Abstract
Myocardial infarction (MI) causes most of the mortality worldwide. Coronary obstruction–caused myocardial ischemic injury leads to permanent loss of the myocardium. Subsequent compensatory myocardial remodeling and heart failure would result in arrhythmia and even sudden death. The molecular mechanisms of these pathological processes remain to be thoroughly revealed. Circular RNAs (circRNAs) are special types of non-coding RNAs which can durably regulate gene expression and modulate cell fate. They had been reported to mediate ischemic myocardial injury and myocardial remodeling. circRNAs can be loaded into extracellular vesicles and released into extracellular space. More recently, it was uncovered that the extracellular circRNAs can regulate intercellular communications, similar to “first messengers.” The cross-talk mediated by extracellular circRNAs had been demonstrated to play important roles in pathological processes. Here, we would like to review the modulation of extracellular circRNAs in ischemic myocardial injury and myocardial remodeling. We believe the extracellular circRNAs can bring new strategies of MI treatment.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
MI is one of the most lethal diseases. Under continuous myocardial ischemia, the cardiomyocytes would die from hypoxia and nutritional deficiency. Losing excess of cardiomyocytes causes cardiac arrest [1]. So far, thrombolysis is the only efficient therapy to treat MI. However, reperfusion of the occluded coronary artery can induce ischemia/reperfusion (I/R) damage of the myocardium, which further increases the mortality [2]. Even though the acute crisis described above was resolved, the patients would be still in danger during the process of myocardial remodeling: The necrotic myocardium would trigger inflammation, which might cause further cardiac damage. Moreover, the dead cardiomyocytes cannot be supplemented, because mature mammalian cardiomyocytes have negligible proliferative ability. The cardiac fibroblasts without contraction would replace the lost myocardium and form scar tissue. Cardiac fibrosis can maintain structural integrity of the heart, while excessive fibrosis can also lead to decrease of heart function. More seriously, cardiac fibrosis could cause arrhythmia, even sudden death [3, 4]. On the other hand, the surviving cardiomyocytes would become hypertrophic to increase their contractive ability and adapt to the hemodynamic change. Continuous pressure overload induces death of the hypertrophic cardiomyocytes, then gives rise to further myocardial fibrosis, dilated ventricle, and eventual heart failure (HF). At the terminal stage of HF, the patients have rather weak cardiac function and may die from retard of circulation [5, 6]. Therefore, to alleviate ischemic myocardial injury and the following myocardial remodeling, the intracellular signal pathways of programmed cardiomyocyte death, cardiomyocyte hypertrophy, and cardiac fibroblast proliferation should be illuminated.
circRNA is a type of special non-coding RNA. circRNAs are covalently closed single-stranded RNA circles which are generated through pre-mRNA back-splicing or intronic lariats escaping from degradation. circRNAs have long half-life. They can act as microRNA (miRNA) sponge, protein sponge, protein scaffold, and transcriptional activator to lastingly regulate gene expression [7]. They not only function in parental cells, but also exist in extracellular space to mediate cell cross-talk. In this review, we purposed to introduce the significant extracellular circRNAs involved in MI and myocardial remodeling, as well as coronary atherosclerosis, the primary cause of MI. Their molecular mechanisms, especially the potential roles in intercellular communication, were summarized.
Cardiovascular circRNAs Mediate Progression of MI and Cardiac Remodeling In Situ
Since abundant circRNAs had been characterized in heart by RNA deep sequencing techniques [8], critical circRNAs, which regulate the processes of ischemic myocardial injury and cardiac remodeling, are gradually identified: In the period of pre-MI, circRNAs affect the coronary atherosclerosis via regulating viability of vascular smooth muscle cells and endothelial cells. For instance, circANRIL retards ribosome maturation by binding pescadillo homologue 1. Overexpression of circANRIL inhibits proliferation and causes apoptosis of vascular smooth muscle cells [9]. circGNAQ acts as a miR-146a-5p-sponge to inhibit endothelial cell senescence [10]. circRNAs were reported to affect myocardial injury by regulating various types of programmed cardiomyocyte death, including apoptosis, necrosis, and autophagy. For instance, up-regulation of circSLC8A1 in MI and I/R promotes cardiomyocyte apoptosis by restraining miR-133a [11]. circRNA ACR activates the expression of PINK1 to suppress cardiomyocyte autophagy [12]. circRNA CNEACR protects cardiomyocyte from necrosis by modulating HDAC7/Foxa2/RIPK3 axis [13]. In addition to directly affecting cardiomyocytes, circRNAs including circFndc3b can also protect myocardium from ischemic injury by promoting angiogenesis in the infarcted area. Mechanistically, circFndc3b binds to FUS to increase the expression of VEGF, resulting in enhanced viability of cardiac endothelial cells [14]. Some circRNAs were shown to involve in post-MI cardiac remodeling. For example, circSLC8A1 not only promotes cardiomyocyte apoptosis but also induces cardiac hypertrophy and fibrosis [11, 15]. circRNA HRCR controls cardiomyocyte hypertrophy by targeting miR-223 [16]. circYAP can block actin polymerization in cardiomyocyte and cardiac fibroblast by interacting with TMP4 and ACTG, leading to inhibition of cardiac hypertrophy and fibrosis [17]. circNlgn is translatable and encodes a nuclear peptide Nlgn173. Nlgn173 decreases cardiomyocyte survival and increases fibroblast proliferation via activating transcription of ING4 and SGK3 [18]. In summary, most of the significant cardiac circRNAs, including the above-mentioned ones, are expressed in the cytoplasm of cardiac cells. They can control intracellular homeostasis of parental cells by regulating expression of genes involved in cell cycle, metabolism, motility, morphology, death, etc. However, when the cardiac cells were under abnormal conditions, dysregulation of these circRNAs would cause aberrant expression or activity of downstream molecules, resulting in cardiac injury and cardiac remodeling.
