Abstract
The controlled aliasing in parallel imaging results in higher acceleration (CAIPIRINHA) technique can decrease scan time. The purpose of this study was to determine whether an arterial phase scan can be performed in 5 s using the CAIPIRINHA short-scan and a reference scanning technique. The generalized autocalibrating partially parallel acquisition (GRAPPA), the CAIPIRINHA routine (CAIPI-routine), and the CAIPIRINHA short-scanning (CAIPI-short) methods were compared. The scan time for each method was preset to 20 s, 15 s, and 10 s, respectively. The reference scan had a scan time of 5 s. A phantom study was used to compare the influence of artifacts during the reference scan. For comparison, the phantom was moved during the last 5 s. In the clinical studies of suspected chronic liver diseases, magnetic resonance imaging of the liver is usually performed while the patient is breath-hold. The motion artifacts of each method were compared. Artifacts were reduced in reference scans using the CAIPIRINHA method. At 5 s after initiation, the rate of change in the standard deviation value was within 30% compared to that of the original image. Motion artifacts due to the influence of the reference scan when a patient failed to hold their breath did not complicate image evaluation. The proportion of motion artifacts for each sequence was as follows: GRAPPA, 5.8%; CAIPI-routine, 1.9%; and CAIPI-short, 0.7%. The arterial phase can be scanned in 5 s using the CAIPI-short and reference scan techniques.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
1 Introduction
The detection, characterization, and diagnosis of liver tumors are traditionally achieved through magnetic resonance imaging (MRI). For specific determination of the presence or absence of tumor vascularity, dynamic contrast material-enhanced (DCE) imaging is necessary [1,2,3]. Breath-hold DCE and hepatobiliary phase sequences are the key sequences required for performing an MRI of the liver. The image quality of the arterial phase of DCE can be significantly compromised by respiratory motion artifacts caused by incomplete breath-hold. According to previous reports, artifacts render the imaging results non-diagnostic at a rate ranging from 4.8 to 18% [4,5,6,7,8]. In previous studies, these probabilities were reduced [9].
Simultaneous acquisition of spatial harmonics (SMASH) and SENSitivity encoding (SENSE) are both parallel imaging techniques that have facilitated marked reductions in scan times [10, 11]. Generalized autocalibrating partially parallel acquisition (GRAPPA) was developed as a more progressive type of SMASH [12, 13]. GRAPPA uses an autocalibration signal by collecting more data in k-space, with improved accuracy and signal-to-noise ratio (SNR). Based on these improvements, the technique of controlled aliasing in parallel imaging results in higher acceleration (CAIPIRINHA) was developed [14, 15]. CAIPIRINHA samples data points in k-space while creating a shift that suppresses the increase in g-factor. Thus, even when using a high parallel factor during three-dimensional (3D) imaging, it is possible to suppress artifacts.
CAIPIRINHA is used in MRI examinations of the liver and allows for shorter scanning times [16]. This new parallel imaging technique has been used at various anatomical sites in previous studies [17, 18]. CAIPIRINHA allows for a shorter breath-hold time than does GRAPPA. Scan times and aliasing artifacts can be reduced with parallel imaging by setting a high acceleration factor (AF). In this approach, a reference scan is first conducted, and with experience, CAIPIRINHA can obtain good images with this technique without requiring a set breath-hold time during the reference scan. This fact is unknown in many hospitals. To determine whether the use of a reference scan technique with the CAIPIRINHA method facilitates shortening of breath-hold time, we conducted both phantom and clinical studies.
2 Materials and methods
This retrospective study was approved by our institutional review board, who waived the requirement for obtaining informed consent.
2.1 Phantom study
All MR images were acquired using a 1.5-T system (Magnetom Avanto Dot VD13, Siemens Healthcare, Erlangen, Germany) with a phased-array matrix coil (anterior) and a spine matrix coil (posterior) for signal reception. Scan parameters are shown in Table 1. The liver phantom was a cylinder with a diameter of 150 mm and a length of 150 mm. To produce hepatic T1 values, contrast material and agar was used in the phantom (T1-value = 520 ms; T2-value = 68 ms) [19]. A bottle phantom was fixed around the liver phantom (bottle phantom; NiSO4·6H2O, NaCl, length of 200 mm) (Fig. 1). Only the liver phantom was designed to move. Gadoxetate disodium (Primovist, Bayer Pharma, Osaka, Japan) was used as contrast agent.
