Abstract
Exchanges of fish larvae between the inner continental shelf and estuaries can be critical to the functional significance of these habitats as nurseries. We sampled near-surface fishes on the inner continental shelf off New Jersey and in an adjacent estuary during the summer and fall of 2005 and 2006 to evaluate the occurrence and variation in these connections. Very few of the abundant taxa (Anchoa mitchilli, Brevoortia tyrannus, Urophycis regia, Etropus microstomus, Peprilus triacanthus, and Pomatomus saltatrix) were exclusive to either habitat but some did change in relative habitat affinity among seasons. For some species, this was indicative of a departure from the ocean for estuaries (e.g., Micropogonias undulatus) while others used both habitats during the summer and fall (e.g., Syngnathus fuscus, Scophthalmus aquosus, and A. mitchilli). Together, these observations confirm the high degree of connectivity between the near-surface larval fishes from the inner continental shelf and estuaries.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
There have been multiple studies of larval fish assemblages on the continental shelf as well as those in estuaries but the connectivity between these assemblages has been less frequently addressed. However, the connectivity between these assemblages is central to the understanding of the functional significance of both habitats alone and relative to each other. The studies that have occurred have focused on the role of estuarine plumes (Govoni and Grimes 1992; Kingsford and Suthers 1994), surf zones (Strydom and Hartman 2005), and descriptive comparison of estuarine and inner shelf assemblages (Tzeng et al. 2002). In many of these instances, the assemblages between the inner shelf and the estuary were observed but were seldom explained. Several studies noted that the movement into the estuary was behaviorally mediated (Raynie and Shaw 1994) and may result in preferential or multiple settlement sites (Able et al. 2006).
Fish assemblages of the US Mid-Atlantic Bight (Cape Cod to Cape Hatteras) continental shelf waters result from reproduction in estuarine, coastal, shelf and slope waters (Able and Fahay 2010; Colvocoresses and Musick 1984; Grosslein and Azarovitz 1982) and are modified by survival and transport of eggs and larvae from other regions. Larval and juvenile fish distribution and abundance on the continental shelf is also influenced locally by multiple water masses and these, in turn, influence those species transported into estuaries. For example, on the inner continental shelf study area, a general southward flow along the coast is modulated by buoyancy from freshwater (estuarine) inputs and wind stress (Munchow and Chant 2000; Wong 1999; Yankovsky et al. 2000; Zhang et al. 2009). During summer thermal stratification, winds from the southwest cause coastal upwelling, particularly at inlets along the New Jersey coast (Glenn et al. 1996; Neuman 1996). Furthermore, physical processes associated with estuarine plumes such as that from Chesapeake Bay shape larval fish assemblages on small scales (10–100 km, 72 h) (Reiss and McConaugha 1999). Separate plume and shelf components in that study are comparable to the shelf and coastal assemblages identified by Cowen et al. (1993). In addition, estuarine-spawned fish (as classified by Able and Fahay 2010) occur in plume waters on the inner continental shelf (Reiss and McConaugha 1999) and are grouped in the coastal assemblage identified by Cowen et al. (1993). A prior study identified many components of the larvae and settled juveniles that were shared but called for further studies to elucidate the connectivity between these habitats (Able et al. 2006).
Among these numerous sources, the importance of the near-surface water layer to larval and juvenile fish is well documented in regard to its function as a potential transport mechanism (Forward and Tankersley 2001; Hare et al. 2006). Furthermore, as water depths decrease when approaching the shoreline, and in particular near estuarine sills, the near-surface assemblage occupies an increasing proportion of the water column (Marshall and Burchardt 2005). Together, all of the above can influence larval transport into or out of estuaries (e.g., Able and Fahay 2010; Hare et al. 2002). Despite this broad understanding of oceanographic processes and fish distribution, many aspects of larval and pelagic juvenile fish assemblages, and thus the functional significance of these habitats, are poorly known. The purpose of this work is to describe the membership and dynamics of the inner continental shelf assemblage and its reflection on the adjacent estuary, as associated with seasonal environmental dynamics. This provides a framework for further directed inquiry of the mechanisms that couple these habitats, and how they could change in the future. We sampled near-surface fishes in inner continental shelf waters off New Jersey during the summer and fall of 2005 and 2006 and compared these assemblages with simultaneous samples from an adjacent estuary in order to determine how larvae and pelagic juveniles from the continental shelf contribute to assemblages of young fishes in the estuary. We compared important characteristics of the environment in which samples were collected and compared sampled fishes among years and habitats, using multivariate techniques to determine if these could have accounted for assemblage differences.
