medical Inc and Boston Scientific Corporation, have developed DBS systems that are approved for clinical use.
Advances in DBS system technology
The DBS system can be voltage or current controlled. Older DBS systems were voltage controlled, and it is postulated that they delivered variable amounts of current due to electrochemical-induced changes in electrode impedance at the brain-electrode interface.8 Newer current-controlled systems provide constant current stimulation, which may be more clinically efficacious compared with the older voltage-controlled systems.9 In addition, DBS devices can be programmed to provide unipolar or bipolar stimulation. In unipolar mode, the active electrode is set as the cathode and the IPG case is set as the anode. In contrast, bipolar stimulation is produced when at least one of the four electrodes in the DBS system functions as the cathode and at least another one of the four functions as the anode. Newer DBS systems also provide the option to implant rechargeable IPGs, which prolongs their lifespan and results in fewer surgical interventions for replacing the IPG.10,11
Differences between the DBS and cardiac implantable electronic devices (CIED)
Cardiac implantable electronic devices include cardiac pacemakers and implantable cardioverter-defibrillators (ICD). The cardiac pacemaker system is generally more complex and advanced compared with a neurostimulator. The basic components of cardiac pacemakers are similar to the DBS system in that the pacemaker consists of electrodes (one to three leads) implanted in the endocardium or epicardium, an energy source (lithium-iodine battery), and wires that connect the leads to the battery. The primary function of the cardiac pacemaker is to generate a threshold pacing current to evoke cardiac muscle depolarization. The pacing threshold is determined by the device’s programmed amplitude and pulse width settings and lead impedance.12 Table 1 summarizes the pertinent differences that exist between a DBS and CIED.
Anesthetic considerations for patients with a DBS device in situ
Literature search
MEDLINE®, EMBASE™, and Cochrane Database of Systematic Reviews databases were searched to identify human studies published in English from 1974 to December 2015. Keywords used for this search include “deep brain stimulation”, “implantable neurostimulators”, “anesthesiology”, “anesthesia”, “neurosurgery”, and “neurosurgical procedures”. Thirty of the 232 articles initially identified were considered as being relevant to this review. Additional articles of relevance were identified from references cited in the identified literature. The literature search also included product information and available incident reports from Medtronic Inc., St Jude Medical, Inc., Boston Scientific Corporation, Health Canada, the United States Food and Drug Administration, and the European Medicines Agency. Table 2 summarizes the available literature on patients with a DBS device in situ who underwent various surgical and non-surgical procedures.
Anesthetic considerations
The major considerations in the perioperative treatment of patients with an implanted DBS system include the medical condition that warranted DBS insertion, potential for electromagnetic interference with the DBS system (e.g., electrocautery use and magnetic resonance imaging [MRI]), and postoperative evaluation of the patient and device. Table 3 outlines the perioperative anesthetic management of patients with an implanted DBS device undergoing surgery.
Preoperative considerations
In the preoperative setting, patients with an implanted DBS device scheduled for elective surgery should be seen in a preanesthesia assessment clinic, particularly as their underlying medical condition may warrant special considerations. For example, patients with Parkinson’s disease, dystonia, or epilepsy have their own unique perioperative anesthetic considerations.13,14
Intraoperative considerations
Device interactions with the DBS system
Intraoperatively, there is potential for the DBS system to interact with multiple medical devices, including diathermy, electrocautery, peripheral nerve stimulator, external cardiac defibrillator, therapeutic ultrasound, and laser equipment. Some of these devices produce varying degrees of electromagnetic interference that potentially affect the functioning of the neurostimulator, which in turn may result in patient harm. For example, electromagnetic interference can cause direct damage to the IPG, resulting in suppressed or increased stimulation or complete cessation of output.10 Alternatively, induced current can pass through the IPG along the conducting wires, leading to heat generation at the tip of the DBS electrodes and ensuing damage to brain tissue in proximity of the electrodes.15,16 Even when the neurostimulator is turned off, the metallic case, leads, and DBS unit remain conductive, allowing current to pass through. Table 4 summarizes these potential device interactions and the appropriate management for each interaction.
