Abstract
Purpose
Recently, we showed that processing of shed blood with a continuous-flow cell saver during cardiopulmonary bypass resulted in a clinically significant reduction in postoperative cognitive decline (POCD) six weeks after coronary artery bypass graft (CABG) surgery. The current study examined if the early benefit of reduced POCD was sustained in the same patient population at one-year follow-up.
Methods
One hundred seventy patients (cell saver group, n = 84; controls, n = 86) underwent neuropsychological testing at baseline and one year after surgery. The raw scores for each test were converted to Z-scores, and a combined Z-score of ten main variables was then calculated for each study group.
Results
Postoperative cognitive decline was present in 16 of 84 (19%) patients in the cell saver group (95% confidence interval [CI], 10.8 to 27.2) vs 15 of 86 (17.4%) patients in the control group (95% CI, 9.6 to 25.2) (P = 0.786). Six of the 15 patients in the control group with POCD at six weeks had the impairment at one year and five did not; four were lost to follow-up. Three of the six cell saver patients with POCD at six weeks still had impairment at one year, two did not, and one was not tested. Thirteen (15.4%) and nine (10.5%) patients in the cell saver and control groups, respectively, developed new POCD which was not evident at the six-week follow-up.
Conclusions
The short-term preservation of cognitive function in elderly patients using the cell saver management strategy did not translate into a long-term benefit one year after CABG surgery. The presence of progressing cerebrovascular disease may be responsible for the long-term cognitive decline. (ClinicalTrials.gov number, NCT00193999).
Résumé
Objectif
Nous avons récemment montré que la récupération du sang avec un autotransfuseur à débit continu au cours de la circulation extracorporelle aboutissait à une réduction significative de déclin cognitif postopératoire (POCD) six semaines après une intervention chirurgicale pour pontage coronarien. L’étude actuelle a voulu savoir si l’avantage précoce d’une diminution de POCD se maintenait dans la même population après un an de suivi.
Méthodes
Cent soixante-dix patients (groupe autotransfuseur, n = 84; groupe contrôle, n = 86) ont passé des tests neuropsychologiques avant la chirurgie et un an après. Les résultats bruts de chaque test ont été convertis en écarts réduits et un écart réduit combiné des dix principales variables a alors été calculé pour chaque groupe de l’étude.
Résultats
Le déclin cognitif postopératoire était présent chez 16 patients sur 84 (19 %) dans le groupe autotransfuseur (intervalle de confiance [IC] à 95 %: 10,8 à 27,2) contre 15 patients sur 86 (17,4 %) dans le groupe témoin (IC à 95 %: 9,6 à 25,2) (P = 0,786). Six patients parmi les 15 du groupe contrôle présentant un POCD à 6 semaines avaient des fonctions altérées à un an tandis que cinq n’en avaient pas lors du suivi; quatre patients ont été perdus de vue. Trois des six patients du groupe autotransfuseur présentant un POCD à 6 semaines avaient encore une altération des fonctions cognitives à un an tandis que deux n’en avaient plus et qu’un patient n’a pas été testé. Treize (15,4 %) et neuf (10,5 %) patients, respectivement du groupe autotransfuseur et du groupe témoin ont développé un nouveau POCD qui n’apparaissait pas lors du suivi à 6 semaines.
Conclusions
La préservation à court terme des fonctions cognitives chez des patients âgés avec l’utilisation d’une stratégie d’autotransfusion ne s’est pas traduite par un avantage à long terme, un an après chirurgie pour pontage coronarien. L’existence d’une maladie cérébrovasculaire évolutive peut être la cause du déclin cognitif à long terme. (Numéro ClinicalTrials.gov: NCT00193999).
