Abstract
Objective
Melittin is the main peptide in bee venom and causes both persistent spontaneous nociception and pain hypersensitivity. Our recent studies indicated that both transient receptor potential (TRP) vanilloid receptor 1 (TRPV1) and canonical TRPs (TRPCs) are involved in mediating the melittin-induced activation of different subpopulations of primary nociceptive cells. Here, we further determined whether TRPC channels are involved in melittin-induced inflammatory nociceptive responses in behavioral assays.
Methods
The anti-nociceptive and anti-hyperalgesic effects of localized peripheral administration of three doses of the non-selective TRPC antagonist, SKF-96365 (1-{β-[3-(4-methoxyphenyl) propoxy]-4-methoxyphenyl}-1H-imidazole hydrochloride), were evaluated in melittin tests. Pain-related behaviors were rated by counting the number of paw flinches, and measuring paw withdrawal thermal latency (s) and paw withdrawl mechanical threshold (g), over a 1-h time-course.
Results
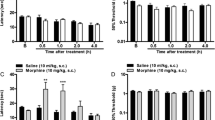
Localized peripheral SKF-96365 given before melittin prevented, and given after melittin significantly suppressed, the melittin-evoked persistent spontaneous nociception. Pre-blockade and post-suppression of activation of primary nociceptive activity resulted in decreased hypersensitivity to both thermal and mechanical stimuli applied to the primary injury site of the ipsilateral hindpaw, despite dose-effect differences between thermal and mechanical hyperalgesia. However, local administration of SKF-96365 into the contralateral hindpaw had no significant effect on any pain-associated behaviors. In addition, SKF-96365 had no effect on baseline threshold for either thermal or mechanical sensitivity under normal conditions.
Conclusion
Besides TRPV1, SKF-96365-sensitive TRPC channels might also be involved in the pathophysiological processing of melittin-induced inflammatory pain and hypersensitivity. Therapeutically, SKF-96365 is equally effective in preventing primary thermal and mechanical hyperalgesia as well as persistent spontaneous nociception. However, this drug is likely to be more effective in the relief of thermal hyperalgesia than mechanical hyperalgesia when applied 5 min after establishment of primary afferent activation.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Clapham DE. TRP channels as cellular sensors. Nature 2003, 426: 517–524.
Pedersen SF, Owsianik G, Nilius B. TRP channels: an overview. Cell Calcium 2005, 38: 233–252.
Ramsey IS, Delling M, Clapham DE. An introduction to TRP channels. Annu Rev Physiol 2006, 68: 619–647.
Putney JW. Physiological mechanisms of TRPC activation. Pflugers Arch 2005, 451: 29–34.
Riccio A, Medhurst AD, Mattei C, Kelsell RE, Calver AR, Randall AD, et al. mRNA distribution analysis of human TRPC family in CNS and peripheral tissues. Brain Res Mol Brain Res 2002, 109: 95–104.
Chung YH, Sun AH, Kim D, Hoon SD, Su KS, Yong KK, et al. Immunohistochemical study on the distribution of TRPC channels in the rat hippocampus. Brain Res 2006, 1085: 132–137.
Elg S, Marmigere F, Mattsson JP, Ernfors P. Cellular subtype distribution and developmental regulation of TRPC channel members in the mouse dorsal root ganglion. J Comp Neurol 2007, 503: 35–46.
Kress M, Karasek J, Ferrer-Montiel AV, Scherbakov N, Haberberger RV. TRPC channels and diacylglycerol dependent calcium signaling in rat sensory neurons. Histochem Cell Biol 2008, 130: 655–667.
Staaf S, Oerther S, Lucas G, Mattsson JP, Ernfors P. Differential regulation of TRP channels in a rat model of neuropathic pain. Pain 2009, 144: 187–199.
Ambudkar IS, Bandyopadhyay BC, Liu X, Lockwich TP, Paria B, Ong HL. Functional organization of TRPC-Ca2+ channels and regulation of calcium microdomains. Cell Calcium 2006, 40: 495–504.
Montell C. Drosophila TRP channels. Pflugers Arch 2005, 451: 19–28.
Selvaraj S, Sun Y, Singh BB. TRPC channels and their implication in neurological diseases. CNS Neurol Disord Drug Targets 2010, 9: 94–104.
Tai Y, Feng S, Du W, Wang Y. Functional roles of TRPC channels in the developing brain. Pflugers Arch 2009, 458: 283–289.
Tai Y, Feng S, Ge R, Du W, Zhang X, He Z, et al. TRPC6 channels promote dendritic growth via the CaMKIV-CREB pathway. J Cell Sci 2008, 121: 2301–2307.
Chen J, Lariviere WR. The nociceptive and anti-nociceptive effects of bee venom injection and therapy: a double-edged sword. Prog Neurobiol 2010, 92: 151–183.
Du YR, Xiao Y, Lu ZM, Ding J, Xie F, Fu H, et al. Melittin activates TRPV1 receptors in primary nociceptive sensory neurons via the phospholipase A2 cascade pathways. Biochem Biophys Res Commun 2011, 408: 32–37.
Habermann E. Bee and wasp venoms. Science 1972, 177: 314–322.
Habermann E. Pharmacologically important substances in the bee- and wasp-venoms. Pharm Unserer Zeit 1974, 3: 145–151.
Lariviere WR, Melzack R. The bee venom test: a new tonic-pain test. Pain 1996, 66: 271–277.
Chen YN, Li KC, Li Z, Shang GW, Liu DN, Lu ZM, et al. Effects of bee venom peptidergic components on rat pain-related behaviors and inflammation. Neuroscience 2006, 138: 631–640.