Materials and Methods
Study Area
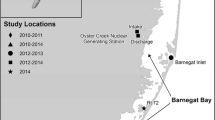
Inner shelf samples were collected along southern New Jersey from Manasquan Inlet to Cape May (Fig. 1). Water depths in the study area range from 3 to 29 m, with the shallowest depths occurring nearshore except for submarine sand ridges such as Beach Haven Ridge, at which the top of the ridge is as shallow as 8 m (Craghan 1995; Vasslides and Able 2008). Estuary samples were collected in Little Sheepshead Creek from a bridge 2.5 km behind Little Egg Inlet in the Great Bay-Little Egg Harbor estuary (Fig. 1). Water depth in Little Sheepshead Creek was 3–4 m. Tidal exchange in the estuarine study area is well described. A drifter study revealed that estuarine waters exiting Little Egg Inlet during ebb turns southward along the coast and remains nearshore (Charlesworth 1968). Atlantic Ocean water flows into the estuary through Little Egg Inlet during flood tides, with portions diverted into the mouth of Little Sheepshead Creek (Chant et al. 2000; Charlesworth 1968). Previous work has shown that ichthyoplankton samples collected from Little Sheepshead Creek are representative of the estuarine assemblage (Chant et al. 2000; Neuman et al. 2002; Witting et al. 1999).
Map of the study area along the coast of New Jersey in the New York Bight. Filled circles represent Methot trawl sample locations on the inner shelf and the filled triangle is the location of the plankton net sampling in Little Sheepshead Creek in the Great Bay-Little Egg Harbor estuary. Inner shelf strata indicated as black lines at inshore (5.5–9.1 m), mid-shore (9.1–18.3 m), and offshore (18.3–27.4 m) depths
Field Sampling
Inner shelf sampling was conducted during the day in early summer (June and first half of July) of 2005, late summer (second half of July, August, and first half of September) of 2005 and 2006, and fall (second half of September and October) of 2006 (Table 1). The sampling was conducted in a stratified random design, with strata defined by bottom depth and latitude as in Byrne (1994) (Fig. 1). Surface salinity and temperature were measured with a handheld instrument (YSI Model 85, Yellow Springs Instruments, Yellow Springs, OH). Depth was determined with a video depth recorder (Furuno Model 256, Furuno, Hyogo, Japan).
Near-surface larvae and pelagic juveniles were sampled with a Methot trawl (Methot 1986) consisting of a square 5 m2 mouth and a 13 m long net (3.5 mm mesh) leading to a tapering canvas collar and 0.165 m diameter (3.5 mm mesh) cod end. At each station, the trawl was towed for 10 min in a large arc to avoid the wake of the research vessel. All tows were made at the surface (0–2 m depth) with the top of the net frame approximately 0.25 m out of the water. A General Oceanics flowmeter (General Oceanics, Inc., Miami, FL) was attached within the frame so that the volume of water sampled could be determined (Taylor et al. 2006). Frame trawls appear efficient at capturing small juvenile fishes based on in situ observations (Itaya et al. 2001). In both years, fish were preserved in 95% ethanol. For large samples, most of the abundant gelatinous zooplankton (scyphomedusa, ctenophores, and larvaceans) were removed immediately. The remaining sample was sorted for fish in the laboratory.
Sampling of larvae and pelagic juvenile fish in the Great Bay-Little Egg Harbor estuary has occurred on a weekly basis year-round since 1989 (Able and Fahay 2010; Witting et al. 1999). A 1-m diameter, circular plankton net (1 mm mesh) fitted with a General Oceanics flowmeter is suspended from a bridge over Little Sheepsheads Creek to a depth of 1.5 m during night-time flood tides in these well-mixed waters (Table 1; Fig. 1). For the study years, three 30-min sets in each deployment were combined for the purpose of this analysis. Surface water temperature was measured with a field thermometer and salinity with a refractometer. While estuarine and inner shelf sampling did not occur on the same day/night in all cases, the regular estuarine sampling routine ensured that these samples were collected within 5 days (+/−) of inner shelf sampling.
Laboratory Procedures
Fish collected in inner continental shelf sampling were processed and later identified to lowest possible taxon and measured to the nearest 0.1-mm standard length (SL) under a dissecting microscope. Larger juveniles were measured to the nearest 1.0-mm fork length or total length (TL) and then converted to SL (see Appendix B in Able and Fahay 1998). Following each estuarine sampling set, samples were immediately sorted in shallow pans and fish were preserved in 95% ethanol. Each fish was then identified and measured to the nearest 0.1-mm SL under a dissecting microscope (see Able et al. 2006). The identification of Urophycis was problematic because of their presence in large numbers in many of the hauls and the possibility of three species occurring in the samples (Able and Fahay 1998). Specimens of Urophycis >7.0 mm that had full dorsal fin development were identified to species based on the number of second dorsal fin rays (Comyns and Grant 1992; Fahay 1983). Scientific nomenclature and the sequence in which families are listed generally follow Nelson et al. (2004). Departures from the latter classification include that for the Pleuronectiformes, where we follow Chapleau (1993) and Cooper and Chapleau (1998), and the Gadiformes, based on Fahay and Markle (1984).
