Abstract
Several meiofaunal samplings along the continental slope of Central America and the Antilles through the Caribbean Sea have revealed a rich kinorhynch fauna of undescribed species. The present contribution includes the description of two new species of the allomalorhagid genera Cristaphyes Sánchez et al., 2016 and Fujuriphyes Sánchez et al., 2016 and two new species of the cyclorhagid genus Echinoderes Claparède, 1863, as well as the first record of the previously known Cristaphyes longicornis (Higgins, 1983), Echinoderes astridae Sørensen, 2014, Echinoderes horni Higgins, 1983, Echinoderes imperforatus Higgins, 1983 and Echinoderes spinifurca Sørensen et al., 2005 for Haiti and the Dominican Republic (Hispaniola Island, Greater Antilles) together with new morphological information of the former. All the new species are formally described. Furthermore, we discuss the possibility of an expansion in the intraspecific morphological variation of C. longicornis, geographical remarks on the Caribbean Kinorhyncha and compare the morphological differences between the newly described species and their most similar congeners.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The phylum Kinorhyncha encompasses a group of marine, holobenthic, free-living, meiofaunal species that inhabit the upper centimetres of oceanic or estuarine sediment from intertidal to abyssal depths (Higgins 1988; Sørensen and Pardos 2008). Currently, the phylum is composed of two classes, Cyclorhagida and Allomalorhagida, based on both morphological and molecular evidence (Sørensen et al. 2015). Kinorhynchs are worldwide distributed, but the available biogeographical data is strongly biased by sampling strategies of the specialists in the phylum that have intensively sampled certain areas to the detriment of others poorly studied, being the North Sea, the Mediterranean Sea, the north-western Atlantic American shoreline and the Sea of Japan and adjacent waters as the most sampled areas of the world so far (Neuhaus 2013).
In this context, it has to be emphasised how little is known about the kinorhynch fauna of the Caribbean region, even though the region generally hosts the greatest marine biodiversity of the western Atlantic Ocean and is a global-scale hotspot of marine species (Roberts et al. 2002; Miloslavich et al. 2010). The first approach to describe the Caribbean kinorhynchs was done by Kirsteuer (1964) with the description of Echinoderes caribiensis Kirsteuer, 1964 from the Mochima National Park, Venezuela. A more extensive sampling campaign was accomplished by Higgins (1983), although this study was limited to the coral reef ecosystem at Carrie Bow Cay, Belize. This contribution increased the number of known Caribbean kinorhynch species to 19, with the description of 5 new species of Cyclorhagida and 13 species of Allomalorhagida (Higgins 1983). More recently, the area of Bocas del Toro, Colón Island and Bastimento Island (Panama) has been extensively studied by Sørensen (2006), Neuhaus et al. (2014) and Pardos et al. (2016a, b), resulting in 5 new species of Cyclorhagida, 2 new species of Allomalorhagida and 7 new reports, bringing the total number of valid kinorhynch species for the Caribbean Sea up to 31.
All the available information on Caribbean Kinorhyncha is limited to the continental shelf, disregarding the Antilles shelves. The main aim of the present project is to describe the still unknown kinorhynch fauna of the aforementioned archipelago through the study of the Caribbean samples obtained by Dr. R. P. Higgins and his colleagues from 1966 to 1991 and deposited at the Smithsonian Institution National Museum of Natural History (NMNH). The present contribution focuses on Hispaniola Island, one of the Greater Antilles, and includes the descriptions of four new species of kinorhynchs and the first report of Cristaphyes longicornis (Higgins, 1983), Echinoderes astridae Sørensen, 2014 and Echinoderes spinifurca Sørensen et al., 2005 for the area, with additional details on morphology of C. longicornis. Further papers will deal with the remaining Caribbean samples.
Materials and methods
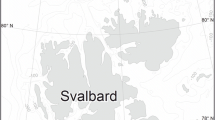
Hispaniola Island is a part of the Greater Antilles of the Caribbean Sea (western Atlantic Ocean) and located between Cuba and Puerto Rico, separated by the Windward Passage and the Mona Passage respectively (Fig. 1a). Samples were taken at several intertidal and subtidal localities, of which seven yielded the analysed specimens in the present study (Table 1). Stations L1 to L6 are situated off the northern coast of the island, while Station L7 is located in the southern part (Fig. 1b). Detailed data on samples and localities is summarised in Table 1. Sampling was performed using a meiobenthic dredge (Fleeger et al. 1988), and meiofauna was separated using the bubble and blot method (Higgins 1964; Sørensen and Pardos 2008). Meiofaunal specimens were fixed in 4% formalin and preserved in propylene glycol.
Unmounted kinorhynchs were observed under a Motic® SMZ-168 stereo zoom microscope and picked up with an Irwin loop. For light microscopy (LM), animals were dehydrated through a graded series of 25%, 50%, 75% and 100% glycerine to be mounted on a glass slide or a Cobb’s aluminium slide holder with Fluoromount G® and sealed with Depex®. The mounted specimens were studied and photographed using an Olympus© BX51-P microscope with differential interference contrast (DIC) optics equipped with an Olympus© DP-70 camera. For scanning electron microscopy (SEM), specimens were transferred to 70% ethanol and then progressively dehydrated through a series of 80%, 90%, 95% and 100% ethanol. Hexamethyldisilazane (HMDS) was used for chemical drying through a HMDS-ethanol series. Specimens were coated with gold and mounted on aluminium stubs to be examined with a JSM® 6335-F JEOL SEM at the ICTS Centro Nacional de Microscopía Electrónica (Complutense University of Madrid, Spain). Identification to genus level of the cyclorhagid kinorhynchs was done according to the dichotomous key provided by Sørensen and Pardos (2008), whereas the allomalorhagid kinorhynchs were identified following the genus diagnoses provided by Sánchez et al. (2016). Line drawings and images plates composition was done using Adobe® Photoshop 6.0 and Illustrator CC-2014 software.
Results
A total of ten species belonging to four genera and three families were recorded along the Hispaniola Island coastline (Table 1). Of these, four species are herein newly described and five are newly reported for Great Antilles.
Class Allomalorhagida Sørensen et al., 2015
Family Pycnophyidae Zelinka, 1896
Genus Cristaphyes Sánchez et al., 2016
Cristaphyes retractilis sp. nov.
(Figs. 2, 3 and 4 and Tables 2 and 3)
Line art illustrations of Cristaphyes retractilis sp. nov. a Male, ventral view; b Male, dorsal view; c Female, segments 1–2, ventral view; d Male, segments 10–11, ventral view. Scale: 100 μm. bsj ball-and-socket joint, dcr dorsal cuticular ridge, dpl dorsal placid, gco glandular cell outlet, ldgco laterodorsal glandular cell outlet, ldse laterodorsal seta, ldss laterodorsal sensory spot, lvse, lateroventral seta, mdp middorsal process, ms muscular scar, pdse paradorsal seta, pf pectinate fringe, plse paralateral seta, ps penile spine, S segment followed by number of corresponding segment, sdgco subdorsal glandular cell outlet, sdss subdorsal sensory spot, spf secondary pectinate fringe, vcr ventral cuticular ridge, vlse ventrolateral seta, vmgco ventromedial glandular cell outlet, vmse ventromedial seta, vmss ventromedial sensory spot, vmt ventromedial tube, vpl ventral placid
Light micrographs showing trunk overview and details in the segments and the sexual dimorphism of Cristaphyes retractilis sp. nov. a Female, dorsal overview of trunk; b Female, ventral overview of trunk; c Female, dorsal overview of segment 1, showing the strongly denticulated anterior margin of the tergal plate; d Female, middorsal, paradorsal, subdorsal and laterodorsal regions on right half of tergal plates of segments 1–2; e Female, detail of the middorsal, paradorsal, subdorsal and laterodorsal regions on left half of tergal plate of segment 2; f Female, lateroventral region on left half of sternal plates of segment 2. g Male, ventromedial region of sternal plates of segments 1–2; h Female, ventrolateral and ventromedial regions on right half of sternal plates of segments 2–3; i Female, middorsal, paradorsal, subdorsal and laterodorsal reigons on left half of tergal plates of segments 7–8; j Female, ventrolateral and ventromedial regions of left half of sternal plates of segments 8–9; k Female, ventrolateral and ventromedial regions of right half of sternal plates of segment 10. l Epibiontic Ciliophora on the cuticle surface on the laterodorsal section of segment 9. Scales: a, b: 100 μm; c–l: 20 μm. cr cuticular ridge, ldse laterodorsal seta, lvse lateroventral seta, mdp middorsal process, pdse paradorsal seta, vlse ventrolateral seta, vmse ventromedial seta, vmt ventromedial tube; sensory spots are marked as dashed circles, and glandular cell outlets as continuous circles
Scanning electron micrographs showing overviews and details in the cuticular trunk morphology of Cristaphyes retractilis sp. nov. a Dorsal overview of trunk; b Ventral overview of trunk; c Middorsal, paradorsal, subdorsal and laterodorsal regions of right half of tergal plate of segment 3; d Ventrolateral and ventromedial regions on left half of sternal plates of segments 2–3; e Detail of ventromedial seta and sensory spot of segment 3; f Detail of middorsal process and paradorsal setae of segment 4; g Detail of middorsal process and paradorsal setae of segment 8; h Ventromedial region on left half of sternal plates of segment 9; i Lateroventral and ventrolateral regions on left half of tergal and sernal plates of segment 10. Scales: a, b: 100 μm; c, d, h–j: 10 μm; e–g: 5 μm. ldse laterodorsal seta, lvse lateroventral seta, mdp middorsal process, pdse paradorsal seta, vlse ventrolateral seta, vmse ventromedial seta; sensory spots are marked as dashed circles
Material examined
Type material. Holotype, adult male, collected on 06 November 1980 at Monte Cristi Bay, Dominican Republic, Hispaniola Island, western Atlantic Ocean: 19° 53′ 12′′ N, 071° 40′ 00′′ W (L3) (Table 1; Fig. 1b) at 3–4 m depth in muddy sand; mounted in Fluoromount G®, deposited at NMNH under accession number: USNM 1490909. Paratypes, four adult males and five adult females; six of them with same collecting data as holotype, mounted in Fluoromount G® and deposited at NMNH under accession numbers: USNM 1490910–1,490,915; two of them collected on 03 November 1980 at Puerto Blanco Harbour, Dominical Republic, Hispaniola Island, western Atlantic Ocean: 19° 54′ 24′′ N, 70° 56′ 24′′ W (L2) (Table 1; Fig. 1b) at 3 m depth in silty mud; mounted in Fluoromount G®, deposited at NMNH under accession numbers: USNM 1490916–1490917; one of them collected on 04 November 1980 at La Isabela, Dominican Republic, Hispaniola Island, western Atlantic Ocean: 19°53′ 18′′ N, 71° 05′ 36′′ W (Table 1; Fig. 1b) at 4 m depth in silty mud; mounted in Fluoromount G®, deposited at NMNH under accession number: USNM 1490918.
Non-type material. Twenty-two additional specimens from the same localities as the holotype and paratypes (17 mounted for LM, 5 mounted for SEM), also deposited at NMNH under accession numbers: USNM 1490919–1490936.
Diagnosis
Cristaphyes with middorsal processes on segments 1–9. Pairs of paradorsal setae on segments 2, 4, 6 and 8, laterodorsal setae on segments 2–9, paralateral setae on segment 1, lateroventral setae on segments 2, 4, 6, 8 and 10, ventrolateral setae on segments 1, 3–8 and 10, one pair of ventromedial setae on segments 2–8 and two pairs of ventromedial setae on segment 9. Setae in each paradorsal pair differ in length and shape: one short and thick, the other one longer and progressively tapering towards end. Laterodorsal setae mesially shifted on uneven segments compared to those of even segments. Segments 10 and 11 retractable into segment 9. Lateral terminal spines absent.
Etymology
From the Latin “retractilis”, meaning retracting, which refers to the segments 10 and 11 that often appear retracted into the precedent segments.
