Abstract
Background
Patients with normal myocardial perfusion imaging (MPI) have a good prognosis. However, pre-clinical coronary and extracoronary atherosclerosis may exist in the absence of myocardial ischemia.
Methods
154 Egyptian patients (mean age 53 years) underwent whole-body non-contrast CT following normal MPI.
Results
Atherosclerosis in the form of calcification was observed in ≥1 vascular bed in 115 of 154 (75%) patients. This included the iliofemoral (62%), abdominal aorta (53%), thoracic aorta (47%), coronary (47%), and carotid (25%) vascular beds. Mean total body calcium score was 3172 ± 530 AU. Extracoronary atherosclerosis in patients with a zero coronary artery calcium (CAC) score was common, occurring in the above-listed beds 42%, 36%, 29%, and 7% of the time, respectively. CAC was rarely present without iliofemoral or abdominal aortic calcification.
Conclusion
Quantitative assessment of calcification in different vascular beds demonstrates that extracoronary atherosclerosis is common in patients who have normal MPI. Atherosclerotic calcifications are most common in the iliofemoral arteries and abdominal aorta, which typically predate coronary calcifications. An imaging strategy to detect extracoronary atherosclerosis could lead to greater understanding of the natural history of atherosclerosis in its long pre-clinical phase and possibly to earlier preventive strategies.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Myocardial ischemia and acute myocardial infarction are late-stage manifestations of atherosclerotic coronary artery disease (CAD). Atherosclerosis is a ubiquitous chronic systemic disease beginning early in life, affecting arteries throughout the body, long before significant stenoses or occlusions appear. Strategies for primary prevention very early in the disease cascade prior to the advent of life-threatening events are needed, and could be based on identification of pre-clinical atherosclerosis.1,2,3 We previously found multi-vessel bed atherosclerosis to be common in ancient human mummies from Africa, Asia, and North and South America, despite their non-western lifestyle and a dearth of traditional risk factors.4 The detection of arterial calcification in systemic arteries outside the coronary circulation is well recognized, not as a risk factor for atherosclerosis, but rather a prima facie diagnostic feature of the disease, often in a pre-clinical state when more individualized and intensive preventive treatment could be employed.2,4,5,6,7,8
In order to assess the prevalence of pre-clinical atherosclerosis in the vascular beds of a contemporary Egyptian population, whole-body CT scans were performed in a cohort of individuals without known CAD and no evidence of myocardial ischemia on MPI.
Methods
Study Population
One hundred and fifty-four consecutive outpatients without known CAD, whose SPECT MPI, performed for standard clinical indications, was normal, were recruited at an outpatient imaging center in Cairo, Egypt (Alfa Scan) from April 2014 to April 2015 to undergo a whole-body CT scan for research purposes. Informed consent of each study subject was obtained prior to enrollment.
Myocardial Perfusion Imaging
A total of 151 (98%) patients underwent symptom-limited treadmill exercise stress according to the standard Bruce protocol, while 3 (2%) patients underwent dipyridamole stress. Termination of the stress test was performed according to recommendations in the updated guidelines of exercise testing.9
A total of 152/154 subjects underwent same-day stress-only MPI imaging in accordance with ASNC 2016 guidelines.9 The remaining two patients underwent two-day technetium-99m sestamibi gated SPECT MPI. All images were acquired with a dual-head Philips JetStream camera (Philips Healthcare, Milpitas, California) with low-energy, high-resolution collimators. All radionuclide images and associated data were processed according to the standard protocols.10,11 Myocardial perfusion images were interpreted using the standard 17-segment model.9,10,11
At least two experienced nuclear cardiology specialists interpreted each segment semi-quantitatively using 0 for normal uptake, 1, 2, and 3 for mild, moderate, and severe reduction of uptake, respectively, and 4 for absent uptake. Summed stress, summed difference, and summed rest scores (SSS, SDS, and SRS) were calculated. High-risk markers were reported, including increased lung-to-heart ratio, transient left ventricular cavity dilatation, and abnormal regional and global wall motion abnormalities. Normal SPECT MPI studies were defined as follows: ejection fraction above 50%, no left ventricular dilatation, and normal myocardial perfusion images (SSS < 4 and SDS = 0).
