Abstract
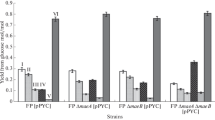
Malic enzymes catalyze the reversible oxidative decarboxylation of L-malate using NAD(P)+ as a cofactor. NADP-dependent malic enzyme (MaeB) from Escherichia coli MG1655 was expressed and purified as a fusion protein. The molecular weight of MaeB was about 83 kDa, as determined by SDS-PAGE. The recombinant MaeB showed a maximum activity at pH 7.8 and 46°C. MaeB activity was dependent on the presence of Mn2+ but was strongly inhibited by Zn2+. In order to understand the physiological roles, recombinant E. coli strains (icd NADP/ΔmaeB and icd NAD/ΔmaeB) containing NADP-dependent isocitrate dehydrogenase (IDH), or engineered NAD-dependent IDH with the deletion of the maeB gene, were constructed using homologous recombination. During growth on acetate, icd NAD/ΔmaeB grew poorly, having a growth rate only 60% that of the wild-type strain (icd NADP). Furthermore, icd NADP/ΔmaeB exhibited a 2-fold greater adaptability to acetate than icd NAD/ΔmaeB, which may be explained by more NADPH production for biosynthesis in icd NADP/ΔmaeB due to its NADP-dependent IDH. These results indicated that MaeB was important for NADPH production for bacterial growth on acetate. We also observed that MaeB activity was significantly enhanced (7.83-fold) in icd NAD, which was about 3-fold higher than that in icd NADP, when switching from glucose to acetate. The marked increase of MaeB activity was probably induced by the shortage of NADPH in icd NAD. Evidently, MaeB contributed to the NADPH generation needed for bacterial growth on two carbon compounds.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Bartolucci, S., R. Rella, A. Guagliardi, C.A. Raia, A. Gambacorta, M. De Rosa, and M. Rossi. 1987. Malic enzyme from archaebacterium Sulfolobus solfataricus. Purification, structure, and kinetic properties. J. Biol. Chem. 262, 7725–7731.
Bologna, F.P., C.S. Andreo, and M.F. Drincovich. 2007. Escherichia coli malic enzymes: Two isoforms with substantial differences in kinetic properties, metabolic regulation, and structure. J. Bacteriol. 189, 5937–5946.
Chang, G.G. and L. Tong. 2003. Structure and function of malic enzymes, a new class of oxidative decarboxylases. Biochemistry 42, 12721–12733.
Datsenko, K.A. and B.L. Wanner. 2000. One-step inactivation of chromosomal genes in Escherichia coli K-12 using PCR products. Proc. Natl. Acad. Sci. USA 97, 6640–6645.
Detarsio, E., C.S. Andreo, and M.F. Drincovich. 2004. Basic residues play key roles in catalysis and NADP(+)-specificity in maize (Zea mays L.) photosynthetic NADP(+)-dependent malic enzyme. Biochem. J. 382, 1025–1030.
Doležal, P., S. Vaňáčová, J. Tachezy, and I. Hrdý. 2004. Malic enzymes of Trichomonas vaginalis: two enzyme families, two distinct origins. Gene 329, 81–92.
Fukuda, W., Y.S. Ismail, T. Fukui, H. Atomi, and T. Imanaka. 2005. Characterization of an archaeal malic enzyme from the hyperthermophilic archaeon Thermococcus kodakaraensis KOD1. Archaea 1, 293–301.
Garrido-Pertierra, A., C. Martinez Marcos, M. Martin Fernandez, and M. Ruiz-Amil. 1983. Properties and function of malate enzyme from Pseudomonas putida. Biochimie. 65, 629–635.
Gourdon, P., M.F. Baucher, N.D. Lindley, and A. Guyonvarch. 2000. Cloning of the malic enzyme gene from Corynebacterium glutamicum and role of the enzyme in lactate metabolism. Appl. Environ. Microbiol. 66, 2981–2987.
Johnson, J.L. and R.J. Brooker. 1999. A K319N/E325Q double mutant of the lactose permease cotransports H+ with lactose — Implications for a proposed mechanism of H+ lactose symport. J. Biol. Chem. 274, 4074–4081.