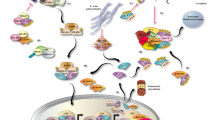
Extracellular Vesicle–Delivered circRNAs Stably Exist in Extracellular Fluids and Mediate Cell Communications
Comparing with linear RNAs such as mRNAs, long non-coding RNAs (lncRNAs), and PIWI-interacting RNAs (piRNAs), circRNAs can tolerate degradation by exonuclease and be retained for a prolonged time [19]. Therefore, circRNAs are found to be stable and abundant in extracellular space [20]. Until now, they have been widely detected in body fluid including plasma [21], saliva [22], gastric juice [23], bile [24], seminal fluid [25], urine [26], and milk [27]. It is clear that circRNAs are delivered into extracellular fluids by extracellular vesicles (EVs). EVs are extracellular lipid bilayer vesicles and typically classified into the subtypes including exosomes, microvesicles, and apoptotic bodies according to their sizes [28,29,30]. Exosomes are the smallest type of EVs with diameters ranging 30 to 100 nm. They are initially formed as intraluminal vesicles in multivesicular bodies. Fusion of multivesicular bodies with the plasma membrane makes intraluminal vesicles be released into extracellular space and transformed into exosomes. Exosomes are the most widely studied and well-understood EVs. They are synthesized by all types of cells. Exosomes contain cargos of parental molecules, such as proteins, lipids, and nucleic acids. Functionally, exosome can regulate intercellular communication. The exosomes distributed in extracellular fluid are absorbed by recipient cells via endocytosis or fusing with plasma membrane of recipient cells. The exosome cargos are together delivered into the cytoplasm of recipient cells and play regulatory roles [31]. Most of the extracellular circRNAs were reported to be localized in exosomes. Comparing with other linear types of non-coding RNAs which exist in EVs and regulate cell communication [32,33,34], circRNAs have a significant advantage of higher stability. The ratio of circRNA level to linear RNA level in exosomes was extremely higher than that in parental cells, suggesting the predominant roles of exosomal circRNAs in intercellular cross-talk [20]. Although the exosomal circRNAs derive from parental cells and show tissue or cell-type specificity, landscape of circRNAs in exosomes is different from that in parental cells, implying that there might be some mechanisms of selectively sorting circRNAs into exosomes [20]. Until now, the mechanisms are still unclear. It had been reported that transport of other types of non-coding RNAs into exosomes was modulated by RNA-binding proteins (RBPs). The RBPs recognize specific motifs in RNAs and facilitate RNA sorting [35,36,37,38]. RBP is also shown to be involved in packing circRNAs into exosomes. For instance, hnRNPA2B1 is the RBP which can transport RNAs into exosomes. It binds to circNEIL3 in glioblastoma multiforme, which is a type of glioma. Knockdown of hnRNPA2B1 in glioblastoma multiforme resulted in up-regulation of circNEIL3 in cells and down-regulation of circNEIL3 in exosomes [39]. However, it still remains to be widely verified the roles of RBPs in sorting exosomal circRNAs. In some cases, circRNAs were more highly enriched in EVs than parental cells [20]. The variety and abundance of cargos in EVs are also affected by physiological and pathological conditions. Cell stress was reported to enhance exosome secretion in a p53-dependent manner [40]. The level of extracellular circRNAs was obviously changed in some diseases. Therefore, the dysregulated circRNAs in exosomes can serve as a new type of biomarkers in disease diagnosis [41, 42]. Actually, EV circRNAs have already been widely reported to affect the progression of diseases, including various types of cancers [43], diabetes, diabetic complications [44, 45], intervertebral disc degeneration (IDD) [46], and stroke [47, 48]. The type of recipient cells can either be the same with or different from the parental cells of EVs. Mechanistically, EV circRNAs primarily function as miRNA sponge in recipient cells. The internalized EV circRNAs can target complementary cytoplasmic miRNAs in recipient cells, resulting in destabilization or inactivation of these miRNAs. The expression of miRNA-targeted genes can be up-regulated by EV circRNAs [44,45,46, 48]. EV circRNAs can also bind to proteins in recipient cells, affecting the protein interaction, stability, and downstream regulatory network [39, 49]. In addition, EV circRNAs have also been reported to interact with specific phosphorylated site in protein and maintain the protein phosphorylation, activating the downstream signal pathway [50]. In short, circulating EV circRNAs are internalized by recipient cells and regulate proliferation, metabolism, migration, and death of the recipient cells by affecting gene expression, protein interaction, and signal transduction (Fig. 1). It can be concluded that EV-delivered circRNAs function as a new type of “first messenger.” For example, the cross-talk between vascular pericytes and endothelial cells can mediate vascular homeostasis in a cPWWP2A-dependent way. Uptake of pericyte-derived cPWWP2A by endothelial cells prevents vascular dysfunction by inhibiting the activity of miR-579. The interaction between cPWWP2A and miR-579 results in up-regulation of angiopoietin 1, occludin, and SIRT1 in endothelial cells [44]. Glioma-derived circNEIS3 can promote macrophage infiltration into the tumor microenvironment and immunosuppressive polarization of macrophage, leading to gliomagenesis. circNEIS3 is highly expressed in glioma and can be packaged into exosomes. Exosomal circNEIS3s are internalized by macrophages in the tumor microenvironment and bind to IGF2BP3 in macrophages, inhibiting ubiquitin-dependent degradation of IGF2BP3 [39]. Glioma-derived exosomal circGLIS3 was also reported to promote glioma progression by mediating the communication between glioma and endothelial cell. circGLIS3 can cover the phosphorylated site T567 in Ezrin and maintain phosphorylation level of this site in endothelial cells, promoting angiogenesis of glioma [50].
General regulatory mechanism of extracellular circRNAs. circRNAs are transported into EVs and released into extracellular space by parental cells. The EVs distributed in extracellular space are internalized by recipient cells via endocytosis or fusing with plasma membrane of recipient cells. The internalized circRNAs can either target miRNAs to regulate the expression of downstream genes, or target proteins to affect interaction or stability of proteins in recipient cells
Potency of Differentially Changed Extracellular circRNAs in MI
Accumulated studies have uncovered the expression profiles of circRNAs in human and murine heart samples with ischemic cardiac injury or cardiac remodeling [8, 51,52,53]. To better explore the underlying mechanisms of circRNAs in heart disease, the extracellular circRNAs in peripheral fluid had also been studied (Fig. 2).
In 2016, Vausort et al. firstly reported a myocardial infarction–associated circular RNA (MICRA), which is transcribed from zinc finger protein 609 (ZNF609) gene. They analyzed peripheral blood samples of 642 patients with acute myocardial infarction (AMI). The level of MICRA was significantly reduced in AMI samples and evaluated to highly correlated with LV dysfunction [54]. The following researches further thoroughly tested circRNAs in blood samples of MI patients. In the study performed by Yang et al., the expression of circRNAs in peripheral blood of 4 AMI patients and 4 mild coronary artery stenosis patients were detected by microarray. Comparing with mild samples, 64 circRNAs were up-regulated and 90 circRNAs were down-regulated in AMI samples, respectively. Overall, down-regulated circRNAs were more frequently detected. Among the down-regulated circRNAs, circRNA_068655, circRNA_104761, and circRNA_104765 were screened out to be potential biomarkers for AMI diagnosis due to their stable expression in blood and significant decrease in AMI samples. Especially, the level of circRNA_104761 (a circular transcript of UBAP2 gene) showed more significant decrease in larger scale of blood samples. It was evaluated to have high sensitivity and specificity for AMI diagnosis. Moreover, circRNA_104761 is localized in cytoplasm of AC16 human cardiomyocyte and protects cardiomyocyte from hypoxia-induced apoptosis. The expression of circRNA_104761 in cardiomyocyte can be depressed by hypoxia [55]. Zhao et al. analyzed the circRNAs in coronary blood samples of 3 AMI patients and 3 participants without AMI. The expression profiles of circRNAs in AMI samples are quite different from that in control samples. Altogether, 89 up-regulated and 53 down-regulated circRNAs in AMI samples were identified [56]. Another circRNA profiling by microarray identified 13,804 circRNAs in plasma of 3 AMI patients and 3 healthy subjects. In total, 650 circRNAs were differentially expressed in AMI plasma and control plasma. Among these circRNAs, 535 circRNAs were up-regulated and 115 circRNAs were down-regulated in AMI samples [57]. These profiles of circRNAs in AMI blood samples presented by different groups were not consistent. It might be caused by individual difference, sampling locations, sample size, diagnostic criteria, RNA quality, and detection methods. Hemocytes in whole blood samples would also affect the result. Nevertheless, the difference between circRNAs in fluids of AMI patients and control fluids was likely to depend on dysregulation of EV-delivered circRNAs. Ge et al. specifically isolated EVs from mice hearts which were subjected to I/R. It was observed that the amount of EVs in heart was increased at the first 24-h post-reperfusion. The result of RNA sequencing identified 119 down-regulated and 66 up-regulated circRNAs in EVs of I/R heart [58].