Diathermy, electrocautery, and electrosurgery:
The terms diathermy, electrocautery, and electrosurgery are often used interchangeably, but differences exist between these terms. Specifically, diathermy refers to generation of heat within body tissue from a high frequency electromagnetic current. Physiotherapists, chiropractors, and sports injury therapists often use this technique for muscle relaxation and treatment of joint conditions.17 Electrocautery refers to generation of heat within a metal wire electrode by passing current through it; this current does not usually enter the patient’s body.17 Electrocautery is usually used for tissue hemostasis or varying degrees of tissue destruction, such as removal of benign skin lesions. Electrosurgery is commonly used during surgery, and it encompasses a range of modalities utilizing high frequency alternating current at the electrode tip to cut, coagulate, or desiccate tissue.17 In general, electrosurgical units have two different electrode configurations, unipolar and bipolar mode. In unipolar mode, the current generated through the electrode enters into the patient’s body and the electrical circuit is completed when the current reaches the grounding pad. In contrast, the electrical current in bipolar mode is confined to tissue between the two electrodes of the electrosurgical unit. Bipolar mode of electrosurgery has been shown to be safer for use in patients with implanted neurostimulators.18-21
There are two case reports of patients with an implanted neurostimulator who suffered serious brain injuries due to heat generation at the tip of the DBS electrodes after use of diathermy for dental treatment.15,16 The manufacturer subsequently issued a product advisory to caution against the use of all forms of diathermy treatment in patients with a neurostimulator.10,22
Regional anesthesia:
In patients with a movement disorder, inactivation of the DBS device could result in recurrence of symptoms that may be worse than the patient’s baseline symptoms. This may be challenging for both the patient and surgeon during procedures performed under regional anesthesia. Providing adequate sedation may help diminish some of the symptoms and facilitate reasonable surgical conditions. Proposed sedation regimes include midazolam (0.5-1 mg iv), propofol infusion (25-75 µg·kg−1·min−1 iv), and dexmedetomidine (0.2-0.7 µg·kg−1·hr−1 iv), and these medications should be titrated to individual patient response. Two case reports have demonstrated the safety of using a peripheral nerve stimulator in patients with an implanted DBS device. In both cases, a brachial plexus block was performed with guidance from a peripheral nerve stimulator, and the DBS generator remained on during the procedure.23,24 Safety precautions required in such procedures include ensuring that the path of electrical stimulation does not pass through the DBS system and that the puncture site is not in proximity to the wires of the DBS device.23,25 Alternatively, the performance of regional blocks with ultrasound guidance obviates the potential interaction between the peripheral nerve stimulator and the DBS device.
Ultrasonic equipment, radiation therapy, laser:
Ultrasound has been increasingly used as a therapeutic modality due to its capability of generating effects such as heat and mechanical stress.26 This technology is commonly used during phacoemulsification for cataract removal, extracorporeal lithotripsy for kidney stone removal, and harmonic scalpel for surgical cutting and cauterization. Diagnostic ultrasound can be safely performed in patients with an implanted neurostimulator, but the manufacturer recommends that the transducer should not be placed directly over the implanted device. The use of phacoemulsification for cataract removal has been shown to be safe in two case reports.27,28 Performance of lithotripsy is not recommended by the manufacturer due to potential damage to the neurostimulator circuitry from high-output ultrasonic frequencies. Nevertheless, its use is not contraindicated, and if lithotripsy must be performed, the beam should not be directed within 15 cm of the neurostimulator.29 Radiation therapy should not be administered within the vicinity of the DBS device. When administered, the amount of exposure should be limited, a lead shield should be used to protect the device, and the device should be checked after every treatment.29,30 Similarly, when laser therapy is used, the neurostimulator should be turned off and the laser should be directed away from the device.29 As there is a limited amount of safety data on the use of the above medical devices in patients with an implanted neurostimulator, the indications for their use should be carefully considered and the risks should be discussed in detail with the patient. General precautionary measures include directing the equipment away from the neurostimulator when in use, turning off the DBS device during the procedure, and checking the device following the procedure.
External cardiac defibrillators:
External cardiac defibrillation and cardioversion may be lifesaving and should not be withheld from patients with an implanted DBS device. A single case report showed that external cardioversion with 300 J did not affect the functioning of the DBS system.31 To minimize damage to the DBS system, the manufacturer (Medtronic Inc.) recommends using the lowest clinically appropriate output setting and positioning the paddles as far away as possible from the neurostimulator and perpendicular to the DBS system.29 Once again, the neurostimulator should be checked after shock delivery.