Li KC, Chen J. Altered pain-related behaviors and spinal neuronal responses produced by s.c. injection of melittin in rats. Neuroscience 2004, 126: 753–762.
Ding J, Xiao Y, Lu D, Du YR, Cui XY, Chen J. Effects of SKF- 96365, a TRPC inhibitor, on melittin-induced inward current and intracellular Ca2+ rise in primary sensory cells. Neurosci Bull 2011, 27: 135–142.
Chen J, Luo C, Li H, Chen H. Primary hyperalgesia to mechanical and heat stimuli following subcutaneous bee venom injection into the plantar surface of hindpaw in the conscious rat: a comparative study with the formalin test. Pain 1999, 83:67–76.
Merritt JE, Armstrong WP, Benham CD, Hallam TJ, Jacob R, Jaxa-Chamiec A, et al. SKF 96365, a novel inhibitor of receptormediated calcium entry. Biochem J 1990, 271: 515–522.
Putney JW Jr. Pharmacology of capacitative calcium entry. Mol Interv 2001, 1: 84–94.
Singh A, Hildebrand ME, Garcia E, Snutch TP. The transient receptor potential channel antagonist SKF96365 is a potent blocker of low-voltage-activated T-type calcium channels. Br J Pharmacology 2010, 160: 1464–1475.
Zhu X, Jiang M, Birnbaumer L. Receptor-activated Ca2+ influx via human Trp3 stably expressed in human embryonic kidney (HEK)293 cells. Evidence for a non-capacitative Ca2+ entry. J Biol Chem 1998, 273: 133–142.
Boulay G, Zhu X, Peyton M, Jiang M, Hurst R, Stefani E, et al. Cloning and expression of a novel mammalian homolog of Drosophila transient receptor potential (Trp) involved in calcium entry secondary to activation of receptors coupled by the Gq class of G protein. J Biol Chem 1997, 272: 29672–29680.
Clapham DE, Julius D, Montell C, Schultz G. International Union of Pharmacology. XLIX. Nomenclature and structure-function relationships of transient receptor potential channels. Pharmacol Rev 2005, 57: 427–450.
Nilius B, Prenen J, Vennekens R, Hoenderop JG, Bindels RJ, Droogmans G. Pharmacological modulation of monovalent cation currents through the epithelial Ca2+ channel ECaC1. Br J Pharmacol 2001, 34: 453–462.
Harteneck C, Gollasch M. Pharmacological modulation of diacylglycerol-sensitive TRPC3/6/7 channels. Curr Pharm Biotechnol 2011, 12: 35–41.
Lu ZM, Xie F, Fu H, Liu MG, Cao FL, Hao J, et al. Roles of peripheral P2X and P2Y receptors in the development of melittininduced nociception and hypersensitivity. Neurochem Res 2008, 33: 2085–2091.
Yu YQ, Chen J. Activation of spinal extracellular signaling-regulated kinases by intraplantar melittin injection. Neurosci Lett 2005, 381: 194–198.
Li MM, Yu YQ, Fu H, Xie F, Xu LX, Chen J. Extracellular signalregulated kinases mediate melittin-induced hypersensitivity of spinal neurons to chemical and thermal but not mechanical stimuli. Brain Res Bull 2008, 77: 227–232.
Hao J, Liu MG, Yu YQ, Cao FL, Li Z, Lu ZM, et al. Roles of peripheral mitogen-activated protein kinases in melittin-induced nociception and hyperalgesia, Neuroscience 2008, 152: 1067–1075.
Trebak M, Vazquez G, Bird GJ, Putney JW Jr. The TRPC3/6/7 subfamily of cation channels. Cell Calcium 2003, 33: 451–461.
Eder P, Groschner K. TRP3/6/7: Topical aspects of biophysics and pathophysiology. Channels 2008, 2: 94–99.
Alvarez J, Coulombe A, Cazorla O, Ugur M, Rauzier JM, Magyar J, et al. ATP/UTP activate cation-permeable channels with TRPC3/7 properties in rat cardiomyocytes. Am J Physiol Heart Circ Physiol 2008, 295: H21–28.
Dietrich A, Kalwa H, Fuchs B, Grimminger F, Weissmann N, Gudermann T. In vivo TRPC functions in the cardiopulmonary vasculature. Cell Calcium 2007, 42: 233–244.
Gottlieb P, Folgering J, Maroto R, Raso A, Wood TG, Kurosky A, et al. Revisiting TRPC1 and TRPC6 mechanosensitivity. Pflugers Arch 2008, 455: 1097–1103.
Spassova MA, Hewavitharana T, Xu W, Soboloff J, Gill DL. A common mechanism underlies stretch activation and receptor activation of TRPC6 channels. Proc Natl Acad Sci U S A 2006, 103: 16586–16591.
Alessandri-Haber N, Dina OA, Chen X, Levine JD. TRPC1 and TRPC6 channels cooperate with TRPV4 to mediate mechanical hyperalgesia and nociceptor sensitization. J Neurosci 2009, 29: 6217–6228.
Author information
Authors and Affiliations
Corresponding authors
Additional information
These authors contributed equally to this work.
Rights and permissions
About this article
Cite this article
Ding, J., Zhang, JR., Wang, Y. et al. Effects of a non-selective TRPC channel blocker, SKF-96365, on melittininduced spontaneous persistent nociception and inflammatory pain hypersensitivity. Neurosci. Bull. 28, 173–181 (2012). https://doi.org/10.1007/s12264-012-1213-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12264-012-1213-y