Potency of Differentially Changed Extracellular circRNAs in Cardiac Remodeling
The circRNAs in blood samples of patients with cardiac hypertrophy were also profiled. Guo et al. analyzed the transcriptomes in plasma of 15 patients with hypertrophic cardiomyopathy (HCM) and 7 healthy controls. Comparing with controls, there are 391 circRNAs being up-regulated and 384 circRNAs being down-regulated in HCM samples [59]. Sonnenschein et al. screened 3 circRNAs which are abundant in peripheral blood, including circDNAJC6, circMBOAT2, and circTMEM56. Their levels in serum of 64 patients with HCM and 3 healthy individuals were compared. The result showed that all these three circRNAs were decreased in HCM samples. Remarkably, the level of these circRNAs in serum was negatively correlated with left ventricular outflow tract gradient and interventricular septum thickness in HCM patients [60]. It was noteworthy that circDNAJC6 and circMBOAT2 had been reported in more previous studies. Down-regulation of circDNAJC6 in heart samples with dilated cardiomyopathy (DCM) had been observed [53]. In addition, the level of circMBOAT2 in plasma was also shown to significantly decrease after cardiac endurance training, indicating the correlation with physical exercise parameters. CircMBOAT2 is expressed in types of cardiac cells, including endothelial cells, smooth muscle cells, induced pluripotency stem cell–derived cardiomyocytes, and cardiac fibroblasts, but mostly enriched in cardiac fibroblasts [61]. Maybe circMBOAT2 was involved in cardiac stress adaption. The exact function and molecular mechanism of circDNAJC6 and circMBOAT2 remain to be explored.
Persistent atrial fibrillation (PsAF) is mainly caused by cardiac fibrosis. To explore the regulatory roles of circRNAs in persistent atrial fibrillation, Gao et al. tested circRNA expression in peripheral blood plasma of paroxysmal atrial fibrillation and PsAF patients by microarray. In total, 92 differently expressed circRNAs in PsAF were identified. After further validation, 7 circRNAs were selected as the significantly changed candidates. According to functional prediction, 5 of these 7 circRNAs were annotated to involve in MAPK and TGF-β signaling pathways, which are critical in progression of cardiac fibrosis. Among the 5 circRNAs, the level of hsa_circ_0004104 in plasma of PsAF patients were significantly down-regulated and negatively correlated with TGF-β1 level. In addition, hsa_circ_0004104 was predicted to target miRNAs involved in cardiac fibrosis, implying its cardiac fibrosis-related function [62].
Dilated cardiomyopathy (DCM) is a transitional stage from MI to HF. Costa et al. analyzed the expression profiles of circRNAs in plasma samples of 130 subjects, including patients with idiopathic DCM, ischemic DCM or familial DCM, as well as healthy controls. The level of hsa_circ_0089762 was specifically elevated in ischemic DCM, which discriminated ischemic DCM from other types of DCM and healthy individuals. Moreover, the plasmic level of hsa_circ_0089762 was negatively associated with diastolic blood pressure and left ventricular ejection fraction. Prediction showed that hsa_circ_0089762 possibly targeted numerous miRNAs, including hsa-miR-21, hsa-miR-183, hsa-miR-361-3p, hsa-miR-384, hsa-miR-873, hsa-miR-938, hsa-miR-1249, and hsa-miR-1283, as well as 128 proteins related to actin cytoskeleton, endoplasmic reticulum-nuclei signaling pathways and pre- and post-Golgi vesicle transportation [63].
In terminal HF stage, the circulating circRNAs also showed aberrant expression. The study completed by Han et al. identified 56 significantly changed circRNAs in peripheral blood of patients with HF. Among these circRNAs, hsa_circ_0097435 was most obviously up-regulated in peripheral blood as well as plasmic exosomes of HF patients. Functionally, hsa_circ_0097435 could promote cardiomyocyte apoptosis, suggesting its contribution to cardiac damage and HF [64]. Another circRNA microarrays analysis also depicted the expression of circRNAs in plasma of three HF patients and three healthy controls. Four hundred seventy-seven up-regulated and 219 down-regulated circRNAs were detected in HF samples. Among these circRNAs, the increase of hsa_circ_0112085, hsa_circ_0062960, hsa_circ_0053919, and hsa_circ_0014010 was significant. Moreover, the level of hsa_circ_0062960 was highly correlated with that of B-type natriuretic peptide (BNP) [65].
Extracellular circRNAs Can Transmit Pro-apoptotic Signal Among Cardiomyocytes
Dysregulation of extracellular circRNAs in MI and cardiac remodeling suggested that EV-delivered circRNAs might be involved in the pathological progression. It is interesting that the pro-apoptotic circRNAs in cardiomyocytes were consistently and remarkably increased in extracellular fluid of MI and cardiac remodeling patients, such as circRNA_104761 [52], CDR1as [66, 67], circPostn [68], circACAP2 [69], and MFACR [70, 71]. Perhaps these EV-delivered apoptotic circRNAs could be released from cardiomyocytes under apoptotic stress and internalized by the neighboring cardiomyocytes. For instance, circSKA3 was reported to promote apoptosis of AC16 cardiomyocyte induced by doxorubicin (DOX). The expression of circSKA3 was not only increased in DOX-treated cardiomyocytes but also increased in exosomes derived from the DOX-treated cardiomyocytes. The exosomes delivered by DOX-treated cardiomyocytes could be internalized by other cardiomyocytes and increase the level of circSKA3 in recipient cardiomyocytes [72]. It has been known that apoptotic cells could suppress inflammation in surrounding tissue via releasing some metabolites [73]. These extracellular pro-apoptotic circRNAs might induce apoptosis of recipient cardiomyocytes under ischemia and prevent necrosis, as well as the subsequent inflammation. It was also possible that these extracellular circRNAs convey signaling to other types of cells rather than cardiomyocytes during MI. Uptake of these circRNAs by cardiac cells, such as endothelial cells and fibroblast, might have an effect independent of apoptosis. In brief, the parental cells and recipient cells of the EV-delivered circRNAs remain to be explored. It is exciting that intercellular regulatory roles of several EV-delivered circRNAs in MI and cardiac remodeling have been uncovered (Fig. 3). The detail will be talked about in the following sections.
Cardiac Protective Potency of Mesenchymal Stem Cell–Derived EV-circRNAs
Exosomes delivered by mesenchymal stem cells (MSC-EV) have been demonstrated to have function of cardiac repairing. Utilizing MSC-EVs could reduce myocardial infarction area and cardiac remodeling level caused by cardiac ischemia [74, 75]. However, the detailed mechanisms were unclear. Zhang et al. showed that the protective effect of human umbilical cord mesenchymal stromal cell-derived extracellular vesicles (HuMSC-EVs) potentially depended on circRNAs in HuMSC-EVs. Incubation of AC16 cardiomyocytes exposed to hypoxia/reoxygenation (H/R) with HuMSC-EVs could significantly increase viability of cardiomyocytes. By means of RNA-seq, 66 differentially expressed circRNAs were detected in HuMSC-EV-incubated cardiomyocytes, comparing with control cardiomyocytes. Further validation indicated that 7 of 10 most significantly increased circRNAs were also abundant in HuMSC-EVs. GO analysis showed that the up-regulated circRNAs might be involved in the biological processes of cellular response to hypoxia, decreased oxygen levels and oxygen levels. KEGG analysis suggested that they possibly target genes associated with several biological pathways, including vascular endothelial growth factor (VEGF), long-term potentiation and glucagon signaling pathway. Moreover, the top 10 up-regulated circRNAs were predicted to target miRNAs involved in VEGF signaling pathway [76]. These results provided potential evidence that circRNAs in MSC-EVs were cardiac protective.
It was also reported that human umbilical cord mesenchymal stem cells–derived exosomes (UMSC-exos) prevent pyroptosis and repair ischemic muscle injury by releasing circHIPK3. The result of circRNA-seq showed that the expression level of circHIPK3 was reduced in skeletal muscle tissue of mice lower limb with acute ischemia. Injection of UMSC-exo can increase circHIPK3 level and promote recovery of injured muscle. Mechanistically, UMSC-exo-delivered circHIPK3 were internalized by skeletal muscle cells, then circHIPK3 can inhibit pyroptosis and inflammation via regulating miR-421/FOXO3a axis [77]. Although the effect of UMSC-exo-delivered circHIPK3 has not been directly tested in ischemic myocardial injury, cardiomyocyte-delivered circHIPK3 has been reported to promote angiogenesis activity of endothelial cells (the detail will be discussed in the next section) [78, 79]. It is possible that MSC-EVs also protected myocardium from ischemic injury via circHIPK3.