CT Imaging Protocol
CT scanning was performed on either a Toshiba Aquilion One 320 (Toshiba America Medical Systems, Tustin, California) (N = 96) or a Philips ICT 256 (Philips Healthcare, Bothell, Washington) (N = 58). Gated coronary CT scans were performed with 2.5 cm slice thickness, 50% overlap, 120 kV, 50 to 100 mA, and 90 to 180 mAs tube current, automatically selected by Toshiba Sure Exposure Version V4.74ER016 or Philips iDose Version V3. Low-dose, whole-body CT scanning (from mandible to mid-calf) was obtained with a spiral acquisition, 1 mm slice thickness, 128 × 0.625 mm collimation, and pitch .899, kV 100 to 120. CAC was quantified using the Agatston scoring method with a Vitrea Workstation, Version 4 (Toshiba Medical Systems Europe, The Netherlands) and was confirmed when needed in OsiriX, Version 3.9 (Pixmeo, Switzerland) 64-bit image processing software.12 Thoracic aorta (above the diaphragm, TAo), abdominal aorta (below the diaphragm, AAo), and iliofemoral (ILF) calcium scores were also performed with OsiriX. A Philips Extended Brilliance Workspace CT workstation (Philips Healthcare, Bothell, Washington) was used for calcium scoring of the carotids. TAo, AAo, and ILF calcium were scored according to previously described methods and using coronary calcium scoring software.4,5,6,13 Hounsfield units ≥130 were used as the calcification threshold. Total body calcium (TBC) is the sum of calcium scores in the five vascular beds. The AAo and ILF calcifications of one CT scan could not be quantified because of inadequate image quality due to patient factors.
Statistical Approach
Results were collected, tabulated, and statistically analyzed using SPSS version 17 (Softonic International, San Francisco, California). Differences between continuous variables and categorical variables were tested with unpaired t tests and χ2 tests. For inter-group comparison, an ANOVA test was used. Binary logistic regression was used to determine the strength of the relationship between one dependent variable and a series of independent variables. Coronary (CA) and carotid (CR) vascular beds were defined as event-related vascular beds. A P value of <0.05 was considered statistically significant.
Results
The study population is characterized in Table 1. Mean age was 53.0 ± 10.6 years (range 22 to 79 years), 108 (70%) were male, and 91/154 (59%) complained of chest pain. Traditional risk factors of dyslipidemia, hypertension, tobacco use, and diabetes were prevalent. Patients with known CAD were excluded. Mean exercise duration (Bruce protocol) was 7.7 ± 2.6 minutes with a range of 1 to 16.6 minutes. Twenty-eight of 151 patients (19%) had an abnormal exercise ECG response and 5 (3%) complained of chest pain during exercise testing. CT mean radiation dose was 6.2 ± 1.8 millisieverts.
Subclinical atherosclerosis was common. Of the 154 patients, 115 (75%) had ≥1 vascular bed calcification involvement. This was observed in 81/108 (75%) of men and 34/46 (74%) of women. The ILF and the AAo were the most frequently involved beds with calcifications increasing in frequency in an ascending “south to north fashion” in both sexes. The CR and CA were least affected, as illustrated in Figure 1. TBC score increased with age in an exponential fashion in men and women (Figure 2A, B). Beginning at age 50, the rate of rise was greater in women than men. The occurrence of calcification in all vascular beds also increased with increasing age. However, there was a substantial delay in the development of calcification in the CR and CA beds relative to the other beds (Figure 3). In these “event-related” arterial beds, the appearance of calcifications lagged the AAo and ILF beds by ≈10 years (Table 2; Figure 3).

Adapted with permission from Laclaustra et al19
Prevalence of arterial calcification in five vascular beds among men and women.