Data Analysis
To test for differences between temperature, salinity, and water density in each season, an analysis of variance (ANOVA) with the Tukey adjustment for pairwise comparisons was completed. All significance tests assumed alpha = 0.05. In addition, several multivariate techniques were utilized to describe the near-surface assemblage structure. Density of fish (number of individuals per 1,000 m3) captured was determined for each fish species in each tow for the Methot trawls on the inner shelf and for each plankton net deployment (three pooled tows; total number captured over three sets/total volume of water filtered over three sets) in the estuary. Empty samples and a sample containing only one individual of one species were removed as they do not contribute to our understanding of the similarity (or difference) between samples. Data were fourth root transformed to reduce the effect of very abundant species in relation to rare ones in multivariate analysis (Clarke and Warwick 2001). A sample similarity matrix was constructed using the Bray Curtis index and depicted as a non-metric multidimensional scaling (MDS) ordination. This technique orders samples by rank similarity along their two most important latent gradients. MDS has advantages over other ordination techniques in that it requires no assumptions about the distribution of fish density, and it has a greater ability to accurately represent complex relations among samples in two-dimensional space (Clarke and Warwick 2001). This same matrix was then utilized in an analyses of similarity (ANOSIM) to evaluate the degree of similarity/dissimilarity between the gears in each season, and the similarity of percentages procedure was then used to determine which species contributed most to the differences between the habitats within each season. Lastly, dominant taxa characterizing assemblages from each habitat during each season were depicted using dominance plots. In order to assess the impact of utilizing different diameter meshes between the ocean and the estuary, the relative abundances of 5-mm size classes (0–135 mm) were determined for each Methot trawl tow and plankton net deployment. A resemblance matrix was then constructed based on Euclidean distance between the samples and utilized in the ANOSIM procedure as described above.
The environmental data (surface salinity, surface temperature, water depth, day of the year, location along a north–south axis, and distance from the shoreline) were standardized and centered. A second sample similarity matrix was then constructed based on the Euclidean distances between the samples but this time based on environmental data independent of species inclusion. The sample ranks of this environmental similarity matrix and the earlier Bray–Curtis fish abundance matrix were compared using a Mantel test, where a rank correlation coefficient between −1 ≤ ρ ≤ 1 is calculated, with the negative end indicating complete opposition and the positive end indicating complete agreement (BVSTEP procedure, selecting for the Spearman’s rank correlation method). Statistical analysis applied SAS (v9.1, SAS Inst., Inc., Cary, NC) and Primer-E (v6, Plymouth Marine Laboratory, Plymouth, UK).
Results
Environmental Characteristics
Mean seasonal surface water temperatures were generally consistent across years and inner shelf and estuarine habitats and ranged from 15.5°C to 26°C during the summer and fall sampling period (Fig. 2a). Surface water temperatures were similar between years for each habitat in a given season with the exception that ocean temperatures in fall differed between years (Table 2). Mean seasonal surface salinity ranged from 27.8 to 30.8 in the ocean and 27.1 to 30.9 in the estuary, with the overall pattern varying between habitat and year (Fig. 2b). Significant differences in salinity between years were found in the ocean in the fall and in the estuary in early summer (Table 2). Together, the changes in salinity and temperature yielded significantly different water density in early and late summer for both habitats but not in fall for either habitat.
Mean seasonal temperature (a), salinity (b), and fish density (c) by year and habitat (inner shelf vs. estuary). See Fig. 1 for sampling locations and text for rationale for seasonal periods
Larval and Juvenile Fish Characteristics
Forty taxa from 30 genera were captured in inner continental shelf samples in the summer and fall, with Anchoa mitchilli, Brevoortia tyrannus, Urophycis regia, Etropus microstomus, Peprilus triacanthus, and Pomatomus saltatrix, all exceeding densities of 1.0 per 1,000 m3 in at least one season for all years combined (Table 3). Fifty-three taxa from 41 genera were captured in the estuary over the same seasons, with A. mitchilli, Micropogonias undulatus, Syngnathus fuscus, Gobiosoma bosc, B. tyrannus, and Menidia menidia among the most abundant and 13 others exceeding a density of 1.0 per 1,000 m3 in at least one season for all years combined (Table 3).
Mean fish density varied between habitats and seasons (Fig. 2c). An ANOVA revealed that there were no significant differences between years for each habitat and season combination, allowing for the pooling of years. For both habitats, the early summer sampling period had the lowest mean density (Methot trawl on the inner shelf = 5.6 individuals per 1,000 m3 and plankton net in estuary = 174.7 individuals per 1,000 m3), with U. regia as the dominant species on the inner shelf and S. fuscus and engraulids (A. mitchilli and Anchoa spp.) dominating in the estuary (Table 3; Fig. 3). The occurrence of numerous A. mitchilli, M. undulatus, Anchoa spp., and G. bosc in late summer greatly increased the diversity of fishes in the estuary at that time, and these persisted to a lesser extent into fall, dominating the estuarine assemblage (Table 3). This seasonal trend was not reflected on the inner shelf, where A. mitchilli and M. undulatus were outnumbered in late summer by E. microstomus and P. triacanthus. In the fall, A. mitchilli were abundant and dominant on the inner shelf, possibly a consequence of their migration out of the estuary (Table 3).
Individuals ranging from 6 to 132 mm were captured on the inner shelf (n = 2,840) and individuals ranging from 2.6 to 139 mm were captured in the estuary (n = 5,936) (Fig. 4). The overall pattern in length frequency distribution between the two habitats differed significantly (ANOSIM Global R = 0.033, p = 0.0189). The smallest fish (2–3 mm) were captured exclusively in the estuary with the plankton net. The modal size in the estuary was also smaller (8 mm) than that on the inner shelf (14 mm). Comparison across seasons shows that B. tyrannus and U. regia were the most abundant species around the Methot trawl length frequency mode (14 ± 2 mm) and M. undulatus, A. mitchilli, and G. bosc were the most abundant around the plankton net length frequency mode (8.0 ± 2 mm). The density of all larvae was generally greater in the estuary across all weeks (Fig. 2c) and especially for sizes smaller than 40 mm (Fig. 4).
