Abstract
Thailand is currently ranked as the world’s fourth largest sugarcane producer after Brazil, India and China and the world’s second largest sugar exporter after Brazil. In the past five years (2015–2020), average sugarcane yield in Thailand was around 63 tonnes/hectare, with an average commercial cane sugar (CCS) content of 12.41%. An estimated 330,000 sugarcane growers are now cultivating sugarcane in Thailand. In the 2017/18 milling season, crop production reached a peak of 134.9 million tonnes of sugarcane with 1.58 million hectares of cultivated area. However, the production has since shown a declining trend due to recurring drought and a shift in farmers’ preference for more profitable crops. Since its inception in 1938, sugarcane breeding has continuously increased the cane productivity by releasing improved varieties with the most notable one, Khon Kaen 3 (KK3), which now covers ~85% of the sugarcane production in Thailand. To continue improve the productivity and sustainability of the Thailand sugar industry, current breeding programs are mainly focusing on cane yield and sugar content, with increasing emphasis on fiber yield, pest and disease resistance, environmental adaptability (e.g., sandy soil, clay soil, drought conditions), ratooning ability and suitability for mechanical harvesting. The development and integration of biotechnology will no doubt contribute to further improvement of desirable traits. This review covers the main characteristics of the national sugar industry, key institutions currently involved in breeding and its supporting research, history and achievements of sugarcane breeding programs, current methods used in breeding programs as well as major cultivars and germplasm collections. The history and current status of the research and application of genetics and biotechnology for sugarcane improvement are also presented.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Main Characteristics of the National Industry
Sugarcane is ranked third in economic importance in Thailand among the major crops, after rice and rubber trees (Fig. 1). In the past 5 years, it accounted around 7–8% (about 1.77–1.83 million hectares) of the total agricultural land (Ministry of Agriculture and Cooperatives 2020; Office of the Cane and Sugar Board 2020), and grown in 47 provinces. The planting areas are located in the north, north-eastern, central and eastern regions of Thailand with approximately 25%, 46%, 24% and 5% of the total cultivated area in each of these regions respectively (Office of the Cane and Sugar Board 2019).
The sugar industry plays an important role in the economic development of Thailand. Among sugarcane producing countries, Thailand is currently the world’s fourth largest cane producer and the second largest exporter of refined sugar (Workman 2020). The exported value was approximately 3.36 billion USD for sugar and sugar confectionery in 2019, which represented 12.6% of the national export balance for food and beverages (UN Comtrade 2020). All refined sugar produced in Thailand is supplied by the local sugarcane production. The policies and strategies of the sugar industry in Thailand are driven and coordinated by the Office of the Cane and Sugar Board (OCSB) which is under the jurisdiction of the Ministry of Industry (Sukyai et al. 2016).
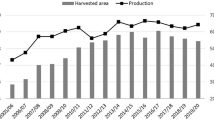
In the last decade, Thailand’s sugarcane production has increased dramatically. The national production gains were mainly attributed to the increase of the cane cultivated area driven by a strong support from the OSCB policies and an attractive selling price for farmers compared to other crops. The national cane production was 30 million tonnes on average in the 1980s, increasing to 50 million tonnes on average in the 1990s, and jumping to 100 million tonnes on average during the period 2010–2019 (Fig. 2). As a result, the number of sugar mills operating in Thailand constantly increased during the last 20 years, from 46 mills in 2002 to 57 mills in 2019.
Number of sugar mills during the period 2002–2019 (left) and the area of cultivated cane in Thailand between 1982/83 and 2019/20 (right). Source: (Office of the Cane and Sugar Board 2020; Thai Sugar Millers Corporation Limited 2018)
During the 2017/18 milling season, production reached a peak with 134.93 million tonnes of cane (Office of the Cane and Sugar Board 2020), mainly attributed to favorable weather conditions. This represented a 45% increase compared to the previous milling season (Table 1). However, during the 2019/20 milling season, cane production dropped to 74.9 million tonnes (approximately 43%). This major decline of the cane production was attributed mainly to two factors: severe drought conditions resulted in a major decline in yield of approximately 35% compared to the previous year and a significant proportion of farmers shifting to alternative crops with more profitable revenues such as cassava, corn or Jasmin rice (United States Department of Agriculture 2019).
Currently 80% of the sugar produced in Thailand (7–11 million tonnes/year) is exported while 20% (2.5–2.7 million tonnes/year) is consumed domestically. Domestic consumption of sugar is expected to grow by only 0.7% in 2020 because of rising health awareness and the imposition of sugar tax on the beverage industry. The beverage industry is the major consumer of sugar (44% of total domestic consumption), followed by food (26%) and dairy products (22%) (Krungsri Research 2020). The sector will face several challenges over the next few years such as the application of Taxes on non-alcoholic drinks based on sugar content and the development of the market for non-sugar-based sweeteners.
In the past five years (2015/16–2019/20), Thailand’s sugarcane yield was on average around 63 tonnes/hectare, with an average commercial cane sugar (CCS) content of 12.41%, from an estimated number of 330,000 sugarcane growers (Table 2).
In Thailand, farmers generally start planting sugarcane from November to March, depending on the regions, when the temperatures are lower and the soil moisture is optimum for planting. Cane is then harvested 10–12 months later when the crushing season starts. Generally, the sugarcane crushing season in Thailand extends for about 180 days starting from November or December and ending on March or April of the following year. Sugarcane is mainly grown under rain-fed conditions and less than 10% of the cultivated area irrigated, with an average of two ratoon crops.
The majority of sugarcane farms are classed as being small to medium in size: 52% are less than 10 hectares and 31% between 10 and 32 hectares, with an average annual production capacity of 1000–2000 tonnes. Only 17% of farms are considered large (over 32 hectares), with an annual production above 2000 tonnes (Khon Kaen University 2014). Recently, large sugarcane farms are using mechanized solutions for planting, weeding, harvesting and de-trashing to facilitate their work and to overcome labor shortage to some extent. At present, mechanization covers almost 100% of the land preparation, about 80% of the planting process, but only 20% of the harvesting process. That is, 80% of the crop is harvested manually, which is labor intensive and costly. The mechanization of harvesting is expected to increase by 50% within the next 5 years.
Despite the fact that the burning reduces sugar quality and quantity, burning of the cane field before harvesting is a common practice on approximately 60–80% of the cultivated area in order to facilitate manual harvesting, save money on labor costs and for management of leaf trash. Sugarcane burning is now considered as a serious problem in Thailand. It is blamed for creating smog and hazardous fine dust in some provinces with large sugarcane plantations. Since 2019/20, the Thai government has implemented a plan to support sugarcane farmers with additional measures to harvest fresh cane and discourage burnt cane in order to reduce pollution. The plan seeks to force sugar mills to accept only 30% of burnt sugarcane for milling in 2019/20, with a further reduction to 20% for the 2020/21 crop year and to no more than 5% for the year 2021/22. As a result of these restriction policies, green cane harvesting in 2019/20 was 50.35% higher than in 2018/19 (Office of the Cane and Sugar Board 2020).
