Abstract
Purpose
Outbreak of SARS-CoV2 and the COVID-19 pandemic had posed a threat to the healthcare system. The world had witnessed the failure of the healthcare system in the diagnosis, surveillance, and development of therapeutics effective against COVID-19. Hence, research emphasis has been given to technological inventions in developing robust and effective tools to fight against respiratory viral outbreaks, including SARS-CoV2.
Methods
The scientific literature was searched and retrieved from PubMed, PubMed Central, the Cochrane Central Register of Controlled Trials (CENTRAL), and Scielo databases. The scientific literature was screened and selected based on recent studies in the area of nanotechnology based inventions and applications in developing diagnostics, surveillance tools, PPE kits, therapeutics, and vaccines.
Results
Based on the scientific literature, there are enormous developments in designing robust diagnostics, surveillance tools, therapeutics, and vaccines. These findings show an increasing demand for nanotechnology-based inventions to tackle zoonotic spillover. Nanotechnology remains a growing area as interdisciplinary science and technology- enable a variety of inventions used in the COVID-19 pandemic. PPE design is a classic example where nanotechnology-driven materials are extensively used. Masks and other fabrics were also developed using nanotechnology-based materials.
Conclusion
The study provides insights into nanotechnology based inventions in the fight against respiratory viral pathogens, including SARS-CoV2, and associated diseases. The study showed the application of nanotechnology in the development of diagnostics, surveillance tools, therapeutics, and vaccines.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
1.1 Outbreak of nSARS-CoV2 and COVID19 pandemic
First-ever reported case of nSARS-CoV2 in December 2019 at Wuhan, China rapidly spread globally and resulted in a global pandemic [1]. There are several theories regarding virus origin however Bats are considered a prominent source of nSARS-CoV2. The virus is rapidly transmitted in the population via aerosol and formites modes and causes acute respiratory distress syndrome [2]. On January 30, 2020, World Health Organization (WHO) declared the outbreak a public health emergency of international concern. Later, WHO declared the spread of nSARS-CoV2 and the disease caused as COVID19 global pandemic on March 11, 2020 [3]. The infection with nSARS-CoV2 results in viral pneumonia where upper and lower respiratory tissue is more commonly affected named coronavirus disease 2019 (COVID19) [4]. Coronaviruses (CoVs) are classified based on crown-shaped spikes present on their surfaces and these CoVs are classified into four main genera namely, alpha-coronavirus (α-CoV), beta-coronavirus (β-CoV), gamma-coronavirus (γ-CoV), and delta-coronavirus (δ-CoV). Coronavirus belong to Coronaviridae family, Nidovirales order where beta coronavirus are more likely cause ARDS and COVID19 [5]. Earlier, two more coronavirus outbreaks were reported in the past where Severe Acute Respiratory Syndrome Coronavirus (SARS-CoV) in 2002–2004 and the Middle East Respiratory Syndrome Coronavirus (MERS-CoV) in 2012–2014 belong to beta coronavirus [6].
Previous coronaviruses outbreaks including SARS-CoV and MERS-CoV represent positive-strand RNA viruses and show significant similarity with nSARS-CoV2 [7]. Beta Coronavirus genome 31 kb encodes spike protein (S-protein), an envelope protein (E-protein), membrane glycoprotein (M-protein), and Nucleocapsid protein (N-protein) [8]. Like other beta coronaviruses, nSARS-CoV2 infects the upper and lower respiratory tract via Angiotensin-Converting Enzyme 2 (ACE2). The receptor-binding domain (RBD) of nSARS-CoV2 differs from other previous coronaviruses i.e. SARS-CoV and MERS-CoV. Infection caused by nSARS-CoV2 trigger massive immune response both innate and adaptive [9]. The severe infection of nSARS-CoV2 triggers the release of interleukins-1β, 2, 6, 7, 8, and 10 (IL-1β, IL-2, IL-6, IL-7, IL-8, and IL-10), granulocyte-colony stimulating factor (GSF), tumor necrosis factor-α (TNF-α), interferon-γ, induced protein (IP-10), monocyte chemo attractant protein-1 (MCP-1) and macrophage inflammatory-protein 1-α (MIP-1α) [10, 11]. Effective clinical diagnosis of nSARS-CoV2 involves the use of antigen and real-time PCR-based tests however, diagnosis also involves X-Rays and CT scans. During the pandemic, several therapeutic were used clinically anti-inflammatory, antiviral, immune modulators, and plasma replacement therapy shown significant beneficial outcomes [12, 13].
2 COVID19 pandemic and nanotechnology
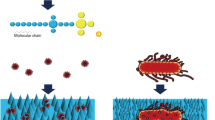
Outbreak of nSARS-CoV2 and COVID19 is novel and hence new approaches become inevitable to diagnose, sterilize, and find effective therapeutics and vaccines for the management of pandemic. Nanotechnology-based inventions including nano-designs, formulations, platforms for vaccines, and adjuvant for therapeutic had showed promising results in the fight against COVID 19 [14]. Nanotechnology-based inventions response was prompt and effective not only in the management of the COVID19 pandemic but also in the containment of the virus, proving mask and PPE to control virus spread and transmission. Nanotechnology-based inventions, formulations, and designs offer several advantages over conventional methods in the fight against the COVID 19 pandemic (Fig. 1) [15]. One and most promising advantage of Nanotechnology inventions is size and ease in functionalizing groups for desired properties. Using a nanotechnology-based platform where environmental sensing; air and water screening provide substantial data useful in the viral spread and dynamic of the pandemic [16].
2.1 Nanotechnology-based materials
Since the time of the outbreak of nSARS-CoV2 and COVID19 pandemic nanotechnology response was remarkable results in various products such as disinfectants, fabric with antiviral properties used for the design of personal protective types of equipment/gear (PPE) for medical and non-medical uses [17, 18]. Though the use of nanotechnology-based inventions/products is not limited to the development of PPE, technology played a pivotal role in diagnostic, therapeutic, and vaccine development [19]. Highly transmissible nSARS-CoV2 requires disinfectant and protective gear such as masks and PPE Kits to control the spread of the virus and control COVID19 [20]. Like most other infectious coronaviruses, nSARS-CoV2 also infects and transmits via aerosol droplets primarily however formites (surface contaminations) are another mode of transmission. The first step in fighting against virus spread and the COVID19 pandemic remain the dissemination of the virus present in the air and surface hence need for an effective disinfectant is inevitable [21]. There are two distinct approaches; one by the spray of functionalized nano-designs with antiviral properties on normal fabric and the second using nanomaterial with antiviral properties for fabric design to develop antiviral material to fight against COVID19 [22]. Earlier studies showed that Goldshield 5 used for spray-on surgical masks offers antibacterial protection specifically against gram-positive and gram-negative bacteria [23]. Goldshield 5 is first-generation approved material under US-FDA regulation as antibacterial coating material [24,25,26].