Inner Shelf vs. Estuary Assemblages
Species assemblages were differentiated by season and habitat, with year having limited influence (Fig. 5). Estuarine samples scored similar to each other along MDS axes but were differentiated from inner shelf samples (Fig. 5a). The ANOSIM procedure quantified sample overlap at R = 0.397 and this was significant (p = 0.001). A number of abundant species occurred in both years, as reflected by high overlap in samples displayed by year (Fig. 5b) and a low non-significant R value of 0.016 (p = 0.123) in ANOSIM. Seasonal differentiation (ANOSIM (R = 0.353, p = 0.001)) is more apparent than annual differentiation and is similar to that of the habitat score (Fig. 5c). Assemblage differences between habitats within each season were pronounced and driven by the presence (or high abundance) of a suite of species in one habitat and their absence (or near absence) in the other (Tables 3 and 4). In early summer, the dissimilarity between assemblages was driven by the abundances of S. fuscus, B. tyrannus, Menidia sp., S. aquosos, and A. mitchilli and Anchoa sp. in the estuary and U. regia on the inner shelf. In late summer, A. mitchilli, Anchoa sp., S. fuscus, G. bosc, M. undulatus, and E. microstomus abundances in the estuary accounted for over 50% of the differences between the assemblages. In the fall, higher estuarine abundances of A. mitchilli, M. undulatus, and C. boleosoma along with the increased inner shelf abundances of B. tyrannus combined to differentiate the estuary assemblage from that of the inner shelf. The environmental variable that best correlated with the species distribution was distance from the shoreline (R = 0.191). The remaining environmental variables did not contribute significantly to the species-environment correlation.
Discussion
Environmental Characteristics
The near-surface waters sampled in the estuary were similar to the inner shelf in hydrography within seasons and year, suggesting a common origin and limited physiological constraints for the larval fish assemblages in these two adjacent systems. However, the freshwater input into Great Bay was occasionally reflected in slightly lower salinities in the estuary than on the inner shelf due to rainfall events (>30 mm of rain in a 24-h period) in the days preceding sampling. Actual daily differences in salinity were minimal because the estuary sampling regime was limited to incoming tides which transported inner shelf water into the estuary. Another hydrographic source of potential connectivity between the inner shelf and the estuary is upwelling. Typically, during summer thermal stratification, winds from the southwest result in upwelling of cold water along the New Jersey coast, and this is well documented at Little Egg Inlet (Glenn et al. 1996; Neuman 1996; Neuman et al. 2002). During the summer and fall of the years studied, there was no evidence of upwelling; however in 2006 there was a prolonged period of cooler water that encompassed a single week of our sampling (see http://rucool.marine.rutgers.edu/index.php/COOL-Data/COOL-Data.html). While this temperature dip represented a departure from the regional water temperatures, it affected both sampling areas, and therefore may not have affected the comparison of samples between the two locations. Prior analysis of estuarine larvae before, during and after upwelling events produced mixed results relative to faunal group and species but no pronounced effect of upwelling at this site (Able and Fahay 2010; Neuman et al. 2002).
Ichthyoplankton Characteristics
The similarity in occurrences and range of lengths of species collected on the inner shelf and in the estuary suggested that both sampling approaches were effective in collecting the pelagic stages of larval and juvenile fishes. The difference in magnitude of fish concentrations was likely due primarily to the high abundance of A. mitchilli larvae in the estuary resulting from intense spawning during summer (Able and Fahay 2010; Witting et al. 1999). Overall, the similarities in the temporal pattern of abundance and overlapping length composition between the inner continental shelf and adjacent estuary suggest that many aspects of the inner continental shelf and estuarine larval and pelagic juvenile fish fauna are shared across this geographic scale, as occurs for benthic juvenile fishes (Able et al. 2006; Martino and Able 2003). However, peak density on the inner shelf occurred in fall compared to late summer in the estuary, due to increases in the abundance of continental shelf spawners such as Urophycis spp. and P. saltatrix (Able and Fahay 2010; Morse et al. 1987). Conversely, one of the late summer estuary dominants, G. bosc, is known to be primarily a summer estuarine spawner (Able and Fahay 2010). Another dominant species, C. regalis, spawns in both the estuary and the inner shelf (Lowerre-Barbieri et al. 1996).