Placement of phantom and schematic of the movement. a A bottle phantom was fixed around the phantom. b Movement was made in one reciprocation in head–foot direction for 2 s to measure the influence of movement on the phantom image (20 mm). The phantom was moved by a motor. a Placement of phantom (b) schematic of motion
To investigate the influence of a breath-hold, the phantom was moved in the head–foot direction (20 mm) for each of the imaging methods (Fig. 1). The timing of each move was as follows: A, 5 s after the start of the routine method (CAIPI-routine); B, 5 s before the end of the CAIPI-routine; C, 5 s after the start of the short-scanning method (CAIPI-short); D, 5 s before the end of the CAIPI-short; E, 5 s after the start of GRAPPA; and F, original image (i.e., the image in which the phantom was not moved was defined as the original image). Scans were performed using the GRAPPA, CAIPI-routine, and CAIPI-short methods. Axial-section imaging was performed, and the phase-encoding direction was anterior–posterior. To see the uniformity of the signal, the center of the phantom was measured with a region of interest (ROI) of 12 cm in diameters and standard deviation (SD) was also obtained. Average values were calculated from the respective timings of 10 imaging scans. Movement was made in one reciprocation for 2 s to measure the influence of movement on the phantom image in the head–foot direction (20 mm). The amount of movement of the phantom speed was determined in a previous study [20].
The phantom image at each time point (A–F) was evaluated, and the rates of change of SDs were compared. The ROI of the phantom center was used to measure SD. The rate of change of SD was evaluated using the average value. The rate of change of SD was defined as follows:
2.2 Clinical study
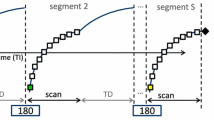
All patients were administrated gadoxetate disodium (Primovist; Bayer Pharma, Osaka, Japan) intravenously at a weight-based dose of 0.025 mmol/kg of body weight (0.1 mL/kg; maximum volume, 10 mL), followed by 30 mL of saline. All the injection rates were 1 mL/s. Arterial phase images were acquired using a real-time bolus display method (CARE Bolus, Siemens Healthcare) after visual detection of the contrast material at the celiac artery [4]. Routine gadoxetate disodium-enhanced liver MR examination, including transverse multi-phase contrast-enhanced T1-weighted breath-hold sequences, was conducted using a fat-suppressed 3D-volume interpolated breath-hold examination (VIBE). Scans were performed using a fat-suppressed 3D-VIBE sequence with different parallel imaging techniques: GRAPPA with acceleration factor (AF) = 2 and CAIPIRINHA with AF = 3 (three-fold acceleration in the partition-encoding direction). The scan time of the CAIPI-routine was set to 15 s. CAIPI-short had a scan time of 10 s. Both the CAIPI-routine and CAIPI-short began with a reference scan that comprised the first 5 s. The timing flow chart for each sequence is shown in Fig. 2.
Flow chart showing timing. The scan time of the GRAPPA was set to 20 s. This setting simultaneously collected the reference scan and main scan. The scan time of the CAIPI-routine was set to 15 s. CAIPI-short had a scan time of 10 s. Both the CAIPI-routine and CAIPI-short methods began with a reference scan during the first 5 s. a Integrated of GRAPPA (b) CAIPI-routine (c) CAIPI-short. GRAPPA generalized autocalibrating partially parallel acquisition; CAIPI-routine, CAIPIRINHA routine method; CAIPI-short, CAIPIRINHA short-scanning method
The integrated of the GRAPPA method could not separate the reference scan from the remaining scan; however, the CAIPIRINHA technique allowed such separation. The slice thickness of CAIPI-routine scans was preset at 2.5 mm, and that of CAIPI-short scans at 3.5 mm. Scan times for the GRAPPA, CAIPA-routine, and CAIPI-short were 20, 15, and 10 s, respectively. Other parameters are shown in Table 1.
Between October 2013 and April 2016, a total of 554 patients with suspected focal liver lesions underwent gadoxetate disodium-enhanced liver MRI. The numbers of patients who underwent GRAPPA, CAIPI-routine, and CAIPI-short were 207, 206, and 141, respectively. The numbers of cirrhosis patients who underwent GRAPPA, CAIPI-routine, and CAIPI-short were 110, 101, and 90, respectively. The numbers of first experience of examination who underwent GRAPPA, CAIPI-routine, and CAIPI-short were 71, 49, and 39, respectively. The experience of examination was first or not, and the change in image quality was compared. Other study populations are shown in Table 2. This investigation was performed retrospectively. Patient images were assigned either to non-transient severe motion (TSM) or the TSM group, according to the criteria listed below.