Effect of the DBS system on other medical devices
Electrical signals from the DBS system have been shown to cause artifacts that affect the functioning of diagnostic and therapeutic cardiac equipment.
Electrocardiogram (ECG):
Electrocardiogram recording can be affected by electrical signals generated from the DBS system; however, this interference resolves when the neurostimulator is turned off.32-35 It is important to point out that, when the device is turned off, recurrence of symptoms may introduce movement-related artifacts that preclude optimal ECG recording.
Cardiac pacemakers and ICDs:
The insertion of cardiac pacemakers and/or ICDs in patients with neurostimulators, and vice versa, is not contraindicated and has been performed successfully.31,36-40 Nevertheless, precautions should be taken because of potential interactions between these devices, including inappropriate sensing and response by the cardiac pacemaker, inappropriate sensing of tachyarrhythmia by the ICD resulting in discharges, and inactivation of or adjustment to the neurostimulator settings.29,40 A thorough preoperative assessment by a multidisciplinary team (i.e., cardiologist, DBS specialist, and anesthesiologist) is necessary to optimize the patient’s clinical status and to adjust settings of the underlying medical devices as required. To minimize interactions between the cardiac pacemaker and the DBS device, the cardiac pacemaker should be programmed to bipolar sensing mode to avoid oversensing and inappropriate response.36-38 Similarly, use of bipolar sensing electrodes for the ICD system also reduces oversensing. It is unlikely that inappropriate ICD discharges would occur at DBS outputs of up to 5 V.31,39,40 In addition, ICD discharges up to 35 J do not seem to affect the functioning of the DBS device, although a case was reported whereby shock delivery converted the generator output to the “off” state with an amplitude of 0 V.40 Therefore, the functioning of the neurostimulator should be checked after the ICD delivers a shock. In order to reduce electromagnetic interference, it is also recommended not to insert the IPGs of the two systems in close proximity.37
Magnets can interfere with the functioning and programming of both cardiac and neurostimulator IPGs, and their use should be avoided, if possible, to prevent unintentional reprogramming or suspension of these devices. Instead, the respective device-specific telemetric programmer should be utilized for programming the device.39 If an external magnet is required intraoperatively to inactivate the defibrillator function of the ICD, precautions should be taken to avoid placing it in close proximity to the DBS device. Lastly, detailed cardiac investigations, e.g., Holter monitoring, should be performed whenever adjustments are made to the DBS device settings to ensure consistent functionality of the cardiac pacemaker device.
Perioperative risk of hardware-related infection
The incidence of hardware-related infection after DBS implantation (primary insertion and/or IPG replacement) varies from 0-15%.41-46 This wide variation is secondary to differences in the definition of hardware infection, the follow-up period after implantation, and the calculation for the incidence of infection based on the number of patients or procedures. Staphylococcus aureus is the commonest microorganism found in cultures.42,43,46 Currently, there is a lack of established guidelines for the treatment of hardware-related infections after DBS insertion, but treatment options include antibiotic therapy with or without partial/complete removal of the device.43,44,46
There is also a paucity of available data to substantiate an increased risk of hardware infection for patients with an implanted DBS system undergoing unrelated surgery (i.e., surgeries that do not involve implantation or replacement of the IPG). Furthermore, there is a lack of evidence-based guidelines regarding specific antibiotic therapy. As such, clear recommendations cannot be made regarding specific antibiotic therapy, and protocol likely varies from centre to centre. In cases where the proposed surgery is distant from the hardware, routine antibiotic prophylaxis for the proposed surgery is likely sufficient. In those cases where the proposed surgery involves re-opening spaces with implanted DBS hardware, our recommendation is to do this in consultation with the DBS surgical team. Should a patient have a surgical site infection and/or develop sepsis, both the DBS surgical team and the infectious disease physician should be consulted so that appropriate therapy can be initiated.46
Postoperative considerations
At the end of the procedure, the neurostimulator should be turned on to the original settings. If general anesthesia was administered, we recommend that the device be turned on before reversal of anesthesia to avoid recurrence of symptoms when the patient is awake.19,24,47 It has been suggested that cortical arousal may occur with subthalamic nucleus (STN) stimulation due to possible involvement of extrathalamic arousal systems in the human sleep-wake cycle.47-49 In a case report by Singh et al., a patient with bilateral STN DBS insertion for Parkinson’s disease underwent a laparoscopic cholecystectomy under general anesthesia. During reversal of anesthesia, the patient did not arouse and entropy levels remained low despite an end-tidal desflurane concentration of 0.4 vol% (MAC 0.1). At this time, the DBS system was reactivated, followed by a sudden increase in entropy values and spontaneous eye opening.47
Non-surgical procedures
Magnetic resonance imaging
The use of MRI in patients with an implanted DBS device was previously an absolute contraindication due to electromagnetic interactions that could potentially result in patient morbidity. The MRI system produces three types of electromagnetic fields: 1) static magnetic field that is constantly present, 2) gradient magnetic field that is present only during a scan, and 3) radiofrequency (RF) field that is present only during a scan and produced by a variety of transmission RF coils.