This analysis identifies several shared species between the estuary and the inner shelf. The habitats are closely related, in part, as the result of ontogenetic movements of larvae between these habitats and settlement of several species with subsequent removal/loss from the ichthyoplankton assemblages. For example, one species generally identified as a shelf spawner, M. undulatus (smallest described at 2.0 mm), appeared in our samples with a distribution concentrated in the estuary, though still substantially abundant on the inner shelf in fall. Previous studies on the continental shelf found larval M. undulatus across the entire shelf of the Middle Atlantic Bight with a northern limit at Delaware Bay (Able and Fahay 1998). However, the M. undulatus larvae we collected in this study were relatively small (<18.7 mm TL), suggesting that spawning occurred north of Delaware Bay. This is consistent with reports of numerous mature adults off New Jersey and increasing larval abundance in the estuary in recent years (Hare and Able 2007). Elsewhere, larval M. undulatus have been found distributed in association with the Chesapeake Bay plume water (Reiss and McConaugha 1999). Another inner shelf spawner with a broad distribution but abundance highest in the estuary was B. tyrannus. The consistent patterns for these and other species likely reflect movement from the inner shelf into estuaries (Able and Fahay 1998, 2010; Warlen et al. 2002). Other species can be found both in the inner shelf and the estuary (e.g., S. fuscus, S. aquosus, and A. mitchilli) (Able and Fahay 2010). Individually, the dominant taxa revealed substantial dynamic variation in their patterns of abundance/length but still present a clearer picture of potential spawning and larval and pelagic juvenile distributions for a variety of fishes on the inner continental shelf than previous shelf studies (Cowen et al. 1993; Morse et al. 1987).
Furthermore, the sources of larvae and juveniles are variable. In some cases, larvae from south of Cape Hatteras are transported north into the study area (Grothues et al. 2002; Hare and Cowen 1991, 1996; Warlen et al. 2002). In other cases, some larvae, such as those for Clupea harengus, may be transported south from more northern areas and into the sampling area (Able and Fahay 1998, 2010). For pelagic juveniles, their distribution is the result of their swimming ability (Boehlert and Mundy 1988; Hare et al. 1999). For example, some juvenile P. saltatrix may rapidly swim across the continental shelf following the breakdown of the shelf-slope front (Hare and Cowen 1996, 1997) and then into estuaries.
Some caution should be used in interpreting the data from this study because samples from the deeper portions of the inner continental shelf were limited to near-surface and thus may not be able to characterize all components of the larval and pelagic juvenile fish fauna. As an example, in another study in the same area, which focused on surfzone and nearshore collections during May–July (Able et al. 2010), smaller larvae collected included several dominant species not found in this study including Tautoga onitis, Lophius americanus, Prionotus evolans, Enchelyopus cimbrius, Hippoglossina oblonga, and Prionotus carolinus. These differences in species composition are likely the result of smaller mesh sizes and earlier sampling (May) in that study. Another possible source of variation is the potential difference between sampling on the inner shelf during the day and the estuary at night. While avoidance of sampling gear during the day is presumably more problematic, the larger Methot trawl may have reduced gear avoidance (Itaya et al. 2001) and thus lessened potential bias in day versus night sampling.
Inner Shelf vs. Estuary Assemblages
This study revealed a seasonal turnover in both estuarine and inner shelf fish species assemblages from early summer through fall. A similar transition period was also reported in earlier accounts of the Little Egg Inlet and adjacent estuary based on some of the same larval (Martino and Able 2003; Witting et al. 1999) and juvenile (Hagan and Able 2003) taxa. Seasonal components, i.e., temperature, and day of the year, appear to most influence the available pool of species while distance from shore serves as a means of separating the species distribution along an estuary to inner shelf gradient. This separation of taxa along an inshore-offshore direction is still apparent when sampling occurs at larger scales (10–100 km, Cowen et al. 1993). Like the present study, Cowen et al. (1993) categorized Anchoa spp. as belonging to the coastal assemblage but classified P. saltatrix, P. triacanthus, Urophycis spp., and E. microstomus as part of the deeper, shelf assemblage (see also Reiss and McConaugha 1999) although they also were consistent components of inner shelf samples in this study. Species behavior may account, in part, for these differences. Larger larvae and pelagic juveniles may be more capable of crossing hydrographic boundaries (Boehlert and Mundy 1988; Hare and Cowen 1996; Hare et al. 2005) and thus changing the composition of the assemblage with respect to water mass (Cowen et al. 1993). The factors influencing the ability to cross hydrographic boundaries on the continental shelf may also influence ingress of larvae and pelagic juveniles from the inner shelf into the estuary. Near inlets, including Little Egg Inlet, tidal effects play an important role in advecting water (Chant et al. 2000; Charlesworth 1968) in addition to the dominant forces driving flow on the inner shelf, namely buoyancy and alongshore winds (Epifanio and Garvine 2001; Reiss and McConaugha 1999).
As seen in this study, some shelf spawned taxa such as Urophycis spp. and P. triacanthus are important components of the inner or mid-shelf assemblage (Martino and Able 2003; Steves et al. 1999; Vasslides and Able 2008) but do not become conspicuous components of the estuarine larval and benthic juvenile assemblage (Hagan and Able 2003; Martino and Able 2003; Witting et al. 1999). Other species, like M. undulatus and E. microstomus, enter inlets and do become common components of the estuarine larval and benthic juvenile assemblage (Able et al. 2006; Martino and Able 2003; Witting et al. 1999). Thus, on spatial scales of 1–10 km, the larval fish assemblage adjacent to inlets may remain segregated into inner shelf and outer shelf components, with species crossover dependent on physical processes modified by behavior or transport of recently hatched individuals (e.g., Forward and Tankersley 2001; Govoni and Pietrafesa 1994; Reiss and McConaugha 1999). These crossovers may result in ingress to the estuary, and therefore contribute to the annually consistent, seasonal ingress of larval fishes through Little Egg Inlet into Great Bay (Able et al. 2006; Able and Fahay 2010; Witting et al. 1999). Although the physical processes driving circulation nearshore (within 20 km) have different temporal and spatial scales than those discussed by Cowen et al. (1993) on the outer shelf and continental slope, each may be important in shaping the larval assemblage. The comparisons at the species level can be confounded because some of the dominant species (e.g., U. regia, P. triacanthus, and P. saltatrix) are represented by multiple cohorts which have independent spawning seasons and perhaps different transport processes and behaviors that are cued to the different seasons in which they are produced (Able and Fahay 1998, 2010).