Major Constraints of Sugarcane Production
Diseases and Pests
Approximately 40 diseases, including major and minor diseases, caused by fungi, bacteria, viruses, and phytoplasmas have been recorded on cultivated sugarcane in Thailand (Srisink et al. 2016). Among these, phytoplasma-caused diseases and smut are currently the most serious diseases. Three phytoplasma organisms are reported to cause three different phytoplasmal disease symptoms. Sugarcane white leaf (SCWL) and sugarcane grassy shoot (SCGS) diseases are caused by two different but closely related phytoplasmas that are naturally spread by the same leafhoppers (Sakuanrungsirikul et al. 2012; Rao et al. 2012; Marcone 2002; Hanboonsong et al. 2006). Both diseases show distinctive white leaf symptoms but differ in that SCGS shows profuse tillering. Since their first report in 1952 in Thailand, these two diseases have caused significant economic losses. The control of these diseases is now considered a national priority.
Sugarcane green grassy shoot (SCGGS) is another phytoplasmal caused disease that was first recognized in 1995 in U-Thong1 variety in a field crop research center in central Thailand. The severely infected plant shows grassy tillering like SCGS but without leaf chlorosis (Pliansinchai and Suchato 1995; Pliansinchai et al. 1997, 1998). This disease is caused by a phytoplasma of a different group from the SCWL and SCGS, based on the studies using sequencing of the 16S (Sakuanrungsirikul et al. 2012). The transmission vector is still unknown so far. SCGGS can cause great loss as infected plants generally do not produce stalks. This disease is mostly spreading in the west and central area with 10–20% incidence currently in the infected fields. To date, no phytoplasma resistant variety is available despite efforts to develop effective screening methods for the insect vector resistant variety (Roddee et al. 2018a). The use of healthy plant materials, roguing diseased plants and adjusting planting time are important measures for reducing the incidence of these phytoplasmal related diseases.
Ratoon stunting disease (RSD), a bacterial disease caused by the bacteria; Leifsonia xyli subsp. xyli, is not considered as a major disease in Thailand. This disease was reported to cause yield loss up to 41% in sugarcane worldwide (Comstock 2008). The surveying of RSD in the central and the northeast plantations using two PCR detection techniques revealed very low incidence of the infected materials. Positive materials were found in some old varieties maintained in research centers while samples collected from farmers’ fields elsewhere showed negative results (Saengsai et al. 2018). Hot water treatment at 50–52 °C of seed canes before planting is practiced in some propagation stations, which can reduce the disease spreading incidence (Srisink et al. 2014; Yulianti et al. 2020).
Smut is a fungal disease caused by Sporisorium scitamineum. In the past, it was considered as a non-significant disease. But in the recent years, Khon Kaen3 (KK3), a moderately smut tolerant variety, dominated the cane planting area, covering more than 80% of sugarcane plantations (Jaisil et al. 2020). Interestingly, more smut infected fields are being reported recently in KK3 variety. The production loss is significant in the ratoon crops especially after severe drought condition. The production loss can be expected at 10–50% in the severe cases.
Wilt and red rot are caused by the fungi; Fusarium moniliforme and Collectotrichum falcatum, respectively. These diseases are found mostly in irrigated cane areas though with less than 10% incidence of the diseased plants. The prevalence of wilt is higher than red rot. Estimated production loss is less than 30% in the severe cases.
Borers, stem boring grub, sugarcane whitefly and termite are important pests and their management is considered as a priority (Srisink et al. 2016). Borers are the most significant insect pests affecting productivity, with estimated yield losses being up to approximately 30–50%. Five borers infest sugarcane all through its growth stages. Early shoot borer can cause 7% CCS reduction in mature cane (Pitak 2009) while Stem boring grub (Dorysthenes (Lophosternus) bugueti Guerin) can cause 13–43% cane yield and 11–46% sugar yield reduction in plant cane, and 54% and 57% reduction of cane and sugar yields respectively in first ratoon cane (Srisink et al. 2016).
Severe outbreaks of Cicada (Platypleuraces piticola Boulard) were reported in the central and western regions of Thailand during the long dry spell period, with losses up to 50% in ratoon cane. However, this insect is currently still considered as a minor pest in Thailand due to its limited area of dispersion.
Integrated pest management including the use of natural enemies and chemicals are being applied to effectively control insect pests in Thailand.
Abiotic Environmental Constraints
Water availability in sugarcane plantations in Thailand is variable, and with less than 10% of the cultivated area being irrigated, is affected strongly by climate. Almost every year, sugarcane production in Thailand is affected by drought periods, causing an estimated annual average loss of 10 million tons of cane (about 10% of the national cane production) (Kanjanavisut and Rattanasiwakoon 2020). Drought frequently occurs in the sugarcane growing regions of Thailand, especially in the north-eastern and central regions. In 2019, the cumulative annual rainfall in Thailand was about 1343 mm. (Fig. 3), a decrease of about 8% compared to 2018, and resulting in severe impact on the sugarcane production.
Status of and Industry/Government View on Genetically Modified (GM) Sugarcane
Thailand embraced agricultural biotechnology earlier than its neighboring countries. Much research and development has focused on producing technology tools to promote Thailand’s agriculture into the new era of industrial revolution. However, Thailand’s policies on biotechnology are still uncertain. Although Thailand was the first country in the region to adopt national biosafety guidelines for laboratory work and field testing since 1992, the implementation of biosafety legislation and adoption of the GM crops now lags those of neighboring countries. Thailand started field trials of GM crops in the early 1990s. However, intensive pressure from activists from April 3, 2001 led the government cabinet deciding to prohibit all open field trials of transgenic plants in Thailand. No GM crops have been approved for field trials since 2003. Transgenic trials are permitted only for research purposes in approved laboratories and/or greenhouses. While restrictions are placed on GM production, import of GM crops continue, including GM soybeans, GM maize, GM cotton and GM processed food (National Center for Genetic Engineering and Biotechnology 2010; Napasintuwong 2010, 2019).
GM technology is a controversial issue in Thailand and thus a cautious approach has been taken in the application of genetic engineering technology in agriculture. Not surprisingly, relatively few research programs have been undertaken related to genetically modified sugarcane. Among these, the study of transformation and expression of chinase gene, herbicide resistance, and sucrose accumulation genes in sugarcane (Saccharum officinarum L.) (Bunnak et al. 2011; Chanprame 2011), were carried out only at laboratory scale with no further implementation in the field so far. Although the undecided policies and regulations have prohibited the progress of the potential benefits from transgenic technology, the use of modern technology tools such as marker-assisted selection (MAS) is allowed while sugarcane genomics and transcriptomics research are being supported by the government.
Key Institutes Currently Involved in Breeding and Supporting Research
Currently, major sugarcane breeding projects are being conducted by both governmental agencies and private sectors, as listed in Table 3. The major funding source for sugarcane breeding programs is the government.