The calcium scores in the TAo, AAo, and ILF beds were considerably higher than those in the CA and CR beds (Table 2). Upon reaching age 70, almost all arterial beds were involved. The number of vascular beds involved increased in a progressive fashion with increasing age for both sexes (Online Appendix Figure 1).
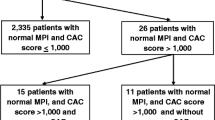
Figure 4A demonstrates the prevalence of calcification in the four non-CA vascular beds based on the presence or absence of CAC and Figure 4B demonstrates the mean calcium score similarly stratified. Even when the coronary calcium score was 0, calcification was frequently present in other vascular beds with the ILF bed most often involved. The frequency of calcification in patients with CAC = 0 was 42% ILF, 36% AAo, 29% TAo, and 7% for CR beds (Figure 4A). A total of 44/83 (53%) of patients with a CAC = 0 had calcifications in other beds. The mean TBC for patients with CAC = 0 was 831 AU (Figure 4B).
Online Appendix Figures 2A and B demonstrate that patients with calcium in the CA and CR arteries frequently had calcification in non-event-related beds and that mean calcification scores were much higher in these beds compared to patients without calcium in the event-related beds.
Lastly, the mean calcium score of four vascular beds similarly increased when stratified by increasing degree of CAC as shown in Figure 5, P < 0.001.
Discussion
This study showed that patients without known CAD and a normal MPI frequently have subclinical atherosclerosis on whole-body CT in the form of arterial calcification. Even in patients without CAC, atherosclerosis was frequent in other vascular beds. Atherosclerosis was most frequent in the ILF bed and progressed with age in an ascending “south to north fashion” to involve the aorta, coronary, and carotid arteries predominantly later in life. Our findings are consistent with those of the PESA study, in which subclinical atherosclerosis, as assessed by 2- and 3-dimensional ultrasound, was frequent in an asymptomatic middle-aged cohort not undergoing MPI. The prevalence of atherosclerosis in the PESA population was comparable to this Egyptian cohort, and most common in the ILF bed (44%), followed by the CR (31%) and AAo (25%) beds, with CAC present in 18%.14 Our data extend the work of Allison and colleagues in which increasing age was a consistent predictor of atherosclerotic disease as assessed by whole-body CT across all beds.15
The distribution and progression of atherosclerosis in autopsy studies was described in the 1950s by Roberts and Wilkins.16,17,18 Atherosclerosis of the common iliac arteries was observed as early as the second to third decades of life.16,17,18
The recent Aragon Workers’ Health Study by Laclaustra et al also suggests that early atherosclerotic involvement of the femoral and iliac arteries, as assessed by ultrasound, might be used as an early, sensitive diagnostic marker for pre-clinical atherosclerosis.19 These investigators observed that femoral subclinical atherosclerosis was associated with a higher risk of CAC than was carotid atherosclerosis. Area under the receiver operating characteristic curve for prediction of positive (≥1) or high (≥300) CAC were 0.706 and 0.810, respectively, for risk factors and femoral plaques. This was statistically significant compared to prediction using traditional risk factors.19
Currently, non-contrast thoracic CT is increasingly used to detect CAC among patients referred for MPI.20 Patients with normal MPI and a CAC of 0 have a better intermediate-term prognosis compared to patients with normal MPI and CAC > 0.13,21,22,23,24,25 The discovery of pre-clinical CAD offers the opportunity to intervene with preventive strategies.26,27,28,29,30
The current study, PESA Study, and the Aragon Workers’ Health studies have demonstrated the frequent presence of atherosclerosis in extracoronary vessel beds in individuals without CAC. Thus, should patients with normal MPI and asymptomatic patients with atherosclerosis in extracoronary beds, but without CAC, be considered for early preventive therapy?14,19,31,32 We currently treat asymptomatic patients using a non-specific risk factor model. This is often implemented using calculators such as the Framingham risk calculator or application-based ACC/AHA 10-year and lifetime risk factor calculator for atherosclerotic cardiovascular disease.33 A patient-centered paradigm based on the presence of atherosclerosis could prove to be more accurate than a population-based risk factor model.34 If such an approach was used, the findings of this study and the PESA and Aragon Workers’ Health studies suggest that the arteries with the earliest onset of atherosclerosis, the ILF and AAo arteries, would be prime targets for assessment.14,19