2.2 Nanotechnology-based materials in designing mask and PPE kits
GS75 is a modified material using a formulation of an organosilane water-stabilized quaternary ammonium chloride formulation in long alkyl chains consisting of a nonionic surfactant, a siloxane molecule that forms a non-polar covalent bond between the surfaces of masks and filters and that will cross-link to the inert materials [27]. The developed material used coating on PPE Kits and Mask for antiviral protection and was reported effective against nSARS-CoV2. The design of antiviral material also seeks metal ions such as Silver and Copper along with other coating materials including benzalkonium chloride, polymers, metal oxides, and functional nanomaterial [28]. The study also showed that the coating with GS75 material on the fabric used for masks and PPE kits provides antiviral protection for 3 days up to 50 °C and was reported safe as no severe toxicity was reported [29, 30]. Pemmada et al. and Imani et al. demonstrated nanotechnology materials used in coating masks, gowns, surgical drapes, textiles, high-touch surfaces, and other personal protective equipment [21, 22]. Similarly, Jung et al. developed and evaluated Copper-Coated Polypropylene Filter Face Mask for antiviral properties [31]. In this study, a commercial Korean Filter (KF) 94 respirator was treated with an oxygen ion beam followed by copper deposition resulting in a ready-to-use mask with the antiviral property (Table 1). Earlier, Sousa et al. demonstrated cupper coating provides antimicrobial and antiviral properties and can be used as coating material effectively [32, 33].
Takeda et al. developed and investigated Copper Iodide Nano-particles to inactivate nSARS-CoV2 in fabric via doped film design [34]. Cupper metal possesses virucidal properties and CuNPs extend virucidal properties at nano-scale ideal for fabric used for masks and PPE kits. Meister et al. developed and demonstrated nano-scale copper and silver film available for coating on fabric in the design of mask and PPE kits with antibacterial and antiviral properties [35]. In a recent study, Souri et al. designed and evaluated an antiviral formulation using nano-Cu conjugated with PLA for coating the mask for antiviral properties [36]. The developed formulation was used for 3D printing also reusable and hence environmentally friendly. In a recent study, Ha et al. investigated the antiviral activity of Cu-NPs where a high-energy electron beam was used to synthesize NPs [37]. The study showed energy induced Cu-NPs showed antiviral activity against the H1N1 influenza virus. Earlier, Gupta et al. reported the antiviral property of Copper NPs cold spray. The study also showed the effectiveness of CuNPs for enveloped viruses over non-enveloped viruses and hence such formulation might be effective against nSARS-CoV2 [38]. Based on experimental findings where nano-copper designs over conventional copper were reported effective in attenuating/killing viruses, CuNPs might be used for the coating to mask and PPE kits. Rabiee et al. studied antiviral activity against the H1N1 influenza virus of ZnO-NPs [39]. The study also report antioxidant, antibacterial and mammalian cell viability of ZnO-NPs where photo-catalytic and biomedical properties were promising.
Yüce and Filiztekin demonstrated the scope and potential of nanomaterial in developing cost effective and robust bioelectronics for the diagnosis of SARS-CoV2 [40]. Here several nano formulations were evaluated in-vitro plate forming units using IO-NPs and reported eight-fold reductions in virus titre. In a recent study, Sarkar et al. evaluated the antiviral potential of different nanoparticles gold nanoparticles, silver nanoparticles, quantum dots, carbon dots, graphene oxide nanoparticles, and zinc oxide nanoparticles [41]. Nanoparticles remain associated with cytotoxicity and emphasis has been given to synthesizing green NPs to combat toxicity. Additionally, surface functionalization is the key approach to minimizing/limiting cytotoxicity [42]. In the COVID19 pandemic, a massive volume of mask and PPE kits were used and are still being used hence green synthesis of NPs provides a sustainable and environment friendly [43]. In a recent study, Prasher and Sharma discussed self-sterilizing surfaces using nanotechnology designs [44]. Tang et al. examined the antiviral property of cationic nanosized cotton fibers ideal for mask and PPE Kit production [45]. The study also demonstrated the antiviral/biocidal activity of nanosized cotton fibers via reactive oxygen species (ROS) generation. Nanosized cotton fibers are associated with strong electrostatic interaction with anionic photosensitizes. These functionalized cotton nano-fibers synthesized using self-propagating 2-diethyl amino ethyl chloride conjugated with photosensitizes restrict microbial growth by 990% and are ideal for fabric used in mask and PPE.
2.3 Nanotechnology in virus detection
Since the time of the outbreak of nSARS-CoV2, detection and diagnosis of infection remain key challenges as it was a novel virus. Subsequently, diagnosis and viral detection become critical for COVID19 management, hence robust and point to care approaches were required [46]. Initially, clinical samples were diagnosed using molecular and immunological profiling where multiple parameters need to examine such as complete blood count (CBC), C Reactive protein level (CRP), lactose dehydrogenase (LDH), Aspartate aminotransferase (AST), alanine aminotransferase (ALT); AST/AL, pro-calcitonin, troponin I. Lymphopenia etc [47]. Simultaneously, imaging techniques such as chest X-Ray and CT were used to understand disease severity rather than infection. Real-time PCR-based diagnosis played a critical role in the early and effective diagnosis of nSARS-CoV2 infection. Immunological profiling precisely pro-inflammatory cytokines including interleukin-1β (IL-1β), IL-1RA, IL-2, IL-6, IL-7, IL-8 (CXCL8), IL-9, IL-10, IL-17, IL-18, tumor necrosis factor (TNF-α), interferon-gamma (IFN-gamma), granulocyte colony-stimulating factor (G-CSF), granulocyte-macrophage colony-stimulating factor (GM-CSF), macrophage inflammatory protein 1 (MIP-1alpha/CCL3), monocyte chemo-attractant protein-1 (MCP-1/CCL2), interferon gamma-induced protein 10 (IP-10/CXCL10), and fibroblast growth factor (FGF) provide a scientific basis of infection and severity of COVID19 as well [48, 49]. Though these techniques are effective but remain associated with several limitations and false-positive results also. Additionally, nSARS-CoV2 and its variants are highly contagious and transmissible hence, a rapid point to care diagnosis was a key in the development of diagnostic kits and methods.