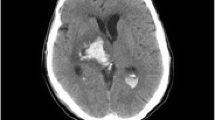
The safety concerns with MRI include excessive heating at the electrode tips of the DBS device due to compounding of the current produced by the gradient and RF magnetic fields within the DBS system, magnetic field interactions, image artifacts and distortion, and functional disruption of the DBS system.50-54 The amount of heat generated within the DBS system is dependent on several factors, including electrical properties of the neurostimulator, field strength of the MRI system, position of the DBS components relative to the RF energy source, type of RF coil used, point of imaging, and the specific absorption rate (SAR).55 The SAR is a measure of the rate at which energy is absorbed by the human body when exposed to an RF electromagnetic field, and this parameter is often used to identify limits for safe MRI. Transient and serious morbidities have been described in two case reports.56,57 In the first report, a patient with bilateral STN DBS insertion experienced unilateral dystonic and ballistic leg movements that lasted several weeks after MRI of the head.56 The second case involved a patient with bilateral STN DBS insertion who became hemiplegic after MRI of the lumbar spine. Subsequent imaging revealed subacute hemorrhage adjacent to the tip of the DBS electrode.57
Some neurostimulators are likely MR compatible, as suggested by studies showing the safe use of MRI under specified conditions in patients with an implanted DBS system.50-55 With advances in DBS technology, some centres now routinely insert DBS devices under intraoperative MR guidance.58 Specific guidelines on the use of MRI in patients with implanted DBS devices vary according to the manufacturer. For example, the use of MRI is currently contraindicated for Boston Scientific DBS systems22 but can be performed, if necessary, in patients with Medtronic DBS systems under specified conditions. The complete MRI guideline for the Medtronic DBS system is available from http://manuals.medtronic.com/wcm/groups/mdtcom_sg/@emanuals/@era/@neuro/documents/documents/contrib_228155.pdf.Footnote 1 Pertinent points from this guideline are outlined in the following text.10,59
Prior to MRI, the treating physician should identify the model of the implanted neurostimulator, the presence of an implanted pocket adaptor, implant status of the lead (internalized or externalized), and the integrity of the system (breaks within the system are potential sites for production of excessive heat), as these factors determine if the DBS system permits scanning the whole body or only the head. In general, patients with a neurostimulator model using a magnetic switch or a DBS system with a pocket adaptor or externalized leads are eligible for head scans only. Once MRI eligibility has been determined, the scan must be performed under specific conditions. To date, only the 1.5 Tesla horizontal closed bore/64 MHz RF MRI system has been shown to be safe for use in patients with an implanted neurostimulator. The safety profile at specific static magnetic fields should not be extrapolated to other systems, even at lower field strengths.55 Neurostimulators should be turned off during imaging, which, in some patients, can lead to recurring symptoms that interfere with adequate image acquisition. In neurostimulator systems with a magnetic switch, the magnetic switch must be turned off and the stimulation amplitude set to 0 V. Other safety precautions include limiting the active scan time, proper patient positioning, and constant communication with the patient to identify early complications. After imaging, the DBS specialist should check the neurostimulator and turn on the device to its original settings.