Description
See Table 2 for measurements and dimensions, and Table 3 for summary of cuticular process, seta, tube, glandular cell outlet and sensory spot locations.
Head and neck. Head with retractable mouth cone and introvert. The collected specimens were not suitable for head examinations, hence data on number and arrangement of scalids and oral styles is not available.
Neck with four dorsal and two ventral sclerotized placids (Fig. 2a–c). Dorsal placids rectangular; mesial ones broader than lateral ones (Fig. 2b). Ventral placids much more elongate and trapezoidal, getting thinner towards the lateral sides (Fig. 2a, c).
Trunk. Trunk with 11 segments (Figs. 2a, b, 3a, b and 4a, b). Segment 1 with one tergal, two episternal and one trapezoidal midsternal plate (Figs. 2a–c, 3a, b and 4a, b); remaining ones with one tergal and two sternal cuticular plates (Figs. 2a–d, 3a, b and 4a, b). Midsternal and tergosternal junctions, as well as junctions between midsternal and episternal plates, as conspicuous lines externally on the cuticle (Figs. 2a, d, 3b and 4b). Tergal cuticular plates slightly bulging middorsally. Sternal plates reach their maximum width at segment 5, but almost constant in width throughout the trunk, slightly tapering at the last two trunk segments (Figs. 2a, b, 3a, b and 4a, b). Sternal cuticular plates are relatively wide in the ratio maximum width to total trunk length (MSW-5:TL average ratio = 32.1%), giving the animal a plump appearance (Figs. 2a, b, 3a, b and 4a, b). Middorsal processes on segments 1–9, keel-shaped, with pointed tips that surpass the posterior segment margins, turning progressively longer towards the posterior end (Figs. 2b, 3a, d, i and 4a, c, f–g). Middorsal process of segment 9 shorter and thinner than previous ones, but also pointed and extending beyond the terminal trunk segment (Fig. 2b). Segments 1–10 with paired, rounded glandular cell outlets in subdorsal positions (Figs. 2b and 3d–e, i). Segments 2–10 with paired cuticular ridges in laterodorsal position followed by small, intracuticular wrinkled glandular cell outlets on their posterior margin (Fig. 2b). Segments 1–10 furthermore with paired, rounded glandular cell outlets in ventromedial position, those on segment 1 being laterally displaced (Figs. 2a, c–d and 3g–h, j–k). Segments 2–10 furthermore with paired ventral cuticular ridges marking the ventrolateral-ventromedial border followed by small, intracuticular wrinkled glandular cell outlets on their posterior margin (Figs. 2a, c, 3h). Cuticular hairs distributed all over the trunk cuticle; tergal plates bear elongate, very thin hairs that become thicker on middorsal processes and shorter towards posterior segment margin (Fig. 4c, f–g); sternal plates bear very short, scale-like, thick hairs that become thinner towards posterior segment margin (Fig. 4d–e, h). Pachycycli and ball-and-socket joints on segments 2–10 (Fig. 2a–d). Apodemes not observed. Primary pectinate fringe finely serrated (Fig. 2a–d), appearing smooth under LM (Fig. 3d–e, g–j); secondary pectinate fringe also finely serrated, wavy (Fig. 2a–c), dorsally protruded to the beginning of middorsal processes; free flaps covering anterior part of subsequent segment. Muscular scars as rounded to oval hairless areas (Fig. 2a–d), quite inconspicuous.
Segment 1 with middorsal process flanked by more densely covered hairy areas that slightly surpasses the posterior segment margin (Figs. 2b and 3d). Midsternal plate forming a midventral convex extension on its posterior edge (Figs. 2a, c, 3b and 4b). Anterolateral margins of the tergal cuticular plate as horn-shaped extensions (Figs. 2a–c, 3a, b and 4a, b). Anterior margin of tergal plate strongly denticulated, followed by a smooth area (Figs. 2b, 3a, c and 4a). Two pairs of sensory spots in subdorsal position; one pair of sensory spots in laterodorsal position, all of them located at the anterior half of the cuticular plate (Figs. 2b and 3d); sensory spots on this and remaining segments rounded to oval, with several rings of cuticular papillae surrounding a central pore (Fig. 4c, e, h, i). Paired setae in paralateral and ventrolateral position (Fig. 2a–c).
Segment 2 with middorsal process as on preceding segment (Figs. 2b and 3d–e). Paired paradorsal setae differing in size: one seta very short (ca. 2 μm long) and relatively thick, the other one longer (ca. 8 μm long) and progressively thinner towards end (Figs. 2b and 3d–e). Paired setae also in laterodorsal, lateroventral and ventromedial positions (Figs. 2a–c, 3d–f, h and 4d); laterodorsal setae located laterally to the muscular scars; ventromedial setae located near the border between the ventrolateral and ventromedial sections (Figs. 2a–c and 4d). Paired sensory spots in subdorsal, laterodorsal and ventromedial positions (Figs. 2a–c and 3d–e, h). Sexually dimorphic male tubes in ventromedial position (Figs. 2a and 3g). Tubes long, flexible, thin-walled, with blunt tip bearing a minute pore.
Segment 3 with middorsal process as on preceding segments (Figs. 2b and 4c). Paired setae in laterodorsal, ventrolateral and ventromedial positions (Figs. 2a, b, 3h and 4c–e); laterodorsal setae mesially displaced compared to those of the precedent segment; ventromedial setae located more lateral than the ventromedial muscular scars (Fig. 2a, b). Two pairs of sensory spots in subdorsal position plus one pair of sensory spots in laterodorsal and ventromedial positions (Figs. 2a, b, 3h and 4c–e).
Segment 4 with middorsal process as on preceding segments (Figs. 2b and 4f). Paired paradorsal setae differing in size: one seta very short (ca. 2 μm long) and relatively thick, the other one longer (ca. 8 μm long) and progressively narrower towards end (Figs. 2b and 4f). Paired setae also in laterodorsal, lateroventral, ventrolateral and ventromedial positions (Fig. 2a, b); laterodorsal setae located laterally to the muscular scars; ventrolateral setae located near the junction between the ventrolateral and ventromedial sections (Fig. 2a, b). Two pairs of sensory spots in subdorsal position plus one pair of sensory spots in laterodorsal and ventromedial positions (Fig. 2a, b).
Segment 5 similar to segment 3 (Fig. 2a, b).
Segment 6 similar to segment 4 (Fig. 2a, b).
Segment 7 similar to segments 3 and 5 (Figs. 2a, b and 3i).
Segment 8 with tergal plate resembling that of segments 4 and 6, but with both paradorsal setae located at the same side of the middorsal process (Figs. 2b, 3i and 4g). Sternal plates identical with those of segments 4 and 6 (Figs. 2a and 3j).
Segment 9 with poorly developed middorsal process that barely surpasses the posterior margin (Fig. 2b). A pair of setae in laterodorsal position, and two pairs in ventromedial position (Figs. 2a, b, 3j and 4 h), one of them close to the border between the ventrolateral and ventromedial sections (Fig. 2a). One pair of sensory spots in subdorsal position; two pairs of sensory spots in laterodorsal and ventromedial positions, the last one located between the ventromedial setae (Figs. 2a, b, 3j and 4h). Nephridiopores not observed. Some specimens with epibiontic Ciliophora protozoans on the dorsal cuticular surface (Fig. 3l).
Segments 10 and 11 often retracted into precedent segment (Figs. 2a, b, 3a, b and 4a, b, i). Segment 10 with paired setae in lateroventral and ventrolateral positions and paired sensory spots in ventromedial position (Figs. 2d, 3k and 4i), all of them near the posterior segment margin. Segment 11 without cuticular appendages (Fig. 2d). Males with two pairs of penile spines and genital pores surrounded by tuft of long hairs in between segments 10 and 11 (Fig. 2d). Lateral terminal spines absent.
Notes on diagnostic and taxonomic features
Cristaphyes retractilis sp. nov. may be easily distinguished from most of its congeners by the lack of lateral terminal spines. Only C. anomalus (Lang, 1953) from Reloncaví Estuary (Chile, eastern Pacific Ocean), C. belizensis (Higgins, 1983) from Carrie Bow Cay (Belize, Caribbean Sea), C. harrisoni (Pardos et al., 2016) from Taboga Island (Panama, eastern Pacific Ocean), C. panamensis (Pardos et al., 2016) from Bastimento Island (Panama, Caribbean Sea), C. phyllotropis (Brown and Higgins, 1983) from Hunter Bay (Australia, western Pacific Ocean), C. rabaulensis (Adrianov, 1999 in Adrianov and Malakhov 1999) from Rabaul (Papua New Guinea, western Pacific Ocean), C. spinosus (Lang, 1949) from Falkland Islands (western Atlantic Ocean) and C. yushini (Adrianov, 1989) from the Seto Inland Sea (Japan, western Pacific Ocean) also lack lateral terminal spines (Lang 1949, 1953; Brown and Higgins 1983; Higgins 1983; Adrianov 1989; Adrianov and Malakhov 1999; Pardos et al. 2016a, b; Sánchez et al. 2016). Cristaphyes retractilis sp. nov. possesses paired paradorsal setae on segments 2, 4, 6 and 8, which is only shared with C. harrisoni (Pardos et al. 2016b; Sánchez et al. 2016), as the remaining species having a different paradorsal setae arrangement (paired on segments 3–6 and 8–9 in C. anomalus; unpaired on segments 2, 6, 8 and 9–10 and paired on segment 4 in C. belizensis; unpaired on segments 3, 5, 7 and 9–10 and paired on segments 2, 4, 6 and 8 in C. panamensis; at least unpaired on segments 2, 5–6 and 8 in C. phyllotropis; and unpaired on segments 2, 4, 6 and 8–9 in C. yushini) (Lang 1953; Brown and Higgins 1983; Higgins 1983; Adrianov 1989; Adrianov and Malakhov 1999; Pardos et al. 2016a; Sánchez et al. 2016). Nevertheless, C. retractilis sp. nov. and C. harrisoni are easily distinguished by the pattern of the ventral setae: the former has paired ventrolateral setae on segments 1, 3–8 and 10, one pair of ventromedial setae on segments 2–8 and two pairs of ventromedial setae on segment 9, whereas the latter is characterised by having paired ventrolateral setae on segments 1, 5 and 10 and paired ventromedial setae on segments 3–8 (Pardos et al. 2016b; Sánchez et al. 2016). The available information on setae arrangement of C. rabaulensis and C. spinosus is scarce (Lang 1949; Adrianov and Malakhov 1999; Sánchez et al. 2016), but the differences with C. retractilis sp. nov. are in the position of lateroventral setae: C. rabaulensis only has lateroventral setae on segment 6 (and possibly on segment 5) and C. spinosus on segments 2, 4 and 9–10 (and possibly on segment 5), while C. retractilis sp. nov. possesses lateroventral setae on all even segments (Lang 1949; Adrianov and Malakhov 1999; Sánchez et al. 2016).
Regarding the general setae arrangement, C. retractilis sp. nov. is most similar to C. carinatus (Zelinka, 1928) from Naples (Italy, Mediterranean Sea) by the presence of several pairs of setae in ventrolateral position (segments 1, 3–8 and 10 in C. retractilis sp. nov. and segments 3–9 in C. carinatus) (Zelinka 1928; Sánchez et al. 2016), as the remaining congeners possess ventrolateral setae only on one, two or three segments (Sánchez et al. 2016). Nevertheless, C. retractilis sp. nov. is characterised by possessing pairs of paradorsal setae on segments 2, 4, 6 and 8, one pair of lateroventral setae on segments 2, 4, 6, 8 and 10 and two pairs of ventromedial setae on segment 9 and by lacking lateral terminal spines, while C. carinatus has pairs of paradorsal setae on segments 1–9, one pair of lateroventral setae on segments 2–10, two pairs of ventromedial setae on segments 3–9 and a pair of lateral terminal spines (Zelinka 1928; Sánchez et al. 2016).