Nanotechnology-based molecular diagnostic approaches showed fast, rapid, and pathogen-specific detection in COVID19. Nanoparticles (NPs) are nanotechnology designs extensively used in nSARS-CoV2 diagnosis in COVID19 compared to other nano-designs. In a recent study, Tavakol et al. advocated the use of different types of NPs in the diagnosis and detection of nSARS-CoV2 [13]. It is evident, that antibody-based approaches and serology largely seek viral genome i.e. ss positive RNA strand where sensitivity, specificity, and accuracy remain a major concern. Xiang et al. demonstrated gold NPs conjugated with IgM/IgG immunoglobulin not only effectively detect viral RNA but also cut the downtime period required for sample collection and processing [13]. Cavalcanti and Nogueira demonstrated in their study rapid viral detection using gold NPs conjugated with IgM/IgG [50]. Another study uses a chiral AuNPs (CAu NPs)-quantum dot (QDs) nano-composite to result in an ultra-sensitive chiro-immunosensor for a large range of viruses including respiratory viral pathogens. The nanotechnology-based platform was also used in previous coronavirus outbreaks for early detection of the virus to develop robust diagnostic tools and kits (Table 2). Different nanoparticles including polymeric NPs, chaperone-mediated ferritin NPs, and spike protein conjugated NPs, self-assembling protein nanoparticles (SANPs), gold nanoparticles (AuNPs), Silver nanoparticles (AgNPs), lumazine synthase NPs and Self adjuvant nanoparticles (SANPs) evaluated for their role in viral detection [51,52,53,54,55,56,57,58,59,60,61,62,63,64,65].
NPs have several advantages over other nano-designs first easy to synthesize, unique in size/dimension, and can accommodate a wide range of conjugating materials [66, 67]. In diagnostic development NPs provide instant detection as described by Moitra et al. using gold NPs based on colorimetric platform results in COVID19 detection in less than 10 min using RNA samples [68]. The study utilizes antisense oligonucleotides (ASOs) specific for N-gene (Nucleocapsid phosphoprotein) of SARS-CoV-2. For viral detection using different NPs two-approach becomes popular one using an optical platform and a second electrochemical. Kim et al. demonstrated a label-free spectrophotometry method for the detection of MERS-CoV in clinical samples. In the study, citrate-capped AuNPs conjugated with thiolated ssDNA probes were used for viral detection [69]. The developed method not only provides rapid viral detection but also allows a large surface area essential for accuracy. In addition to optical methods, electrochemical-based platforms utilize nucleic acid detection, and Lew et al. developed AuNPs-based detection methods not only provide rapid detection but also amplify the signal that helps in the detection of the virus even at low titer [70]. NPs offer a wide range of sample detection for the presence of viruses where samples from sputum, throat, swabs, urine, plasma, feces, oral swabs, whole blood, and saliva can be used effectively [71].
2.4 Nanotechnology-based materials in surface decontaminations
Spread and transmission of nSARS-CoV2 are primarily via aerosol and fomites. Chemical disinfectants precisely chlorides, peroxides, quaternary amines, and alcohols are used for surface disinfection and sterilization of surfaces and personal protective equipment [72]. Though the chemicals disinfectants for nSARS-CoV2 showed promising results in sterilizing surfaces and PPE however several limitations remain associated such as public health and environmental issues [19]. Additionally, the concentration of chemical disinfectants affects the efficacy and activity associated with short timed. Nanotechnology-based formulations were developed for viral sterilization and surface cleaning showed the promising result. Various nano-designs are used to develop disinfectant formulations using NPs; Gold, Silver, Copper, titanium dioxide, etc [73]. These nano-materials offer disinfectant activity against coronavirus via generating reactive oxygen species (ROS), generating photo-dynamic and photo-thermal capabilities [74]. It is evident NPs and other nano-designs are cytotoxic and hence biodegradable nanomaterials such as polymeric lipid-based formulation. Querido et al. developed and evaluated self disinfecting nanoformulation to control infections including nASRS-CoV2 [75]. Vaze et al. explained mechanism of air utilizing engineering water nanostructures based on nano-sanitizers (Table 3) [76].
2.5 Nanotechnology-based vaccine against COVID19
In early 2020 after the nSARS-CoV2 genome sequencing vaccine design was started where several platforms such as protein, Micro RNA molecules, whole virus, and nucleic acid were used. As a result in late 2020 vaccine become available for clinical use (emergency uses). Vaccines elicit both antibody (humoral)-mediated immunity (AMI) and cell-mediated immunity (CMI), long-lived immune responses, and immune memory. Vaccines are associated with premature degradation of light/heavy chain subunits and non-specific delivery reduces efficacy. Silva et al. studied the role of nano-designs and formulations not only to improve the stability of vaccine subunits but also to target delivery [77]. The use of nanotechnology in vaccine development is not new and earlier several vaccine prototypes were developed for viruses such as hepatitis, H1N1 influenza, and coronaviruses as well. Previously Li et al. investigated antigen delivery against the hepatitis B virus using HBsAg-functionalized solid lipid nanoparticles (SLNs) [78]. The result demonstrated a strong immune response including cell-mediated and antigen mediated with HBsAg functionalized solid lipid nanoparticles. The study also reports a higher cellular update and minimal cellular toxicity with HBs-Ag SLNPs. Nanotechnology-based formulation/inventions are also used as the carrier for immunogenic molecules where targeted delivery remains a key concern (Fig. 2). Several findings demonstrated the use of poly lactic-co-glycolic acid (PLGA), polymeric NPs, and calcium phosphate as cargo for immunogenic molecule delivery [79]. Study has showed polyethyleneimine nanoparticles conjugated with spike protein of SARS-CoV showed enhanced immune response in the animal model [80]. Zhao et al. formulated a vaccine candidate (ARCoV) based on a lipid-nanoparticles-encapsulated mRNA (mRNA-LNP) encoding the receptor-binding domain (RBD) of SARS-CoV-2, leading to Th1-biased cellular responses and production of effective neutralizing antibodies against SARS-CoV-2 as shown in mice and non-human primates [55]. A detail of the vaccine designed against nSARS-CoV2 using nanotechnology is summarized in Table 4.