Key Objectives of Breeding Programs
The Office of the Cane and Sugar Board (OCSB) developed a strategic plan for the period 2015–2036, aiming to increase the cane planting area from 1.7 to 2.6 million hectares, cane production from 106 to 180 million tonnes, the average cane yield from 66 to 71 tonnes/hectare and the sugar production from 11.1 to 20.4 million tonnes. This plan was designed to respond to the increasing demand of sugar products and biofuels in South-East Asia as well as to support development of the bio-economy in this region (Sukyai et al. 2016). To comply with a green and sustainable growth of the sugarcane industry, research funding in Thailand is supporting projects aiming to develop new varieties with improved sugar yield and agronomic performance under biotic and abiotic stress conditions.
One of the most important challenges of sugarcane breeding is to improve cane yield per hectare. In Thailand, cane productivity per hectare is still relatively low, compared with other sugar producers and exporting countries (Yadav et al. 2020). To limit the impact of drought periods on the sugarcane production, the development of “drought-tolerant” varieties is now under progress. To enhance the productivity of the Thai sugar industry, current breeding programs are also aiming at developing varieties with good adaptation, good ratooning ability, suitability for specific locations, pest and disease resistance, and suitability for mechanical harvesting.
Sugarcane produces potentially valuable products beside sugar i.e., bioethanol, bagasse, molasses, vinasses, scrap waxes (Chunhawong et al. 2018). To improve the Thai sugar industry’s productivity, breeding programs conducted in Thailand are now attempting to develop specific varieties for each purpose. The development of varieties specifically suitable for the production of bagasse is now implemented in Thailand. Germplasm and crossing populations are now being screened for varieties with high fiber yields (i.e., high biomass) and breeding programs are also being conducted for that specific purpose.
History and Achievements of Breeding Programs
The first sugarcane breeding program in Thailand was established in 1938 in Lampang province, in the northern region of Thailand (Department of Agriculture 1982). This project introduced a number of exotic sugarcane clones, especially from the world collection, and utilized them to improve modern sugarcane varieties. Since then, breeding programs focused on the development of new varieties with improved sugar yield and high CCS as a priority. During the period 1983–2019, most commercial sugarcane varieties were released by the DOA and the OCSB through conventional breeding programs. Currently about 85% of the sugarcane production in Thailand is covered by the variety KK3. This variety presents several advantages i.e., high yielding, good ratooning ability, moderately tolerant to drought, wide adaptation and water and nitrogen use efficiencies. It was released in 2008 by the DOA, becoming widely distributed across Thailand since then.
Even though sugarcane breeding programs in Thailand were successful in developing and releasing new varieties, the cane yield and sugar yield of the new varieties has not been significantly different to KK3. Since 2008, no varieties have shown better agronomic performance in comparison with KK3. Even the elite hybrids, Suphan Buri 80 and Khon Kaen 80, obtained from the same parents as KK3, have displayed lower cane and sugar yields compared to KK3. In order to assess the reason for this low gain in yield, heritability of agronomic traits especially for yield and sweetness were studied in sugarcane germplasm collections. Studies are being conducted to evaluate the general combining ability (GCA) and specific combining ability (SCA) for yield and sugar content in germplasm collections. The accessions with highest GCA and SCA are expected to give promising results for future breeding programs.
Thai breeding programs have also targeted the development of varieties with high fiber yield suitable for bioenergy production. Five hybrids generated by crossing commercial varieties with S. spontaneum were registered and released namely, Biotech1 and Biotech2 of KU; and TPJ03-452, TPJ04-713 and TPJ04-768 of the DOA under the DOA and Japan International Research Center for Agricultural Sciences (JIRCAS) cooperative project. The fiber yields of these hybrids were higher than KK3 and K88-92 while sugar yields of TPJ03-452 and TPJ04-768 were similar to commercial cultivars (Ando et al. 2016).
Current Methods used in Breeding Programs
Since the development and application of molecular markers for breeding is still at an early stage, almost all sugarcane improvement programs in Thailand are following the conventional breeding steps: hybridization, selection, yield evaluation and variety release. Breeding programmes mainly focus on the cane yield in tonnes of cane per hectare (TCH), and sugar content measured as CCS, but also on fiber content and pest and disease resistance, environmental adaptability (e.g., sandy soil, clay soil, drought conditions) and yield stability in time (along the successive ratoon generations). The majority of conventional sugarcane breeding program conducted in Thailand take 10–12 years from initial crossing to the release of the variety.
The choice of parents used for crossings initiate the program and is considered as the most important step since it impacts all subsequent stages of the program. Each year, approximately 100–500 parents are selected in each station for crossing. The selection of parents requires knowledge of the agronomic characteristics of the varieties available for hybridization. The sugarcane breeders’ task is to combine the desired traits observed in parents into the resulting population of progeny obtained through crossing. However, this task is not easy in sugarcane as prediction of trait performance is difficult due to the genetic complexity of sugarcane. In Thailand, the selection of parents for crossings is based on their individual phenotypic performance for the trait targeted by the breeding program. As a result, crosses are planned to maximize the production of hybrids with traits of interest. As an example, parents presenting high CCS but low cane yield are preferably used as male while parents presenting low CCS but high cane yield are preferably used as the female parent.
However, according to Fig. 4, the comparison of thai commercial varieties cultivars indicates that sugarcane breeding programs conducted in Thailand during the last 50 years have delivered varieties with improved sugar yields through the improvement of cane yield, while the improvement of the sugar content is limited. This observation could be explained by the fact that the genetic background of commercial varieties in Thailand is relatively narrow in comparison with sugarcane varieties from other sugarcane producing countries (Kimura et al. 2020). To overcome a potential inbreeding depression, the selection of parents in future breeding programmes may integrate the genetic distances between parents evaluated by molecular markers.
Minimum and maximum cane yield (a), sugar yield (b) and Commercial Cane Sugar (c) potential of 40 Thai commercial cane varieties. Values were calculated based on experimental data submitted to the new variety registration system. Source: (Suphan Buri Field Crops Research Center 2017; Khon Kaen Field Crops Research Center 2016; Jaisil and Yammankong 2015)
Moreover, the crossing of sugarcane is severely limited by the nature of its flowering and the asynchronous flowering of targeted parents. Some parents may also fail to produce viable seeds after crossing. The number of parents used for crossings is relatively limited and are often sharing a close genetic background. Since flower initiation and spikelet fertility are related to environmental factors such as the temperature and relative humidity, crossing stations are located where the environment is suitable to pollen shedding and fertility. In the north-eastern region of Thailand, crossing is conducted at research stations at Phurua (Loei province) and Tha-Phra (Khon Kaen province). In the central region, crossing is done at Thong-PhaPhoom, Sai Yok, Tha-Muang (Kanchana Buri province) and Kampaengsaen (Nakhon Prathom province). Among these 6 major breeding stations, the flowering rate varies from 25% (Kampaengsaen) to almost 100% (Thong-PhaPhoom) according to the local conditions offered by the station during the flowering period (i.e., rainfall, temperature, altitude) (Chatwachirawong 2006). Female parents of each cross are usually marcotted before the flag leaf stage and cut at late tip emergence stage before being used in crosses. The marcotted arrowing shoots are then cut from the field to make crosses in a nursery house.