Limitations
This study is a single-center study involving a selected population of a moderate number of symptomatic patients referred for MPI. It thus represents patients referred for MPI rather than the general population. Also, while all MPI exams were normal in the patients in our study, MPI does not have 100% sensitivity to detect ischemia. Thus, some of our patients may indeed have had symptomatic obstructive CAD.35 Calcium scoring is also an indirect surrogate for atherosclerotic disease and clinical outcomes are not available in our particular patient cohort. While the study raises the possibility that utility of CT as a modality for calcium scoring outside of the heart may be useful for the early diagnosis of atherosclerosis, this approach involves radiation exposure and the challenge of incidental findings.36 Without further work, it is not possible to evaluate the risk/benefit ratio of such a strategy. Other non-CT approaches for diagnosing extracardiac, pre-clinical atherosclerosis might be developed and more fully evaluated in such a strategy. For example, ultrasonography of the iliofemoral arteries, as performed by the PESA and Aragon Workers’ Health study investigators, or potential novel techniques might be considered.
Conclusions
Quantitative assessment of calcification in different vascular beds demonstrates that many patients without known CAD and normal myocardial perfusion, even those with CAC = 0, often have significant atherosclerosis in extracoronary vessel beds. Atherosclerosis appeared first in the iliofemoral bed and then, in an ascending, “south to north fashion,” to other arterial beds. ILF calcification correlates strongly with coronary atherosclerosis. Imaging the extracoronary vascular beds provides a sensitive method for identifying pre-clinical atherosclerosis, prior to the development of CAC. The evaluation of atherosclerosis prior to its onset in the coronary arteries may lead to better understanding of the progression and natural history of atherosclerosis in its long, pre-clinical phase. It also raises the potential for a more patient-centered approach to prevention compared to the traditional use of CAC or targeting preventive measures based on more non-specific risk factors.
New Knowledge Gained
Subclinical extracoronary atherosclerosis was identified in many patients with normal MPI and without coronary calcium. Atherosclerosis most frequently appeared first in iliofemoral bed and typically progressed in an ascending fashion. Assessment of extracoronary calcification creates the opportunity to consider a more patient-specific paradigm than traditional risk factor evaluation models. Further investigation of the utility and practicality of imaging of such a paradigm is warranted.
Abbreviations
- ILF:
-
Iliofemoral
- AAo:
-
Abdominal aorta
- TAo:
-
Thoracic aorta
- CRA:
-
Carotid arteries
- TBC:
-
Total body calcium score
- CA:
-
Coronary arteries
References
Cohn JN. Prevention of cardiovascular disease. Trends Cardiovasc Med 2015;25:436-42.
Wann S. Dying with cardiovascular disease, not of it. Trends Cardiovasc Med 2015;25:443-44.
Naghavi M, Falk E, Hecht HS, et al. From vulnerable plaque to vulnerable patient–Part III: Executive summary of the screening for heart attack prevention and education (SHAPE) task force report. Am J Cardiol 2006;98:2H-15H.
Thompson RC, Allam AH, Lombardi GP, et al. Atherosclerosis across 4000 years of human history: The Horus study of four ancient populations. The Lancet 2013;381:1211-22.
Allam AH, Thompson RC, Wann L, Miyamoto MI, Thomas GS. Computed tomographic assessment of atherosclerosis in ancient egyptian mummies. JAMA 2009;302:2091-94.
Allam AH, Thompson RC, Wann LS, et al. Atherosclerosis in ancient Egyptian mummies: The Horus study. JACC Cardiovasc Imaging 2011;4:315-27.
Bild DE. Multi-ethnic study of atherosclerosis: Objectives and design. Am J Epidemiol 2002;156:871-81.
Demer LL, Tintut Y. Vascular calcification: Pathobiology of a multifaceted disease. Circulation 2008;117:2938-48.