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Postoperative cognitive decline (POCD) is a well-recognized phenomenon after cardiac surgery. Cerebral embolization, systemic inflammatory response, and cerebral hypoperfusion are usually implicated as likely etiologic factors for short-term POCD after cardiac surgery. The incidence of short-term POCD varies among studies, but the reported rates are 46-53% at discharge1 , 2 and 15-36% at six weeks1 , 3 , 4 after coronary artery bypass graft (CABG) surgery. The presence of early POCD measured at hospital discharge has been shown to be a significant predictor of long-term cognitive dysfunction five years after surgery.1 , 2 However, it has also been suggested that the presence of cerebrovascular disease at baseline and the subsequent development of new cerebral infarcts in the elderly patient may play an important role in late cognitive decline irrespective of surgery and cardiopulmonary bypass (CPB).5 - 8
Recently, we showed that processing of shed blood with a continuous-flow cell saver during CPB resulted in a clinically significant reduction in POCD six weeks after surgery.4 The reduced cognitive decline in the cell saver group was likely attributed to the lower lipid cerebral embolic load and the modified systemic inflammatory response with the application of cell saver.
The purpose of the current study was to determine if the early benefit of reduced POCD was sustained in the same patient population at the one-year follow-up.
Methods
After approval by the University Health Network Research Ethics Board (10th Floor, Room 1056, 700 University Ave., Toronto, ON, M5G 1Z5, August 16, 2001, REB 01-0420-B), an informed consent was obtained from patients > 60 yr of age who were scheduled for elective CABG surgery. Exclusion criteria included a surgical procedure in addition to CABG, redo CABG surgery, emergent surgery, severe kidney or liver disease (creatinine > 133 mg·dL−1 and bilirubin > 2 mg·dL−1), symptomatic cerebrovascular disease, history of stroke, transient ischemic attacks, or atrial fibrillation. We also excluded patients who were unable to complete the preoperative assessment and those who could not speak English. Patients were randomly allocated to either a cell saver or a control (cardiotomy suction) group according to a computer-generated randomization code in blocks of a predetermined size. Patients were followed for up to one year after surgery.
Perioperative management strategies were described in detail in the original publication.4 Briefly, the continuous-flow cell saver (Fresenius Corporation, Concord, CA, USA) was used to process shed blood before returning it back to the patient, and cardiotomy suction was used in a standard closed venous reservoir where cardiotomy blood was collected and reinfused through the arterial circuit back to the patient. Both the cell saver and the cardiotomy suction were used during the same time periods, i.e., from the time full heparinization (ACT > 400 sec) was established to after the initial dose of protamine was given.
Neuropsychological testing
A trained psychometrist blinded to the treatment arm assignment conducted the neuropsychological testing one week before (baseline) surgery as well as six weeks and one year after surgery. The battery of tests included 12 tests. Ten main variables were chosen a priori to be used in the analyses: 1) Rey Auditory Verbal Learning Test, 2) Rey Visual Design Learning Test, 3) Halstead-Reitan Trail Making Tests Parts A and B (Trails B - Trails A), 4) Grooved Pegboard Test, 5) Wechsler Memory Scale (WMS) Digit Span Forward, 6) WMS Digit Span Backward, 7) WMS Spatial Span Forward, 8) WMS Spatial Span Backward, 9) Choice and Simple Reaction Time Tests (Choice Reaction Time - Simple Reaction Time), and 10) Verbal Fluency Test. If a lower score showed improved performance, the directional data were reversed so that all improvements gave positive change scores.
The same analysis of the neuropsychological testing was applied as previously described.4 In brief, to estimate the change in performance from baseline to one year after surgery, the raw scores for each test were converted to Z-scores. A Z-score was calculated for each main variable in each patient by subtracting the preoperative score from the postoperative score and dividing the difference by the preoperative standard deviation of that variable.9 Subsequently, a combined Z-score was calculated by the summation of all of the individual test scores. Patients with a negative score of < 1 were considered to have POCD. The primary outcome was the dichotomous cognitive deterioration variable based on the combined cognitive score. Tests not completed were treated as omissions and not as failures.