Each year, about 60,000–500,000 seedlings are produced from about 600–3000 crosses, across 6 major breeding stations. Most seedlings produced are derived from crossing between Saccharum hybrids (bi-parental) and/or elite clones x commercial, and/or commercial x commercial parents, meanwhile, less than 5% of crosses were done between Saccharum hybrids and S. spontaneum or Erianthus arundinaceus, essentially at Tha-Phra and Thong-PhaPhoom crossing stations. All seedlings are then distributed to selection research stations for subsequent selection steps. The major stations involved in the sugarcane improvement are listed in Table 3. More details about general breeding procedures in Thailand are given in Table 4. At the end of the selection program (about 12 years after crosses are made), when a clone meets the characteristics targeted, variety registration and release is carried out.
The new cultivar release and recommendation mainly can be approved by government and its organization/industry committees as well. The new variety developed by the DOA follows specific rules, such as number of tested locations and years, yield potential level based on the control cultivars, environmental adaptation, major diseases susceptibility, water and nitrogen use efficiencies and farmers’ adoption. For each new variety registered, characteristics are provided by the breeder and approved by the vote of the Plant Breeding Research Committee of the DOA with expert representatives from public and private institutions.
The official registration of new variety follows the procedures under the Plant Varieties Protection Act B.E. 2542 (1999) regulated by the DOA of MOAC and a certificate is issued by the Director-General of DOA to grant plant breeders’ rights and provide legal protection. Meanwhile, the release of new commercial varieties by breeding organizations and private companies also happens without the certification by the DOA. However, in that case, the new variety cannot benefit from legal protection.
On average, Thai commercial sugarcane varieties have displayed a cane yield of 95–116 tonnes/hectare and an average sugar yield per unit area between 12 and 15 tonnes/hectare. However, in terms of sugar content, the CCS of Thai commercial sugarcane varieties has not improved much between 1983 and 2019, with only one unit (%) CCS increase.
The success of a sugarcane breeding program relies on several factors such as the number of seedlings tested, phenotypic data accuracy and the selection method used. In the case of KK3, the breeding program started with more than 100 crosses resulting in a total of 20,000 seedlings. Since only KK3 was selected, the selection ratio was of 1: 20,000 (0.005%) and it took about 12–13 years from crossing to variety release (Sakuanrungsirikul 2020).
Since 1994, the Thai government has invested approximately 1.80 million USD (National Research Council of Thailand 2017a) in KK3 Research and Development (R&D) projects. There were positive and significant economic impacts, including environment and social benefits (Praneetvatakul et al. 2018). Some of the most obvious economic benefits are reduced production cost and increased sugar production/tonne cane. In 2016, the Net Present Value (NPV) of KK3 R&D to the Thai sugar industry was approximately 201 million USD. The benefit incidence proportion to cane grower, sugar mills and seed cane dealer was about 88%, 9% and 3%, respectively with the Benefit Cost Ratio (BCR) and Internal Rate of Return (IRR) about 160.58 and 44%, respectively (National Research Council of Thailand 2017b).
Recently Thai breeders have been concerned about the limited genetic diversity among cultivars and seek to broaden the genetic base of parental germplasm through introgression hybridization. Successive crosses between promising commercial varieties and wild relatives are being conducted. Interspecific crosses, especially using S. spontaneum, have given rise to modern commercial hybrid varieties (Ando et al. 2016; Da Silva 2017). The introgression of genes from other species has the potential to improve the vigor, resilience and pest and disease resistance of commercial varieties.
To broaden the genetic diversity of commercial varieties and to improve their resilience against diseases and drought stress, intergeneric hybridization with related genera such as E. arundinaceus were implemented (Terajima et al. 2019) under the JIRCAS and KKFCRC of DOA cooperation project using delayed heading techniques. The intergeneric progenies with drought tolerance and good ratooning ability are still under evaluation and no hybrids have been released yet for commercial purpose.
Current Major Cultivars
The varieties KK3 and LK92-11 are the two most widely grown varieties in Thailand (Jaisil et al. 2020). These two varieties cover more than 93% of the cultivated sugarcane area (Table 5). As indicated above, Thai breeders are now concerned about the lack of diversity and associated vulnerability of these varieties to potential diseases and the possible impact to the Thai sugar industry.
Germplasm Collection and Development
Several institutions across Thailand are managing sugarcane germplasm collection, not only for hybridization but also for food security. The conservation of accessions is done in field nurseries, and in some cases by cryopreservation. The number and composition of accessions varies greatly among breeding institutions (Table 6). Each accession included in the collection is characterized with specific morphological and physiological traits. The agronomic performances are also being tested over time (plant and ratoon stages) and in different environments. In some cases, genetic markers are used to evaluate the genetic distance between accessions.
The import of sugarcane material from a foreign country is strictly regulated and must follow the criteria, procedure and conditions under the notification of the Plant Quarantine Act B.E. 2507 (No. 5) B.E. 2550 dated 26 April B.E. 2550 from the Ministry of Agriculture and Cooperatives to prevent the introduction of new sugarcane pest and diseases in Thailand. Because of the procedure and the strict conditions required, only a few international exchanges of sugarcane varieties have been reported between Thailand and other countries, and most of the exchanges of varieties are being done between Thai breeding institutions, at the national scale.
Molecular Genetics and Biotechnology
Over the past 60 years, total sugarcane production has increased across the globe, but the rate of sugar yield improvement appears to have plateaued (Yadav et al. 2020). Better understanding of sugarcane genomics and transcriptomics is expected to help break the yield improvement plateau that conventional breeding is facing in Thailand. In 2000 the Thai government began strongly supporting R&D, persuading the private sector to invest in in commercial research and giving financial incentives (tax incentives, soft loans and subsidies), in order to obtain commercial research innovation matching user demands and social returns (Tangkitvanich 2002). The sugarcane industry is one of the agricultural research priorities. Several research projects related to the genetic improvement of the sugarcane have recently been funded, including one aimed at producing a transcriptome dataset of the commercial variety KK3 (Piriyapongsa et al. 2018).
Application of Biotechnology to Sugarcane in Thailand—History and Current Status
Since the mid 1970s, with the development of biotechnology and molecular biology techniques, several research projects aiming to improve the performance of sugarcane varieties were conducted in Thailand.
Tissue Culture Techniques
The propagation of clean seed cane for commercial purposes is mainly practiced by the KU, DOA, OCSB, the Department of Agricultural Extension (DOAE), KKU and most sugar companies. For about 10% of the cultivated area, disease-free seed canes are distributed to farmers to limit the propagation of major sugarcane diseases at the national and international scale. Micropropagation in sugarcane is achieved mainly through axillary shoot formation, adventitious shoot formation, and somatic embryogenesis (Tiwari et al. 2011).