Electroconvulsive therapy (ECT)
The safety concerns of performing ECT in patients with implanted neurostimulators include generation of heat at the DBS electrodes from induction of RF current by the electrical charge, functional disruption of the DBS system, and displacement of electrodes from induced seizure activity.60-62 Three case reports showing safe administration of ECT recommend the following precautionary measures: turning the neurostimulator off, placement of the ECT electrodes as far away as possible from the DBS electrodes, and use of the lowest possible energy for seizure induction.50-54
Limitations
The main limitation of our review is the lack of hypothesis-driven scientific evidence to support the safe use of certain medical devices or the performance of certain procedures in patients with implanted neurostimulators, as most data were obtained from case reports/series and manufacturer guidelines. There is also a possibility of bias as not all incident reports may be published and critical events may not be made available by the manufacturers. Nevertheless, it would be impractical or difficult to perform randomized-controlled trials to obtain information such as critical incidents and potential device interactions. As such, case reports/series, manufacturer guidelines, and critical incident reports remain the major sources of relevant information.
Conclusions
Deep brain stimulation technology has expanded and advanced considerably over the past 25 years. Current DBS systems have smaller IPGs, offer rechargeable IPG options, and are capable of providing variable frequencies and strengths of stimulation. There is now increasing interest in the development of closed-loop DBS systems,63,64 DBS devices with directional leads,11,65,66 and computer-guided programming of DBS systems.63,67,68 Deep brain stimulation is now a standard of care for patients with an expanding range of neurological conditions, and the anesthesiologist will more commonly encounter patients with these devices. Thus, it is necessary for the anesthesiologist to be aware of the pertinent issues to prevent harm to the patient. The principles of anesthetic management include identification of the type of device, involvement of the DBS physician for specific precautions and device management, turning off the DBS device intraoperatively, practising precautions for safe use of electrosurgical equipment, and checking the device postoperatively.
Notes
The MRI guideline for the new St Jude Medical DBS system is not available as the device is currently not commercially available in Canada.
References
Kalia SK, Sankar T, Lozano AM. Deep brain stimulation for Parkinson’s disease and other movement disorders. Curr Opin Neurol 2013; 26: 374-80.
American Society of Anesthesiologists. Practice advisory for the perioperative management of patients with cardiac implantable electronic devices: pacemakers and implantable cardioverter-defibrillators: an updated report by the American Society of Anesthesiologists Task Force on Perioperative Management of Patients with Cardiac Implantable Electronic Devices. Anesthesiology 2011; 114: 247-61.
Healey JS, Merchant R, Simpson C, et al. Canadian Cardiovascular Society/Canadian Anesthesiologists’ Society/Canadian Heart Rhythm Society joint position statement on the perioperative management of patients with implanted pacemakers, defibrillators, and neurostimulating devices. Can J Cardiol 2012; 28: 141-51.
Benabid AL, Pollak P, Louveau A, Henry S, de Rougemont J. Combined (thalamotomy and stimulation) stereotactic surgery of the VIM thalamic nucleus for bilateral Parkinson disease. Appl Neurophysiol 1987; 50: 344-6.
Venkatraghavan L, Manninen P. Anesthesia for deep brain stimulation. Curr Opin Anaesthesiol 2011; 24: 495-9.
Lozano AM, Lipsman N. Probing and regulating dysfunctional circuits using deep brain stimulation. Neuron 2013; 77: 406-24.
Montgomery EB Jr, Gale JT. Mechanisms of action of deep brain stimulation (DBS). Neurosci Biobehav Rev 2008; 32: 388-407.
Miocinovic S, Lempka SF, Russo GS, et al. Experimental and theoretical characterization of the voltage distribution generated by deep brain stimulation. Exp Neurol 2009; 216: 166-76.
Lempka SF, Johnson MD, Miocinovic S, Vitek JL, McIntyre CC. Current-controlled deep brain stimulation reduces in vivo voltage fluctuations observed during voltage-controlled stimulation. Clin Neurophysiol 2010; 121: 2128-33.
Medtronic for Healthcare Professionals. Deep brain stimulation for movement disorders. Available from URL: http://professional.medtronic.com/pt/neuro/dbs-md/ (accessed October 2016).
Boston Scientific. Vercise™ DBS system. Available from URL: http://www.bostonscientific.com/en-EU/products/deep-brain-stimulation-systems/Vercise-DBS.html (accessed October 2016).