Cardiomyocyte Regulates Endothelial Cell via Exosomal circHIPK3
EVs mediate communication between cardiomyocyte and cardiac endothelial cell. Cardiomyocyte-derived EVs were discovered to ameliorate MI by inducing angiogenesis and recovering microcirculation in infarcted area. They can be internalized by cardiac endothelial cells and promote proliferation of the recipient endothelial cells [80]. It was suggested that some molecules within EVs might play important roles. Intracellular circHIPK3 had been demonstrated to regulate proliferation of endothelial cells and neonatal cardiomyocytes [81, 82]. The expression of circHIPK3 in cardiomyocytes could be induced by oxidative stress [83]. CircHIPK3 was also proved to act as a messenger, which could transmit angiogenesis signal from cardiomyocyte to endothelial cell post-MI. Uptake of neonatal mouse cardiomyocyte-derived exosomal circHIPK3 would increase viability of cardiac microvascular endothelial cells (CMVECs). Cardiomyocytes with hypoxic stimulation–derived exosomes showed higher circHIPK3 level and stronger protective effect on recipient CMVECs. Increase of circHIPK3 in CMVECs via internalizing cardiomyocyte-derived exosomes could inhibit miR-29a, leading to activation of IGF-I and VEGFA signal [78, 79]. This is a possible compensatory mechanism triggering cardiac angiogenesis, when cardiac tissue undergoes ischemia.
circRNAs Mediate Cross-Talk Between Macrophages and Cardiac Fibroblasts
Inflammation plays important roles in myocardial injury and myocardial fibrosis during MI [4]. There is communication between macrophages and cardiac fibroblasts. In the infarcted area of myocardium, infiltrated macrophages can activate cardiac fibroblasts by secreting transforming growth factor-β (TGF-β) and interleukin-6 (IL-6). Recently, Wang et al. indicated that M2 macrophage (M2M) can also mediate cardiac fibrosis via EV-delivered circRNAs. They observed that EVs released by M2Ms enhanced cardiac fibrosis post-MI in vivo, as well as proliferation of cardiac fibroblasts in vitro. Comparing with M0 macrophage–secreted EVs, which appeared in early stage of MI, a series of circRNAs were detected to be up-regulated in M2M-secreted EVs. Among the circRNAs in M2M-derived EVs, circUbe3a was most significantly up-regulated. Uptake of M2M-derived EVs obviously increased the level of circUbe3a in recipient cardiac fibroblasts. The proliferation, migration, and phenotypic transformation of recipient cardiac fibroblasts were promoted through circUbe3a/miR-138-5p/RhoC axis [84]. This study clearly revealed that circRNAs mediate novel communications between macrophages and cardiac fibroblasts. It also indicated that extracellular circUbe3a transmits a compensatory signal of promoting cardiac fibrosis to maintain cardiac structure post-MI.
Extracellular circRNAs in Coronary Atherosclerosis
Coronary atherosclerosis is the essential cause of cardiac ischemia. Therefore, it is also necessary to discuss the potential effect of extracellular circRNAs in coronary atherosclerosis. The levels of circRNAs including hsa_circ_0005540 [85], hsa_circ_0001445 [86], hsa_circ_0001879, hsa_circ_0004104 [87], and hsa_circ_0124644 [88] were detected to significantly increase in blood samples or plasmic exosomes of patients with coronary artery stenosis. They showed close correlation with coronary atherosclerosis and coronary artery disease. Hsa_circ_0124644 can aggravate oxidized low-density lipoprotein-induced endothelial injury by regulating miR-149-5p/PAPP-A pathway in human vascular endothelial cells [89]. Hsa_circ_0004104 was not only involved in PsAF and cardiac fibrosis [62], but also showed pro-atherosclerosis potency. Overexpression of hsa_circ_0004104 in THP-1-derived macrophages can up-regulate pro-atherosclerosis genes and downregulate anti-atherosclerosis genes [87]. It might establish a potential connection between coronary atherosclerosis and cardiac dysfunction.
Conclusion and Perspective
The extracellular circRNA-mediated intercellular communications, including cardiomyocyte-cardiomyocyte, cardiomyocyte-endotheliocyte, and M2M-cardiac fibroblast, have been shown to play important roles in ischemic myocardial injury and cardiac remodeling. The EV-delivered circRNAs were released from various types of cardiac cells and received by same or other types of cells, regulating cell proliferation, migration, and viability like first messengers. There are numerous circRNAs in extracellular fluids remaining to be further explored. Their changed expression level suggested that they might involve in cell cross-talk during ischemic myocardial injury and cardiac remodeling. The critical ones can not only be selected as biomarkers in diagnosis, but also deserved to be developed into drugs. However, a circRNA might show distinct functions and mechanisms in different types of recipient cells. For instance, MICRA mentioned above (also called circZNF609) inhibits cell migration, tube formation and promote apoptosis in endothelial cells. The expression of MICRA in endothelial cells is induced by hypoxia stress [90]. MICRA was also reported to decrease in peripheral blood leukocytes of patients with coronary artery disease. Overexpression of MICRA in macrophages showed an anti-inflammatory effect [91]. Modification of the EV-membrane with specific protein or peptide will effectively help EVs target specific type of cell or tissue [92, 93]. In addition, the cross-talk between heart and other organs also plays non-negligible roles in ischemic cardiac injury. Extracellular miRNAs have been reported to mediate the cross-talk between adipose tissue and ischemic myocardium. For example, exosomal miRNAs generated by adipose-derived stem cell including miR-146a and miR-320d can protect cardiomyocyte from hypoxia-induced apoptosis [94, 95]. On the other hand, miR-1956 derived from ischemic myocardium promotes proliferation of adipose-derived mesenchymal stem cells [96]. However, EV circRNA–mediated cross-talk between other organs and the heart has not been discovered yet. It remains to be further detected.
References
Libby, P. (2013). Mechanisms of acute coronary syndromes and their implications for therapy. The New England journal of medicine, 368(21), 2004–2013.
Eltzschig, H. K., & Eckle, T. (2011). Ischemia and reperfusion–from mechanism to translation. Nature medicine, 17(11), 1391–1401.
Porter, K. E., & Turner, N. A. (2009). Cardiac fibroblasts: At the heart of myocardial remodeling. Pharmacology & therapeutics, 123(2), 255–278.
Prabhu, S. D., & Frangogiannis, N. G. (2016). The Biological Basis for Cardiac Repair After Myocardial Infarction: From Inflammation to Fibrosis. Circulation research, 119(1), 91–112.
Shimizu, I., & Minamino, T. (2016). Physiological and pathological cardiac hypertrophy. Journal of molecular and cellular cardiology, 97, 245–262.
McMurray, J. J., & Pfeffer, M. A. (2005). Heart failure. Lancet (London, England), 365(9474), 1877–1889.
Chen, L. L. (2020). The expanding regulatory mechanisms and cellular functions of circular RNAs. Nature reviews. Molecular cell biology, 21(8), 475–490.
Werfel, S., Nothjunge, S., Schwarzmayr, T., Strom, T. M., Meitinger, T., & Engelhardt, S. (2016). Characterization of circular RNAs in human, mouse and rat hearts. Journal of molecular and cellular cardiology, 98, 103–107.
Holdt, L. M., Stahringer, A., Sass, K., Pichler, G., Kulak, N. A., Wilfert, W., Kohlmaier, A., Herbst, A., Northoff, B. H., Nicolaou, A., Gäbel, G., Beutner, F., Scholz, M., Thiery, J., Musunuru, K., Krohn, K., Mann, M., & Teupser, D. (2016). Circular non-coding RNA ANRIL modulates ribosomal RNA maturation and atherosclerosis in humans. Nature communications, 7, 12429.