In summary, the collection of near-surface larval and pelagic juvenile fishes sampled on the inner continental shelf and an adjacent estuary through the summer and fall provides further clarification of the coastal assemblage previously described by Cowen et al. (1993) and helps differentiate those taxa that spawn in estuaries and on the inner shelf from those that spawn in deeper water during this time of the year. The inner shelf assemblage shares many species with the assemblage found in estuaries because (1) many species occupy both estuarine and shelf habitats, (2) others leave the inner shelf and enter the estuary to settle into juvenile habitats, and (3) some larvae that result from spawning in the estuary may be outwelled onto the inner shelf with the buoyant plumes typical of these systems.
References
Able, K.W., and M.P. Fahay. 1998. The first year in the life of estuarine fishes in the Middle Atlantic Bight. New Brunswick: Rutgers University Press.
Able, K.W., and M.P. Fahay. 2010. Ecology of estuarine fishes: temperate waters of the western north Atlantic. Baltimore: The Johns Hopkins University Press.
Able, K.W., M.P. Fahay, D.A. Witting, R.S. McBride, and S.M. Hagan. 2006. Fish settlement in the ocean vs. estuary: Comparison of pelagic larval and settled juvenile composition and abundance from southern New Jersey, USA. Estuarine, Coastal and Shelf Science 66: 280–290.
Able, K.W., D.H. Wilber, A. Muzeni-Corino, and D.G. Clarke. 2010. Spring and summer larval fish assemblages in the surf zone and nearshore off northern New Jersey, USA. Estuaries and Coasts 33: 211–222.
Boehlert, G.W., and B.C. Mundy. 1988. Roles of behavioral and physical factors in larval and juvenile fish recruitment to estuarine nursery areas. American Fisheries Society Symposium 3: 51–67.
Byrne, D. 1994. Stock assessment of New Jersey’s nearshore recreational fisheries resources. In Proceedings of the workshop on the collection and use of trawl survey data for fisheries management, ed. T. Berger, 36–42. Washington: Atlantic States Marine Fisheries Commission.
Chant, R.J., M.C. Curran, K.W. Able, and S.M. Glenn. 2000. Delivery of winter flounder (Pseudopleuronectes americanus) larvae to settlement habitats in coves near tidal inlets. Estuarine, Coastal and Shelf Science 51: 529–541.
Chapleau, F. 1993. Pleuronectiform relationships: A cladistic reassessment. Bulletin of Marine Science 52(1): 516–540.
Charlesworth, L.J., Jr. 1968. Bay, inlet and nearshore marine sedimentation: Beach Haven-Little Egg Inlet region, New Jersey (260 pp). Ph.D. dissertation, The University of Michigan.
Clarke, K.R., and R.M. Warwick. 2001. Change in marine communities: An approach to statistical analysis and interpretation, 2nd ed. Plymouth: PRIMER-E.
Colvocoresses, J.A., and J.A. Musick. 1984. Species associations and community composition of Middle Atlantic Bight continental shelf demersal fishes. Fishery Bulletin 82: 295–313.
Comyns, B.H., and G.C. Grant. 1992. Identification and distribution of Urophycis and Phycis (Pisces, Gadidae) larvae and pelagic juveniles in the U.S. Middle Atlantic Bight. Fishery Bulletin 91: 210–223.
Cooper, J.A., and F. Chapleau. 1998. Monophyly and relationships of the family Pleuronectidae (Pleuronectiformes), with a revised classification. Fishery Bulletin 96(4): 686–726.
Cowen, R.K., J.A. Hare, and M.P. Fahay. 1993. Beyond hydrography: can physical processes explain larval fish assemblages within the Middle Atlantic Bight? Bulletin of Marine Science 53(2): 567–587.
Craghan, M. 1995. Topographic changes and sediment characteristics at a shoreface sand ridge—Beach Haven Ridge, New Jersey (96 pp.). Master’s thesis, Rutgers University.
Epifanio, C.E., and R.W. Garvine. 2001. Larval transport on the Atlantic continental shelf of North America: A review. Estuarine, Coastal and Shelf Science 52: 51–77.
Fahay, M.P. 1983. Guide to early stages of marine fishes occurring in the western North Atlantic Ocean, Cape Hatteras to the southern Scotian Shelf. Journal of Northwest Atlantic Fisheries Science 4: 1–423.