Korpas D. Basic principles of cardiac pacemaker technology. In: Korpas D, editor. Implantable Cardiac Devices Technology. New York: Springer Science + Business Media New York; 2013. p. 7-12.
Poon CC, Irwin MG. Anaesthesia for deep brain stimulation and in patients with implanted neurostimulator devices. Br J Anaesth 2009; 103: 152-65.
Venkatraghavan L, Luciano M, Manninen P. Review article: anesthetic management of patients undergoing deep brain stimulator insertion. Anesth Analg 2010; 110: 1138-45.
Nutt JG, Anderson VC, Peacock JH, Hammerstad JP, Burchiel KJ. DBS and diathermy interaction induces severe CNS damage. Neurology 2001; 56: 1384-6.
Ruggera PS, Witters DM, von Maltzahn G, Bassen HI. In vitro assessment of tissue heating near metallic medical implants by exposure to pulsed radio frequency diathermy. Phys Med Biol 2003; 48: 2919-28.
Riordan AT, Gamache C, Fosko SW. Electrosurgery and cardiac devices. J Am Acad Dermatol 1997; 37: 250-5.
Davies RG. Deep brain stimulators and anaesthesia. Br J Anaesth 2005; 95: 424.
Khetarpal M, Yadav M, Kulkarni D, Gopinath R. Anaesthetic management of a patient with deep brain stimulation implant for radical nephrectomy. Indian J Anaesth 2014; 58: 461-3.
Weaver J, Kim SJ, Lee MH, Torres A. Cutaneous electrosurgery in a patient with a deep brain stimulator. Dermatol Surg 1999; 25: 415-7.
Martinelli PT, Schulze KE, Nelson BR. Mohs micrographic surgery in a patient with a deep brain stimulator: a review of the literature on implantable electrical devices. Dermatol Surg 2004; 30: 1021-30.
Boston Scientific Corporation. Vercise™ Deep Brain Stimulation System Physician Manual. Marlborough: Boston Scientific Corporation; 2014.
Minville V, Chassery C, Benhaoua A, Lubrano V, Albaladejo P, Fourcade O. Nerve stimulator-guided brachial plexus block in a patient with severe Parkinson’s disease and bilateral deep brain stimulators. Anesth Analg 2006; 102: 1296.
Gandhi R, Chawla R. Anaesthetic management of shoulder arthroscopic repair in Parkinson’s disease with deep brain stimulator. Indian J Anaesth 2014; 58: 309-11.
Venkatraghavan L, Chinnapa V, Peng P, Brull R. Non-cardiac implantable electrical devices: brief review and implications for anesthesiologists. Can J Anesth 2009; 56: 320-6.
Miller DL, Smith NB, Bailey MR, et al. Overview of therapeutic ultrasound applications and safety considerations. J Ultrasound Med 2012; 31: 623-34.
Parsloe CF, Twomey JM. Safety of phacoemulsification in a patient with an implanted deep brain neurostimulation device. Br J Ophthalmol 2005; 89: 1370-1.
Ozturk F, Osher RH. Phacoemulsification in a patient with a deep brain stimulator. J Cataract Refract Surg 2006; 32: 687-8.
Medtronic Inc. Medtronic DBS Therapy - Information for Prescribers. Minneapolis:Medtronic Inc; Available from URL: http://professional.medtronic.com/pt/neuro/dbs-md/prod/#.V_45RsksDVd (accessed October 2016).
Medtronic Inc. Frequently asked questions. Available from URL: http://www.medtronicneuro.com.au/faq_pain_neurostimulation.html (accessed October 2016).
Rosenow JM, Tarkin H, Zias E, Sorbera C, Mogilner A. Simultaneous use of bilateral subthalamic nucleus stimulators and an implantable cardiac defibrillator. Case report. J Neurosurg 2003; 99: 167-9.
Garg R, Borthakur B, Pawar M. Management of patient with deep brain stimulator for emergency laparotomy. J Neurosurg Anesthesiol 2011; 23: 168.
Martin WA, Camenzind E, Burkhard PR. ECG artifact due to deep brain stimulation. Lancet 2003; 361: 1431.
Constantoyannis C, Heilbron B, Honey CR. Electrocardiogram artifacts caused by deep brain stimulation. Can J Neurol Sci 2004; 31: 343-6.