Wu, W. P., Zhou, M. Y., Liu, D. L., Min, X., Shao, T., Xu, Z. Y., Jing, X., Cai, M. Y., Xu, S., Liang, X., Mo, M., Liu, X., & Xiong, X. D. (2021). circGNAQ, a circular RNA enriched in vascular endothelium, inhibits endothelial cell senescence and atherosclerosis progression. Molecular therapy. Nucleic acids, 26, 374–387.
Li, M., Ding, W., Tariq, M. A., Chang, W., Zhang, X., Xu, W., Hou, L., Wang, Y., & Wang, J. (2018). A circular transcript of ncx1 gene mediates ischemic myocardial injury by targeting miR-133a-3p. Theranostics, 8(21), 5855–5869.
Zhou, L. Y., Zhai, M., Huang, Y., Xu, S., An, T., Wang, Y. H., Zhang, R. C., Liu, C. Y., Dong, Y. H., Wang, M., Qian, L. L., Ponnusamy, M., Zhang, Y. H., Zhang, J., & Wang, K. (2019). The circular RNA ACR attenuates myocardial ischemia/reperfusion injury by suppressing autophagy via modulation of the Pink1/ FAM65B pathway. Cell death and differentiation, 26(7), 1299–1315.
Gao, X. Q., Liu, C. Y., Zhang, Y. H., Wang, Y. H., Zhou, L. Y., Li, X. M., Wang, K., Chen, X. Z., Wang, T., Ju, J., Wang, F., Wang, S. C., Wang, Y., Chen, Z. Y., & Wang, K. (2021). The circRNA CNEACR regulates necroptosis of cardiomyocytes through Foxa2 suppression. Cell death and differentiation. https://doi.org/10.1038/s41418-021-00872-2.Advanceonlinepublication
Garikipati, V., Verma, S. K., Cheng, Z., Liang, D., Truongcao, M. M., Cimini, M., Yue, Y., Huang, G., Wang, C., Benedict, C., Tang, Y., Mallaredy, V., Ibetti, J., Grisanti, L., Schumacher, S. M., Gao, E., Rajan, S., Wilusz, J. E., Goukassian, D., Houser, S. R., … Kishore, R. (2019). Circular RNA CircFndc3b modulates cardiac repair after myocardial infarction via FUS/VEGF-A axis. Nature communications, 10(1), 4317.
Lim, T. B., Aliwarga, E., Luu, T., Li, Y. P., Ng, S. L., Annadoray, L., Sian, S., Ackers-Johnson, M. A., & Foo, R. S. (2019). Targeting the highly abundant circular RNA circSlc8a1 in cardiomyocytes attenuates pressure overload induced hypertrophy. Cardiovascular research, 115(14), 1998–2007.
Wang, K., Long, B., Liu, F., Wang, J. X., Liu, C. Y., Zhao, B., Zhou, L. Y., Sun, T., Wang, M., Yu, T., Gong, Y., Liu, J., Dong, Y. H., Li, N., & Li, P. F. (2016). A circular RNA protects the heart from pathological hypertrophy and heart failure by targeting miR-223. European heart journal, 37(33), 2602–2611.
Wu, N., Xu, J., Du, W. W., Li, X., Awan, F. M., Li, F., Misir, S., Eshaghi, E., Lyu, J., Zhou, L., Zeng, K., Adil, A., Wang, S., & Yang, B. B. (2021). YAP Circular RNA, circYap, Attenuates Cardiac Fibrosis via Binding with Tropomyosin-4 and Gamma-Actin Decreasing Actin Polymerization. Molecular therapy : The journal of the American Society of Gene Therapy, 29(3), 1138–1150.
Du, W. W., Xu, J., Yang, W., Wu, N., Li, F., Zhou, L., Wang, S., Li, X., He, A. T., Du, K. Y., Zeng, K., Ma, J., Lyu, J., Zhang, C., Zhou, C., Maksimovic, K., & Yang, B. B. (2021). A Neuroligin Isoform Translated by circNlgn Contributes to Cardiac Remodeling. Circulation research, 129(5), 568–582.
Suzuki, H., Zuo, Y., Wang, J., Zhang, M. Q., Malhotra, A., & Mayeda, A. (2006). Characterization of RNase R-digested cellular RNA source that consists of lariat and circular RNAs from pre-mRNA splicing. Nucleic acids research, 34(8), e63.
Li, Y., Zheng, Q., Bao, C., Li, S., Guo, W., Zhao, J., Chen, D., Gu, J., He, X., & Huang, S. (2015). Circular RNA is enriched and stable in exosomes: A promising biomarker for cancer diagnosis. Cell research, 25(8), 981–984.
Li, P., Chen, S., Chen, H., Mo, X., Li, T., Shao, Y., Xiao, B., & Guo, J. (2015). Using circular RNA as a novel type of biomarker in the screening of gastric cancer. Clinica chimica acta; international journal of clinical chemistry, 444, 132–136.
Bahn, J. H., Zhang, Q., Li, F., Chan, T. M., Lin, X., Kim, Y., Wong, D. T., & Xiao, X. (2015). The landscape of microRNA, Piwi-interacting RNA, and circular RNA in human saliva. Clinical chemistry, 61(1), 221–230.
Shao, Y., Li, J., Lu, R., Li, T., Yang, Y., Xiao, B., & Guo, J. (2017). Global circular RNA expression profile of human gastric cancer and its clinical significance. Cancer medicine, 6(6), 1173–1180.
Xu, Y., Leng, K., Yao, Y., Kang, P., Liao, G., Han, Y., Shi, G., Ji, D., Huang, P., Zheng, W., Li, Z., Li, J., Huang, L., Yu, L., Zhou, Y., Jiang, X., Wang, H., Li, C., Su, Z., … Cui, Y. (2021). A Circular RNA, Cholangiocarcinoma-Associated Circular RNA 1, Contributes to Cholangiocarcinoma Progression, Induces Angiogenesis, and Disrupts Vascular Endothelial Barriers. Hepatology (Baltimore, Md.), 73(4), 1419–1435.
Dong, W. W., Li, H. M., Qing, X. R., Huang, D. H., & Li, H. G. (2016). Identification and characterization of human testis derived circular RNAs and their existence in seminal plasma. Scientific reports, 6, 39080.
Ma, H., Xu, Y., Zhang, R., Guo, B., Zhang, S., & Zhang, X. (2019). Differential expression study of circular RNAs in exosomes from serum and urine in patients with idiopathic membranous nephropathy. Archives of medical science : AMS, 15(3), 738–753.
Zeng, B., Chen, T., Luo, J., Xie, M., Wei, L., Xi, Q., Sun, J., & Zhang, Y. (2020). Exploration of Long Non-coding RNAs and Circular RNAs in Porcine Milk Exosomes. Frontiers in genetics, 11, 652.
van der Pol, E., Böing, A. N., Harrison, P., Sturk, A., & Nieuwland, R. (2012). Classification, functions, and clinical relevance of extracellular vesicles. Pharmacological reviews, 64(3), 676–705.
Stahl, P. D., & Raposo, G. (2019). Extracellular Vesicles: Exosomes and Microvesicles, Integrators of Homeostasis. Physiology (Bethesda, Md.), 34(3), 169–177.
Hauser, P., Wang, S., & Didenko, V. V. (2017). Apoptotic Bodies: Selective Detection in Extracellular Vesicles. Methods in molecular biology (Clifton, N.J.), 1554, 193–200.
Pegtel, D. M., & Gould, S. J. (2019). Exosomes. Annual review of biochemistry, 88, 487–514.
Mori, M. A., Ludwig, R. G., Garcia-Martin, R., Brandão, B. B., & Kahn, C. R. (2019). Extracellular miRNAs: From Biomarkers to Mediators of Physiology and Disease. Cell metabolism, 30(4), 656–673.