Fahay, M.P., and D.F. Markle. 1984. Gadiformes: development and relationships. In Ontogeny and systematics of fishes, eds. H.G. Moser, W.J. Richards, D.M. Cohen, M.P. Fahay, A.W. Kendall, Jr., and S.L, Richardson, 265–283. American Society of Ichthyologists and Herpetologists Special Publication No. 1. Lawrence: Allen Press.
Forward Jr., R.B., and R.A. Tankersley. 2001. Selective tidal-stream transport of marine animals. Oceanography and Marine Biology: An Annual Review 39: 305–353.
Glenn, S.M., M.F. Crowley, D.B. Haidvogel, and Y.T. Song. 1996. Underwater observatory captures coastal upwelling events off New Jersey. Transactions of the American Geophysical Union 77(25): 233–236.
Govoni, J.J., and C.B. Grimes. 1992. The surface accumulation of larval fishes by hydrodynamic convergence within the Mississippi River plume front. Continental Shelf Research 12(11): 1265–1276.
Govoni, J.J., and L.J. Pietrafesa. 1994. Eulerian views of layered water currents, vertical distribution of some larval fishes, and inferred advective transport over the continental shelf off North Carolina, USA, in winter. Fisheries Oceanography 3(2): 120–132.
Grosslein, M.D., and T.R. Azarovitz. 1982. Fish distribution. MESA New York Bight Atlas Monograph 15. Albany: New York Sea Grant Institute.
Grothues, T.M., R.K. Cowen, L.J. Pietrafesa, F. Bignami, G.L. Weatherly, and C.N. Flagg. 2002. Flux of larval fish around Cape Hatteras. Limnology and Oceanography 47(1): 165–175.
Hagan, S.M., and K.W. Able. 2003. Seasonal changes of the pelagic fish assemblage in a temperate estuary. Estuarine, Coastal and Shelf Science 56(1): 15–29.
Hare, J.A., and K.W. Able. 2007. Mechanistic links between climate and fisheries along the east coast of the United States: Explaining population outbursts of Atlantic croaker (Micropogonias undulatus). Fisheries Oceanography 16(1): 31–45.
Hare, J.A., and R.K. Cowen. 1991. Expatriation of Xyrichtys novacula (Pisces: Labridae) larvae: Evidence of rapid cross-slope exchange. Journal of Marine Research 49: 801–823.
Hare, J.A., and R.K. Cowen. 1996. Transport mechanisms of larval and pelagic juvenile bluefish (Pomatomus saltatrix) from South Atlantic Bight spawning grounds to Middle Atlantic Bight nursery habitats. Limnology and Oceanography 41(6): 1264–1280.
Hare, J.A., and R.K. Cowen. 1997. Size growth, development, and survival of the planktonic larvae of Pomatomus saltatrix (Pisces: Pomatomidae). Ecology 78(8): 2415–2431.
Hare, J.A., J.A. Quinlan, F.E. Werner, B.O. Blanton, J.J. Govoni, R.B. Forward, L.R. Settle, and D.E. Hoss. 1999. Larval transport during winter in the SABRE study area: results of a coupled vertical larval behaviour-three-dimensional circulation model. Fisheries Oceanography 8(2 Suppl.): 57–76.
Hare, J.A., J.H. Churchill, R.K. Cowen, T.J. Berger, P.C. Cornillon, P. Dragos, S.M. Glenn, J.J. Govoni, and T.N. Lee. 2002. Routes and rates of larval fish transport from the southeast to the northeast United States continental shelf. Limnology and Oceanography 47(6): 1774–1789.
Hare, J.A., S. Thorrold, H. Walsh, C. Reiss, A. Valle-Levinson, and C. Jones. 2005. Biophysical mechanisims of larval fish ingress into Chesapeake Bay. Marine Ecology Progress Series 303: 295–310.
Hare, J.A., H.J. Walsh, and M.J. Wuenschel. 2006. Sinking rates of late-stage fish larvae: Implications for larval ingress into estuarine nursery habitats. Journal of Experimental Marine Biology and Ecology 330(2): 493–504.
Itaya, K., Y. Fujimori, D. Shiode, I. Aoki, T. Yonezawa, S. Shimizu, and T. Miura. 2001. Sampling performance and operational quality of a frame trawl used to catch juvenile fish. Fisheries Science 67: 436–443.
Kingsford, M.J., and I.M. Suthers. 1994. Dynamic estuarine plumes and fronts: importance to small fish and plankton in coastal waters of NSW, Australia. Continental Shelf Research 14(6): 655–672.
Lowerre-Barbieri, S.K., M.E. Chittenden Jr., and L.R. Barbieri. 1996. The multiple spawning pattern of weakfish in the Chesapeake Bay and Middle Atlantic Bight. Journal of Fish Biology 48: 1139–1163.
Marshall, H.G., and L. Burchardt. 2005. Neuston: its definition with a historical review regarding its concept and community structure. Archiv fuer Hydrobiologie 164(4): 429–448.
Martino, E., and K.W. Able. 2003. Fish assemblages across the marine to low salinity transition zone of a temperate estuary. Estuarine, Coastal and Shelf Science 56(5–6): 967–985.
Methot, R.D. 1986. Frame trawl for sampling pelagic juvenile fish. California Cooperative Oceanic Fisheries Investigations 27: 267–278.