The image quality of the GRAPPA, CAIPI-routine, and CAIPI-short methods obtained during the arterial phase was compared. Motion artifacts from the evaluations were also compared. For all 554 patients, images obtained in the dynamic arterial phases were anonymized, randomized, and reviewed by two readers, each with more than 20 years of experience in interpretation of abdominal MR images.
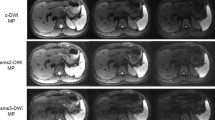
Each arterial phase image was rated on a five-point scale as follows: 5 = no motion artifact, 4 = minimal motion artifact, 3 = moderate motion artifact, 2 = severe motion artifact, and 1 = extensive motion artifact (Fig. 3). TSM was defined by a mean motion score of at least 2. Motion artifact scores were averaged across the two readers to produce a mean motion score for each arterial phase.
Image analysis according to motion artifacts. Each arterial phase image was rated on a five-point scale as follows: a no motion artifact; b minimal motion artifact; c moderate motion artifact; d severe motion artifact; and e extensive motion artifact. Scores are A = 5, B = 4, C = 3, D = 2, and E = 1
2.3 Comparison of breath-hold and breath-hold shortening methods under clinical conditions
Next, clinical images of the first and follow-up examinations were compared. All examinations were performed using the CAIPI-routine. The breath-holding time from before the reference scan to the arterial phase of the DCE were defined as the all-breath-hold method (ABH) and the time in the middle of the reference scan was defined as the breath-hold shortening method (BHS). The first examination method used ABH, and the follow-up examinations used BHS (mean days ± SD, 206 ± 118 days; range 9–390 days). The timing of breath-hold of BHS and ABH is shown in Fig. 4. Motion artifact scores of these two conditions were also compared. The number of patients was 30, including 19 male and 15 cirrhosis patients. The median age, body weight, and body mass index (BMI) were 69 ± 11.3 years, 63 ± 13.2 kg, and 22.7 ± 3.6, respectively.
2.4 Statistical analysis
Statistical analysis was performed using SPSS Statistics for Windows, Version 17.0 (SPSS, Inc., Chicago, IL). The average values of the rate of change of SD were compared using the Wilcoxon signed-rank test. Patient characteristics and differences with respect to mean age, weight, and body mass index were compared using the Kruskal–Wallis test or Mann–Whitney U test. Differences in the first experience of examination and Child–Pugh score were compared using the χ2 test. Motion artifact scores of ABH and BHS were compared using the Wilcoxon signed-rank test. The two readers were compared using kappa coefficients. P value of 0.05 or smaller was considered significant for all the hypothesis tests. [5].
3 Results
3.1 Phantom study
Figure 5 shows the rate of change of SD for the results of the phantom study. At 5 s after the start of the CAIPI-routine and CAIPI-short scans (Fig. 5a, c), the rate of change of SD was within 30% of that of the original image. There were significant differences between the rate 5 s after the start and 5 s before the end of the CAIPI-routine (Fig. 5a, b) and CAIPI-short methods (Fig. 5c, d), respectively, (both, P < 0.01).
Rate of change of standard deviation. Rate of change of standard deviation (SD) is shown for the results of the phantom study. A significant difference was found between results at 5 s after initiation and 5 s before termination of scanning (CAIPI-routine: P < 0.01; CAIPI-short: P < 0.05). CAIPI-routine CAIPIRINHA routine method, CAIPI-short CAIPIRINHA short-scanning method, GRAPPA generalized auto-calibrating partially parallel acquisition, SD standard deviation
Integrated of GRAPPA was approximately 40% (E).
3.2 Clinical study
Tables show the motion artifact scores (Tables 3,4, and 5). TSMs for the various sequences were 5.8% (GRAPPA, 12/207), 1.9% (CAIPI-routine, 4/206), and 0.7% (CAIPI-short, 1/141). In addition, the kappa coefficients of the two readers for the motion scores were 0.791 (GRAPPA), 0.772 (CAIPI-routine), and 0.803 (CAIPI-short). The two readers’ opinions were typically in agreement. Artifact scores improved with decreased scan time. There were no significant differences between patient characteristics of the non-TSM and TSM groups in CAIPI-routine or CAIPI-short. However, there were significant differences in weight (P = 0.04) and first experience of examination (P = 0.03) between the non-TSM and TSM groups in GRAPPA.