The ability to retract segments 10 and 11 into the preceding segments has not previously been observed in any Cristaphyes, although it has been described for other kinorhynchs, including Echinoderes applicitus Ostmann et al., 2012 from Java (western Pacific Ocean), E. maxwelli (Omer-Cooper, 1957) from South Africa, E. strii Pardos et al., 2016 from Pedro González Island (Panama, eastern Pacific Ocean) and Pycnophyes alexandroi Pardos et al., 2016 from Taboga Island (Panama, eastern Pacific Ocean) (Omer-Cooper 1957; Ostmann et al. 2012; Pardos et al. 2016a, b). This character is hardly explained as an artefact of the fixation or the preservation process because it has been frequently found in many specimens of samples from different localities, as also observed by Pardos et al. (2016b). Finally, the presence of two pairs of ventromedial setae on the same segment is only present in the newly described species (segment 9), C. belizensis (Higgins, 1983) from Carrie Bow Cay, Caribbean Sea (segments 4–9) and C. carinatus (segments 3–9) (Zelinka 1928; Higgins 1983; Sánchez et al. 2016).
Cristaphyes cf. longicornis
Material examined.
Non-type material. Seven adult females, four prepared for LM with Fluoromount G® and three prepared for SEM, collected on 03 November 1980 at Puerto Blanco Harbour, Dominican Republic, Hispaniola Island, western Atlantic Ocean: 19° 54′ 24′′ N, 70° 56′ 24′′ W (L2) (Table 1; Fig. 1b) at 3 m depth in silty mud, deposited at NMHN under accession numbers: USNM 1490937–1490941.
Description
The morphology of the examined specimens generally followed the original description of Cristaphyes longicornis (Higgins, 1983). Hence, only deviations from the original description are mentioned in the following.
Segment 1 with paired ventrolateral sensory spots located near the posterior margin of segment (Fig. 5a). Segments 6 and 8 with paired lateroventral setae (Fig. 5b), as reported in the original description but not observed in the type material by Sánchez et al. (2016). Segment 10 lacking lateroventral setae, bearing a pair of widened ventrolateral sensory spots located near the posterior margin of segment (Fig. 5c). Secondary pectinate fringe on segments 1–11 developed as three transvers finely serrated fringes, two of them near the anterior segments margins, the other one almost reaching the posterior margins (Fig. 5d). Conspicuous paraventral apodemes present on segments 9–10. Cuticular hairs on segments 1–11 very small, scale-like, basally widened, abruptly tapering, randomly distributed through tergal and sternal cuticular plates (Fig. 5e), densely covering a middorsal bulging on segment 10 (Fig. 5f).
Scanning electron micrographs showing details in the cuticular trunk morphology of Cristaphyes cf. longicornis. a Ventrolateral region on right half of sternal plates of segment 1; b Lateroventral region on left half of sternal plates of segments 5–8; c Ventrolateral region on left half of sternal plates of segment 10; d Middorsal, paradorsal and subdorsal regions of tergal plate of segment 3, arrows mark the secondary pectinate fringes; e Detail of cuticular hairs of segment 9 sternal plate; f Detail of cuticular, hairy bulging of segment 10. Scales: a, c, d, f: 10 μm; b: 20 μm; e: 2 μm. avlss anterior ventrolateral sensory spot, lvse lateroventral seta, pvlss posterior ventrolateral sensory spot, vlse ventrolateral seta, vlss ventrolateral sensory spot; sensory spots are marked as dashed circles
Notes on diagnostic and taxonomic features
Several Cristaphyes specimens were studied and herein reported as C. cf. longicornis, as they agree with the main diagnostic characters of the species: trunk tapering slightly beginning with segment 7, lateral terminal spines long and recurved at tip, middorsal processes on segments 2–10, unpaired paradorsal setae on segments 4, 6 and 8, laterodorsal setae on segments 2–9, paralateral setae on segment 1, lateroventral setae on segments 2, 4 and 10, ventrolateral setae on segments 5 and 10, ventromedial setae on segments 1 and 3–9 and males with sexually dimorphic tubes in ventromedial position on segment 2 (Higgins 1983; Sánchez et al. 2016).
Some morphological differences from the original description were observed, including the presence of paired ventrolateral sensory spots located near the posterior margin of segment 1 and the absence of lateroventral setae on segment 10. Additionally, Higgins (1983) determined the presence of lateroventral setae on segments 2, 4, 6 and 8, but according to the revision of the type material by Sánchez et al. (2016), C. longicornis possesses these structures only on segments 2 and 4. The specimens herein reported as C. cf. longicornis also possess lateroventral setae on segments 6 and 8, as originally described.
The observed morphological discrepancies may have passed unnoticed to Higgins (1983) and Sánchez et al. (2016) likely because they are barely seen under LM and the badly preserved type material, preventing proper observation of the cuticular characters. However, intraspecific variation in the arrangement of sensory spots of segment 1 and lateroventral setae may exist, defining morphologically different populations through the Caribbean Basin. Moreover, the presence of paired posterior ventrolateral sensory spots on segment 1, paired lateroventral setae on segments 6 and 8 and the absence of paired lateroventral setae on segment 10 in the specimens collected from Hispaniola Island could indicate the existence of a new species. However, we do not consider this morphological variation as sufficient to erect a new species, especially when taking the bad preservation stage of the type specimens into account.
Genus Fujuriphyes Sánchez et al., 2016
Fujuriphyes dalii sp. nov.
(Figs. 6, 7 and 8 and Tables 4 and 5)
Line art illustrations of Fujuriphyes dalii sp. nov. a Male, ventral view; b Male, dorsal view; c Female, segments 1–2, ventral view; d Male, segments 10–11, ventral view. Scale: 50 μm. ap apodeme, bsj ball-and-socket joint, dcr dorsal cuticular ridge, dpl dorsal placid, gco glandular cell outlet, ldse laterodorsal seta, ldss laterodorsal sensory spot, lts lateral terminal spine, lvse lateroventral seta, mdgco middorsal glandular cell outlet, ms muscular scar, pdse paradorsal seta, pdss paradorsal sensory spot, pf pectinate fringe, plne paralateral nephridiopore, plse paralateral seta, ps penile spine, S segment followed by number of corresponding segment, sdgco subdorsal glandular cell outlet, sdss subdorsal sensory spot, spf secondary pectinate fringe, type3ss type 3 sensory spot, vcr ventral cuticular ridge, vlse ventrolateral seta, vlss ventrolateral sensory spot, vmgco ventromedial glandular cell outlet, vmse ventromedial seta, vmss ventromedial sensory spot, vpl ventral placid
Light micrographs showing trunk overview and cuticular details in the segments of Fujuriphyes dalii sp. nov. a Ventral overview of trunk; b Dorsal overview of trunk; c Dorsal placids; d Ventral placids; e Dorsal overview of segment 1, showing the strongly denticulated anterior margin of tergal plate followed by a crenulated area with high longitudinal ridges and regions of the tergal plate superficially spotted; f Dorsal view of segment 1; g Ventral view of segment 1; h Ventrolateral and ventromedial regions on left half of sternal plates of segments 2–4; i Middorsal, paradorsal, subdorsal and laterodorsal regions on right half of tergal plates of segments 2–4; j Lateroventral region on right half of tergal plates of segments 2–4; k Ventrolateral and ventromedial regions on left half of sternal plates of segment 5; l Ventrolateral and ventromedial regions on left half of sternal plates of segment 10; m Middorsal, paradorsal, subdorsal and laterodorsal regions on right half of tergal plates of segments 9–10; n Dorsal view of segment 11; o Detail of the left lateral terminal spine. Scales: a, b, 100 μm; c–n, 20 μm; o, 50 μm. ldse laterodorsal seta, lvse lateroventral seta, ms muscular scar, pdse paradorsal seta, plse paralateral seta, S segment followed by number of corresponding segment, vlse ventrolateral seta, vmse ventromedial seta; sensory spots are marked as dashed circles, and glandular cell outlets as continuous circles
Scanning electron micrographs showing overviews and details in the cuticular trunk morphology of Fujuriphyes dalii sp. nov. a Dorsal overview of trunk; b Ventral overview of trunk; c Ventromedial region on left half of sternal plates of segment 2, showing the sensory spot and the female sexually dimorphic seta; d Middorsal and paradorsal regions of tergal plate of segment 4, showing the middorsal glandular cell outlet, the paradorsal seta and the paradorsal sensory spots; e Ventrolateral region on left half of sternal plates of segment 4, showing the ventromedial sensory spot and the ventrolateral seta; f Subdorsal and laterodorsal regions on right half of tergal plate of segment 11, showing the type 3 sensory spots. Scales: a, b: 100 μm; c–e: 10 μm; f: 5 μm. mdgco middorsal glandular cell outlet, pdse paradorsal seta, vlse ventrolateral seta, vmse ventromedial seta; sensory spots are marked as dashed circles, and setae and glandular cell outlets as continuous circles
Material examined
Type material. Holotype, adult male, collected on 02 November 1980 at 200 m east of Puerto Plata Harbour, Dominican Republic, Hispaniola Island, western Atlantic Ocean: 19°48′ 12′′ N, 70° 42′ 00′′ W (L1) (Table 1; Fig. 1b) at 4–5 m depth in brown sandy mud; mounted in Fluoromount G®, deposited at NMHN under accession number: USNM 1490942. Paratypes, 8 adult males and 8 adult females; 14 of them with same collecting data as holotype, mounted in Fluoromount G® and deposited at NMHN under accession numbers: USNM 1490943-1490956; 2 of them collected on 06 November 1980 at Monte Cristi Bay, south-western of Capra Island, Dominican Republic, Hispaniola Island, western Atlantic Ocean: 19° 53′ 12′′ N, 71° 40′ 00′′ W (L3) (Table 1; Fig. 1b), at 3–4 m depth in muddy sand associated with assemblages of Thalassia sp., mounted in Fluoromount G®, deposited at NMHN under accession numbers: USNM 1490957-1490958.
Non-type material. Two additional specimens from the same locality of the holotype, mounted for SEM, also deposited at NMHN under accession number USNM 1490959.
Diagnosis
Fujuriphyes without middorsal processes or elevations; anterior margin of first segment strongly denticulated, followed by a crenulated area with elevated longitudinal ridges; ball-and-socket joints present on segments 2–5; unpaired paradorsal setae on segments 2, 4, 6 and 8, paired laterodorsal setae on segments 2–9, paired paralateral setae on segment 1, paired lateroventral setae on segments 2, 4 and 10, paired ventrolateral setae on segments 1, 3–7 and 10 (two pairs on segment 5), and a pair of ventromedial setae on segment 8–9.
Etymology
The name is dedicated to the prominent Spanish surrealist Salvador Dalí (1904–1989), whose peculiar moustache resembles the shape of the lateral terminal spines of the species.
Description
See Table 4 for measurements and dimensions, and Table 5 for summary of seta, spine, nephridiopore, glandular cell outlet and sensory spot locations.
Head and neck. Head with retractable mouth cone and introvert. The collected specimens were not suitable for head examinations, hence data on number and arrangement of scalids and oral styles is not available.
Neck with four dorsal and two ventral sclerotized placids (Figs. 6a–c and 7c, d). Dorsal placids rectangular, wide (Figs. 6b and 7c); mesial ones broader, with a small indentation in the middle of the posterior margin (Figs. 6b and 7c); lateral ones narrower (Figs. 6b and 7c). Ventral placids also rectangular but much more elongate, getting narrower towards the lateral sides (Figs. 6a, c and 7d).