Piccoli, M. T., Gupta, S. K., Viereck, J., Foinquinos, A., Samolovac, S., Kramer, F. L., Garg, A., Remke, J., Zimmer, K., Batkai, S., & Thum, T. (2017). Inhibition of the Cardiac Fibroblast-Enriched lncRNA Meg3 Prevents Cardiac Fibrosis and Diastolic Dysfunction. Circulation research, 121(5), 575–583.
Li, B., Hong, J., Hong, M., Wang, Y., Yu, T., Zang, S., & Wu, Q. (2019). piRNA-823 delivered by multiple myeloma-derived extracellular vesicles promoted tumorigenesis through re-educating endothelial cells in the tumor environment. Oncogene, 38(26), 5227–5238.
Mukherjee, K., Ghoshal, B., Ghosh, S., Chakrabarty, Y., Shwetha, S., Das, S., & Bhattacharyya, S. N. (2016). Reversible HuR-microRNA binding controls extracellular export of miR-122 and augments stress response. EMBO reports, 17(8), 1184–1203.
Shurtleff, M. J., Temoche-Diaz, M. M., Karfilis, K. V., Ri, S., & Schekman, R. (2016). Y-box protein 1 is required to sort microRNAs into exosomes in cells and in a cell-free reaction. eLife, 5, e19276.
Villarroya-Beltri, C., Gutiérrez-Vázquez, C., Sánchez-Cabo, F., Pérez-Hernández, D., Vázquez, J., Martin-Cofreces, N., Martinez-Herrera, D. J., Pascual-Montano, A., Mittelbrunn, M., & Sánchez-Madrid, F. (2013). Sumoylated hnRNPA2B1 controls the sorting of miRNAs into exosomes through binding to specific motifs. Nature communications, 4, 2980.
Santangelo, L., Giurato, G., Cicchini, C., Montaldo, C., Mancone, C., Tarallo, R., Battistelli, C., Alonzi, T., Weisz, A., & Tripodi, M. (2016). The RNA-Binding Protein SYNCRIP Is a Component of the Hepatocyte Exosomal Machinery Controlling MicroRNA Sorting. Cell reports, 17(3), 799–808.
Pan, Z., Zhao, R., Li, B., et al. (2022). EWSR1-induced circNEIL3 promotes glioma progression and exosome-mediated macrophage immunosuppressive polarization via stabilizing IGF2BP3. Molecular cancer, 21(1), 16.
Yu, X., Harris, S. L., & Levine, A. J. (2006). The regulation of exosome secretion: A novel function of the p53 protein. Cancer research, 66(9), 4795–4801.
Wang, S., Zhang, K., Tan, S., Xin, J., Yuan, Q., Xu, H., Xu, X., Liang, Q., Christiani, D. C., Wang, M., Liu, L., & Du, M. (2021). Circular RNAs in body fluids as cancer biomarkers: The new frontier of liquid biopsies. Molecular cancer, 20(1), 13.
Fanale, D., Taverna, S., Russo, A., & Bazan, V. (2018). Circular RNA in Exosomes. Advances in experimental medicine and biology, 1087, 109–117.
Wang, Y., Liu, J., Ma, J., Sun, T., Zhou, Q., Wang, W., Wang, G., Wu, P., Wang, H., Jiang, L., Yuan, W., Sun, Z., & Ming, L. (2019). Exosomal circRNAs: Biogenesis, effect and application in human diseases. Molecular cancer, 18(1), 116.
Liu, C., Ge, H. M., Liu, B. H., Dong, R., Shan, K., Chen, X., Yao, M. D., Li, X. M., Yao, J., Zhou, R. M., Zhang, S. J., Jiang, Q., Zhao, C., & Yan, B. (2019). Targeting pericyte-endothelial cell crosstalk by circular RNA-cPWWP2A inhibition aggravates diabetes-induced microvascular dysfunction. Proceedings of the National Academy of Sciences of the United States of America, 116(15), 7455–7464.
Shi, R., Jin, Y., Hu, W., Lian, W., Cao, C., Han, S., Zhao, S., Yuan, H., Yang, X., Shi, J., & Zhao, H. (2020). Exosomes derived from mmu_circ_0000250-modified adipose-derived mesenchymal stem cells promote wound healing in diabetic mice by inducing miR-128-3p/SIRT1-mediated autophagy. American journal of physiology. Cell physiology, 318(5), C848–C856.
Song, J., Chen, Z. H., Zheng, C. J., Song, K. H., Xu, G. Y., Xu, S., Zou, F., Ma, X. S., Wang, H. L., & Jiang, J. Y. (2020). Exosome-Transported circRNA_0000253 Competitively Adsorbs MicroRNA-141-5p and Increases IDD. Molecular therapy. Nucleic acids, 21, 1087–1099.
Yang, L., Han, B., Zhang, Z., Wang, S., Bai, Y., Zhang, Y., Tang, Y., Du, L., Xu, L., Wu, F., Zuo, L., Chen, X., Lin, Y., Liu, K., Ye, Q., Chen, B., Li, B., Tang, T., Wang, Y., et al. (2020). Extracellular Vesicle-Mediated Delivery of Circular RNA SCMH1 Promotes Functional Recovery in Rodent and Nonhuman Primate Ischemic Stroke Models. Circulation, 142(6), 556–574.
Chen, W., Wang, H., Zhu, Z., Feng, J., & Chen, L. (2020). Exosome-Shuttled circSHOC2 from IPASs Regulates Neuronal Autophagy and Ameliorates Ischemic Brain Injury via the miR-7670-3p/SIRT1 Axis. Molecular therapy. Nucleic acids, 22, 657–672.
Zhang, X., Chen, L., Xiao, B., Liu, H., & Su, Y. (2019). Circ_0075932 in adipocyte-derived exosomes induces inflammation and apoptosis in human dermal keratinocytes by directly binding with PUM2 and promoting PUM2-mediated activation of AuroraA/NF-κB pathway. Biochemical and biophysical research communications, 511(3), 551–558.
Li, Y., Chen, J., Chen, Z., Xu, X., Weng, J., Zhang, Y., Mo, Y., Liu, Y., Wang, J., & Ke, Y. (2021). CircGLIS3 Promotes High-Grade Glioma Invasion via Modulating Ezrin Phosphorylation. Frontiers in cell and developmental biology, 9, 663207.
Tan, W. L., Lim, B. T., Anene-Nzelu, C. G., Ackers-Johnson, M., Dashi, A., See, K., Tiang, Z., Lee, D. P., Chua, W. W., Luu, T. D., Li, P. Y., Richards, A. M., & Foo, R. S. (2017). A landscape of circular RNA expression in the human heart. Cardiovascular research, 113(3), 298–309.
Jakobi, T., Siede, D., Eschenbach, J., Heumüller, A. W., Busch, M., Nietsch, R., Meder, B., Most, P., Dimmeler, S., Backs, J., Katus, H. A., & Dieterich, C. (2020). Deep Characterization of Circular RNAs from Human Cardiovascular Cell Models and Cardiac Tissue. Cells, 9(7), 1616.
Siede, D., Rapti, K., Gorska, A. A., Katus, H. A., Altmüller, J., Boeckel, J. N., Meder, B., Maack, C., Völkers, M., Müller, O. J., Backs, J., & Dieterich, C. (2017). Identification of circular RNAs with host gene-independent expression in human model systems for cardiac differentiation and disease. Journal of molecular and cellular cardiology, 109, 48–56.
Vausort, M., Salgado-Somoza, A., Zhang, L., Leszek, P., Scholz, M., Teren, A., Burkhardt, R., Thiery, J., Wagner, D. R., & Devaux, Y. (2016). Myocardial Infarction-Associated Circular RNA Predicting Left Ventricular Dysfunction. Journal of the American College of Cardiology, 68(11), 1247–1248.