3.3 Comparison of breath-hold and breath-hold shortening methods under clinical conditions
Both the BHS and ABH rendered image evaluation difficult for one patient (1/30, 3.3%). The motion artifact score was 4.68 for the BHS and 4.72 for the ABH; the difference in these scores was not significant (P = 0.27). This showed that image quality was the same for both the ABH and BHS methods (Fig. 6). Motion artifacts did not affect image capture with either method, and high-quality images were obtained.
4 Discussion
The present study demonstrated the effectiveness of shortening the scan time and the influence of the reference scan. The phantom study, the reference scan was not found to depend on the rate of change of SD of motion artifacts. As CAIPIRINHA uses a separate reference scan and main scan, it is inferred that reference scan information is not reflected in the image. However, the main scan can be used to fill all k-space after the reference scan. Therefore, motion artifacts may be introduced during the main scan. For this reason, the patient is not required to hold his or her breath during the reference scan. A decrease in scan time may significantly reduce motion artifacts. A long breath-hold is not essential during a CAIPIRINHA reference scan which implies that the scan can be performed with a short breath-hold. Our findings suggested that accurate measurement of the arterial phase is possible during a 5-s breath-hold.
The parallel imaging method allows for a shorter scan time and does not require additional expensive hardware. The use of a CAIPIRINHA sampling strategy was previously shown to reduce signal-to-noise ratio losses and residual aliasing artifacts; these improvements have been explored in a clinical setting. However, the influence of the reference scan of the CAIPIRINHA approach has not been investigated [21, 22]. Integrated of GRAPPA is the traditionally used technique; however, this method does not allow for segregation of the reference scan. Integrated of GRAPPA simultaneously collects a reference scan and a main scan. GRAPPA allows for the selection of separate mode and integrated mode. However, the reference mode of CAIPIRINHA is only compatible with the separate mode. Additionally, the reference scan can acquire only the first part of the scan. Motion artifacts are one of the primary causes of image degradation during DCE imaging of the abdomen. In previous studies, shorter scan times have been shown to ameliorate motion artifacts [16]. Our study revealed similar results in that decreased scan time was associated with decreased frequency of motion artifacts with all the three methods (GRAPPA, CAIPI-routine, and CAIPI-short).
Five seconds after the start of CAIPIRINHA, phantom experiments showed a low rate of change of SD for motion artifacts. With CAIPIRINHA, this timing was equivalent to that observed for the reference scan. In addition, BHS artifacts were not included in the scan. It was, therefore, possible to obtain a high-quality image and time reference scan, even when the breath-hold time is short. Because, the timing of the reference scan is separate in CAIPIRINHA and is not required to fill the k-space, motion artifacts are not created.
In phantom experiments, at 5 s before the end of CAIPIRINHA, there were motion artifacts and the rate of change of SD was high. Therefore, in the clinical case, breath-hold must be done perfectly to the end. In comparison, GRAPPA produced artifacts even 5 s after the initiation of scanning. Reference scans for GRAPPA create a sensitivity map of the collected data, which stretches several lines from the k-space center [12]. GRAPPA incorporates a reference scan into the primary scan. In addition, the amount of k-space data from the integrated GRAPPA was larger, because it made the fit by producing motion artifacts throughout the entire scan. Instead of shortening the scan time, the resolution of CAIPI-short is lower compared to CAIPI-routine. This should be taken into consideration. The DCE of the arterial phase is an important examination method for evaluation of tumor blood flow.
The current study has proven the importance of the reference scan. However, this study has several limitations. First, tumor detectability was not evaluated, due to the retrospective nature of the study. To study this, it would be necessary to perform an examination at the same time using both the approaches, but this was not possible in this retrospective study. Second, we allowed free breathing, rather than setting the phantom to a specific deep breath movement. However, it has been inferred from both phantom and clinical studies that there is no relationship between the amount of movement and the reference scan. Finally, due to the retrospective nature of the study, the results were gathered over a lengthy period of time.