Mutagenesis
The application of mutagenesis techniques on sugarcane in Thailand started in 1984 with the application of gamma radiation on sugarcane buds aiming to develop smut resistant lines (Srising and Thumtong 1984). Then, a mutagenesis study conducted by Ngampongsai et al. (1992) reported the exposure of callus and plantlets of sugarcane derived from Q83 and U-Thong 1 with various doses of gamma radiation, and an average better regeneration of the variety Q83 compared to the variety U-Thong1 after exposure. In 2007, a study conducted by Punyadee et al. (2007) successfully performed in-vitro selection of sugarcane cell lines resistant to the herbicide imazapyr. Kerdkhong et al. (2018) conducted a study of the mutations in somaclones compared to the wild type using simple sequence repeat (SSR) markers. Among all SSRs, 3 primers showed different polymorphisms from the wild type plant and were considered as useful to separate somaclones into genetic clusters. Chaisan et al. (2018) reported the exposure to gamma radiations and Ethyl-methane sulfonate (EMS) of the cultivar KK3 to produce clones with improved drought tolerance. After greenhouse and field evaluations under drought conditions, 14 clones were successfully characterized as drought tolerant which are now intended for use in breeding programs.
Somaclonal variation, with or without irradiation or chemical mutagens, has generated beneficial traits of interest in sugarcane such as agronomic features and increased disease tolerance. Nevertheless, progress in the adoption of such technology in the generation of mutant accessions has been extremely limited. Moreover, considering their weak agronomical performances compared to the wild type, no mutant varieties have been used for commercial purpose yet.
Transgenic Research
Recent research work in Thailand has reported the transformation of sugarcane with the BT gene Cry1A(b) to confer improved resistance against the sugarcane stem borer (Islam et al. 2016). Genetic transformation was also applied for the creation of clones with improved tolerance to the herbicide glyphosate (Chanprame 2011). However, currently, the use of genetic transformation in sugarcane is reserved only for research purposes, and evaluation of GE clones in field trials are not yet to be permitted by the Thai government, as noted above. Genome editing techniques have also been initiated in Thailand. Only a single attempt of gene editing on sugarcane has been reported in Thailand so far, aiming at the improvement of the sugar content (Chanprame 2011).
Use of DNA Markers in Sugarcane Breeding in Thailand–Current Status
The use of genetic markers to secure the management of germplasm is starting in both governmental and private sectors. The DOA, KU, OCSB and MitrPhol Innovation and Research Center, are currently fingerprinting each clone in their germplasm collections with microsatellite markers to secure its management over time and identify possible mislabeling. These markers are also being used to confirm the genetic identity of unknown varieties, or at least to measure genetic distance with varieties already present in the germplasm. In 2007, 3,199 SSRs markers were developed by data mining on partial sugarcane EST database for genetic mapping purposes (Thipmongkol et al. 2007). In 2013, Random Amplified Polymorphic DNA (RAPD) markers were tested for the selection of S. spontaneum and true hybrids S. spontaneum x S. officinarum (Chaiyabut et al. 2013). Microsatellite markers have also been used to identify true hybrids between Saccharum and Erianthus genus at KKFCRC under the DOA and JIRCAS cooperation project (Terajima et al. 2019). At present, 100 promising clones are in yield evaluation.
Expressed sequence tag-simple sequence repeats (EST-SSRs) are considered valuable for breeding programs, since they may be directly associated with the genes expressing a particular trait. In 2009, QTL mapping of sugar content related traits was performed on a population of hybrids S. officinarum x S. spontaneum using AFLP, SSRs markers. Markers associated with the sucrose synthase gene were found of interest for further application in sugarcane marker assisted breeding (Poomipant et al. 2009). In 2015, Intron Length Polymorphism (ILP) markers associated with the sucrose metabolism were developed and evaluated for the selection of hybrids with higher sugar content (Pongsiripat et al. 2015). In 2019, an association mapping study was successfully performed on a set of 200 accessions using EST-SSRs markers related to sugar-related traits (Ukoskit et al. 2019). Two EST-SSR markers with a putative relationship with uridine diphosphate (UDP) glycosyltransferase, and beta-amylase were associated with pol and sugar yield. These two functional markers are now being used for marker-assisted breeding programs in Thailand. Recently, Khanbo et al. (2021) performed a candidate gene association mapping based on 109 genes. The study identified favorable alleles of the candidate genes involved in signaling and transcriptional regulation. The results will support the genetic improvement of sugarcane and may help to clarify the genetic architecture of sugar-related traits.
Pakphom et al. (2011) reported the development of a set of markers related to the stem diameter and the fiber percentage. At MitrPhol Innovation and Research Center, microsatellite markers related to high biomass have been developed and are now being validated on crossing populations.
Several molecular techniques have been applied to study genetic responses to drought. Research conducted at the National Center for Genetic Engineering and Biotechnology (BIOTEC) in collaboration with MitrPhol Innovation and Research Center aimed to identify candidate genes associated with drought tolerance (Nawae et al. 2020). This work aimed to follow the transcriptional changes of genes between a drought-sensitive and a drought-tolerant variety in response to drought stress. Expression profiles suggested that the tolerant sugarcane has a more effective genetic response than the sensitive genotype at the initiation of drought stress, with a higher expression of genes involved in water retention, antioxidant secondary metabolite biosynthesis, and oxidative and osmotic stress response. Dehydration Responsive Element Binding (DREB) genes are coding for transcription factors produced by plants in response to stress conditions such as salinity, drought, and cold stress. In Thailand, Chanprame et al. (2019) isolated and characterized one of these DREB genes from sugarcane (ScDREB2) believed to be associated with salinity tolerance.
A project is being operated to develop varieties with improved tolerance to the sugarcane white leaf disease (SCWLD). Since the genetic determinism of defense mechanisms against SCWLD is still poorly understood, the project is aiming to identify candidate genes by studying transcriptional changes between susceptible and more tolerant varieties of sugarcane in response to an infection by Candidatus Phytoplasma (unpublished work). Comparative transcriptome analysis has revealed about 1500 differentially expressed genes (DEGs). Amongst those, a set of candidate genes with gene ontology (GO) terms and metabolic functions related to plant defense mechanisms was defined. After validation of their expression pattern, candidate genes with the most promising profiles will be tested on crossing populations for future characterization of SCWLD resistant/tolerant sugarcane varieties.
Genomic selection (GS) is a breeding method in which individuals are selected based on their predicted breeding values that are calculated from genome-wide DNA marker profiles (Meuwissen et al. 2001). Until now, only a few studies have investigated GS for sugarcane (Yadav et al. 2020). The development of SNP markers associated with drought tolerance via target sequencing is being done at Thammasat University in collaboration with the BIOTEC and MitrPhol Innovation and Research Center. After validation, these markers might constitute a reference set for future genomic selection applications in sugarcane. Also, SNP markers were developed by Genotyping-By-Sequencing (GBS) at the KKFCRC. The association of these markers with major agronomic traits of interest is still under progress.