Trunk. Trunk with 11 segments (Figs. 6a, b, 7a, b and 8a, b). Segment 1 with one tergal, two episternal and one trapezoidal midsternal plate; remaining segments with one tergal and two sternal cuticular plates (Figs. 6a–d, 7a, b and 8a, b). Midsternal and tergosternal junctions as conspicuous lines externally on the cuticle (Figs. 6a, c–d, 7a and 8b). Sternal plates reach their maximum width at segment 5, but almost constant in width throughout the trunk, slightly tapering at the last trunk segments (Figs. 6a, b, 7a, b and 8a, b). Sternal cuticular plates wide in ratio of maximum sternal width to trunk lenght (MSW-5:TL average ratio = 33.2%), giving the animal a plump appearance (Figs. 6a, b, 7a, b and 8a, b). Middorsal processes and elevations absent. Segments 2–9 with minute glandular cell outlets in middorsal position (Figs. 6b, 7i, m and 8d). Segments 2–10 furthermore with paired, oval, elongated glandular cell outlets in subdorsal position (Figs. 6b, and 7f, i, m). Segments 2–10 also with paired cuticular ridges in laterodorsal position followed by small, intracuticular wrinkled glandular cell outlets located on their inner posterior margin (Fig. 6b). Segments 1–10 furthermore with paired, rounded to oval glandular cell outlets in ventromedial position (first pair laterally displaced to ventrolateral position) (Figs. 6a, c and 7g–h, k, l). Segments 2–10 furthermore with paired ventral cuticular ridges marking the ventrolateral-ventromedial limit followed by small, intracuticular wrinkled glandular cell outlets located on their inner posterior margin (Fig. 6a, c). Cuticular hairs only on the tergosternal junction. Pachycycli and ball-and-socket joints on segments 2–5 (Fig. 6a, b). Paraventral apodemes on segments 9–10 (Fig. 6a). Pectinate fringe finely serrated (Figs. 6a–d), appears smooth under LM (Fig. 7f–i, k–m); secondary pectinate fringe also finely serrated (Fig. 6a, b); free flaps extend around the posterior segment margins. Muscular scars oval (Fig. 6a–d), scarcely detectable on most segments.
Segment 1 without middorsal cuticular processes and elevations. Anterolateral margins of the tergal cuticular plate as horn-shaped extensions (Figs. 6a–c, 7a, b, g and 8a, b). Anterior margin of tergal plate strongly denticulated, followed by a crenulated area with high longitudinal ridges (Figs. 6b, 7e and 8a). Paired setae in paralateral and ventrolateral positions (Figs. 6a–c and 7f–g). Paired sensory spots in paradorsal, subdorsal, laterodorsal and ventromedial positions, all of them located at the anterior half of the cuticular plates (Figs. 6a–c and 7f, g); sensory spots on this and remaining segments rounded to oval, with several rings of cuticular papillae surrounding a central pore (Fig. 8c–e).
Segment 2 without middorsal cuticular processes and elevations. Unpaired seta in paradorsal position (Figs. 6b and 7i). Paired setae in laterodorsal and lateroventral positions (Figs. 6a–c and 7i, j); laterodorsal pair located a bit more lateral than the muscular scar (Fig. 6b). Two pairs of sensory spots in subdorsal position; one pair of sensory spots in laterodorsal and ventromedial positions (Figs. 6a–c, 7h, i and 8c). Both males and females without ventromedial tubes, females with paired ventromedial setae (Figs. 6c, 7h and 8c).
Segment 3 without middorsal cuticular processes and elevations. Paired setae in laterodorsal and ventrolateral positions (Figs. 6a, b and 7h, i). Paired sensory spots in paradorsal, subdorsal, laterodorsal and ventromedial positions (Figs. 6a, b and 7h, i).
Segment 4 without middorsal cuticular processes and elevations. Unpaired seta in paradorsal position (Figs. 6b, 7i and 8d). Paired setae in laterodorsal, lateroventral and ventrolateral positions (Figs. 6a, b, 7h–j and 8e). Paired sensory spots in paradorsal, subdorsal, laterodorsal and ventromedial positions (Figs. 6a, b, 7h, i and 8d, e).
Segment 5 without middorsal cuticular processes and elevations. A pair of setae in laterodorsal position; two pairs of setae in ventrolateral position (Figs. 6a, b and 7k). A pair of sensory spots in paradorsa, subdorsal, laterodorsal and ventromedial positions (Figs. 6a, b and 7k).
Segment 6 without middorsal cuticular processes and elevations. Unpaired seta in paradorsal position (Fig. 6b). Paired setae in laterodorsal and ventrolateral positions (Fig. 6a, b). Paired sensory spots in paradorsal, subdorsal, laterodorsal and ventromedial positions (Fig. 6a, b).
Segment 7 similar to segment 3 (Fig. 6a, b).
Segment 8 without middorsal cuticular processes and elevations. Unpaired seta in paradorsal position (Fig. 6b). Paired setae in laterodorsal and ventromedial positions (Fig. 6a, b). Paired sensory spots in paradorsal, subdorsal, laterodorsal and ventromedial positions (Fig. 6a, b).
Segment 9 without middorsal cuticular processes and elevations. Protonephridial opnenings in paralateral position (Fig. 6b). Paired setae in laterodorsal and ventromedial positions; ventromedial setae laterally shifted, not aligned with segment 8 ventromedial setae (Fig. 6a, b). Two pairs of sensory spots in subdorsal position plus one pair of sensory spots in laterodorsal and ventromedial positions (Figs. 6a, b and 7m).
Segment 10 without middorsal cuticular processes and elevations. Paired setae in lateroventral and ventrolateral positions (the latter flanked by the sensory spots) (Figs. 6a, b and 7l, m). A pair of sensory spots in subdorsal and laterodorsal positions (the latter near the posterior margin of the tergal plate); two pairs of sensory spots in ventrolateral position (Figs. 6a, b and 7l,m).
Segment 11 with three pairs of type 3 sensory spots, one pair in subdorsal and two pairs in laterodorsal positions (Figs. 6b, 7n and 8f). Posterior ventral margin conspicuously serrated. Males with two pairs of penile spines and genital pores surrounded by tuft of long hairs in between segments 10 and 11 (Fig. 6d). Lateral terminal spines long (LTS:TL average ratio = 30.2%), stout, wide, apparently flexible (Figs. 6a, b, 7a, b, o and 8a, b).
Notes on diagnostic and taxonomic features
Currently, the genus accomodates six species: two from the Caribbean Sea, Fujuriphyes deirophorus (Higgins, 1983) and Fujuriphyes distentus (Higgins, 1983), one from the Gulf of Mexico, Fujuriphyes viserioni Sánchez, et al. 2019, one from the East China Sea, Fujuriphyes longispinosus Sánchez and Yamasaki, 2016, one from the Black and Mediterranean Seas, Fujuriphyes ponticus (Reinhard, 1881) and one from the Mediterranean Sea, Fujuriphyes rugosus (Zelinka, 1928) (Reinhard 1881; Zelinka 1928; Higgins 1983; Sánchez et al. 2012, 2016, 2019; Sánchez and Yamasaki 2016). Fujuriphyes dalii sp. nov. is easily distinguished from its congeners by the presence of paralateral setae on segment 1, lateroventral setae on segments 2, 4 and 10, a single pair of ventrolateral setae on segments 1, 3–7 and 10, with an extra pair on segment 5, ventromedial setae only on segments 8–9 and by the absence of male ventromedial tubes on segment 2 (see Reinhard 1881; Zelinka 1928; Higgins 1983; Sánchez and Yamasaki 2016; Sánchez et al. 2019 for original description of all known species of the genus and appendix of Sánchez et al. 2016 for updated morphological characters).
Fujuriphyes dalii sp. nov. lacks middorsal cuticular processes and elevations, whereas the remaining congeners posseess middorsal elevations on segments 1–9 (F. deirophorus and F. distentus), on segments 2–9 (F. ponticus and F. rugosus), on segment 3 (F. viserioni) or, at least, on segments 1–6 (F. longispinosus) (Reinhard 1881; Zelinka 1928; Higgins 1983; Sánchez and Yamasaki 2016; Sánchez et al. 2016, 2019). Regarding the setae arrangement, the recently described F. longispinosus from Nagannu Island, Japan (East China Sea) shows most resemblance to F. dalii sp. nov., as both species have two pairs of ventrolateral setae on segment 5 and a relatively low number of ventromedial setae (F. dalii sp. nov. possesses a pair of ventromedial setae on segments 8–9 and F. longispinosus is characterised by having paired, ventromedial setae on segments 2 and 9) (Sánchez and Yamasaki 2016; Sánchez et al. 2016). However, the arrangement of ventrolateral setae of F. dalii sp. nov. (one pair on segments 1, 3, 4, 6, 7 and 10, and two pairs on segment 5) is unique among its congeners. Furthermore, F. dalii sp. nov. lacks male-specific ventromedial tubes on segment 2 as F. longispinosus, F. ponticus and F. rugosus (Reinhard 1881; Zelinka 1928; Sánchez and Yamasaki 2016; Sánchez et al. 2016), whereas F. deirophorus and F. distentus possess these structures (Higgins 1983; Sánchez et al. 2016). Moreover, F. dalii sp. nov. has lateral terminal spines as F. longispinosus, F. ponticus and F. rugosus (Reinhard 1881; Zelinka 1928; Sánchez and Yamasaki 2016; Sánchez et al. 2016), while F. deirophorus and F. distentus lack lateral terminal spines (Higgins 1983; Sánchez et al. 2016).
Class Cyclorhagida (Zelinka, 1896) Sørensen et al. 2015
Family Echinoderidae Zelinka, 1894
Genus Echinoderes Claparède, 1863.
Echinoderes brevipes sp. nov.
(Figs. 9 and 10 and Tables 6 and 7)
Line art illustrations of Echinoderes brevipes sp. nov. a Female, ventral view; b Female, dorsal view; c Male, segments 10–11, ventral view; d Male, segments 10–11, dorsal view. Scale: 50 μm. dpl dorsal placid, lat lateral accessory tube, ldss laterodorsal sensory spot, ldt laterodorsal tube, ltas lateral terminal accessory spine, lts lateral terminal spine, lvs lateroventral spine, lvt lateroventral tube, mdgco1 middorsal type 1 glandular cell outlet, mds middorsal spine, mlss midlateral sensory spot, mvpl midventral placid, ne nephridiopore, pdgco1 paradorsal type 1 glandular cell outlet, pdss paradorsal sensory spot, pf pectinate fringe, ps penile spine, sdss subdorsal sensory spot, sdt subdorsal tube, slgco2 sublateral type 2 glandular cell outlet, te tergal extension, tp trichoscalid plate, vlss ventrolateral sensory spot, vlt ventrolateral tube, vmgco1 ventromedial type 1 glandular cell outlet
Light micrographs showing trunk overview and cuticular details in the segments of Echinoderes brevipes sp. nov. a Dorsal overview of trunk; b Ventral overview of trunk; c Dorsal view of introvert and neck, showing the dorsal placids and some trichoscalids, spinoscalids and outer oral styles; d Ventral view of introvert and neck, showing the ventral placids and some spinoscalids and outer oral styles; e Dorsal view of segments 1–2; f Ventral view of segments 1–2; g Dorsal overview of segments 4–10; h Ventral overview of segments 4–9; i Middorsal, paradorsal, subdorsal and laterodorsal regions of tergal plate of segment 7; j Middorsal, paradorsal, subdorsal and laterodorsal regions of tergal plate of segment 8; k Lateroventral region on left half of tergal plate of segments 6–9; l Middorsal, paradorsal, subdorsal and laterodorsal regions of tergal plate of segment 10; m Detail of a lateral terminal spine. Scales: a, b: 50 μm; c–m: 20 μm. dpl dorsal placid, lat lateral accessory tube, ldt laterodorsal tube, lts lateral terminal spine, lvs lateroventral spine, lvt lateroventral tube, mds middorsal spine, mvpl midventral placid, oos outer oral style, psps primary spinoscalid, S segment followed by number of corresponding segment, sdt subdorsal tube, slgco2 sublateral type 2 glandular cell outlet, sps spinoscalid, trs trichoscalid, vlt ventrolateral tube; type 1 glandular cell outlets are marked as continuous circles and sensory spots as dashed circles
Material examined.