Frysinger RC, Quigg M, Elias WJ. Bipolar deep brain stimulation permits routine EKG, EEG, and polysomnography. Neurology 2006; 66: 268-70.
Senatus PB, McClelland S 3rd, Ferris AD, et al. Implantation of bilateral deep brain stimulators in patients with Parkinson disease and preexisting cardiac pacemakers. Report of two cases. J Neurosurg 2004; 101: 1073-7.
Capelle HH, Simpson RK, Kronenbuerger M, Michaelsen J, Tronnier V, Krauss JK. Long-term deep brain stimulation in elderly patients with cardiac pacemakers. J Neurosurg 2005; 102: 53-9.
Ozben B, Bilge AK, Yilmaz E, Adalet K. Implantation of a permanent pacemaker in a patient with severe Parkinson’s disease and a preexisting bilateral deep brain stimulator. Int Heart J 2006; 47: 803-10.
Obwegeser AA, Uitti RJ, Turk MF, et al. Simultaneous thalamic deep brain stimulation and implantable cardioverter-defibrillator. Mayo Clin Proc 2001; 76: 87-9.
Tavernier R, Fonteyne W, Vandewalle V, de Sutter J, Gevaert S. Use of an implantable cardioverter defibrillator in a patient with two implanted neurostimulators for severe Parkinson’s disease. Pacing Clin Electrophysiol 2000; 23: 1057-9.
Voges J, Waerzeggers Y, Maarouf M, et al. Deep-brain stimulation: long-term analysis of complications caused by hardware and surgery–experiences from a single centre. J Neurol Neurosurg Psychiatry 2006; 77: 868-72.
Sillay KA, Larson PS, Starr PA. Deep brain stimulator hardware-related infections: incidence and management in a large series. Neurosurgery 2008; 62: 360-6.
Bhatia R, Dalton A, Richards M, Hopkins C, Aziz T, Nandi D. The incidence of deep brain stimulator hardware infection: the effect of change in antibiotic prophylaxis regimen and review of the literature. Br J Neurosurg 2011; 25: 625-31.
Piacentino M, Pilleri M, Bartolomei L. Hardware-related infections after deep brain stimulation surgery: review of incidence, severity and management in 212 single-center procedures in the first year after implantation. Acta Neurochir (Wien) 2011; 153: 2337-41.
Pepper J, Zrinzo L, Mirza B, Foltynie T, Limousin P, Hariz M. The risk of hardware infection in deep brain stimulation surgery is greater at impulse generator replacement than at the primary procedure. Stereotact Funct Neurosurg 2013; 91: 56-65.
Bjerknes S, Skogseid IM, Sæhle T, Dietrichs E, Toft M. Surgical site infections after deep brain stimulation surgery: frequency, characteristics and management in a 10-year period. PLoS One 2014; 9: e105288.
Singh M, Venkatraghavan L. Cortical arousal with deep brain stimulation after general anesthesia for laparoscopic cholecystectomy. J Neurosurg Anesthesiol 2016. DOI:10.1097/ANA.0000000000000389.
Krone L, Frase L, Piosczyk H, et al. Top-down control of arousal and sleep: fundamentals and clinical implications. Sleep Med Rev 2016. DOI:10.1016/j.smrv.2015.12.005.
Kundishora AJ, Gummadavelli A, Ma C, et al. Restoring conscious arousal during focal limbic seizures with deep brain stimulation. Cereb Cortex 2016. DOI:10.1093/cercor/bhw035.
Gleason CA, Kaula NF, Hricak H, Schmidt RA, Tanagho EA. The effect of magnetic resonance imagers on implanted neurostimulators. Pacing Clin Electrophysiol 1992; 15: 81-94.
Tronnier VM, Staubert A, Hähnel S, Sarem-Aslani A. Magnetic resonance imaging with implanted neurostimulators: an in vitro and in vivo study. Neurosurgery 1999; 44: 118-25.
Finelli DA, Rezai AR, Ruggieri PM, et al. MR imaging-related heating of deep brain stimulation electrodes: in vitro study. AJNR Am J Neuroradiol 2002; 23: 1795-802.