The sequencing and annotation of the sugarcane genome will lead to a greater understanding of the morphology and physiology of sugarcane. In Thailand, the development and integration of biotechnology will no doubt lead to further improvement of desirable traits and support the sustainability of the sugar industry.
Other Perspectives of Thailand Sugarcane Breeding
Long breeding cycles and a narrow genetic base of the local germplasm collections have limited the development of varieties with desirable agronomic and milling traits. To overcome the plateaus limiting the improvement of the cane yield or sugar content, the efficiency and accuracy of the selection have to be improved. To meet this objective, breeding programs will tend to improve the evaluation of breeding values for parental selection and integrate high-throughput genomics and phenomics screening tools to shorten the length of the breeding cycle. Most sugarcane breeding research works conducted in Thailand are now aiming at a better understanding of the association between genotypes and phenotypes. New varieties must include features such as a better adaptation to specific agro-climatic environments. New varieties must also be suitable to mechanical harvesting. Increased shedding of dead leaves in new varieties will help to reduce the burning of cane and hence reduce the emission of PM2.5. These directions will support development of more profitable new varieties for specific uses and, as a result, will help the sugar industry in Thailand to face future challenges.
References
Ando, S., Y. Terajima, S. Tagane, M. Sato, K. Ishiki, M. Matsuoka, Y. Takagi, A. Sugimoto, W. Ponragdee, T. Sansayawichai, and A. Tippayawat. 2016. New sugarcane varieties using wild sugarcane and collaboratively bred in Thailand. Annual report 2015 (April 2015-March 2016). Japan international research center for agricultural sciences.
Department of Agriculture. 1982. History of sugarcane breeding in Thailand. The progress of plant breeding in the department of agriculture, 2–3 October 1982. Department of agriculture extension. Bangkok, Thailand.
Bunnak, S., P. Jaisil, and P. Therakulpisuth. 2011. Transformation and expression of chitinase gene to genetic modified sugarcane (Saccharum officinarum L.). Research report. Khon Kaen University.
Chaisan, T., W. Wannarat, and J. Authapun. 2018. Creation of genetic variation and selection for drought tolerance in sugarcane. In ISSCT 12th Germplasm & Breeding workshop.
Chaiyabut, A., S. Poeaim, and K. Distabanjong. 2013. Genetic Relationship in Saccharum spontaneum and Their Hybrids by RAPD. National genetics conference 2013 (NGC2013). Thai J. Genet. 2013, S(1): 206–209
Chanprame, S. 2011. Transformation of herbicide resistant and sucrose accumulation genes into selected sugarcane cell. Final report on project to create knowledge and develop sugarcane under the master plan project in basic intelligence: Long-term project year 2009: Volume 1 Varieties and information. Kasetsart University. 323–328.
Chanprame, S., T. Promkhlibnil, S. Suwanno, and C. Laksana. 2019. Isolation characterization and expression of transcription factor ScDREB2 from wild, commercial and interspecific hybrid sugarcane in salinity condition. Journal of Plant Biotechnology 46: 97–105. https://doi.org/10.5010/JPB.2019.46.2.097.
Chatwachirawong, P. 2006. Development of sugarcane crossing station at Ban Tiphuyae, Tambol Charae, Amphoe Thongphaphum, Kanchanaburi Province (phase 1): (Final report: research development design and engineering project). Nakhon Pathom (Thailand): national science and technology development agency, Bangkok (Thailand). National center for genetic engineering and biotechnology.
Chunhawong, K., T. Chaisan, and S. Rungmekarat. 2018. Sugar industry and utilization of its by-products in Thailand: an overview. Sugar Tech 20(2):111–115. https://doi.org/10.1007/s12355-018-0599-x.
Comstock, J.C. 2008. Sugarcane yield loss due to ratoon stunt. Journal Association Sugar Cane Technologists 28: 22–31.
Da Silva, J.A. 2017. The Importance of the wild Cane saccharum spontaneum for bioenergy genetic breeding. Sugar Tech 19: 229–240. https://doi.org/10.1007/s12355-017-0510-1.
Hanboonsong, Y., W. Ritthison, C. Choosai, and P. Sirithorn. 2006. Transmission of sugarcane white leaf phytoplasma by yamatotettix flavovittatus, a new leafhopper vector. Journal of Economic Entomology. 99 (5): 1531–1537.
Hydro-informatics institute, 2018. Thailand water situation in 2018. https://www.thaiwater.net/current/YearlyReport2018/home.html. Accessed 31 August 2020.
Islam, N., C. Laksana, and S. Chanprame. 2016. Agrobacterium-mediated transformation and expression of BT gene in transgenic sugarcane. Journal of the International Society for Southeast Asian Agricultural Sciences 22: 84–95.
Jaisil, P. and S. Yammankong. 2015. Sugarcane variety in Thailand. Khon Kaen University. Khon Kaen. Khon Kaen Publishing Part., Ltd.
Jaisil, P., P. Songsri, N. Jongrangklang, J. Kruangpathi, and K. Sutapakdee.2020. National evaluation of elite sugarcane clones suitable for growing areas Phase III (Period II), Final report. Khon Kaen Univeristy.
Khon Kaen Field Crops Research Center. 2016. Sugarcane variety. Pre-Congress tour 2016, 29th 2016 ISSCT, Thailand. 2–3 December 2016, Khon Kaen Field Crops Research Center, Department of Agriculture.
Kanjanavisut, K. and P. Rattanasiwakoon. 2020. EIC evaluates that the 2020 drought could extend to June with severe impacts on sugarcane, off-season rice, and cassava. 2 March 2020. https://www.scbeic.com/en/detail/product/6660. Accessed 12 Sept 2020.
Kerdkhong, C., S. Prammanee, and R. Lersrutaiyotind. 2018. Mutation identification in sugarcane somaclones using Simple Sequence Repeat Markers (SSR). Journal of Food Health and Bioenvironmental Science 11: 13–22.
Khanbo, S., S. Tangphatsornruang, J. Piriyapongsa, W. Wirojsirasak, P. Punpee, P. Klomsa-ard, and K. Ukoskit. 2021. Candidate gene association of gene expression data in sugarcane contrasting for sucrose content. Genomics 113: 229–237. https://doi.org/10.1016/j.ygeno.2020.12.014.
Khon Kaen University. 2014. Project on production cost management and knowledge transfer to reduce farmers’ sugarcane production costs in Crop year 2014/15, Final Report. Khon Kaen University.
Kimura, T., Y. Ueta, M. Mori, S. Tsuruta, Y. Terajima, S. Ando, M. Umeda, T. Hattori, Y. Tarumoto, T. Subtira, B. Ruttawat, S. Chanachai, W. Pongragdee, and S. Sakuanrungsirikul. 2020. Analysis of genetic diversity among sugarcane genetic resources in Thailand. The International plant and animal genome XXVIII. San Diego, USA.
Krungsri Research. 2020. Industry Horizon. https://www.krungsri.com/bank/getmedia/795c2645-d7fc-4e58-8a9e-749fe208e67c/IH_Industry_Horizon_200409_EN.aspx. Accessed 11 October 2020.