Type material. Holotype, adult male, collected on 08 May 1976 off Santo Domingo, Dominican Republic, Hispaniola Island, Caribbean Sea: 18° 28′ 00′′ N, 69° 57′ 00′′ W (L7) (Table 1; Fig. 1b) at 0.7–1.0 m depth in an unknown sediment; mounted in Fluoromount G®, deposited at NMNH under accession number: USNM 1490960. Paratypes, two adult females and two adult males; all of them same collecting data as holotype, mounted in Fluoromount G® and deposited at NMNH under accession numbers: USNM 1490961–1,490,964.
Diagnosis
Echinoderes with middorsal spines on segments 4, 6, 8, increasing in length posteriorly; subdorsal and ventrolateral tubes plus sublateral glandular cell outlets type 2 on segment 2; lateroventral tubes on segment 5 and lateroventral spines on segments 6–9; lateral accessory tubes on segments 6–8; laterodorsal tubes on segments 8 and 10. Lateral terminal spines very short.
Etymology
From the Latin “brevis”, meaning short, and “pes”, meaning foot, which refers to the possession of conspicuously very short lateral terminal spines compared to the total trunk length of the species.
Description
See Table 6 for measurements and dimensions, and Table 7 for summary of spine, tube, nephridiopore, glandular cell outlet and sensory spot locations.
Head and neck. Head with retractable mouth cone and introvert (Fig. 10a–d). Internal part of mouth cone with several rings of inner oral styles; exact number, arrangement and morphology of inner oral styles not determined. External part of mouth cone with nine outer oral styles (Fig. 10c, d). Outer oral styles alternate in size between slightly longer and slightly shorter ones (Fig. 10c, d). Five long styles appear anterior to the odd numbered introvert sections, whereas four slightly shorter ones appear anterior to the even numbered ones, except in the middorsal section 6 where a style is missing (Fig. 10c, d). Outer oral styles with two jointed subunits, with a rectangular basis bearing a short fringe at its base, and a triangular, hook-like distal structure (Fig. 10c, d).
Introvert with seven rings of cuticular spinoscalids (Fig. 10c, d). Ring 01 with ten primary spinoscalids consisting of a short basal sheath and a distal end piece (Fig. 10c, d). Basal sheath with a proximal long fringe situated very close to the insertion point, bearing several flexible, elongated fringe tips, followed by a smooth part bearing another fringe with several long, flexible tips (Fig. 10c, d). Distal piece of the primary spinoscalids wide, rounded in cross-section, smooth, hook-like, with blunt tip (Fig. 10c, d). Remaining rings bear spinoscalids laterally compressed, with a long, smooth basis and elongate, thin, hook-like distal piece (Fig. 10c, d). Exact number, arrangement and detailed morphology of these spinoscalids not determined as they tended to be collapsed when mounted.
Neck with 16 trapezoidal placids, wider at base, with a distinct joint between the neck and segment 1 (Figs. 9a, b and 10c, d); midventral one widest (ca. 12 μm wide at base) (Figs. 9a and 10d), remaining ones alternate between wider and narrower (6–9 μm at base) (Figs. 9b and 10c). Placids closely situated together at base, distally separated by cuticular folds (Figs. 9a, b and 10c, d). A ring of six long, hairy trichoscalids associated with the placids of the neck is present, attached to small trichoscalid plates (Fig. 10c, d).
Trunk. Trunk with 11 segments (Figs. 9a, b and 10a, b). Segments 1–2 as closed cuticular rings; remaining ones with one tergal and two sternal cuticular plates (Figs. 9a, b and 10a, b). Midsternal junction as conspicuous line on the cuticle; tergosternal junctions mostly subcuticular (Figs. 9a, c and 10a, b). Tergal plates of anterior segments slightly bulging middorsally; posterior ones more flattened, giving the animal a tapering outline in lateral view. Sternal plates reach their maximum width at segment 5, progressively tapering towards the last trunk segments (Figs. 9a, b and 10a, b). Sternal plates relatively narrow compared to the total trunk length (MSW-5:TL average ratio = 24.6%), giving the animal a slender appearance (Figs. 9a, b and 10a, b).
Segment 1 without spines and tubes. Cuticular hairs distributed in two irregular, transvers rows mainly in the posterior half of the plate, that merge together in a single transvers row from ventrolateral to ventromedial area (Fig. 9a, b). Cuticular hairs on this and remaining segments long, filiform. Unpaired type 1 glandular cell outlet in middorsal position (Figs. 9b and 10e). Paired sensory spots in subdorsal and ventrolateral positions (Figs. 9a, b and 10e, f). Posterior segment margin straight, showing a pectinate fringe with a very weak serration (Fig. 9a, b).
Segment 2 with paired, thick tubes in subdorsal and ventrolateral positions (Figs. 9a,b and 10e, f). Cuticular hairs distributed in two irregular, transvers rows only on the posterior half of the plate, that merge together in an irregular ventral patch showing no definite pattern (Fig. 9a, b). Unpaired type 1 glandular cell outlet in middorsal position; paired type 2 glandular cell outlets in sublateral position (Figs. 9a, b and 10e, f). Paired sensory spots in laterodorsal and midlateral positions (Figs. 9b and 10e). Posterior segment margin and pectinate fringe as on the precedent segment (Fig. 9a, b).
Segment 3 with cuticular plates lacking spines and tubes. Cuticular hairs dorsally arranged in four wavy, transvers rows that increase in length posteriorly, plus a pair of paraventral patches (Fig. 9a, b). Unpaired type 1 glandular cell outlet in middorsal position; paired type 1 glandular cell outlets in ventromedial position (Fig. 9a, b). Paired sensory spots in subdorsal position (Fig. 9b). Primary pectinate fringe well-developed, with regular tips (Fig. 9a, b).
Segment 4 with a middorsal spine exceeding the posterior edge of the segment but not reaching the posterior margin of the following segment (Figs. 9b and 10g). Cuticular hairs arranged in five wavy, transvers rows that increase in length posteriorly, plus a pair of paraventral patches (Fig. 9a, b). Paired type 1 glandular cell outlets in paradorsal and ventromedial positions (Figs. 9a, b and 10g, h). Paired sensory spots in paradorsal position, located posteriorly to middorsal spine near the posterior margin of the segment (Figs. 9b and 10g). Pectinate fringe as on the precedent segment (Fig. 9a, b).
Segment 5 with paired, long, narrow tubes in lateroventral position (Figs. 9a and 10h). Cuticular hairs arranged in 5–6 wavy, transvers rows that increase in length posteriorly, plus a pair of paraventral patches (Fig. 9a, b). Unpaired type 1 glandular cell outlet in middorsal position; paired type 1 glandular cell outlets in ventromedial position (Figs. 9a, b and 10g, h). Paired sensory spots in subdorsal position (Figs. 9b and 10g). Pectinate fringe as on the precedent segment (Fig. 9a, b).
Segment 6 with a middorsal spine longer than that of segment 4, exceeding the posterior margin of the following segment, and paired, robust, lateroventral spines (Figs. 9a, b and 10g, h, k). Paired, long, narrow tubes in lateral accessory position (Figs. 9a and 10h, k). Cuticular hairs as on the preceding segment (Fig. 9a, b). Paired type 1 glandular cell outlets in paradorsal and ventromedial positions (Figs. 9a, b and 10g, h). Paired sensory spots in paradorsal position, located posteriorly to middorsal spine near the posterior margin of the segment (Figs. 9b and 10g). Pectinate fringe as on the precedent segment (Fig. 9a, b).
Segment 7 with paired, robust, lateroventral spines (Figs. 9a and 10h, k) and long, narrow tubes in lateral accessory position (Figs. 9a and 10h, k). Cuticular hairs as on the preceding segment (Fig. 9a, b). Unpaired type 1 glandular cell outlet in middorsal position; paired type 1 glandular cell outlets in ventromedial position (Figs. 9a, b and 10g, i). Paired sensory spots in subdorsal position (Figs. 9b and 10g, i). Pectinate fringe as on the preceding segment (Fig. 9a, b).
Segment 8 with a middorsal spine much longer than that of segment 6, almost reaching the posterior margin of the last trunk segment, and paired, robust, lateroventral spines (Figs. 9a, b and 10g, h, j, k). Paired, long, narrow tubes in laterodorsal and lateral accessory positions (Figs. 9a, b and 10g, h, j, k). Cuticular hairs arranged in 7–8 wavy, transvers rows that increase in length posteriorly, plus a pair of paraventral patches (Fig. 9a, b). Paired type 1 glandular cell outlets in paradorsal and ventromedial positions (Figs. 9a, b and 10g, h, j). Paired sensory spots in paradorsal position, located posteriorly to middorsal spine near the posterior margin of the segment (Fig. 9b and 10g, j). Pectinate fringe as on the preceding segment (Fig. 9a, b).
Segment 9 with paired, robust, lateroventral spines (Figs. 9a and 10h, k). Cuticular hairs arranged in 9–10 wavy, transversal rows that increase in length posteriorly, plus a pair of paraventral patches (Fig. 9a, b). Paired type 1 glandular cell outlets in paradorsal and ventromedial positions (Figs. 9a, b and 10g, h). Paired sensory spots in subdorsal position (Figs. 9b and 10g). Nephridiopore as a very small sieve plate, in lateral accessory position (Figs. 9a and 10h). Pectinate fringe as on the preceding segment (Fig. 9a, b).
Segment 10 with paired, long, narrow tubes in laterodorsal position in both sexes (Figs. 9b, d and 10l). Cuticular hairs arranged in 7–9 wavy, transvers rows that increase in length posteriorly, forming two separated dorsal patches on the tergal plate (Fig. 9a, b). Two unpaired type 1 glandular cell outlets in middorsal position; paired type 1 glandular cell outlets in ventromedial position, mesially displaced, not aligned with the previous ventromedial type 1 glandular cell outlets (Figs. 9a–d and 10g). Paired sensory spots in subdorsal position (Figs. 9b). Posterior segment margin deeply curved, extending in the ventromedial area, with a pectinate fringe as on the preceding segment (Fig. 9a–d).
Segment 11 with lateral terminal spines very thick and short (LTS:TL average ratio = 12.5%), robust and distally pointed, showing a hollow central cavity (Figs. 9a–d and 10m). Females with a pair of lateral terminal accessory spines, even shorter, about two thirds of length of lateral terminal spines (Fig. 9a, b). Males with three pairs of penile spines arising laterally under the pectinate fringe of the precedent segment; ventral and dorsal penile spines (ps1 and ps3) filiform, midlateral penile spine (ps2) shorter and coarser (Fig. 9c, d). Tergal plate with a middorsal, hairy protuberance, and two relatively long, distally pointed tergal extensions (Fig. 9b, d).
Notes on diagnostic and taxonomic features
Echinoderes brevipes sp. nov. has middorsal spines on segments 4, 6 and 8 as well as short, robust lateral terminal spines (LTS). There are only three species with the same pattern of features: Echinoderes abbreviatus Higgins, 1983 from Carrie Bow Cay (Belize, Caribbean Sea), Echinoderes belenae Pardos et al., 2016 from Taboga Island (Panama, eastern Pacific Ocean) and Echinoderes rociae Pardos et al., 2016 from Bocas del Toro, Bastimento Island (Panama, Caribbean Sea) (Higgins 1983; Pardos et al. 2016a, b). Nevertheless, Echinoderes brevipes sp. nov. can be unequivocally distinguished from the aforementioned congeners by the arrangement of the remaining spines and tubes. The four species share the presence of lateroventral spine/tubes on segments 5–9 (Higgins 1983; Pardos et al. 2016a, b), but E. abbreviatus and E. belenae have lateral accessory tubes only on segment 8 (Higgins 1983; Pardos et al. 2016b), and E. rociae on segments 7–8 (Pardos et al. 2016a), whereas the newly described species possesses lateral accessory tubes on segments 6–8.