Morse, W.W., M.P. Fahay, and W.G. Smith. 1987. MARMAP surveys of the continental shelf from Cape Hatteras, North Carolina to Cape Sable, Nova Scotia (1977–1984). Atlas No. 2 Annual distribution patterns of fish larvae. National Oceanic and Atmospheric Administration, Technical Memorandum NMFS-F National Marine Fisheries Service. Northeast Fisheries Science Center 47: 1–215.
Munchow, A., and R.J. Chant. 2000. Kinematics of inner shelf motions during the summer stratified season off New Jersey. Journal of Physical Oceanography 30: 247–268.
Nelson, J.S., E.J. Crossman, H. Espinosa-Perex, L.T. Findley, C.R. Gilbert, R.N. Lea, and J.D. Williams. 2004. Common and scientific names of fishes from the United States, Canada, and Mexico. American Fisheries Society Special Publication 29. Bethesda, Maryland.
Neuman, M. 1996. Evidence of upwelling along the New Jersey coastline and the south shore of Long Island, New York. Bulletin of the New Jersey Academy of Science 41(1): 7–13.
Neuman, M.J., K.W. Able, and S.M. Glenn. 2002. The effects of upwelling on larval fish occurrence and abundance in the Jacques Cousteau National Estuarine Research Reserve at Mullica River-Great Bay (JCNERR). Institute of Marine and Coastal Sciences, Jacques Cousteau Technical Report #100-17. New Brunswick: Rutgers University.
Raynie, R.C., and R.F. Shaw. 1994. Ichthyoplankton abundance along a recruitment corridor from offshore spawning to estuarine nursery ground. Estuarine, Coastal and Shelf Science 39: 421–450.
Reiss, C.S., and J.R. McConaugha. 1999. Cross-frontal transport and distribution of ichthyoplankton associated with Chesapeake Bay plume dynamics. Continental Shelf Research 19: 151–170.
Steves, B.P., R.K. Cowen, and M.H. Malchoff. 1999. Settlement and nursery habitats for demersal fishes on the continental shelf of the New York Bight. Fishery Bulletin 98: 167–188.
Strydom, N.A., and B.D. Hartman. 2005. Estuary-dependence of larval fishes in a non-estuary associated South African surf zone: evidence for continuity of surf assemblages. Estuarine, Coastal and Shelf Science 63: 101–108.
Taylor, D.T., P.M. Rowe, and K.W. Able. 2006. Habitat use of the inner continental shelf off southern New Jersey by summer-spawned bluefish (Pomatomus saltatrix). Fishery Bulletin 104: 593–604.
Tzeng, W.-N., Y.-T. Wang, and C.-W. Chang. 2002. Spatial and temporal variations of the estuarine larval fish community on the west coast of Taiwan. Marine and Freshwater Research 53: 419–430.
Vasslides, J.M., and K.W. Able. 2008. Importance of shoreface sand ridges as habitat for fishes off the northeast coast of the United States. Fishery Bulletin 106: 93–107.
Warlen, S.M., K.W. Able, and E. Laban. 2002. Recruitment of larval Atlantic menhaden (Brevoortia tyrannus) to North Carolina and New Jersey estuaries: evidence for larval transport northward along the east coast of the United States. Fishery Bulletin 100(3): 609–623.
Witting, D.A., K.W. Able, and M.P. Fahay. 1999. Larval fishes of a Middle Atlantic Bight estuary: assemblage structure and temporal stability. Canadian Journal of Fisheries and Aquatic Science 56: 222–230.
Wong, K.-C. 1999. The wind driven currents on the Middle Atlantic Bight inner shelf. Continental Shelf Research 19: 757–773.
Yankovsky, A.E., R.W. Garvine, and A. Munchow. 2000. Mesoscale currents on the inner New Jersey shelf driven by the interaction of buoyancy and wind forcing. Journal of Physical Oceanography 30(9): 2214–2230.
Zhang, W., J. Wilkin, and R. Chant. 2009. Modeling of the pathways and mean dynamics of river plume dispersal in New York Bight. Journal of Physical Oceanography 39: 1167–1183.
Acknowledgments
We appreciate the experience of Captain J. Zlotnik and R. Hoden while sampling aboard R/V Arabella. G. Bell, D. Bottinelli, B. Rokeach, R. Nichols, J. Conwell, and M. Greaney assisted in the collection and laboratory workup of samples. A. Muzeni and J. Lamonaca provided expertise in larval fish identification. R. Hagan and S. Zeck collected many of the estuary samples. Anonymous reviewers and an associate editor markedly improved the quality of the manuscript with careful comments. This study was funded through a Bluefish/Striped Bass Research Program (Rutgers University and National Marine Fisheries Service collaborative program). We are grateful to all of the above. This paper is contribution no. 2011-2 of the Rutgers University Institute of Marine and Coastal Sciences.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Able, K.W., Grothues, T.M., Rowe, P.M. et al. Near-Surface Larval and Juvenile Fish in Coastal Habitats: Comparisons Between the Inner Shelf and an Estuary in the New York Bight During Summer and Fall. Estuaries and Coasts 34, 726–738 (2011). https://doi.org/10.1007/s12237-011-9399-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12237-011-9399-1