2.6 Nanotechnology-based therapeutics to combat COVID19
The application is nanotechnology is vast and in the COVID19 pandemic nanotechnology designs, formulations and inventions become part of the drug development process to combat disease [82, 83]. These formulations/designs are being used for two major objectives one as the carrier for drug delivery and the second as therapeutic (Fig. 2). Nano-designs easily penetrate respiratory epithelia and precisely NPs due to nanoscale size and ease in functionalization offer a wide range of physicochemical properties. Indeed, nanotechnology-based cargoes are ideal for targeted drug delivery however in the COVID19 pandemic another avenue of nanotechnology was explored i.e. therapeutic [84]. Nanotechnology remains associated with novel drug development against nSARS-CoV2 and evaluation of repurposing therapeutic as well. Loczechin et al. investigated the therapeutic potential of functionalized carbon quantum dots (CQDs) against human coronaviruses [83]. The mechanism proposed that CQDs restrict the entry of HCoV-229E due to the functional group attached to CQDs. The study also reported CQDs to inhibit viral replication and reduce titer volume significantly. Huang et al. investigated MERS-CoV inactivation using gold nanorods (AuNRs) where a series of heptad repeat 1 (HR1) peptide inhibitors restrict the entry of MERS-CoV2 to the host cell. Several findings have demonstrated that nSARS-CoV2 and ACEII receptor require a conformational change for viral entry into the host cell [81]. Abo-zeid et al. demonstrated that Fe2O3 NPs strongly interacts with S1-RBD of the SARS-CoV-2 and restrict viral entry to the host cell significantly [85].
Key therapeutics that remains associated with the treatment of COVID19 includes IFN-α, Lopinavir/ritonavir, Ribavirin, Chloroquine phosphate, and Arbidol. Additionally, Kaletra (Lopinavir/Ritonavir), hydroxychloroquine, oseltamivir phosphate, and Azithromycin have shown promising results as antiviral/anti-inflammatory agents against nSARS-CoV2. PegIntron® and Pegasys® are two FDA-approved NPs containing PEG-interferon alfa 2b and PEG-interferon 2a for the treatment of COVID19 [86]. Additionally, Chitosan NPs were used for INFα conjugated with Lopinavir/ritonavir were used for the management of COVID19. Another finding demonstrated that Glutathione-capped Ag2S nano-clusters were also potent nanomaterial for coronavirus suppressing. Ag NPs absorb the –SH groups of viral proteins and could serve as an effective therapeutic against nSARS-CoV2 [87]. Similarly, the study demonstrated a strong antiviral activity of graphene oxide (GO) with Ag NPs study against Feline coronavirus (FCoV). There is a grown list of nano-formulations (Table 5) that are being used/under trial for the treatment of COVID19 and or serving as a carrier for therapeutic including polymeric NPs, chaperone-mediated ferritin nanoparticles, Nano-bodies, Self-Assembling Protein Nanoparticles (SAPN), an adenoviral vector encoding Ad5, spike protein nanoparticles, VLPs, AuNPs, AgNPs, and Lumazine synthase NPs [81, 88].
3 Challenges and prospects
The key challenge with nanotechnology-based inventions and formulations intended for clinical uses is toxicity. Further surface functionalization often opted to reduce toxicity however difficult to remove. Another major challenge that remains associated with the use of nanotechnology is the non-availability of pretested formulations as novel SARS-CoV is new and diagnosis, detection, surveillance, therapeutic and vaccine uses need extensive preclinical and clinical studies to profile safety and efficacy profile. On several occasions, these nanotechnology-based inventions and formulations seem effective in animal models but fail when evaluated in the clinical setting. Nanotechnology-based formulations in the management of the COVID19 pandemic are still evolving and changing the genomic structure of nSARS-CoV2 continuously posing limitations on the effectiveness of the technology. In the last two years with the COVID19 pandemic and previously evaluated nano-formulations with other coronaviruses SARS-CoV and MERS-CoV along with influenza H1N1 and hepatitis showed a promising future in a wide range of applications including diagnosis and detection, design of biosensor for surveillance and monitoring, antiviral fabric and coating to mask and PPE, development of novel therapeutic and repurposing of existing therapeutic and vaccine development. Sterilization, surface decontamination, sanitization of skin, etc. pumped/released tonnes of the chemical into the environment does have serious complications to human health and the environment. Hence a sustainable approach remains the utmost priority where nano-formulation with reuses and self-degradation capabilities are required. Certainly, nanotechnology-based inventions/formulations and designs are superior in many aspects to conventional approaches, and in the future, more effective and robust materials will be available to fight against infection.
4 Conclusion
The outbreak of nSARS-CoV2 in December 2019 tuned in the global pandemic COVID19 affected the lives of millions of people worldwide and is still underway. The fight against COVID19 and the spread of nSARS-CoV 2 and its variants are multidimensional where the diagnosis of new cases, surveillance of the environment (air and water), development of robust and rapid diagnostic kits, therapeutic and vaccine do require integration of modern technology. Nanotechnology-based formulations, designs, and inventions had shown promising results in the fight against nSARS-CoV2 (causative agent) and COVID19 (disease). The last two yeast nanotechnology-based formulations/inventions/designs fulfilled the critical need for the designing of efficient vaccines to prevent virus infection, early and fast diagnosis by the high sensitivity and selectivity diagnostic kits, and effective antiviral and protective therapeutics to decline and eliminate the viral load and side effects derived from tissue damages. Nanotechnology response in the COVID19 pandemic was outstanding including new diagnostic methods/tools, antiviral nano-formulation, coated fabric for mask and PPE, new drug development along with repurposing and vaccine development. Nanotechnology not only fulfills current requirements to fight against COVID19 pandemic but also offers a sustainable and environmentally friendly technology. Though there are a few limitations with nanotechnology-based products such as cytotoxicity and these limitations are being addressed via functionalization of nano-designs and formulation. Modern medicine coupled with nanotechnology provides a wide range of application and fight against not only nSARS-CoV2 but also other viral/microbial pathogens.