Sample size justification and statistical analysis
Sample size calculations were based on the prevalence of POCD at six weeks after surgery and were described in detail in the original publication.4
Comparability of both groups was tested with the use of Chi square statistics or Fisher’s exact test on qualitative variables, as appropriate, and the Student’s t test on quantitative variables. For the primary analysis of dichotomous cognitive outcome, a sum of the individual Z-scores (a combined Z-score) of the ten main variables was compared between the two groups with a Chi square test. Confidence intervals (CI) for proportions were calculated at 95%. A P value < 0.05 was considered significant. All analyses were performed on an intention-to-treat basis. Statistical analysis was conducted with the use of SPSS® computer software (SPSS Inc., Chicago, IL, USA).
Results
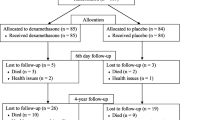
In the original trial, 226 patients (cell saver group, n = 112; control group, n = 114) underwent baseline neuropsychological testing. Ninety-nine patients in each group were evaluated for the presence of POCD at six weeks after surgery. The observed proportion of patients with POCD was six of 99 (6.1%) patients in the cell saver group and 15 of 99 (15.2%) patients in the control group, difference, 9.1% (95% CI, 0.4 to 18.1). Eighty-four of 112 (75%) patients in the cell saver group and 86 of 114 (75%) controls returned for one-year neuropsychological testing (Fig. 1). One year after surgery, postoperative cognitive decline was present in 16 of 84 (19%) patients in the cell saver group (95% CI, 10.8 to 27.2) and 15 of 86 (17.4%) patients in the control group (95% CI, 9.6 to 25.2) (P = 0.786) (difference, -1.6%; 95% CI, -13.3 to 10.1) (Fig. 2). Both groups were similar with respect to demographic data and surgical characteristics (Table 1).
Twenty-one patients in the cell saver group and 17 patients in the control group who completed the neuropsychological testing at six weeks did not return for the one-year follow-up. Six patients in the cell saver group and four patients in the control group who did not complete the assessment at six weeks returned for the one-year testing. Six of the 15 patients in the control group with POCD at six weeks still had the impairment at one year and five did not; four were lost to follow-up. Three of the six patients in the cell saver group with POCD at six weeks still had the impairment at one year, two did not, and one was not tested. Thirteen (15.4%) and nine (10.5%) patients in the cell saver and control groups, respectively, developed new POCD which was not evident at the six-week follow-up. The raw neuropsychological test scores are shown in Table 2.
Discussion
The current study showed that the short-term (six weeks) preservation of cognitive function4 in elderly patients using the cell saver management strategy during CPB did not translate into a long-term benefit one year after CABG surgery. Although the lack of long-term preservation of cognitive function after cardiac surgery is disappointing, these findings clearly raise a number of questions regarding the etiology and pathophysiology of POCD. First, do early and late POCDs have different etiologies? Second, is cardiac surgery a form of stressor that identifies patients who would have developed POCD regardless of the surgical intervention? Third, are the perioperative efforts to reduce early POCD worthwhile if the long-term benefits are not sustainable?
It is important to emphasize that the majority of patients who had POCD at the one-year follow-up developed new cognitive dysfunction that was not evident at six weeks after surgery. These findings are supportive of our current contention that different etiologies are responsible for early and late POCD. However, an alternative hypothesis is also plausible, i.e., that the pathophysiologic neurologic sequelae processes (e.g., an apoptotic change initiated by perioperative insults and inflammation) that may have been present at surgery manifested themselves only at the one-year follow-up.