Marcone, C. 2002. Phytoplasma diseases of sugarcane. Sugar Tech. 4 (3 & 4): 79–85.
Meuwissen, T.H.E., B.J. Hayes, and M.E. Goddard. 2001. Prediction of total genetic value using genome-wide dense marker maps. Genetics 157 (4): 1819–1829.
Ministry of Agriculture and Cooperatives. 2020. Thailand agricultural area in 2017. Ministry of Agriculture and Cooperatives (MOAC). https://data.moac.go.th/. Accessed 20 March 2020.
Napasintuwong, O. 2010. The role of agricultural biotechnology policies in Thailand’s economy. Asian Biotechnology and Development Review 12 (1): 1–19.
Napasintuwong, O. 2019. Current status of agricultural biotechnology in Thailand. FFTC Agricultural Policy Platform (FFTC-AP). Food and Fertilizer Technology for the Asian and Pacific Region. Ap.fftc.org.tw/article/1383. 27 Jan 2021.
National Research Council of Thailand. 2017a. Evaluation research investment on cane and sugar, oil palm and livestock. https://www.nrct.go.th/e-publish1/evaluate-research-investment2/evaluate-research-investment2-upd.pdf. Accessed 20 March 2020.
National Research Council of Thailand. 2017b. Impact of research investment on major economic crops. https://nriis.nrct.go.th/FileUpload/AttatchFile/News/256011271140502970129.pdf. Accessed 20 March 2020.
Nawae, W., J.R. Shearman, S. Tangphatsornruang, P. Punpee, T. Yoocha, D. Sangsrakru, C. Naktang, et al. 2020. Differential expression between drought-tolerant and drought-sensitive sugarcane under mild and moderate water stress as revealed by a comparative analysis of leaf transcriptome. Peer J 8: e9608. https://doi.org/10.7717/peerj.9608.
Ngampongsai, S., W. Sriphisut, and P. Suriyapan. 1992. Use of radiation and tissue culture techniques to induce mutation in sugarcane. Suphan Buri Field Crops Research Center, Thailand.
Office of the Cane and Sugar Board. 2020. Annual Report 2019. Office of the Cane and Sugar Board, Ministry of Industry. Bangkok, Text and Journal Publication Press.
Pakphom, Y., R. Lersrutaiyothin, and S. Anusonpornperm. 2011. Evaluation of molecular markers relating to fiber percentage and stem diameter of sugarcane. Kamphaengsaen Academic Journal (thailand) 3 (3): 1–17.
Piriyapongsa, J., P. Kaewprommal, S. Vaiwsri, S. Anuntakarun, W. Wirojsirasak, P. Punpee, P. Klomsa-ard, P.J. Shaw, W. Pootakham, T. Yoocha, D. Sangsrakru, S. Tangphatsornruang, S. Tongsima, and S. Tragoonrung. 2018. Uncovering full-length transcript isoforms of sugarcane cultivar Khon Kaen 3 using single-molecule long-read sequencing. Peer J: e5818. https://doi.org/10.7717/peerj.5818.
Pitak, N. 2009. Control and management of sugarcane borers. Diseases and pests control and management training course. April 28–30, 2009. Khon Kaen field crops research center, Department of agriculture. 50p.
Pliansinchai, U., and W. Suchato. 1995. Sett transmission of sugarcane green grassy shoot disease. In: Proceedings 2nd national plant protection congress, Oct. 9–11, 1995, Chiangmai, Thailand, pp. 234–238.
Pliansinchai, U., N. Surinthu, S. Prammanee, T. Thanawon, and M. Yimsaard. 1997. Ethiology Symptomatology of Grassy shoot syndrome of sugarcane and heat therapy. In Proceedings of the 35th Kasetsart University annual conference: Plant, Agricultural extension and communication and agro-Industry. 3–5 Feb. 1997. Bangkok, pp. 339–406.
Pliansinchai, U., U. Laebwan, and W. Suchato. 1998. Sugarcane green grassy shoot disease: Epidermiology, transmission and control. In: Proceedings of the 3rd National cane and sugar conference, Nov. 25–27, 1998, Bangkok, Thailand, 24 (Abstract).
Pongsiripat, N., and K. Ukoskit. 2015. Development of ILP markers from genes related to sucrose metabolism in sugarcane and genetic structure of sugarcane. Thai of Journal Genetics 8 (2): 111–122.
Poomipant, P., P. Thipmongkolcharoen, G. Phusudsawang. P. Chatwachirawong and K. Ukoskit. 2009. Construction of a genetic linkage map of simplex and double simplex markers in sugarcane. In Proceedings of the 16 National Genetics: Genetics for national energy crisis. Pathum Thani (Thailand): Genetics Society of Thailand, Bangkok (Thailand). pp. 330.
Praneetvatakul, S., K. Vijitsrikamol, C. Lerdyosuk, and B. Kontong. 2018. Impact pathway of sugarcane and sugar researches in Thailand. Journal of the Association of Researchers Vol. 23 No. 2 (2561). https://so04.tci-thaijo.org/index.php/jar/article/view/241902/164474.
Punyadee, P., M. Throngros, and T. Pornprom. 2007. Biochemical mechanism of resistance to imazapyr in sugarcane cell selections. Thai Journal of Agricultural Science 40: 133–141.
Rao, G.P., S. Mall, and C. Marcone. 2012. Recent biotechnological approaches in diagnosis and management of sugarcane phytoplasma diseases. In Functional plant science and biotechnology. Recent trends in biotechnology and microbiology, vol. 6, ed. R.K. Gaur, et al., 19–29. New York: global Science Books.
Roddee, J., Y. Kobori, and Y. Hanboonsong. 2018a. Characteristics of sugarcane white leaf phytoplasma transmission by the leafhopper Matsumuratettix hiroglyphicus. Entomologia Experimentalis et Applicata. https://doi:https://doi.org/10.1111/eea.12741
Roddee, J., Y. Kobori, and Y. Hanboonsong. 2018b. Multiplication and distribution of sugarcane white leaf phytoplasma transmitted by the leafhopper, Matsumuratettix hiroglyphicus (Matsumura) (Hemiptera: Cicadellidae), in Infected Sugarcane. Sugar Tech 20. Springer India: 445–453. https://doi:https://doi.org/10.1007/s12355-017-0559-x.
Saengsai, W., S. Sakuanrungsirikul, and P. Yong-Bao. 2018. Detection of Leifsonia xyli subsp. xyli in sugarcane in the Northeastern and Central Thailand using PCR techniques. Khon Kaen Agric J. 46 supplement 2: 202–211.
Sakuanrungsirikul, S. 2020. Future research perspectives: problem and solution assessment on increasing sugarcane productivity. Field and renewable energy crops research institute annual seminar 2020. Field and renewable energy crops research institute, Department of agriculture.