Change history
02 July 2024
A Correction to this paper has been published: https://doi.org/10.1007/s12553-024-00891-2
References
Baloch S, Baloch MA, Zheng T, Pei X. The coronavirus disease 2019 (COVID-19) pandemic. Tohoku J Exp Med. 2020;250(4):271–8. https://doi.org/10.1620/tjem.250.271.
Mahalmani VM, Mahendru D, Semwal A, et al. COVID-19 pandemic: a review based on current evidence. Indian J Pharmacol. 2020;52(2):117–29. https://doi.org/10.4103/ijp.IJP_310_20.
Cucinotta D, Vanelli M. WHO declares COVID-19 a pandemic. Acta Biomed. 2020;91(1):157–60. https://doi.org/10.23750/abm.v91i1.9397.
Muralidar S, Ambi SV, Sekaran S, Krishnan UM. The emergence of COVID-19 as a global pandemic: understanding the epidemiology, immune response and potential therapeutic targets of SARS-CoV-2. Biochimie. 2020;179:85–100. https://doi.org/10.1016/j.biochi.2020.09.018.
Velavan TP, Meyer CG. The COVID-19 epidemic. Trop Med Int Health. 2020;25(3):278–80. https://doi.org/10.1111/tmi.13383.
Rauf A, Abu-Izneid T, Olatunde A, et al. COVID-19 pandemic: epidemiology, etiology, conventional and non-conventional therapies. Int J Environ Res Public Health. 2020;17(21):8155. https://doi.org/10.3390/ijerph17218155.
Lotfi M, Hamblin MR, Rezaei N. COVID-19: transmission, prevention, and potential therapeutic opportunities. Clin Chim Acta. 2020;508:254–66. https://doi.org/10.1016/j.cca.2020.05.044.
Helmy YA, Fawzy M, Elaswad A, Sobieh A, Kenney SP, Shehata AA. The COVID-19 pandemic: a comprehensive review of taxonomy, genetics, epidemiology, diagnosis, treatment, and control. J Clin Med. 2020;9(4):1225. https://doi.org/10.3390/jcm9041225.
Li H, Liu SM, Yu XH, Tang SL, Tang CK. Coronavirus disease 2019 (COVID-19): current status and future perspectives. Int J Antimicrob Agents. 2020;55(5):105951. https://doi.org/10.1016/j.ijantimicag.2020.105951.
Dhama K, Khan S, Tiwari R, et al. Coronavirus Disease 2019-COVID-19. Clin Microbiol Rev. 2020;33(4):e00028-20. https://doi.org/10.1128/CMR.00028-20.
Liu YC, Kuo RL, Shih SR. COVID-19: The first documented coronavirus pandemic in history. Biomed J. 2020;43(4):328–33. https://doi.org/10.1016/j.bj.2020.04.007.
Xiang J, Yan M, Li H, Liu T, Lin C, Huang S, Shen C. Evaluation of enzyme-linked immunoassay and colloidal gold-immunochromatographic assay kit for detection of novel coronavirus (SARS-Cov-2) causing an outbreak of pneumonia (COVID-19). MedRxiv. 2020. https://doi.org/10.1101/2020.02.27.20028787.
Tavakol S, Zahmatkeshan M, Mohammadinejad R, et al. The role of nanotechnology in current COVID-19 outbreak. Heliyon. 2021;7(4):e06841. https://doi.org/10.1016/j.heliyon.2021.e06841.
Gutiérrez Rodelo C, et al. Zinc associated nanomaterials and their intervention in emerging respiratory viruses: journey to the field of biomedicine and biomaterials. Coord Chem Rev. 2022;457:214402. https://doi.org/10.1016/j.ccr.2021.214402.
Jacofsky D, Jacofsky EM, Jacofsky M. Understanding antibody testing for COVID-19. J Arthroplasty. 2020;35(7S):S74–81. https://doi.org/10.1016/j.arth.2020.04.055.
Cano-Vicent A, Tuñón-Molina A, Martí M, et al. Antiviral face mask functionalized with solidified hand soap: low-cost infection prevention clothing against enveloped viruses such as SARS-CoV-2. ACS Omega. 2021;6(36):23495–503. https://doi.org/10.1021/acsomega.1c03511.
Larsen GS, et al. Polymer, additives, and processing effects on N95 filter performance. ACS Appl Polym Mater. 2021;3:1022–31.
Zhou J, Hu Z, Zabihi F, Chen Z, Zhu M. Progress and perspective of antiviral protective material. Adv Fiber Mater. 2020;2:123–39.
Muthiah G, Sarkar A, Roy S, et al. Nanotechnology toolkit for combating COVID-19 and beyond. Chem Nano Mat. 2022;8(4):e202100505. https://doi.org/10.1002/cnma.202100505.
Foffa I, Losi P, Quaranta P, Cara A, Al Kayal T, D’Acunto M, Presciuttini G, Pistello M, Soldani G. A copper nanoparticles-based polymeric spray coating: nanoshield against Sars-Cov-2. J Appl Biomater Funct Mater. 2022;20:22808000221076326. https://doi.org/10.1177/22808000221076326.
Pemmada R, Zhu X, Dash M, Zhou Y, Ramakrishna S, Peng X, Thomas V, Jain S, Nanda HS. Science-based strategies of antiviral coatings with viricidal properties for the COVID-19 like pandemics. Mater (Basel). 2020;13(18):4041. https://doi.org/10.3390/ma13184041.
Imani SM, Ladouceur L, Marshall T, Maclachlan R, Soleymani L, Didar TF. Antimicrobial nanomaterials and coatings: current mechanisms and future perspectives to control the spread of viruses including SARS-CoV-2. ACS Nano. 2020;14(10):12341–69. https://doi.org/10.1021/acsnano.0c05937.
Tseng CC, Pan ZM, Chang CH. Application of a quaternary ammonium agent on surgical face masks before use for pre-decontamination of nosocomial infection-related bioaerosols. Aerosol Sci Technol. 2016;50:199–210.