It is well known that the signs of systemic atherosclerotic disease are associated with global cognitive decline in elderly patients.10 , 11 Leary et al.7 reported that 3-5% of the general population in their 70 s are likely to have a new brain infarct each year as detected by magnetic resonance imaging (MRI) scans. Indeed, a link between brain infarcts identified by MRI scans and cognitive decline has been established in large-scale population-based longitudinal studies.12 , 13 A recent editorial by Hammon and Stump8 estimated that the rate of first-time one-year neurological events in the CABG patient population would range from 10-15%. Furthermore, evidence is emerging that preexisting cerebrovascular disease may play a pivotal role in both short- and long-term POCD.14 - 17 Consequently, it is not surprising that the rates of new brain infarcts in previous studies and the new onset of POCD in the current report were similar at the one-year follow-up.
Age and the baseline cognitive index are generally considered the most powerful predictors of both short- and long-term cognitive decline in cardiac as well as non-cardiac surgical populations.1 , 3 , 18 Importantly, several groups of investigators have found similar rates of long-term cognitive decline in age-adjusted non-surgical controls. In the late 1990 s, Hlatky et al. showed that long-term cognitive function was similar after CABG surgery and coronary angioplasty in the majority of patients.19 The International Study of Postoperative Cognitive Dysfunction (ISPOCD) group20 showed that POCD was present in 10.4% of elderly patients one to two years after non-cardiac surgery. This rate of cognitive decline was almost identical (10.6%) to the age-adjusted non-surgical controls tested at the same time interval. At one, three, and six-year follow-ups, Selnes et al. observed similar rates of late cognitive decline in patients undergoing coronary revascularization procedures and those who were of similar age with coronary artery disease who had not undergone CABG surgery.17 , 21 After adjusting for age, sex, education, and baseline comorbidity, there was no significant difference between the rates of cognitive decline in patients after CABG surgery and control subjects without coronary artery disease at five-year follow-up.22 Likewise, using healthy and non-surgical control groups, Sweet et al. could not show a higher late cognitive decline in CABG patients.23
Similarities in late cognitive decline between patients with and without surgery may suggest that age may simply be a surrogate measure for the preexisting cerebral vascular disease which is considered a major contributor to POCD. Even more so, late cognitive decline after CABG surgery may not be specific to employment of CPB, but rather, it may be related to the presence of cerebrovascular disease. However, it seems likely that certain subgroups of patients may have a more favourable course of cognitive recovery depending on the effects that their baseline cognitive function and education levels may have on aging their arterial and immune systems. In the current study, it is significant that the POCD, which was present in one-third of the patients at six weeks, had resolved at the one-year follow-up.
While short-term POCD after cardiac surgery can likely be attributed to macro- and micro-embolism, cerebral hypoperfusion, and systemic inflammatory response, the presence of progressive cerebrovascular disease is likely a contributor to (along with other factors) long-term POCD. Although it appears that etiologies for short- and long-term cognitive impairment differ, the fact remains that one-half of the patients who were found to have POCD at six weeks still had the impairment at one year. This finding supports our contention that implementation of strategies aimed to reduce short-term cognitive impairment might also benefit patients in the long term. Perception of general health varies directly with cognitive functioning. Consequences of cognitive decline in the elderly after non-cardiac surgery include poorer quality of life, risk of leaving the labour market prematurely, dependency on social transfer payments, and increased mortality.18 , 24 Similarly to non-cardiac surgery, it has been shown that patients with POCD had poorer quality of life when assessed at one year and five years after otherwise successful cardiac surgery.25 , 26 Lower five-year overall cognitive function scores were associated with lower general health and a less productive working status. These findings have significant social and financial implications. Unfortunately, there is no known treatment of POCD once it occurs. Therefore, development of reliable and feasible preventive strategies for both immediate and delayed postoperative neurocognitive complications is of paramount importance.
One of the common limitations of longitudinal cohort studies is patient dropout rates on the follow-up assessments. In the current study, a similar number of patients in the cell saver and control groups (21% vs 17%, respectively) failed to return for the one-year follow-up. However, there was no difference between groups with respect to demographic data or surgical characteristics. Furthermore, the focus of the current study was on an elderly patient population undergoing primary CABG surgery. Consequently, our findings should be applied with caution in younger patient populations undergoing more complex cardiac surgical procedures.