Furthermore, the middorsal spines (MDS) of E. brevipes sp. nov. are remarkably longer than those of the aforementioned congeners (MDS4 average: 16.2 μm in E. abbreviatus, 17.0 μm in E. belenae, 13.0 μm in E. rociae and 30.6 μm in E. brevipes sp. nov.; MDS6 average: 23.2 μm in E. abbreviatus, 21.0 μm in E. belenae, 16.0 μm in E. rociae and 44.0 μm in E. brevipes sp. nov.; MDS8 average: 34.8 μm in E. abbreviatus, 25.0 μm in E. belenae, 20.0 μm in E. rociae and 76.6 μm in E. brevipes sp. nov.) (Higgins 1983; Pardos et al. 2016a, 2016b.
Echinoderes parahorni sp. nov.
(Figs. 11, 12 and 13 and Tables 8 and 9)
Line art illustrations of Echinoderes parahorni sp. nov. a Male, ventral view; b Male, dorsal view; c Female, segments 10–11, ventral view; d Female, segments 10–11, dorsal view. Scale: 50 μm. dpl dorsal placid, lat lateral accessory tube, ldss laterodorsal sensory spot, ldt laterodorsal tube, ltas lateral terminal accessory spine, lts lateral terminal spine, lvs lateroventral spine, lvt lateroventral tube, mdgco1 middorsal type 1 glandular cell outlet, mvpl midventral placid, ne nephridiopore, pdgco1 paradorsal type 1 glandular cell outlet, pdss paradorsal sensory spot, pf pectinate fringe, ps penile spine, sdgco2 subdorsal type 2 glandular cell outlet, sdss subdorsal sensory spot, te tergal extension, tp trichoscalid plate, vlss ventrolateral sensory spot, vlt ventrolateral tube, vmgco1 ventromedial type 1 glandular cell outlet, vmss ventromedial sensory spot
Light micrographs showing trunk overview and cuticular details in the segments of Echinoderes parahorni sp. nov. a Dorsal overview of trunk; b Ventral overview of trunk; c Dorsal view of neck, showing the dorsal placids and the trichoscalid plates; d Ventral view of neck, showing the ventral placids and the trichoscalid plates; e Dorsal overview of segments 1–2; f Ventral overview of segments 1–2; g Dorsal overview of segments 3–10; h Ventral overview of segments 5–10; i Lateroventral region on right half of tergal plates of segments 6–9; j Ventrolateral and ventromedial regions on right half of sternal plates of segments 3–9. Scales: a, b: 50 μm; c–j: 20 μm. dpl dorsal placid, lat lateral accessory tube, lvs lateroventral spine, lvt lateroventral tube, mvpl midventral placid, S segment followed by number of corresponding segment, sdgco2 subdorsal glandular cell outlet type 2, tp trichoscalid plate, vlt ventrolateral tube; sensory spots are marked as dashed circles, and type 1 glandular cell outlets as continuous circles
Scanning electron micrographs showing overviews and details in the cuticular trunk morphology of Echinoderes parahorni sp. nov. a Dorsal overview of trunk; b Ventral overview of trunk; c Middorsal, subdorsal and laterodorsal regions on left half of cuticular plate of segments 1–2; d Detail of a ventromedial sensory spot of segment 5; e Ventrolateral region on right half of cuticular plate of segment 2, showing a ventrolateral tube; f Lateroventral region on right half of sternal plates of segments 5–6, showing the lateroventral spines; g lateroventral and lateral accessory regions on right half of sternal plates of segment 8, showing a lateroventral spine and a lateral accessory tube; h detail of the segment 9 nephridiopore. Scales: a, b: 40 μm; c, f, g: 10 μm; d, h: 1 μm; e: 4 μm. lat lateral accessory tube, lvs lateroventral spine, lvt lateroventral tube, sdgco2 subdorsal glandular cell outlet type 2; sensory spots are marked as dashed circles, and type 1 glandular cell outlets as continuous circles
Synonymy
Echinoderes horni Sørensen et al. 2005: p. 507.
Echinoderes horni Herranz et al. 2014a: p. 3, 10, 23–24; Table 1, 2 and 3; Figs. 1b, 8, 9 and 10a, c.
Echinoderes horni Herranz et al. 2014b: p. 71, 78, 86.
Echinoderes horni Sørensen et al. 2015: p. 4; Table 1, including the 18S RNA sequence EU669453 submitted to GenBank.
Material examined.
Type material. Holotype, adult male, collected on 06 Nov 1980 off Monti Cristi Bay, Dominican Republic, Hispaniola Island, western Atlantic Ocean: 19° 53′ 12′′ N, 71° 40′ 00′′ W (L3) (Table 1; Fig. 1b) at 3–4 m depth in muddy sand associated with assemblages of Thalassia sp., mounted in Fluoromount G®, deposited at NMNH under accession number: USNM 1490965. Paratypes, 18 adult males and 25 adult females, all of them same collecting data as holotype, mounted in Fluoromount G® and deposited at NMNH under accession numbers: USNM 1490966-1491008; one adult male and one adult female collected on 03 Nov 1980 off Puerto Blanco, Dominican Republic, Hispaniola Island, western Atlantic Ocean: 19° 54′ 24′′ N, 70° 56′ 24′′ W (L2) (Table 1; Fig. 1b) at 3 m depth in silty mud, mounted in Fluoromount G®, deposited at NMNH under accession numbers: USNM 1491009-1491010; four adult females collected on 07 Nov 1980 off Icaquitos Bay, Dominican Republic, Hispaniola Island, western Atlantic Ocean: 19° 53′ 12′′ N, 71° 38′ 30′′ W (L5) (Table 1; Fig. 1b) at 2 m depth in muddy sand associated with assemblages of Thalassia sp., mounted in Fluoromount G®, deposited at NMNH under accession numbers: USNM 1491011-1491018; one adult male collected on 10 Nov 1980 off Cabo Haitiano, Haiti, Hispaniola Island, western Atlantic Ocean: 19° 46′ 12′′ N, 72° 11′ 00′′ W (L6) (Table 1; Fig. 1b) at 3–5 m depth in mud, mounted in Fluoromount G®, deposited at NMNH under accession number: USNM 1491019.
Non-type material. Four specimens from the same locality of the holotype (mounted for SEM), also deposited at NMNH under accession number: USNM 1491020.
Additional material. Nine additional specimens from Fort Pierce, Florida, western Atlantic Ocean (27° 29.96′ N, 80° 12.67′ W) at 15 m depth, collected in May 2006 by Dr. M. V. Sørensen and Dr. T. M. Jespersen, mounted in Fluoromount G®, deposited at the Natural History Museum of Denmark under accession numbers: NHMD 288580-188585. Two additional specimens from Fort Pierce, Florida, western Atlantic Ocean (27° 29.56′ N, 80° 12.23′ W) at 15 m depth, collected in June 22, 2003 by Dr. M. V. Sørensen and Dr. R. M. Kristensen, mounted in Fluoromount G®, also deposited at the Natural History Museum of Denmark under accession numbers: NHMD 288578-288579.
Diagnosis
Echinoderes lacking middorsal spines; ventrolateral tubes and subdorsal type 2 glandular cell outlets on segment 2; lateroventral tubes on segment 5, laterodorsal tubes on segment 10 (very reduced in females) and lateroventral spines on segments 6–9; lateral accessory tubes on segment 8.
Etymology
From the Ancient Greek παρά (“pará”), meaning next to or similar to, which refers to the similar morphological features that the newly described species shares with its congener E. horni.
Description
See Table 8 for measurements and dimensions, and Table 9 for summary of spine, tube, nephridiopore, glandular cell outlet and sensory spot locations.
Head and neck. Head with retractable mouth cone and introvert (Fig. 13a, b). Internal part of mouth cone with several rings of inner oral styles; exact number, arrangement and morphology of inner oral styles not determined. External part of mouth cone with nine outer oral styles (Fig. 13a, b). Outer oral styles alternate in size between slightly longer and slightly shorter ones (Fig. 13a, b). Five long styles appear anterior to the odd numbered introvert sections, whereas four slightly shorter ones appear anterior to the even numbered ones, except in the middorsal section 6 where a style is missing (Fig. 13a, b). Outer oral styles with two jointed subunits, with a rectangular basis bearing a short fringe at its base, and a triangular, hook-like distal portion (Fig. 13a, b).
Introvert with seven rings of cuticular spinoscalids (Fig. 13a, b). Ring 01 with ten primary spinoscalids, each one with long, rectangular basal sheath and distal end piece (Fig. 13a, b). Basal sheath with very long fringe situated near its basis that exceeds the joint between the two subunits, bearing several flexible and elongated fringe tips. Distal piece narrow, oval in cross-section, smooth, hook-like, with blunt tip. Remaining rings bear spinoscalids laterally compressed, all with basal sheath and elongate, thin, hook-like distal piece (Fig. 13a, b). Exact number, arrangement and detailed morphology of these spinoscalids not determined as they tended to be collapsed when mounted for LM; furthermore, specimens mounted for SEM did not have the introvert completely extended, so full examination of this structure was not possible.
Neck with 16 trapezoidal placids, wider at base, with distinct joint between the neck and segment 1 (Figs. 11a, b and 12c, d); midventral placid widest (ca. 9–10 μm wide at base) (Figs. 11a and 12d), remaining ones alternating between slightly wider and slightly narrower, varying between 5 and 7 μm at base (Figs. 11b and 12c). Placids closely situated together at base, distally separated by flexible cuticular folds (Figs. 11a, b and 12c, d). A ring of six, long, hairy trichoscalids associated with the neck is present, attached to small trichoscalid plates (Figs. 11a, b and 12c, d).
Trunk. Trunk with 11 segments (Figs. 11a, b, 12a, b and 13a-b). Segments 1–2 as closed cuticular rings; remaining ones with one tergal and two sternal cuticular plates (Figs. 11a, b, 12a, b and 13a, b). Midsternal and tergosternal junctions as conspicuous lines on the cuticle (Figs. 11a, c, 12b and 13b). Tergal plates of anterior segments slightly bulging middorsally; posterior ones more flattened, giving the animal a tapering outline in lateral view. Sternal plates reach their maximum width at segment 5, progressively tapering towards the last trunk segments (Figs. 11a, b, 12a, b and 13a, b). Sternal plates relatively narrow in ratio to the total trunk length (MSW-5:TL average ratio = 20.9%), giving the animal a slender general aspect (Figs. 11a, b, 12a, b and 13a, b).
Segment 1 without spines and tubes. Cuticular hairs irregularly distributed through the cuticular surface, without any recognisable pattern (Fig. 11a, b). Cuticular hairs on this and following segments long, bracteates, filiform (Fig. 13c–h). Unpaired type 1 glandular cell outlet in middorsal position (Figs. 11b, 12e and 13c); paired type 1 glandular cell outlets in ventrolateral position (Figs. 11a and 12f). Paired sensory spots in subdorsal and laterodorsal positions (Figs. 11b, 12e and 13c). Sensory spots on this and remaining segments with a single pore surrounded by few, short micropapillae and flanked by one or two non-bracteate cuticular hairs (Fig. 13d). Posterior segment margin straight, showing a pectinate fringe with a very weak serration (Fig. 11a, b).
Segment 2 with paired, narrow tubes located in ventrolateral position (Figs. 11a, 12f and 13e). Cuticular hairs distributed in three continuous, transvers rows only in the posterior half of the plate (Fig. 11a, b). Unpaired type 1 glandular cell outlet in middorsal position (Figs. 11b and 12e); paired type 1 glandular cell outlets in ventromedial position (Figs. 11a and 12f). Paired type 2 glandular cell outlet in subdorsal position (Figs. 11b, 12e and 13c). Paired sensory spots in laterodorsal and ventromedial positions (Figs. 11b, 12e and 13c). Posterior segment margin and pectinate fringe as on the precedent segment (Fig. 11a, b).