Liebeskind LS, Allred GD, U.S. Patent and Trade Office. Methods for using water-stabalized organosilanes. 2003. (U.S. Patent No. 6,632,805).
Liebeskind LS, Allred GD, U.S. Patent and Trademark Office. Water-stabilized organosilane compounds and methods for using the same. 2001. (U.S. Patent No. 6,221,944).
Liebeskind LS, Allred GD, U.S. Patent and Trademark Office. Water-stabilized organosilane compounds and methods for using the same. 1999. (U.S. Patent No. 5,959,014).
Paranthaman MP, Peroutka-Bigus N, Larsen KR, et al. Effective antiviral coatings for deactivating SARS-CoV-2 virus on N95 respirator masks or filters. Mater Today Adv. 2022;14:100228. https://doi.org/10.1016/j.mtadv.2022.100228.
Balagna C, Perero S, Percivalle E, Nepita EV, Ferraris M. Virucidal effect against coronavirus SARS-CoV-2 of a silver nanocluster/silica composite sputtered coating. Open Ceramics. 2020;1.
Damokhi A, Yousefinejad S, Fakherpour A, Jahangiri M. Improvement of performance and function in respiratory protection equipment using nanomaterials. J Nanopart Res. 2022;24(4):76. https://doi.org/10.1007/s11051-022-05460-0.
Deng W, Sun Y, Yao X, et al. Masks for COVID-19. Adv Sci (Weinh). 2022;9(3):e2102189. https://doi.org/10.1002/advs.202102189.
Jung S-Y, Kang KW, Lee E-Y, Seo D-W, Kim H-L, Kim H, Kwon T, Park H-L, Kim H, Lee S-M. Heterologous prime–boost vaccination with adenoviral vector and protein nanoparticles induces both Th1 and Th2 responses against Middle East respiratory syndrome coronavirus. Vaccine. 2018;36(24):3468–76.
He L, Tai W, Li J, Chen Y, Gao Y, Li J, Sun S, Zhou Y, Du L, Zhao G. Enhanced ability of oligomeric nanobodies targeting MERS coronavirus receptor-binding domain. Viruses. 2019;11(2):166.
Sousa BC, Massar CJ, Gleason MA, et al. On the emergence of antibacterial and antiviral copper cold spray coatings. J Biol Eng. 2021;15:8. https://doi.org/10.1186/s13036-021-00256-7.
Takeda Y, Jamsransuren D, Nagao T, Fukui Y, Matsuda S, Ogawa H. Application of copper iodide nanoparticle-doped film and fabric to inactivate SARS-CoV-2 via the virucidal activity of cuprous ions (Cu+). Appl Environ Microbiol. 2021;87(24):e0182421. https://doi.org/10.1128/AEM.01824-21.
Meister TL, Fortmann J, Breisch M, Sengstock C, Steinmann E, Köller M, Pfaender S, Ludwig A. Nanoscale copper and silver thin film systems display differences in antiviral and antibacterial properties. Sci Rep. 2022;12(1):7193. https://doi.org/10.1038/s41598-022-11212-w.
Souri M, Chiani M, Farhangi A, et al. Anti-COVID-19 Nanomaterials: directions to improve prevention, diagnosis, and treatment. Nanomaterials (Basel). 2022;12(5):783. https://doi.org/10.3390/nano12050783. Published 2022 Feb 25.
Ha T, Pham TTM, Kim M, Kim YH, Park JH, Seo JH, Kim KM, Ha E. Antiviral activities of high energy e-beam induced copper nanoparticles against H1N1 influenza virus. Nanomaterials (Basel). 2022;12(2):268. https://doi.org/10.3390/nano12020268.
Gupta V, Mohapatra S, Mishra H, et al. Nanotechnology in cosmetics and cosmeceuticals-a review of latest advancements. Gels. 2022;8(3):173. https://doi.org/10.3390/gels8030173.
Rabiee N, Bagherzadeh M, Ghadiri AM, Kiani M, Aldhaher A, Ramakrishna S, Tahriri M, Tayebi L, Webster TJ. Green synthesis of ZnO NPs via Salvia hispanica: evaluation of potential antioxidant, antibacterial, mammalian cell viability, H1N1 influenza virus inhibition and photocatalytic activities. J Biomed Nanotechnol. 2020;16(4):456–66. https://doi.org/10.1166/jbn.2020.2916.
Yüce M, Filiztekin E, Özkaya KG. COVID-19 diagnosis-a review of current methods. Biosens Bioelectron. 2021;172:112752. https://doi.org/10.1016/j.bios.2020.112752.
Sarkar J, Das S, Aich S, Bhattacharyya P, Acharya K. Antiviral potential of nanoparticles for the treatment of Coronavirus infections. J Trace Elem Med Biol. 2022;72:126977. https://doi.org/10.1016/j.jtemb.2022.126977.
Rakowska PD, Tiddia M, Faruqui N, et al. Antiviral surfaces and coatings and their mechanisms of action. Commun Mater. 2021;2:53. https://doi.org/10.1038/s43246-021-00153-y.
Tiwari AK, Mishra A, Pandey G, Gupta MK, Pandey PC. Nanotechnology: a potential weapon to fight against COVID-19. Part Part Syst Charact. 2022;39(1):2100159. https://doi.org/10.1002/ppsc.202100159.
Prasher P, Sharma M. Nanotechnology-based self-sterilizing surfaces and their potential in combating COVID-19. Nanomed (Lond). 2021;16(14):1183–6. https://doi.org/10.2217/nnm-2021-0079.
Tang P, Zhang Z, El-Moghazy AY, Wisuthiphaet N, Nitin, Sun G. Daylight-induced antibacterial and antiviral cotton cloth for offensive personal protection. ACS Appl Mater Interfaces. 2020;12(44):49442–51.
Sundberg K, Champagne VK, McNally B, Helfritch D, Sisson RD. Effectiveness of nanomaterial copper cold spray surfaces on inactivation of influenza A virus. J Biotechnol Biomater. 2015. https://doi.org/10.4172/2155-952X.1000205.
Kumar R, Nayak M, Sahoo GC, Pandey K, Sarkar MC, Ansari Y, Das VNR, Topno RK, Bhawna, Madhukar M, Das P. Iron oxide nanoparticles based antiviral activity of H1N1 influenza a virus. J Infect Chemother. 2019;25(5):325–9. https://doi.org/10.1016/j.jiac.2018.12.006.