Rezai AR, Finelli D, Nyenhuis JA, et al. Neurostimulation systems for deep brain stimulation: in vitro evaluation of magnetic resonance imaging-related heating at 1.5 tesla. J Magn Reson Imaging 2002; 15: 241-50.
Kahan J, Papadaki A, White M, et al. The safety of using body-transmit MRI in patients with implanted deep brain stimulation devices. PLoS One 2015; 10: e0129077.
Rezai AR, Phillips M, Baker KB, et al. Neurostimulation system used for deep brain stimulation (DBS): MR safety issues and implications of failing to follow safety recommendations. Invest Radiol 2004; 39: 300-3.
Spiegel J, Fuss G, Backens M, et al. Transient dystonia following magnetic resonance imaging in a patient with deep brain stimulation electrodes for the treatment of Parkinson disease. Case report. J Neurosurg 2003; 99: 772-4.
Henderson JM, Tkach J, Phillips M, Baker K, Shellock FG, Rezai AR. Permanent neurological deficit related to magnetic resonance imaging in a patient with implanted deep brain stimulation electrodes for Parkinson’s disease: case report. Neurosurgery 2005; 57: E1063.
Ostrem JL, Ziman N, Galifianakis NB, et al. Clinical outcomes using ClearPoint interventional MRI for deep brain stimulation lead placement in Parkinson’s disease. J Neurosurg 2016; 124: 908-16.
Medtronic Inc. MRI guidelines for Medtronic deep brain stimulation systems. Minneapolis: Medtronic Inc; 2015. Available from URL: http://manuals.medtronic.com/wcm/groups/mdtcom_sg/@emanuals/@era/@neuro/documents/documents/contrib_228155.pdf (accessed October 2016).
Moscarillo FM, Annunziata CM. ECT in a patient with a deep brain-stimulating electrode in place. J ECT 2000; 16: 287-90.
Chou KL, Hurtig HI, Jaggi JL, Baltuch GH, Pelchat RJ, Weintraub D. Electroconvulsive therapy for depression in a Parkinson’s disease patient with bilateral subthalamic nucleus deep brain stimulators. Parkinsonism Relat Disord 2005; 11: 403-6.
Bailine S, Kremen N, Kohen I, et al. Bitemporal electroconvulsive therapy for depression in a Parkinson disease patient with a deep-brain stimulator. J ECT 2008; 24: 171-2.
Miocinovic S, Somayajula S, Chitnis S, Vitek JL. History, applications, and mechanisms of deep brain stimulation. JAMA Neurol 2013; 70: 163-71.
Shah RS, Chang SY, Min HK, Cho ZH, Blaha CD, Lee KH. Deep brain stimulation: technology at the cutting edge. J Clin Neurol 2010; 6: 167-82.
St Jude Medical. Movement disorders. Available from URL: http://media.sjm.com/newsroom/media-kits/neurological-disease/united-states/default.aspx (accessed October 2016).
Pollo C, Kaelin-Lang A, Oertel MF, et al. Directional deep brain stimulation: an intraoperative double-blind pilot study. Brain 2014; 137: 2015-26.
Butson CR, Cooper SE, Henderson JM, McIntyre CC. Patient-specific analysis of the volume of tissue activated during deep brain stimulation. Neuroimage 2007; 34: 661-70.
Frankemolle AM, Wu J, Noecker AM, et al. Reversing cognitive-motor impairments in Parkinson’s disease patients using a computational modelling approach to deep brain stimulation programming. Brain 2010; 133: 746-61.
Acknowledgment
The authors thank Marina Englesakis, library information specialist, for her assistance in the literature search.
Conflicts of interest
None declared.
Editorial responsibiltiy
This submission was handled by Dr. Steven Backman, Associate Editor, Canadian Journal of Anesthesia.
Author contributions
Tze Yeng Yeoh was involved with the literature review and writing the manuscript. Lashmi Venkatraghavan, Suneil Kalia, and Pirjo Manninen contributed to critical analysis and editing the final manuscript.
Funding
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yeoh, T.Y., Manninen, P., Kalia, S.K. et al. Anesthesia considerations for patients with an implanted deep brain stimulator undergoing surgery: a review and update. Can J Anesth/J Can Anesth 64, 308–319 (2017). https://doi.org/10.1007/s12630-016-0794-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12630-016-0794-8