Sakuanrungsirikul, S., T. Wongwarat, S. Srisink, and P. Theerakulpisut. 2012. Genetic diversity of phytoplasma associated with diseases of sugarcane and some grasses in Thailand based on sequencing analysis of the 16S–23S rRNA intergenic spacer region. Khon Kaen Agric J. 40 (Supplement 3): 231–240.
Scortecci, K.C., S. Creste, T. Calsa Jr., A.X. Mauro, M.G.A. Landell, A. Figueira, and A.B. Vagner. 2012. Challenges, opportunities and recent advances in sugarcane breeding. Plant Breeding. https://doi.org/10.5772/28606.
Srising, S., and P. Thumtong. 1984. Mutation breeding in sugarcane for smut resistance by gamma ray on sugarcane bud. Research report in 1984: fiber crop, sugarcane, tobacco, Department of agriculture. Bangkok Thailand, 1984.- ISBN 974–7619–93–8. p. 388
Srisink, S., S. Sakuanrungsirikul, W. Suchato, and W. Yodprang. 2014. Hot water treatment for white leaf control. Research and development project report 2014. Department of agriculture. https://www.doa.go.th/research/attachment.php?aid=1032. Accessed 28 January 2021.
Srisink, S., W. Suchato, A. Boontham, W. Wandee, S. Poonpan, and W. Yodprang. 2016. Integrated sugarcane pest and disease management. Suphan Buri field crops research center. Edition 1. 52pp. http://www.sugarzone.in.th/cane/suphan_cane/book_cane1.pdf. Accessed 16 Oct 2020.
Office of the Cane and Sugar Board. 2019. Sugarcane plantation areas in Thailand crop year 2018/19 report. Office of the cane and sugar board, Ministry of Industry. https://www.ocsb.go.th/upload/journal/fileupload/923-9040.pdf. Accessed 20 Mar 2020.
Suphan Buri Field Crops Research Center. 2017. Sugarcane varieties release from suphan buri field crops research center (SPFCRC). Office of agricultural research and development, Region 5, Department of agriculture. Suphan buri, U-Thong publishing part., Ltd.
Sukyai, P., N. Yingkamhaeng, N.T. Lam, V. Tangsatianpan, C. Watcharinrat, G. Vanitjinda, W. Vanichsriratana, and K. Sriroth. 2016. Research and development prospects for sugarcane and sugar industry in Thailand. Sugar Tech 18(6):583–587. https://doi.org/10.1007/s12355-016-0495-1.
Sundar, A.R., E.L. Barnabas, P. Malathi, and R. Viswanathan. 2012. A mini-review on smut disease of sugarcane caused by Sporisorium scitamineum. Botany 53: 107–128. https://doi:https://doi.org/10.5772/34116.
Tagane, S., A. Sugimoto, Y. Terashima, Y. Egawa, K. Ishiki, M. Sato, S. Irei, W. Ponragdee, T. Sansayawicha, and A. Tippayawat. 2010. Evaluation and classification of Erianthus germplasm in Thailand. https://www.jircas.go.jp/en/publication/research_results/2010_20.
Tagane, S., M. Yasuda, W. Ponragdee, T. Sansayawichai, and A. Sugimoto. 2011. Cytological study of Erianthus procerus and E. arundinaceus (Gramineae) in Thailand. Cytologia 76(2): 171–175. https://doi.org/https://doi.org/10.1508/cytologia.76.171/_pdf.
Tagane, S., W. Ponragdee, T. Sansayawichai, A. Sugimoto, and Y. Terajima. 2012. Characterization and taxonomical note about Thai Erianthus germplasm collection: the morphology, flowering phenology and biogeography among E. procerus and three types of E. arundinaceus. Genetic Resources and Crop Evolution 59: 769–781. https://doi.org/10.1007/s10722-011-9717-2.
Tangkitvanich, S. 2002. Reorienting commercial research for private sectors. Annual research report seminar 2002. December 12–14, 2002. The Ambassador city Jomtien hotel, Chon Buri. https://tdri.or.th/wp-content/uploads/2013/07/YE2002_5_04.pdf.
Terajima, Y., A. Tippayawat, W. Ponragdee, T. Sansayawichai, S. Irei, A. Sugimoto, H. Takagi, and H. Hayashi. 2019. Application of a delayed heading technique to early-heading erianthus arundinaceus native to thailand for intergeneric crossing with sugarcane. Tropical Agriculture and Development 63 (1): 1–11.
National Center for Genetic Engineering and Biotechnology. 2010. White paper: Updated status and perspective of Thailand on research and development of modern biotechnology and biosafety regulation, 3rd ed. Thailand: Technical Biosafety Committee.
Thipmongkol, P., K. Chanthong, and K. Ukoskit. 2007. Development of SSR markers by data mining from partial sugarcane est database. J Sci Tech 15 (3): 58–65.
Tiwari, A.K., S. Tripathi, M. Lal, M.L. Sharma, and P. Chiemsombat. 2011. Elimination of sugarcane grassy shoot disease through apical meristem culture. Archives of Phytopathology and Plant Protection 44: 1942–1948.
Ukoskit, K., G. Posudsavang, N. Pongsiripat, P. Chatwachirawong, P. Klomsa-ard, P. Poomipant, and S. Tragoonrung. 2019. Detection and validation of EST-SSR markers associated with sugar-related traits in sugarcane using linkage and association mapping. Genomics. Elsevier. https://doi.org/10.1016/j.ygeno.2018.03.019.
UN Comtrade. 2020. Thailand exports of sugars and sugar confectionery. https://tradingeconomics.com/thailand/exports/sugars-sugar-confectionery. Accessed 12 Sept 2020.
Workman, D. 2020. Sugar exports by country 2019. https://www.worldstopexports.com/sugar-exports-country. Accessed Oct 2020.
Yadav, S., P. Jackson, X. Wei, EM. Ross, K. Aitken, E. Deomano, F. Atkin, BJ. Hayes, and KP. Voss-Fels. 2020. Accelerating genetic gain in sugarcane breeding using genomic selection. Agronomy 10: 1–21. https://doi.org/10.3390/agronomy10040585.
Yulianti, T., K.S. Wijayanti, and Supriyono. 2020. Management of ratoon stunting disease by hot water treatment to provide healthy cane seed. In: 1st International Conference on Sustainable Plantation (1st ICSP 2019). IOP Conf. Series: Earth and Environmental Science 418 (2020) 012063. IOP Publishing. https://doi.org/10.1088/1755-1315/418/1/012063.
Acknowledgements
The authors would like to thank Assoc. Prof. Dr. Prasit Jaisil, Mr. Werapon Ponragdee and Mr. Sarewat Juttupornpong for their insightful comments on the breeding and varieties development issues. We also gratefully acknowledge the comments on the pests and diseases topic from Ms. Sunee Srisink and Mr. Rungsi Charoensathaporn.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Khumla, N., Sakuanrungsirikul, S., Punpee, P. et al. Sugarcane Breeding, Germplasm Development and Supporting Genetics Research in Thailand. Sugar Tech 24, 193–209 (2022). https://doi.org/10.1007/s12355-021-00996-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12355-021-00996-2