In conclusion, the current study showed that short-term preservation of cognitive function in elderly patients using the cell saver management strategy during CPB did not translate into a long-term benefit one year after CABG surgery. The presence of progressing cerebrovascular disease may be a likely contributing factor to long-term POCD.
References
Newman MF, Kirchner JL, Phillips-Bute B, et al. Longitudinal assessment of neurocognitive function after coronary-artery bypass surgery. N Engl J Med 2001; 344: 395-402.
Nathan HJ, Rodriguez R, Wozny D, et al. Neuroprotective effect of mild hypothermia in patients undergoing coronary artery surgery with cardiopulmonary bypass: five-year follow-up of a randomized trial. J Thorac Cardiovasc Surg 2007; 133: 1206-11.
Djaiani GN, Phillips-Bute B, Blumenthal JA; Neurologic Outcome Research Group; CARE Investigators of the Duke Heart Center. Chronic exposure to nicotine does not prevent neurocognitive decline after cardiac surgery. J Cardiothorac Vasc Anesth 2003; 17: 341-5.
Djaiani G, Fedorko L, Borger MA, et al. Continuous-flow cell saver reduces cognitive decline in elderly patients after coronary bypass surgery. Circulation 2007; 116: 1888-95.
Mullges W, Babin-Ebell J, Reents W, Toyka KV. Cognitive performance after coronary artery bypass grafting: a follow-up study. Neurology 2002; 59: 741-3.
Selnes OA, Grega MA, Borowicz LM Jr, Royall RM, McKhann GM, Baumgartner WA. Cognitive changes with coronary artery disease: a prospective study of coronary artery bypass graft patients and nonsurgical controls. Ann Thorac Surg 2003; 75: 1377-84.
Leary MC, Saver JL. Annual incidence of first silent stroke in the United Sstates: a preliminary estimate. Cerebrovasc Dis 2003; 16: 280-5.
Hammon JW Jr, Stump DA. Five-year cognitive outcomes: surgical effects or natural progression of vascular disease. J Thorac Cardiovasc Surg 2007; 133: 1133-4.
Rasmussen LS, Larsen K, Houx P, et al. The assessment of postoperative cognitive function. Acta Anaesthesiol Scand 2001; 45: 275-89.
Singh-Manoux A, Britton AR, Marmot M. Vascular disease and cognitive function: evidence from the Whitehall II Study. J Am Geriatr Soc 2003; 51: 1445-50.
Vinkers DJ, Stek ML, van der Mast RC, et al. Generalized atherosclerosis, cognitive decline, and depressive symptoms in old age. Neurology 2005; 65: 107-12.
Longstreth WT Jr, Dulberg C, Manolio TA, et al. Incidence, manifestations, and predictors of brain infarcts defined by serial cranial magnetic resonance imaging in the elderly: the Cardiovascular Health Study. Stroke 2002; 33: 2376-82.
O’Sullivan M, Morris RG, Huckstep B, Jones DK, Williams SC, Markus HS. Diffusion tensor mri correlates with executive dysfunction in patients with ischaemic leukoaraiosis. J Neurol Neurosurg Psychiatry 2004; 75: 441-7.
Goto T, Baba T, Yoshitake A, Shibata Y, Ura M, Sakata R. Craniocervical and aortic atherosclerosis as neurologic risk factors in coronary surgery. Ann Thorac Surg 2000; 69: 834-40.
Ernest CS, Murphy BM, Worcester MU, et al. Cognitive function in candidates for coronary artery bypass graft surgery. Ann Thorac Surg 2006; 82: 812-8.
Silbert BS, Scott DA, Evered LA, Lewis MS, Maruff PT. Preexisting cognitive impairment in patients scheduled for elective coronary artery bypass graft surgery. Anesth Analg 2007; 104: 1023-8.