Kobayashi, K., S. Doi, S. Negoro, I. Urabe, and H. Okada. 1989. Structure and properties of malic enzyme from Bacillus stearothermophilus. J. Biol. Chem. 264, 3200–3205.
Lamed, R. and J.G. Zeikus. 1981. Thermostable, ammonium-activated malic enzyme of Clostridium thermocellum. Biochim. Biophys. Acta. 660, 251–255.
Lance, C. and P. Rustin. 1984. The central role of malate in plant metabolism. Physiol. Veg. 22, 625–641.
Lerondel, G., T. Doan, N. Zamboni, U. Sauer, and S. Aymerich. 2006. YtsJ has the major physiological role of the four paralogous malic enzyme isoforms in Bacillus subtilis. J. Bacteriol. 188, 4727–4736.
Mitsch, M.J., R.T. Voegele, A. Cowie, M. Osteras, and T.M. Finan. 1998. Chimeric structure of the NAD(P)+ — and NADP+- dependent malic enzymes of Rhizobium (Sinorhizobium) meliloti. J. Biol. Chem. 273, 9330–9336.
Murai, T., M. Tokushige, J. Nagai, and H. Katsuki. 1971. Physiological functions of NAD- and NADP-linked malic enzymes in Escherichia coli. Biochem. Biophys. Res. Commum. 43, 875–881.
Neidhardt, F.C., P.L. Bloch, and D.F. Smith. 1974. Culture medium for enterobacteria. J. Bacteriol. 119, 736–747.
Oh, M.K., L. Rohlin, K.C. Kao, and J.C. Liao. 2002. Global expression profiling of acetate-grown Escherichia coli. J. Biol. Chem. 277, 13175–13183.
Sender, P.D., M.G. Martin, S. Peiru, and C. Magni. 2004. Characterization of an oxaloacetate decarboxylase that belongs to the malic enzyme family. FEBS Lett. 570, 217–222.
Seo, S.H., C.H. Rhee, and H.D. Park. 2007. Degradation of malic acid by Issatchenkia orientalis KMBL 5774, an acidophilic yeast strain isolated from Korean grape wine pomace. J. Microbiol. 45, 521–527.
Song, Y.D., J.P. Wynn, Y.H. Li, D. Grantham, and C. Ratledge. 2001. A pre-genetic study of the isoforms of malic enzyme associated with lipid accumulation in Mucor circinelloides. Microbiology 147, 1507–1515.
Spencer, C.C., M. Bertrand, M. Travisano, and M. Doebeli. 2007. Adaptive diversification in genes that regulate resource use in Escherichia coli. PLoS Genet. 3, 83–88.
Stols, L. and M.I. Donnelly. 1997. Production of succinic acid through overexpression of NAD(+)-dependent malic enzyme in an Escherichia coli mutant. Appl. Environ. Microbiol. 63, 2695–2701.
Stueland, C.S., K. Gorden, and D.C. LaPorte. 1988. The isocitrate dehydrogenase phosphorylation cycle. Identification of the primary rate-limiting step. J. Biol. Chem. 263, 19475–19479.
Suye, S.I., Y. Okada, A. Funada, M. Kawagoe, and S. Inuta. 1992. Purification and properties of malic enzyme from Pseudomonas diminuta IFO-13182. J. Ferment. Bioeng. 73, 343–347.
Voegele, R.T., M.J. Mitsch, and T.M. Finan. 1999. Characterization of two members of a novel malic enzyme class. Biochem. Biophys. Acta. 1432, 275–285.
Wheeler, M.C.G., M.A. Tronconi, M.F. Drincovich, C.S. Andreo, U.I. Flugge, and V.G. Maurino. 2005. A comprehensive analysis of the NADP-malic enzyme gene family of arabidopsis. Plant Physiol. 139, 39–51.
Zhu, G., G.B. Golding, and A.M. Dean. 2005. The selective cause of an ancient adaptation. Science 307, 1279–1282.
Author information
Authors and Affiliations
Corresponding author
Additional information
These authors contributed equally to this work.
Rights and permissions
About this article
Cite this article
Wang, B., Wang, P., Zheng, E. et al. Biochemical properties and physiological roles of NADP-dependent malic enzyme in Escherichia coli . J Microbiol. 49, 797–802 (2011). https://doi.org/10.1007/s12275-011-0487-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12275-011-0487-5