Yang, W., Sun, L., Cao, X., Li, L., Zhang, X., Li, J., Zhao, H., Zhan, C., Zang, Y., Li, T., Zhang, L., Liu, G., & Li, W. (2021). Detection of circRNA Biomarker for Acute Myocardial Infarction Based on System Biological Analysis of RNA Expression. Frontiers in genetics, 12, 686116.
Zhao, C., Liu, J., Ge, W., Li, Z., Lv, M., Feng, Y., Liu, X., Liu, B., & Zhang, Y. (2021). Identification of Regulatory circRNAs Involved in the Pathogenesis of Acute Myocardial Infarction. Frontiers in genetics, 11, 626492.
Yin, L., Tang, Y., & Jiang, M. (2021). Research on the circular RNA bioinformatics in patients with acute myocardial infarction. Journal of clinical laboratory analysis, 35(2), e23621.
Ge, X., Meng, Q., Zhuang, R., Yuan, D., Liu, J., Lin, F., Fan, H., & Zhou, X. (2019). Circular RNA expression alterations in extracellular vesicles isolated from murine heart post ischemia/reperfusion injury. International journal of cardiology, 296, 136–140.
Guo, Q., Wang, J., Sun, R., He, Z., Chen, Q., Liu, W., Wu, M., Bao, J., Liu, Z., Wang, J., & Zhang, Y. (2020). Comprehensive Construction of a Circular RNA-Associated Competing Endogenous RNA Network Identified Novel Circular RNAs in Hypertrophic Cardiomyopathy by Integrated Analysis. Frontiers in genetics, 11, 764.
Sonnenschein, K., Wilczek, A. L., de Gonzalo-Calvo, D., Pfanne, A., Derda, A. A., Zwadlo, C., Bavendiek, U., Bauersachs, J., Fiedler, J., & Thum, T. (2019). Serum circular RNAs act as blood-based biomarkers for hypertrophic obstructive cardiomyopathy. Scientific reports, 9(1), 20350.
Meinecke, A., Mitzka, S., Just, A., Cushman, S., Stojanović, S. D., Xiao, K., Mooren, F. C., Fiedler, J., & Thum, T. (2020). Cardiac endurance training alters plasma profiles of circular RNA MBOAT2. American journal of physiology. Heart and circulatory physiology, 319, 1 H13-H21.
Gao, Y., Liu, Y., Fu, Y., Wang, Q., Liu, Z., Hu, R., Yang, X., & Chen, M. (2021). The potential regulatory role of hsa_circ_0004104 in the persistency of atrial fibrillation by promoting cardiac fibrosis via TGF-β pathway. BMC cardiovascular disorders, 21(1), 25.
Costa, M. C., Calderon-Dominguez, M., Mangas, A., Campuzano, O., Sarquella-Brugada, G., Ramos, M., Quezada-Feijoo, M., Pinilla, J., Robles-Mezcua, A., Del Aguila Pacheco-Cruz, G., Belmonte, T., Enguita, F. J., & Toro, R. (2021). Circulating circRNA as biomarkers for dilated cardiomyopathy etiology. Journal of molecular medicine (Berlin, Germany), 99(12), 1711–1725.
Han, J., Zhang, L., Hu, L., Yu, H., Xu, F., Yang, B., Zhang, R., Zhang, Y., & An, Y. (2020). Circular RNA-Expression Profiling Reveals a Potential Role of Hsa_circ_0097435 in Heart Failure via Sponging Multiple MicroRNAs. Frontiers in genetics, 11, 212.
Sun, Y., Jiang, X., Lv, Y., Liang, X., Zhao, B., Bian, W., Zhang, D., Jiang, J., & Zhang, C. (2020). Circular RNA Expression Profiles in Plasma from Patients with Heart Failure Related to Platelet Activity. Biomolecules, 10(2), 187.
Geng, H. H., Li, R., Su, Y. M., Xiao, J., Pan, M., Cai, X. X., & Ji, X. P. (2016). The Circular RNA Cdr1as Promotes Myocardial Infarction by Mediating the Regulation of miR-7a on Its Target Genes Expression. PloS one, 11(3), e0151753.
Chen, C., Shen, H., Huang, Q., & Li, Q. (2020). The Circular RNA CDR1as Regulates the Proliferation and Apoptosis of Human Cardiomyocytes Through the miR-135a/HMOX1 and miR-135b/HMOX1 Axes. Genetic testing and molecular biomarkers, 24(9), 537–548.
Cheng, N., Wang, M. Y., Wu, Y. B., Cui, H. M., Wei, S. X., Liu, B., & Wang, R. (2021). Circular RNA POSTN Promotes Myocardial Infarction-Induced Myocardial Injury and Cardiac Remodeling by Regulating miR-96–5p/BNIP3 Axis. Frontiers in cell and developmental biology, 8, 618574.
Zhang, J., Tang, Y., Zhang, J., Wang, J., He, J., Zhang, Z., & Liu, F. (2021). CircRNA ACAP2 Is Overexpressed in Myocardial Infarction and Promotes the Maturation of miR-532 to Induce the Apoptosis of Cardiomyocyte. Journal of cardiovascular pharmacology, 78(2), 247–252.
Wang, K., Gan, T. Y., Li, N., Liu, C. Y., Zhou, L. Y., Gao, J. N., Chen, C., Yan, K. W., Ponnusamy, M., Zhang, Y. H., & Li, P. F. (2017). Circular RNA mediates cardiomyocyte death via miRNA-dependent upregulation of MTP18 expression. Cell death and differentiation, 24(6), 1111–1120.
Wang, S., Li, L., Deng, W., & Jiang, M. (2021). CircRNA MFACR Is Upregulated in Myocardial Infarction and Downregulates miR-125b to Promote Cardiomyocyte Apoptosis Induced by Hypoxia. Journal of cardiovascular pharmacology, 78(6), 802–808.
Li, B., Cai, X., Wang, Y., Zhu, H., Zhang, P., Jiang, P., Yang, X., Sun, J., Hong, L., & Shao, L. (2021). Circ-SKA3 Enhances Doxorubicin Toxicity in AC16 Cells Through miR-1303/TLR4 Axis. International heart journal, 62(5), 1112–1123.
Green, D. R. (2020). Ghostly metabolic messages from dying cells. Nature, 580(7801), 36–37.
Gnecchi, M., He, H., Noiseux, N., Liang, O. D., Zhang, L., Morello, F., Mu, H., Melo, L. G., Pratt, R. E., Ingwall, J. S., & Dzau, V. J. (2006). Evidence supporting paracrine hypothesis for Akt-modified mesenchymal stem cell-mediated cardiac protection and functional improvement. FASEB journal: Official publication of the Federation of American Societies for Experimental Biology, 20(6), 661–669.
Heldman, A. W., DiFede, D. L., Fishman, J. E., Zambrano, J. P., Trachtenberg, B. H., Karantalis, V., Mushtaq, M., Williams, A. R., Suncion, V. Y., McNiece, I. K., Ghersin, E., Soto, V., Lopera, G., Miki, R., Willens, H., Hendel, R., Mitrani, R., Pattany, P., Feigenbaum, G., Oskouei, B., … Hare, J. M. (2014). Transendocardial mesenchymal stem cells and mononuclear bone marrow cells for ischemic cardiomyopathy: the TAC-HFT randomized trial. JAMA, 311(1), 62–73.
Zhang, C., Wang, H., Li, J., & Ma, L. (2021). Circular RNA Involvement in the Protective Effect of Human Umbilical Cord Mesenchymal Stromal Cell-Derived Extracellular Vesicles Against Hypoxia/Reoxygenation Injury in Cardiac Cells. Frontiers in cardiovascular medicine, 8, 626878.
Yan, B., Zhang, Y., Liang, C., Liu, B., Ding, F., Wang, Y., Zhu, B., Zhao, R., Yu, X. Y., & Li, Y. (2020). Stem cell-derived exosomes prevent pyroptosis and repair ischemic muscle injury through a novel exosome/circHIPK3/ FOXO3a pathway. Theranostics, 10(15), 6728–6742.