Sun H, Jiang C, Wu L, Bai X, Zhai S. Cytotoxicity-related bioeffects induced by nanoparticles: the role of surface chemistry. Front Bioeng Biotechnol. 2019;7:414. https://doi.org/10.3389/fbioe.2019.00414.
Wang S, Sun H, Ang HM, Tadé MO. Adsorptive remediation of environmental pollutants using novel graphene-based nanomaterials. Chem Eng J. 2013;226:336–47. https://doi.org/10.1016/j.cej.2013.04.070.
Cavalcanti IDL, Nogueira MCdBL. Pharmaceutical nanotechnology: which products are been designed against COVID-19? J Nanoparticle Res. 2020;22(9):1–11.
Kevadiya BD, Machhi J, Herskovitz J, Oleynikov MD, et al. Diagnostics for SARS-CoV-2 infections. Nat Mater. 2021;20(5):593–605. https://doi.org/10.1038/s41563-020-00906-z.
Qiu G, Gai Z, Tao Y, Schmitt J, Kullak-Ublick GA, Wang J. Dual-functional plasmonic photothermal biosensors for highly accurate severe acute respiratory syndrome coronavirus 2 detection. ACS Nano. 2020;14(5):5268–77.
Ahmed SR, Nagy É, Neethirajan S. Self-assembled star-shaped chiroplasmonic gold nanoparticles for an ultrasensitive chiro-immunosensor for viruses. RSC Adv. 2017;7(65):40849–57.
Raj VS, Okba NM, Gutierrez-Alvarez J, Drabek D, van Dieren B, Widagdo W, Lamers MM, Widjaja I, Fernandez-Delgado R, Sola I. Chimeric camel/human heavy-chain antibodies protect against MERS-CoV infection. Sci Adv. 2018;4(8):eaas9667.
Zhao G, He L, Sun S, Qiu H, Tai W, Chen J, Li J, Chen Y, Guo Y, Wang Y. A novel nanobody targeting Middle East respiratory syndrome coronavirus (MERS-CoV) receptor-binding domain has potent cross-neutralizing activity and protective efficacy against MERS-CoV. J Virol. 2018;92(18):10–1128.
Wilken L, McPherson A. Application of camelid heavy-chain variable domains (VHHs) in prevention and treatment of bacterial and viral infections. Int Rev Immunol. 2018;37(1):69–76.
Van Heeke G, Allosery K, De Brabandere V, De Smedt T, Detalle L, de Fougerolles A. Nanobodies® as inhaled biotherapeutics for lung diseases. Pharmacol Therapeut. 2017;169:47–56.
Coleman CM, Venkataraman T, Liu YV, Glenn GM, Smith GE, Flyer DC, Frieman MB. MERS-CoV spike nanoparticles protect mice from MERS-CoV infection. Vaccine. 2017;35(12):1586–9.
Naskalska A, Dabrowska A, Nowak P, Szczepanski A, Jasik K, Milewska A, Ochman M, Zeglen S, Rajfur Z, Pyrc K. Novel coronavirus-like particles targeting cells lining the respiratory tract. PLoS ONE. 2018;13(9):e0203489.
Kim H, Park M, Hwang J, Kim JH, Chung D-R, Lee K-S, Kang M. Development of label-free colorimetric assay for MERS-CoV using gold nanoparticles. ACS Sens. 2019;4(5):1306–12.
Teengam P, Siangproh W, Tuantranont A, Vilaivan T, Chailapakul O, Henry CS. Multiplex paper-based colorimetric DNA sensor using pyrrolidinyl peptide nucleic acid-induced AgNPs aggregation for detecting MERS-CoV, MTB, and HPV oligonucleotides. Anal Chem. 2017;89(10):5428–35.
Widagdo W, Okba NM, Li W, De Jong A, De Swart RL, Begeman L, Van Den Brand JM, Bosch B-J, Haagmans BL. Species-specific colocalization of Middle East respiratory syndrome coronavirus attachment and entry receptors. J Virol. 2019;93(16):10–1128.
Kim E, Erdos G, Huang S, Kenniston TW, Balmert SC, Carey CD, Raj VS, Epperly MW, Klimstra WB, Haagmans BL. Microneedle array delivered recombinant coronavirus vaccines: immunogenicity and rapid translational development. EBioMedicine. 2020;55:102743.
Rahman MZ, Hoque ME, Alam MR, et al. Face masks to combat coronavirus (COVID-19)-processing, roles, requirements, efficacy, risk and sustainability. Polym (Basel). 2022;14(7):1296. https://doi.org/10.3390/polym14071296.
Charelli LE, de Mattos GC, de Jesus Sousa-Batista A, Pinto JC, Balbino TA. Polymeric nanoparticles as therapeutic agents against coronavirus disease. J Nanopart Res. 2022;24(1):12. https://doi.org/10.1007/s11051-022-05396-5.
Widagdo W, Okba NM, Li W, De Jong A, De Swart RL, Begeman L, Van Den Brand JM, Bosch B-J, Haagmans BL. Species-specific colocalization of Middle East respiratory syndrome coronavirus attachment and entry receptors. J Virol. 2019;93(16):10–128.
Zhao H, Liu F, Xie W, et al. Ultrasensitive supersandwich-type electrochemical sensor for SARS-CoV-2 from the infected COVID-19 patients using a smartphone. Sens Actuators B. 2020;327:128899.
Moitra P, Alafeef M, Dighe K, Frieman MB, Pan D. Selective naked-eye detection of SARS-CoV-2 mediated by N gene targeted antisense oligonucleotide capped plasmonic nanoparticles. ACS Nano. 2020;14(6):7617–27. https://doi.org/10.1021/acsnano.0c03822.
Kim Y-S, Son A, Kim J, Kwon SB, Kim MH, Kim P, Kim J, Byun YH, Sung J, Lee J. Chaperna-mediated assembly of ferritin-based Middle East respiratory syndrome-coronavirus nanoparticles. Front Immunol. 2018;9:1093.