Segment 3 without spines and tubes. Cuticular hairs arranged in 4–5 wavy, transvers rows that increase in length posteriorly, plus a pair of paraventral patches (Fig. 11a, b). Unpaired type 1 glandular cell outlet in middorsal position; paired type 1 glandular cell outlets in ventromedial positions (Figs. 11a, b and 12g, j). Paired sensory spots in subdorsal and laterodorsal positions (Figs. 11b and 12g). Posterior segment margin and pectinate fringe as on the precedent segment (Fig. 11a, b).
Segment 4 without spines and tubes. Cuticular hairs arranged in 4–5 wavy, transvers rows that increase in length posteriorly, plus a pair of paraventral patches (Fig. 11a, b). Paired type 1 glandular cell outlets in paradorsal and ventromedial positions (Figs. 11a, b and 12g, j). Paired sensory spots in laterodorsal and ventromedial positions (Figs. 11a, b and 12g, j). Posterior segment margin and pectinate fringe as on the precedent segment (Fig. 11a, b).
Segment 5 with paired tubes in lateroventral position (Figs. 11a, 12h and 13f). Cuticular hairs arranged in 5–6 wavy, transverse rows that increase in length posteriorly, plus a pair of paraventral patches (Fig. 11a, b). Unpaired type 1 glandular cell outlet in middorsal position; paired type 1 glandular cell outlets in ventromedial position (Figs. 11a, b and 12g, j). Paired sensory spot in subdorsal, laterodorsal and ventromedial positions (Figs. 11a, b and 12g, h, j). Pectinate fringe as on precedent segment (Fig. 11a, b).
Segment 6 with paired spines on lateroventral position (Figs. 11a, 12h, i and 13f). Cuticular hairs arranged as on precedent segment (Fig. 11a, b). Paired type 1 glandular cell outlets in paradorsal and ventromedial positions (Figs. 11a, b and 12g, h, j). Paired sensory spots in paradorsal, laterodorsal and ventromedial positions, the former located posteriorly to middorsal spine (Figs. 11a, b and 12g, h, j). Pectinate fringe as on precedent segment (Fig. 11a, b).
Segment 7 with paired spines in lateroventral position (Figs. 11a and 12h, i). Cuticular hairs arranged in 5–6 wavy, transverse rows that increase in length posteriorly, plus a pair of paraventral patches (Fig. 11a, b). Unpaired type 1 glandular cell outlet in middorsal position; paired type 1 glandular cell outlets in ventromedial position (Figs. 11a, b and 12g, h, j). Paired sensory spots in paradorsal, subdorsal, laterodorsal and ventromedial positions (Figs. 11a, b and 12g, h, j). Pectinate fringe as on precedent segment (Fig. 11a, b).
Segment 8 with paired spines on lateroventral position plus paired tubes in lateral accessory position (Figs. 11a, 12h, i and 13g). Cuticular hairs arranged in 6–7 wavy, transverse rows that increase in length posteriorly, plus a pair of paraventral patches (Fig. 11a, b). Paired type 1 glandular cell outlets in paradorsal and ventromedial positions (Figs. 11a, b and 12g, h, j). Paired sensory spots in paradorsal, subdorsal, laterodorsal and ventromedial positions (Figs. 11a, b and 12g, h, j). Pectinate fringe as on precedent segment (Fig. 11a, b).
Segment 9 with paired spines on lateroventral position (Figs. 11a and 12h, i). Cuticular hairs arranged in 8–9 wavy, transverse rows of cuticular hairs that increase in length posteriorly, plus a pair of paraventral patches (Fig. 11a, b). Paired type 1 glandular cell outlets in paradorsal and ventromedial positions (Figs. 11a, b and 12g, h, j). Paired sensory spots in paradorsal, subdorsal, laterodorsal, ventrolateral and ventromedial positions (Figs. 11a, b and 12g, h, j). Nephridiopore as a sieve plate, in lateral accessory position (Figs. 11a and 13h).
Segment 10 with paired tubes in laterodorsal position, almost completely reduced in females but still with a cuticular marking that indicates the presence of the structure. Cuticular hairs arranged into 5–6 wavy, transverse rows of cuticular hairs that increase in length posteriorly, not reaching the midventral section (Fig. 11a, b). Two unpaired type 1 glandular cell outlets in middorsal position; paired type 1 glandular cell outlets in ventromedial position (Figs. 11a, b and 12g, h). Paired sensory spots in subdorsal and ventromedial positions, the former mesially shifted, not aligned with precedent subdorsal sensory spots (Figs. 11a, b and 12g, h). Posterior segment margin deeply curved, extending in the ventromedial area, with a pectinate fringe as on the precedent segment (Fig. 11a–d).
Segment 11 with long, narrow lateral terminal spines (LTS:TL average ratio = 63.6%), being swollen in its proximal quarter, distally slender and pointed, showing a hollow central cavity in LM (Figs. 11a, b, 12a, b and 13a, b). Unpaired type 1 glandular cell outlet in middorsal position (Fig. 11b, d). Females with a pair of lateral accessory terminal spines relatively short, about one-fifth of length of lateral terminal spines (Fig. 11c, d). Males with three pairs of penile spines arising laterally under the pectinate fringe of the precedent segment; ventral and dorsal penile spines (ps1 and ps3) filiform, midlateral penile spine (ps2) shorter and thicker (Fig. 11a, b). Tergal plate carrying two relatively short, distally pointed, laterally hairy tergal extensions (Fig. 11b, d).
Notes on diagnostic and taxonomic features
Echinoderes parahorni sp. nov. shares the absence of middorsal spines together with 14 congeners: E. andamanensis Higgins and Rao, 1979; E. applicitus, E. aspinosus Sørensen et al., 2012; E. bengalensis (Timm, 1958); E. caribiensis, E. coulli Higgins, 1977; E. filispinosus Adrianov, 1989; E. horni Higgins, 1983; E. hwiizaa Yamasaki and Fujimoto, 2014; E. komatsui Yamasaki and Fujimoto, 2014; E. malakhovi Adrianov, 1999 in Adrianov and Malakhov, 1999; E. marthae Sørensen, 2014; E. multisetosus Adrianov, 1989 and E. strii (Omer-Cooper 1957; Timm 1958; Kirsteuer 1964; Higgins 1977, 1983; Higgins and Rao 1979; Adrianov 1989; Adrianov and Malakhov 1999; Ostmann et al. 2012; Sørensen et al. 2012; Yamasaki and Fujimoto 2014; Pardos et al. 2016a). Of these, only E. horni from Carrie Bow Cay, Belize (Caribbean Sea) possesses a similar arrangement of spines and tubes with E. parahorni sp. nov., as both have lateroventral tubes on segment 5, lateroventral spines on segments 6–9 and lateral accessory tubes on segment 8 (Higgins 1983). However, E. horni has lateroventral tubes on segment 2 (Higgins 1983), while E. parahorni sp. nov. carries these structures in ventrolateral position. Moreover, E. parahorni sp. nov. is characterised by having type 2 glandular cell outlets in subdorsal position on segment 2 and tubes in laterodorsal position on segment 10, two morphological features that are absent in E. horni (Higgins 1983). Although the morphological description of E. horni is relatively old, Sørensen et al. (2016) revised the type material deposited at NMNH and did not determined discrepancies with the original description of the species. Thus, the morphological differences reported herein clearly support the erection of the new species.
The re-examination of several kinorhynchs previously reported as E. horni from Fort Pierce, Florida (western Atlantic Ocean) determined they are conspecific with E. parahorni sp. nov. adding relevant biogeographical information of this newly described species. Thus, all the ecological, morphological, phylogenetical and biogeographical information previously published for these specimens must be considered for E. parahorni (Sørensen et al. 2005; Herranz et al. 2014a, b; Sørensen et al. 2015).
Discussion
Current knowledge of the kinorhynch Caribbean fauna and new reports for Hispaniola Island
Until now, the kinorhynch fauna of the Caribbean Sea was composed of 12 genera and 31 species, including 2 species without confirmation (Antygomonas cf. paulae and Semnoderes cf. pacificus) plus three determined only to genus level (Campyloderes sp., Cephalorhyncha sp. and Echinoderes sp.) (Sørensen 2006; Pardos et al. 2016a). With the present study, the knowledge of the Caribbean kinorhynchs has been increased with the description of four new species (Cristaphyes retractilis sp. nov., Fujuriphyes dalii sp. nov., Echinoderes brevipes sp. nov. and E. parahorni sp. nov.), and the first report of Echinoderes astridae and E. spinifurca for the area. Echinoderes horni and E. imperforatus Higgins, 1983 are also firstly reported for Hispaniola Island, although previously known in the Caribbean Sea.
Echinoderes astridae was originally described from Araçá Bay, São Sebastião (Brazil), western Atlantic Ocean, in grey mud mixed with sand at 10 m depth (Sørensen 2014). The presence of the species in Monte Cristi Bay and Puerto Blanco (Dominican Republic) represents the first record of the species in Caribbean waters and its first report since the original description. Moreover, the Caribbean sediment samples consisted of mud and silty mud collected at 3–4 m depth. Echinoderes horni and E. imperforatus were described from Twin Cays (Belize), Caribbean Sea, in calcareous sediment with detritus and sand at 1–2 m depth (Higgins 1983). In the present study, both species were collected in Monte Cristi Bay and Jicaquito Bay in muddy sand at 2–4 m depth, being their first record for the Great Antilles. Echinoderes spinifurca was first described off Fort Pierce (Florida), in Dentalium sand at 15 m depth (Sørensen et al. 2005); here, we report this species in Monte Cristi Bay and Puerto Blanco (Dominican Republic) in mud and silty mud at 3–4 m depth. These new records extend the biogeographical and ecological ranges for each species.
Geographic distribution of Caribbean kinorhynchs
Overall, the 35 known Caribbean species of Kinorhyncha are distributed as follows:
- 1.
Ten species only known from Carrie Bow Cay, Belize: Cristaphyes belizensis, Echinoderes abbreviatus, Fujuriphyes deirophorus, Higginsium erismatum (Higgins, 1983), H. trisetosum (Higgins, 1983), Leiocanthus corrugatus (Higgins, 1983), L. ecphantor (Higgins, 1983), Pycnophyes apotomus (Higgins, 1983), P. stenopygus (Higgins, 1983) and Setaphyes iniorhaptus (Higgins, 1983).
- 2.
Five species only known from the Caribbean Panama: Cristaphyes panamensis Pardos et al., 2016, Echinoderes collinae Sørensen, 2006, E. orestauri Pardos et al., 2016 and E. rociae.
- 3.
One species only known from Mochima, Venezuela: Echinoderes caribiensis.
- 4.
Three species only known from Hispaniola Island, Greater Antilles: Cristaphyes retractilis sp. nov., Echinoderes brevipes sp. nov. and Fujuriphyes dalii sp. nov.
- 5.
Six species only found in the Caribbean Sea (more than a single locality): Cristaphyes longicornis, Echinoderes imperforatus, E. wallaceae Higgins, 1983, Fujuriphyes distentus, Leiocanthus emarginatus (Higgins, 1983) and Paracentrophyes praedictus Higgins, 1983.
- 6.
Eleven species found both inside and outside the Caribbean Sea: Antygomonas paulae Sørensen, 2007, Centroderes barbanigra Neuhaus et al., 2014, Echinoderes astridae, E. horni, E. intermedius Sørensen, 2006, E. parahorni sp. nov. E. spinifurca, E. truncatus Higgins, 1983, Pycnophyes alexandroi Pardos et al., 2016, P. beaufortensis Higgins, 1964 and Semnoderes pacificus Higgins, 1967.
The geographic distribution of most of the Caribbean Kinorhyncha is limited to the Caribbean Sea. The Caribbean constitutes one distinctive biogeographical subregion of the Tropical North Western Atlantic Province, but is biogeographically heterogeneous and scarcely isolated (Briggs 1995; Miloslavich et al. 2010). Moreover, nearby regions, including the Atlantic coast of Mexico and South America, have been scarcely studied regarding the kinorhynch fauna. In addition, the new records of Cristaphyes longicornis, Echinoderes astridae, E. horni, E. imperforatus and E. spinifurca for Haiti and the Dominican Republic suggest that the biogeographical range of the previously known Caribbean kinorhynchs could be wider.