Wang, Y., Zhao, R., Liu, W., Wang, Z., Rong, J., Long, X., Liu, Z., Ge, J., & Shi, B. (2019). Exosomal circHIPK3 Released from Hypoxia-Pretreated Cardiomyocytes Regulates Oxidative Damage in Cardiac Microvascular Endothelial Cells via the miR-29a/IGF-1 Pathway. Oxidative medicine and cellular longevity, 2019, 7954657.
Wang, Y., Zhao, R., Shen, C., Liu, W., Yuan, J., Li, C., Deng, W., Wang, Z., Zhang, W., Ge, J., & Shi, B. (2020). Exosomal CircHIPK3 Released from Hypoxia-Induced Cardiomyocytes Regulates Cardiac Angiogenesis after Myocardial Infarction. Oxidative medicine and cellular longevity, 2020, 8418407.
Colliva, A., Braga, L., Giacca, M., & Zacchigna, S. (2020). Endothelial cell-cardiomyocyte crosstalk in heart development and disease. The Journal of physiology, 598(14), 2923–2939.
Shan, K., Liu, C., Liu, B. H., Chen, X., Dong, R., Liu, X., Zhang, Y. Y., Liu, B., Zhang, S. J., Wang, J. J., Zhang, S. H., Wu, J. H., Zhao, C., & Yan, B. (2017). Circular Noncoding RNA HIPK3 Mediates Retinal Vascular Dysfunction in Diabetes Mellitus. Circulation, 136(17), 1629–1642.
Si, X., Zheng, H., Wei, G., Li, M., Li, W., Wang, H., Guo, H., Sun, J., Li, C., Zhong, S., Liao, W., Liao, Y., Huang, S., & Bin, J. (2020). circRNA Hipk3 Induces Cardiac Regeneration after Myocardial Infarction in Mice by Binding to Notch1 and miR-133a. Molecular therapy. Nucleic acids, 21, 636–655.
Qiu, Z., Wang, Y., Liu, W., Li, C., Zhao, R., Long, X., Rong, J., Deng, W., Shen, C., Yuan, J., Chen, W., & Shi, B. (2021). CircHIPK3 regulates the autophagy and apoptosis of hypoxia/reoxygenation-stimulated cardiomyocytes via the miR-20b-5p/ATG7 axis. Cell death discovery, 7(1), 64.
Wang, Y., Li, C., Zhao, R., Qiu, Z., Shen, C., Wang, Z., Liu, W., Zhang, W., Ge, J., & Shi, B. (2021). CircUbe3a from M2 macrophage-derived small extracellular vesicles mediates myocardial fibrosis after acute myocardial infarction. Theranostics, 11(13), 6315–6333.
Wu, W. P., Pan, Y. H., Cai, M. Y., Cen, J. M., Chen, C., Zheng, L., Liu, X., & Xiong, X. D. (2020). Plasma-Derived Exosomal Circular RNA hsa_circ_0005540 as a Novel Diagnostic Biomarker for Coronary Artery Disease. Disease markers, 2020, 3178642.
Vilades, D., Martínez-Camblor, P., Ferrero-Gregori, A., Bär, C., Lu, D., Xiao, K., Vea, À., Nasarre, L., Sanchez Vega, J., Leta, R., Carreras, F., Thum, T., Llorente-Cortés, V., & de Gonzalo-Calvo, D. (2020). Plasma circular RNA hsa_circ_0001445 and coronary artery disease: Performance as a biomarker. FASEB journal : Official publication of the Federation of American Societies for Experimental Biology, 34(3), 4403–4414.
Wang, L., Shen, C., Wang, Y., Zou, T., Zhu, H., Lu, X., Li, L., Yang, B., Chen, J., Chen, S., Lu, X., & Gu, D. (2019). Identification of circular RNA Hsa_circ_0001879 and Hsa_circ_0004104 as novel biomarkers for coronary artery disease. Atherosclerosis, 286, 88–96.
Zhao, Z., Li, X., Gao, C., Jian, D., Hao, P., Rao, L., & Li, M. (2017). Peripheral blood circular RNA hsa_circ_0124644 can be used as a diagnostic biomarker of coronary artery disease. Scientific reports, 7, 39918.
Wang, G., Li, Y., Liu, Z., Ma, X., Li, M., Lu, Q., Li, Y., Lu, Z., Niu, L., Fan, Z., & Lei, Z. (2020). Circular RNA circ_0124644 exacerbates the ox-LDL-induced endothelial injury in human vascular endothelial cells through regulating PAPP-A by acting as a sponge of miR-149-5p. Molecular and cellular biochemistry, 471(1–2), 51–61.
Liu, C., Yao, M. D., Li, C. P., Shan, K., Yang, H., Wang, J. J., Liu, B., Li, X. M., Yao, J., Jiang, Q., & Yan, B. (2017). Silencing Of Circular RNA-ZNF609 Ameliorates Vascular Endothelial Dysfunction. Theranostics, 7(11), 2863–2877.
Liang, B., Li, M., Deng, Q., Wang, C., Rong, J., He, S., Xiang, Y., & Zheng, F. (2020). CircRNA ZNF609 in peripheral blood leukocytes acts as a protective factor and a potential biomarker for coronary artery disease. Annals of translational medicine, 8(12), 741.
Vandergriff, A., Huang, K., Shen, D., Hu, S., Hensley, M. T., Caranasos, T. G., Qian, L., & Cheng, K. (2018). Targeting regenerative exosomes to myocardial infarction using cardiac homing peptide. Theranostics, 8(7), 1869–1878.
Wang, X., Chen, Y., Zhao, Z., Meng, Q., Yu, Y., Sun, J., Yang, Z., Chen, Y., Li, J., Ma, T., Liu, H., Li, Z., Yang, J., & Shen, Z. (2018). Engineered Exosomes With Ischemic Myocardium-Targeting Peptide for Targeted Therapy in Myocardial Infarction. Journal of the American Heart Association, 7(15), e008737.
Pan, J., Alimujiang, M., Chen, Q., Shi, H., & Luo, X. (2019). Exosomes derived from miR-146a-modified adipose-derived stem cells attenuate acute myocardial infarction-induced myocardial damage via downregulation of early growth response factor 1. Journal of cellular biochemistry, 120(3), 4433–4443.
Liu, L., Zhang, H., Mao, H., Li, X., & Hu, Y. (2019). Exosomal miR-320d derived from adipose tissue-derived MSCs inhibits apoptosis in cardiomyocytes with atrial fibrillation (AF). Artificial cells, nanomedicine, and biotechnology, 47(1), 3976–3984.
Gao, L., Mei, S., Zhang, S., Qin, Q., Li, H., Liao, Y., Fan, H., Liu, Z., & Zhu, H. (2020). Cardio-renal Exosomes in Myocardial Infarction Serum Regulate Proangiogenic Paracrine Signaling in Adipose Mesenchymal Stem Cells. Theranostics, 10(3), 1060–1073.
Funding
This work was supported by Natural Science Foundation of Shandong Province (grant number ZR2019ZD28) and National Natural Science Foundation of China (grant number 81900259).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of Interest
The authors declare no competing interests.
Research Involving Human Participants and/or Animals
No human or animal studies were carried out by the authors for this review article.
Additional information
Associate Editor Junjie Xiao oversaw the review of this article.
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Li, M., Ding, W., Liu, G. et al. Extracellular Circular RNAs Act as Novel First Messengers Mediating Cell Cross-Talk in Ischemic Cardiac Injury and Myocardial Remodeling. J. of Cardiovasc. Trans. Res. 15, 444–455 (2022). https://doi.org/10.1007/s12265-022-10219-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12265-022-10219-y