Davenport et al. reported that intravenous gadoxetate disodium can result in acute self-limiting dyspnea, which can have a deleterious effect on arterial phase MR image quality. This occurs significantly more often with intravenous gadoxetate disodium than with intravenous gadobenate dimeglumine. Motion artifacts were often observed during the arterial phase with this approach with gadoxetate disodium as compared to other contrast agents [7]. This problem has been greatly improved by the shorter scan time. Our findings suggested that motion artifacts are influenced by breath time rather than by contrast agent. The most important factor with respect to imaging is likely to be the shorter scan time. GRAPPA results differed significantly according to the weight and age of the patient and between the non-TSM and TSM groups, with respect to the first DCE MRI examination. This result is the same as that of a previous study [4]. However, there was no significant difference between the CAIPI-routine and CAIPI-short methods with respect to patient characteristics of each group. This was because the scan time was extremely short, and the TSMs were decreased considerably. Even though breath-hold is difficult, elderly patients are often imaged using the CAIPI-short technique. The data collected in this study showed that the incidence of motion artifacts in this group was low. The technique described here, i.e., the shortening of scan time by CAIPIRINHA(CAIPI-short), or another high-speed technique, such as a compressed sense and radial scan, may allow completely dynamic scans [23].
5 Conclusions
Use of a reference scan technique with the CAIPIRINHA method facilitates a decrease in the time required for breath-hold, and reduces the scan time to 5 s. Failure of arterial phase scanning was significantly reduced using this approach.
Abbreviations
- ABH:
-
All-breath-hold methods
- AF:
-
Acceleration factor
- BHS:
-
Breath-hold shortening methods
- BMI:
-
Body mass index
- CAIPIRINHA:
-
Controlled aliasing in parallel imaging results in higher acceleration
- CAIPI-routine:
-
CAIPIRINHA routine method
- CAIPI-short:
-
CAIPIRINHA short-scanning method
- DCE:
-
Dynamic contrast material-enhanced
- GRAPPA:
-
Generalized autocalibrating partially parallel acquisition
- MRI:
-
Magnetic resonance imaging
- ROI:
-
Region of interest
- SD:
-
Standard deviation
- SENSE:
-
SENSitivity encoding
- SMASH:
-
Simultaneous acquisition of spatial harmonics
- SNR:
-
Signal-to-noise ratio
- TSM:
-
Transient severe motion
- VIBE:
-
Fat-suppressed 3D-volume interpolated breath-hold examination
- 3D:
-
Three-dimensional
References
Goodwin MD, Dobson JE, Sirlin CB, Lim BG, Stella DL. Diagnostic challenges and pitfalls in MR imaging with hepatocyto-specific contrast agent. Radiographics. 2011;31:1547–68.
Zech CJ, Herrmann KA, Reiser MF, Schoenberg SO. MR imaging in patients with suspected liver metastases: value of liver-specific contrast agent Gd-EOB-DTPA. Magn Reson Med Sci. 2007;6:43–52.
Murakami T, Okada M, Hyodo T. CT versus MR imaging of hepatocellular carcinoma: toward improved treatment decisions. Magn Reson Med Sci. 2012;11:75–82.
Hayashi T, Saitoh S, Tsuji Y, Takahashi J, Tagaya N, Hiramoto M, Fukuzawa K, Tano M, Miyati T, Kumada H. Influence of gadoxetate disodium on oxygen saturation and heart rate during dynamic contrast-enhanced MR imaging. Radiology. 2015;276:756–65.
Davenport MS, Bashir MR, Pietryga JA, Weber JT, Khalatbari S, Hussain HK. Dose-toxicity relationship of gadoxetate disodium and transient severe respiratory motion artifact. AJR. 2014;203:796–802.
Davenport MS, Caoili EM, Kaza RK, Hussain HK. Matched within-patient cohort study of transient arterial phase respiratory motion-related artifact in MR imaging of the liver: gadoxetate disodium versus gadobenate dimeglumine. Radiology. 2014;272:123–31.
Davenport MS, Viglianti BL, Al-Hawary MM, Caoili EM, Kaza RK, Liu PS, Maturen KE, Chenevent TL, Hussain HK. Comparison of acute transient dyspnea after intravenous administration of gadoxetate disodium and gadobenate dimeglumine: effect on arterial phase image quality. Radiology. 2013;266:452–61.
Bashir MR, Castelli P, Davenport MS, Larson D, Marin D, Hussain HK, Jaffe TA. Respiratory motion artifact affecting hepatic arterial phase MR imaging with gadoxetate disodium is more common in patients with a prior episode of arterial phase motion associated with gadoxetate disodium. Radiology. 2015;274:141–8.
Tanimoto A, Higuchi N, Ueno A. Reduction of ringing artifacts in the arterial phase of gadoxetic acid-enhanced dynamic MR Imaging. Magn Reson Med Sci. 2012;11:91–7.
Sodickson DK, Manning WJ. Simultaneous acquisition of spatial harmonics (SMASH): fast imaging with radiofrequency coil arrays. Magn Reson Med. 1997;38:591–603.