Henzlova MJ, Duvall WL, Einstein AJ, Travin MI, Verberne HJ. ASNC imaging guidelines for SPECT nuclear cardiology procedures: Stress, protocols, and tracers. J Nucl Cardiol 2016;23:606-39.
Hendel RC, Berman DS, Di Carli MF, et al. ACCF/ASNC/ACR/AHA/ASE/SCCT/SCMR/SNM 2009 Appropriate use criteria for cardiac radionuclide imaging: A Report of the American College of Cardiology Foundation Appropriate Use Criteria Task Force, the American Society of Nuclear Cardiology, the American College of Radiology, the American Heart Association, the American Society of Echocardiography, the Society of Cardiovascular Computed Tomography, the Society for Cardiovascular Magnetic Resonance, and the Society of Nuclear Medicine. J Am Coll Cardiol 2009;53:2201-29.
Klocke FJ, Baird MG, Lorell BH, et al. ACC/AHA/ASNC guidelines for the clinical use of cardiac radionuclide imaging–executive summary: A report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (ACC/AHA/ASNC Committee to Revise the 1995 Guidelines for the Clinical Use of Cardiac Radionuclide Imaging). Circulation 2003;108:1404-18.
Agatston AS, Janowitz WR, Hildner FJ, Zusmer NR, Viamonte JM, Detrano R. Quantification of coronary artery calcium using ultrafast computed tomography. J Am Coll Cardiol 1990;15:827-32.
Allison MA, Hsi S, Wassel CL, et al. Calcified atherosclerosis in different vascular beds and the risk of mortality. Arterioscler Thromb Vasc Biol 2012;32:140-46.
Fernandez-Friera L, Penalvo JL, Fernandez-Ortiz A, et al. Prevalence, vascular distribution, and multiterritorial extent of subclinical atherosclerosis in a middle-aged cohort: The PESA (progression of early subclinical atherosclerosis) study. Circulation 2015;131:2104-13.
Allison MA, Criqui MH, Wright CM. Patterns and risk factors for systemic calcified atherosclerosis. Arterioscler Thromb Vasc Biol 2004;24:331-36.
Leherbauer L, Sonneck-Koenne C, Zakavi SR, et al. Coronary artery calcium (CAC) score—A prognostic tool in coronary artery disease? Iran J Nucl Med 2011;19:46-51.
Chang SM, Nabi F, Xu J, et al. The coronary artery calcium score and stress myocardial perfusion imaging provide independent and complementary prediction of cardiac risk. J Am Coll Cardiol 2009;54:1872-82.
Chang SM, Nabi F, Xu J, et al. Value of CACS compared with ETT and myocardial perfusion imaging for predicting long-term cardiac outcome in asymptomatic and symptomatic patients at low risk for coronary disease: Clinical implications in a multimodality imaging world. JACC Cardiovasc Imaging 2015;8:134-44.
Laclaustra M, Casasnovas JA, Fernández-Ortiz A, et al. Femoral and carotid subclinical atherosclerosis association with risk factors and coronary calcium: The AWHS study. J Am Coll Cardiol 2016;67:1263-74.
Detrano R, Guerci AD, Carr JJ, et al. Coronary calcium as a predictor of coronary events in four racial or ethnic groups. N Engl J Med 2008;358:1336-45.
Bos D, Ikram MA, Isaacs A, et al. Genetic loci for coronary calcification and serum lipids relate to aortic and carotid calcification. Circ Cardiovasc Genet 2013;6:47-53.
Hacker M, Becker C. The incremental value of coronary artery calcium scores to myocardial single photon emission computer tomography in risk assessment. J Nucl Cardiol 2011;18:700-11 quiz 12-6.
Schepis T, Gaemperli O, Koepfli P, et al. Added value of coronary artery calcium score as an adjunct to gated SPECT for the evaluation of coronary artery disease in an intermediate-risk population. J Nucl Med 2007;48:1424-30.
Thompson RC, McGhie AI, Moser KW, et al. Clinical utility of coronary calcium scoring after nonischemic myocardial perfusion imaging. J Nucl Cardiol 2005;12:392-400.