Lew TTS, Aung KMM, Ow SY, et al. Epitope-Functionalized Gold Nanoparticles for rapid and selective detection of SARS-CoV-2 IgG antibodies [published online ahead of print, 2021 Jun 16]. ACS Nano. 2021;15(7):12286–12267. https://doi.org/10.1021/acsnano.1c04091.
Zhang NN, Li XF, Deng YQ, Zhao H, et al. A thermostable mRNA vaccine against COVID-19. Cell. 2020;182(5):1271–83. https://doi.org/10.1016/j.cell.2020.07.024.
Weiss C, Carriere M, Fusco L, et al. Toward nanotechnology-enabled approaches against the COVID-19 pandemic. ACS Nano. 2020;14(6):6383–406. https://doi.org/10.1021/acsnano.0c03697.
Saravanan M, Mostafavi E, Vincent S, Negash H, Andavar R, Perumal V, Chandra N, Narayanasamy S, Kalimuthu K, Barabadi H. Nanotechnology-based approaches for emerging and re-emerging viruses: special emphasis on COVID-19. Microb Pathog. 2021;156:104908. https://doi.org/10.1016/j.micpath.2021.104908.
Heo KJ, Jeong SB, Shin J, et al. Water-repellent TiO2-organic dye-based air filters for efficient visible-light activated photochemical inactivation against bioaerosols. Nano Lett. 2021;21(4):1576–83.
Querido MM, Aguiar L, Neves P, Pereira CC, Teixeira JP. Self-disinfecting surfaces and infection control. Colloids Surf B Biointerfaces. 2019;178:8–21.
Vaze N, Pyrgiotakis G, McDevitt J, Mena L, Melo A, Bedugnis A, et al. Inactivation of common hospital acquired pathogens on surfaces and in air utilizing engineered water nanostructures (EWNS) based nano-sanitizers. Nanomed Nanotechnol Biol Med. 2019;18:234–42.
Silva JM, Videira M, Gaspar R, Préat V, Florindo HF. Immune system targeting by biodegradable nanoparticles for cancer vaccines. J Control Release. 2013;168(2):179–99. https://doi.org/10.1016/j.jconrel.2013.03.010.
Li J, Helal ZH, Karch CP, Mishra N, Girshick T, Garmendia A, Burkhard P, Khan MI. A self-adjuvanted nanoparticle based vaccine against infectious bronchitis virus. PloS One. 2018;13(9):e0203771.
Sales-Junior PA, Guzman F, Vargas MI, Sossai S, Patarroyo VAM, González CZ, Patarroyo JH. Use of biodegradable PLGA microspheres as a slow release delivery system for the Boophilus microplus synthetic vaccine SBm7462. Vet Immunol Immunopathol. 2005;107(3–4):281–90. https://doi.org/10.1016/j.vetimm.2005.05.004.
Verleysen E, Ledecq M, Siciliani L, et al. Titanium dioxide particles frequently present in face masks intended for general use require regulatory control. Sci Rep. 2022;12:2529. https://doi.org/10.1038/s41598-022-06605-w.
Hu C-MJ, Chang W-S, Fang Z-S, Chen Y-T, Wang W-L, Tsai H-H, Chueh L-L, Takano T, Hohdatsu T, Chen H-W. Nanoparticulate vacuolar ATPase blocker exhibits potent host-targeted antiviral activity against feline coronavirus. Sci Rep. 2017;7(1):1–11.
Mello VM, Eller CM, Salvio AL, Nascimento FF, Figueiredo CM, Silva ESRF, Sousa PSF, Costa PF, Paiva AAP, Mares-Guias MAMM, Lemos ERS, Horta MAP. Effectiveness of face masks in blocking the transmission of SARS-CoV-2: a preliminary evaluation of masks used by SARS-CoV-2-infected individuals. PLoS ONE. 2022;17(2):e0264389. https://doi.org/10.1371/journal.pone.0264389.
Łoczechin A, Séron K, Barras A, et al. Functional carbon quantum dots as medical countermeasures to human coronavirus. ACS Appl Mater Interfaces. 2019;11(46):42964–74. https://doi.org/10.1021/acsami.9b15032.
Huang X, Li M, Xu Y, Zhang J, et al. Novel gold nanorod-based HR1 peptide inhibitor for Middle East respiratory syndrome coronavirus. ACS Appl Mater Interfaces. 2019;11(22):19799–807. https://doi.org/10.1021/acsami.9b04240.
Abo-Zeid Y, Ismail NSM, McLean GR, Hamdy NM. A molecular docking study repurposes FDA approved iron oxide nanoparticles to treat and control COVID-19 infection. Eur J Pharm Sci. 2020;153:105465. https://doi.org/10.1016/j.ejps.2020.105465.
Coleman CM, Venkataraman T, Liu YV, Glenn GM, Smith GE, Flyer DC, Frieman MB. MERS-CoV spike nanoparticles protect mice from MERS-CoV infection. Vaccine. 2017;35(12):1586–9.
Li W, Hulswit RJ, Widjaja I, Raj VS, McBride R, Peng W, Widagdo W, Tortorici MA, Van Dieren B, Lang Y. Identification of sialic acid-binding function for the Middle East respiratory syndrome coronavirus spike glycoprotein. Proc Natl Acad Sci USA. 2017;114(40):E8508–17.
Xu S, Bai X, Wang L. Exploration of photothermal sensors based on photothermally responsive materials: a brief review. Inorg Chem Front. 2018;5(4):751–9.
Acknowledgements
Author would like to thank MetaBioGenX Pvt. Ltd Jhansi, India for proving facility for the study. Author would like to thanks All India Institute of Medical Sciences AIIMS Bhopal, Madhya Pradesh for providing facility and support for the study.
Funding
No funding was received for the present study.
Author information
Authors and Affiliations
Contributions
STK and MKV conceptualize the study, MKV and HS retrieved scientific information, SKT and MKV wrote and edited the draft.
Corresponding author
Ethics declarations
Ethical statement
Not applicable.
Conflict of interest
Author declares no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The original online version of this article was revised: Author name has been corrected.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kumar, S., Singh, H. & Verma, M.K. Nanotechnology based materials and inventions to fight against COVID-19: recent progress in the development of robust diagnostics, surveillance tools, therapeutics and vaccines. Health Technol. (2024). https://doi.org/10.1007/s12553-024-00880-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12553-024-00880-5