Echinoderes horni, E. intermedius, Pycnophyes alexandroi and Semnoderes pacificus have been reported in both the Caribbean and the Pacific Ocean (Higgins 1967; Sørensen 2006; Pardos et al. 2016a, b). The only existing connection between these two bodies of water is the Panama Canal. Although the canal could act as a passageway for marine fauna exchange between the two oceans (Menzies 1968; Schloder et al. 2013; Ros et al. 2014), meiofaunal species encounter great physical, chemical and biological limitations to achieve the migration (Pardos et al. 2016b). Thus, deeper studies would be necessary to clarify the biogeographical status of the aforementioned species.
References
Adrianov AV (1989) The first report on Kinorhyncha of the Sea of Japan. Zool Zh 68:17–27
Adrianov AV, Malakhov VV (1999) Cephalorhyncha of the world ocean. KMK Scientific Press, Moscow
Briggs J (1995) Global biogeography. Elsevier Science, Amsterdam
Brown R, Higgins RP (1983) A new species of Kinorhynchus (Homalorhagida, Pycnophyidae) from Australia with a redescription and range extension of other Kinorhyncha from the South Pacific. Zool Scr 12(3):161–169
Claparède ARE (1863) Zur Kenntnis der Gattung Echinoderes Duj. Beobachtungen über Anatomie und Entwicklungsgeschichte wirbelloser Thiere an der Küste von Normandie angestellt. Verlag von Wilhelm Engelmann, Leipzig
Fleeger JW, Thistle D, Thiel H (1988) Sampling Equipment. In: Higgins RP, Thiel H (eds) Introduction to the study of meiofauna, 1st edn. Smithsonian Institution Press, Washington D.C., pp 115–133
Herranz M, Boyle MJ, Pardos F, Neves RC (2014a) Comparative myoanatomy of Echinoderes (Kinorhyncha): a comprehensive investigation by CLSM and 3D reconstruction. Front Zool 11:31
Herranz M, Sánchez N, Pardos F, Higgins RP (2014b) New Kinorhyncha from Florida coastal waters. Helgol Mar Res 68:59–87
Higgins RP (1964) Three new kinorhynchs from the North Carolina coast. Bull Mar Sci 14:479–493
Higgins RP (1967) The Kinorhyncha of new-Caledonia. In: Expédition Française Sur les Recifs Coralliens de la nouvelle-Calédonie 2, 1st edn. Fondation Singer-Polignac, Paris, pp 75–90
Higgins RP (1977) Two new species of Echinoderes (Kinorhyncha) from South Carolina. Trans Am Microsc Soc 96:340–354
Higgins RP (1983) The Atlantic Barrier Reef ecosystem at Carrie Bow Cay, Belize, II: Kinorhyncha. Smithson Contrib Mar Sci (18):1–131
Higgins RP (1988) Kinorhyncha. In: Higgins RP, Thiel H (eds) Introduction to the study of meiofauna, 1st edn. Smithsonian Institution Press, Washington D.C., pp 328–331
Higgins RP, Rao GC (1979) Kinorhynchs from the Andaman Islands. Zool J Linnean Soc 67:75–85
Kirsteuer E (1964) Zur Kenntnis der Kinorhynchen Venezuelas. Zool Anz 173:388–393
Lang K (1949) Echinoderida. Further zoological results of the Swedish Antarctic expedition 1901–1903 4:1–22
Lang K (1953) Reports of the Lund University Chile expedition 1948-1949. 9. Echinoderida. Acta Univ Lund 49(4):3–8
Menzies RJ (1968) Transport of marine life between oceans through the Panama Canal. Nature 220:802–803
Miloslavich P, Díaz JM, Klein E, Alvarado JJ, Díaz C, Gobin J, Escobar-Briones E, Cruz-Motta JJ, Weil E, Cortés J, Bastidas AC, Robertson R, Zapata F, Martín A, Castillo J, Kazandjian A, Ortiz M (2010) Marine biodiversity in the Caribbean: regional estimates and distribution patterns. PLoS ONE 5(8): e11916.
Neuhaus B (2013) 5. Kinorhyncha (=Echinodera). In: Schmidt-Rhaesa A (ed) Handbook of zoology. Gastrotricha, Cycloneuralia and Gnathifera. Volume 1: Nematomorpha, Priapulida, Kinorhyncha, Loricifera, 1st edn. De Gruyter, Göttingen, pp 181–348
Neuhaus B, Pardos F, Sørensen MV, Higgins RP (2014) New species of Centroderes (Kinorhyncha: Cyclorhagida) from the Northwest Atlantic Ocean, life cycle and ground pattern of the genus. Zootaxa 3901:1–69
Omer-Cooper J (1957) Deux nouvelles especes de Kinorhyncha en Provenance de l'Afrique du Sud. Bull Mens Soc Linn Lyon 26:213–216
Ostmann A, Nordhaus I, Sørensen MV (2012) First recording of kinorhynchs from Java, with the description of a new brackish water species from a mangrove-fringed lagoon. Mar Biodivers 42:79–91
Pardos F, Sánchez N, Herranz M (2016a) Two sides of a coin: the phylum Kinorhyncha in Panama. I Caribbean Panama. Zool Anz 265:3–25
Pardos F, Herranz M, Sánchez N (2016b) Two sides of a coin: the phylum Kinorhyncha in Panama. II Pacific Panama. Zool Anz 265:26–47
Reinhard W (1881) Uber Echinoderes und Desmoscolex der Umgebung von Odessa. Zool Anz 4:588–592
Roberts CM, McClean CJ, Veron JE, Hawkins JP, Allen GR, McAllister DE, Mittermeier CG, Schueler FW, Spalding M, Wells F, Vynne C, Werner TB (2002) Marine biodiversity hotspots and conservation priorities for tropical reefs. Science 295:1280–1284
Ros M, Ashton GV, Lacerda MB, Carlton JT, Vázquez-Luis M, Guerra-García JM, Ruiz GM (2014) The Panama Canal and the transoceanic dispersal of marine invertebrates: evaluation of the introduced amphipod Paracaprella pusilla Mayer, 1890 in the Pacific Ocean. Mar Environ Res 99:204–211
Sánchez N, Yamasaki H (2016) Two new Pycnophyidae species (Kinorhyncha: Allomalorhagida) from Japan lacking ventral tubes in males. Zool Anz 265:80–89
Sánchez N, Herranz M, Benito J, Pardos F (2012) Kinorhyncha from the Iberian Peninsula: new data from the first intensive sampling campaigns. Zootaxa 3402:24–44
Sánchez N, Yamasaki H, Pardos F, Sørensen MV, Martínez A (2016) Morphology disentangles the systematics of a ubiquitous but elusive meiofaunal group (Kinorhyncha: Pycnophyidae). Cladistics 32:479–505
Sánchez N, Sørensen MV, Landers SC (2019) Pycnophyidae (Kinorhyncha: Allomalorhagida) from the Gulf of Mexico: Fujuriphyes viserioni sp. nov. and a redescription of Leiocanthus langi (Higgins, 1964), with notes on its intraspecific variation. Mar Biodivers. https://doi.org/10.1007/s12526-019-00947-x.
Schloder C, Canning-Clode J, Saltonstall K, Strong EE, Ruiz GM, Torchin ME (2013) The Pacific bivalve Anomia peruviana in the Atlantic: a recent invasion across the Panama Canal? Aquat Invasions 8:443–448
Sørensen MV (2006) New kinorhynchs from Panama, with a discussion of some phylogenetically significant cuticular structures. Meiofauna Mar 15:51–77
Sørensen MV (2007) A new species of Antygomonas (Kinorhyncha: Cyclorhagida) from the Atlantic coast of Florida, USA. Cah Biol Mar 48:155–168
Sørensen MV (2014) First account of echinoderid kinorhynchs from Brazil, with the description of three new species. Mar Biodivers 44:251–274
Sørensen MV, Pardos F (2008) Kinorhynch systematics and biology—an introduction to the study of kinorhynchs, inclusive identification keys to the genera. Meiofauna Mar 16:21–73
Sørensen MV, Heiner I, Ziemer O (2005) A new species of Echinoderes from Florida (Kinorhyncha: Cyclorhagida). Proc Biol Soc Wash 118:499–508
Sørensen MV, Rho HS, Min WG, Kim D, Chang CY (2012) An exploration of Echinoderes (Kinorhyncha: Cyclorhagida) in Korean and neighboring waters, with the description of four new species and a redescription of E. tchefouensis Lou, 1934. Zootaxa 3368:161–196
Sørensen MV, Dal Zotto M, Rho HS, Herranz M, Sánchez N, Pardos F, Yamasaki H (2015) Phylogeny of Kinorhyncha based on morphology and two molecular loci. PLoS ONE 10(7): e0133440
Sørensen MV, Herranz M, Landers SC (2016) A new species of Echinoderes (Kinorhyncha: Cyclorhagida) from the Gulf of Mexico, with a redescription of Echinoderes bookhouti Higgins, 1964. Zool Anz 265:48–68
Timm RW (1958) Two new species of Echinoderella (phylum Kinorhyncha) from the Bay of Bengal. J Bombay Nat Hist Soc 55:107–109
Yamasaki H, Fujimoto S (2014) Two new species in the Echinoderes coulli group (Echinoderidae, Cyclorhagida, Kinorhyncha) from the Ryukyu Islands, Japan. ZooKeys 382:27–52
Zelinka C (1894) über die Organisation von Echinoderes. Verh Dtsch Zool Ges 4:46–49
Zelinka C (1896) Demonstration von Tafeln der Echinoderes Monographie. Verh Dtsch Zool Ges 6:197–199
Zelinka C (1928) Monographie der Echinodera. Engelmann, Leipzig
Acknowledgements
We thank Kathryn Ahlfeld and Dr. Jon L. Norenburg from the Smithsonian Institution of Washington for their efficiency in sending the examined material and Dr. Robert P. Higgins for collecting the material and making it available for all researchers. We also thank Dr. M. V. Sørensen and Dr. H. Yamasaki for their useful revision, suggestions and comments that strongly improved the present paper. Cepeda was supported by a predoctoral fellowship of the Complutense University of Madrid (CT27/16-CT28/16).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This article does not contain any studies with animals performed by any of the authors.
Sampling and field studies
All necessary permits for sampling and observational field studies have been obtained by the authors from the competent authorities and are mentioned in the acknowledgements, if applicable.
Data availability statement
Data sharing not applicable to this article as no datasets were generated or analysed during the current study.
Additional information
Communicated by S. Gollner
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is registered in ZooBank under urn:lsid:zoobank.org:pub: D10BC214-CD04-4543-B6C0-9A3A2700CD97.
Cristaphyes retractilis sp. nov. is registered in Zoobank under urn:lsid:zoobank.org:act:3D46B431-40FA-4415-B00C-63811B07252B.
Fujuriphyes dalii sp. nov. is registered in Zoobank under urn:lsid:zoobank.org:act:0C1C372B-1EA0-4B0B-854A-53D5AA947B33.
Echinoderes brevipes sp. nov. is registered in Zoobank under urn:lsid:zoobank.org:act:4E5899A5-85C8-44C1-B3C5-21A71C6C00A7.
Echinoderes parahorni sp. nov. is registered in Zoobank under urn:lsid:zoobank.org:act:F8FA16F5-75FC-47BB-A56D-7A3FF59EF0CE
Rights and permissions
About this article
Cite this article
Cepeda, D., Sánchez, N. & Pardos, F. First extensive account of the phylum Kinorhyncha from Haiti and the Dominican Republic (Caribbean Sea), with the description of four new species. Mar Biodiv 49, 2281–2309 (2019). https://doi.org/10.1007/s12526-019-00963-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12526-019-00963-x