Selnes OA, Grega MA, Bailey MM, et al. Cognition 6 years after surgical or medical therapy for coronary artery disease. Ann Neurol 2008; 63: 581-90.
Monk TG, Weldon BC, Garvan CW, et al. Predictors of cognitive dysfunction after major noncardiac surgery. Anesthesiology 2008; 108: 18-30.
Hlatky MA, Bacon C, Boothroyd D, et al. Cognitive function 5 years after randomization to coronary angioplasty or coronary artery bypass graft surgery. Circulation 1997; 96: II-11-4.
Abildstrom H, Rasmussen LS, Rentowl P, et al. Cognitive dysfunction 1-2 years after non-cardiac surgery in the elderly. ISPOCD Group. International Study of Post-Operative Cognitive Dysfunction. Acta Anaesthesiol Scand 2000; 44: 1246-51.
Selnes OA, Grega MA, Borowicz LM Jr, et al. Cognitive outcomes three years after coronary artery bypass surgery: a comparison of on-pump coronary artery bypass graft surgery and nonsurgical controls. Ann Thorac Surg 2005; 79: 1201-9.
van Dijk D, Moons KG, Nathoe HM, et al. Cognitive outcomes five years after not undergoing coronary artery bypass graft surgery. Ann Thorac Surg 2008; 85: 60-4.
Sweet JJ, Finnin E, Wolfe PL, et al. Absence of cognitive decline one year after coronary bypass surgery: comparison to nonsurgical and healthy controls. Ann Thorac Surg 2008; 85: 1571-8.
Steinmetz J, Christensen KB, Lund T, Lohse N, Rasmussen LS. Long-term consequences of postoperative cognitive dysfunction. Anesthesiology 2009; 110: 548-55.
Newman MF, Grocott HP, Mathew JP, et al. Report of the substudy assessing the impact of neurocognitive function on quality of life 5 years after cardiac surgery. Stroke 2001; 32: 2874-81.
Phillips-Bute B, Mathew JP, Blumenthal JA, et al. Association of neurocognitive function and quality of life 1 year after coronary artery bypass graft (CABG) surgery. Psychosom Med 2006; 68: 369-75.
Acknowledgements
We gratefully acknowledge the support of the Department of Anesthesia, the Division of Cardiac Surgery, and the perfusionists at Toronto General Hospital, University Health Network and the Department of Anesthesia, University of Toronto.
Source of funding
This study was funded by the Heart and Stroke Foundation of Ontario.
Disclosures
None.
Competing interests
None declared.
Author information
Authors and Affiliations
Corresponding author
Additional information
Author contributions
George Djaiani, Ludwik Fedorko, and Jacek Karski contributed to the conception of the study, and Vivek Rao fully supported the plan. George Djaiani, Rita Katznelson, Ludwik Fedorko, Jo Carroll, and Jacek Karski contributed to the study’s design. Vivek Rao, a cardiac surgeon, participated in patient selection. Ludwik Fedorko took an active part in setting up the inclusion and exclusion criteria and was involved in data presentation. George Djaiani supervised the acquisition, analysis, and interpretation of the data and drafted the first version of the article.
Jacek Karski participated in the data analysis, and Jacek Karski, Rita Katznelson, and Vivek Rao were involved with interpretation of the data. Robin Green, a neuropsychologist, actively participated in selection of the neuropsychological testing with respect to the type of tests and the timing of administration. Jo Carroll took an active part in the day-to-day conduct of the trial.
Rights and permissions
About this article
Cite this article
Djaiani, G., Katznelson, R., Fedorko, L. et al. Early benefit of preserved cognitive function is not sustained at one-year after cardiac surgery: a longitudinal follow-up of the randomized controlled trial. Can J Anesth/J Can Anesth 59, 449–455 (2012). https://doi.org/10.1007/s12630-012-9675-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12630-012-9675-y