Wong ND, Gransar H, Shaw L, et al. Thoracic aortic calcium versus coronary artery calcium for the prediction of coronary heart disease and cardiovascular disease events. JACC Cardiovasc Imaging 2009;2:319-26.
Arad Y, Spadaro LA, Roth M, Newstein D, Guerci AD. Treatment of asymptomatic adults with elevated coronary calcium scores with atorvastatin, vitamin C, and vitamin E: The St. Francis Heart Study randomized clinical trial. J Am Coll Cardiol 2005;46:166-72.
Hwang IC, Jeon JY, Kim Y, et al. Statin therapy is associated with lower all-cause mortality in patients with non-obstructive coronary artery disease. Atherosclerosis 2015;239:335-42.
Rozanski A, Gransar H, Shaw LJ, et al. Impact of coronary artery calcium scanning on coronary risk factors and downstream testing the EISNER (early identification of subclinical atherosclerosis by noninvasive imaging research) prospective randomized trial. J Am Coll Cardiol 2011;57:1622-32.
Schenker MP, Dorbala S, Hong EC, et al. Interrelation of coronary calcification, myocardial ischemia, and outcomes in patients with intermediate likelihood of coronary artery disease: A combined positron emission tomography/computed tomography study. Circulation 2008;117:1693-700.
Wayhs R, Zelinger A, Raggi P. High coronary artery calcium scores pose an extremely elevated risk for hard events. J Am Coll Cardiol 2002;39:225-30.
Shah PK. Screening asymptomatic subjects for subclinical atherosclerosis: can we, does it matter, and should we? J Am Coll Cardiol 2010;56:98-105.
Shah PK. Rapid detection of subclinical atherosclerosis: Potential implications for primary prevention in LMIC. Glob Heart 2013;8:91-3.
Stone NJ, Robinson JG, Lichtenstein AH, et al. 2013 ACC/AHA Guideline on the treatment of blood cholesterol to reduce atherosclerotic cardiovascular risk in adults. J Am Coll Cardiol 2014;63:2889-934.
Weintraub WS, Daniels SR, Burke LE, et al. Value of primordial and primary prevention for cardiovascular disease: A policy statement from the American Heart Association. Circulation 2011;124:967-90.
Leppo JA. Comparison of pharmacologic stress agents. J Nucl Cardiol 1996;3:S22-26.
Furtado CD, Aguirre DA, Sirlin CB, et al. Whole-body CT screening: Spectrum of findings and recommendations in 1192 patients. Radiology 2005;237:385-94.
Acknowledgements
We thank statistician Kevin Kennedy, M.S. of the Saint Luke’s Mid America Heart Institute, Kansas City, MO, for his expert assistance.
Disclosure
None.
Author information
Authors and Affiliations
Consortia
Corresponding author
Additional information
The authors of this article have provided a PowerPoint file, available for download at SpringerLink, which summarises the contents of the paper and is free for re-use at meetings and presentations. Search for the article DOI on SpringerLink.com.
Funding Outside funding was not used for data collection, analysis, or manuscript preparation.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Online Appendix Figure 1
Mean age of men and women without arterial calcification, one-to-two vascular beds with calcification, and calcification of greater than three vascular beds (TIFF 1579 kb)
Online Appendix Figure 2
(A) Prevalence of calcification within three arterial beds based on the presence of calcium within the coronary and/or carotid artery vascular beds. (B) Mean calcium score within three arterial beds based on the presence of calcium within the coronary and/or carotid artery vascular beds. Values are mean ± SE. P < 0.001 between three groups (TIFF 3199 kb)
Rights and permissions
About this article
Cite this article
Allam, A.H.A., Thompson, R.C., Eskander, M.A. et al. Is coronary calcium scoring too late? Total body arterial calcium burden in patients without known CAD and normal MPI. J. Nucl. Cardiol. 25, 1990–1998 (2018). https://doi.org/10.1007/s12350-017-0925-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12350-017-0925-9