Jakob PM, Griswold MA, Edelman RR, Sodickson DK. AUTO-SMASH: a self-calibrating technique for SMASH imaging. SiMultaneous acquisition of spatial harmonics. MAGMA. 1998;7:42–54.
Griswold MA, Jakob PM, Heidemann RM, Nittka M, Jellus V, Wang J, Kiefer B, Haase A. Generalized autocalibrating partially parallel acquisitions (GRAPPA). Magn Reson Med. 2002;47:1202–10.
Wintersperger BJ, Nikolaou K, Dietrich O, Rieber J, Nittka M, Reiser MF, Schoenberg SO. Single breath-hold real-time cine MR imaging: improved temporal resolution using generalized autocalibrating partially parallel acquisition (GRAPPA) algorithm. Eur Radiol. 2003;13:1931–6.
Breuer FA, Blaimer M, Heidemann RM, Mueller MF, Griswold MA, Jakob PM. Controlled aliasing in parallel imaging results in higher acceleration (CAIPIRINHA) for multislice imaging. Magn Reson Med. 2005;53:684–91.
Breuer FA, Blaimer M, Mueller MF, Seiberlich N, Heidemann RM, Griswold MA, Jakob PM. Controlled aliasing in volumetric parallel imaging (2D CAIPIRINHA). Magn Reson Med. 2006;55:549–56.
Ogawa M, Kawai T, Kan H, Kobayashi S, Akagawa Y, Suzuki K, Nojiri S, Ozawa Y, Shibamoto Y. Shortened breath-hold contrast-enhanced MRI of the liver using a new parallel imaging technique, CAIPIRINHA (controlled aliasing in parallel imaging results in higher acceleration): a comparison with conventional GRAPPA technique. Abdom Imaging. 2015;40:3091–8.
Fushimi Y, Okada T, Kanagaki M, Yamamoto A, Kanda Y, Sakamoto R, Hojo M, Takahashi JC, Miyamoto S, Togashi K. 3D dynamic pituitary MR imaging with CAIPIRINHA: initial experience and comparison with 2D dynamic MR imaging. Eur J Radiol. 2014;83:1900–6.
Othman AE, Martirosian P, Schraml C, Taron J, Weiss J, Bier G, Schwentner C, Nickel D, Bamberg F, Kramer U, Nikolaou K, Notohamiprodjo M. Feasibility of CAIPIRINHA-Dixon-TWIST-VIBE for dynamic contrast-enhanced MRI of the prostate. Eur J Radiol. 2015;84:2110–6.
Stanisz GJ, Odrobina EE, Pun J, Escaravage M, Graham SJ, Bronskill MJ, Henkelman RM. T1, T2 relaxation and magnetization transfer in tissue at 3T. Magn Reson Med. 2005;54:507–12.
Muro I, Takahara T, Horie T, Honda M, Kamiya A, Okumura Y, Hanaki A, Imai Y. Influence of respiratory motion in body diffusion weighted imaging breathing. Jpn Radiol Tecnol. 2005;61:1551–8.
Wright KL, Harrell MW, Jesberger JA, Landeras L, Nakamoto DA, Thomas S, Nickel D, Kroeker R, Griswold MA, Gulani V. Clinical evaluation of CAIPIRINHA: comparison against a GRAPPA standard. J Magn Reson Imaging. 2014;39:189–94.
Yu MH, Lee JM, Yoon J, Kiefer B, Han JK, Choi B. Clinical application of controlled aliasing in parallel imaging results in a higher acceleration (CAIPIRINHA)-volumetric interpolated breathhold (VIBE) sequence for gadoxetic acid-enhanced liver MR imaging. J Magn Reson Imaging. 2013;38:1020–6.
Hersh C, Li F, Tobias KB, Andrew BR, Ruth PL, James SB, Daniel KS, Ricardo O. Free-breathing contrast-enhanced multiple MRI of the liver using a combination of compressed sensing, parallel imaging, and golden-angle radial sampling. Invest Radiol. 2013;48:10–6.
Acknowledgements
The authors would like to thank Yasutaka Tanuma for help collecting the data for this study.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare that they have no conflicts of interest.
Ethical approval
The IRB approved this study and waived the requirement that patients provide informed consent. This article does not contain any animal studies.
Informed consent
Informed consent was obtained from all participants for whom identifying information was included in this article.
About this article
Cite this article
Tsuji, Y., Saitoh, S., Takahashi, J. et al. Influence of the reference scan and scan time on the arterial phase of liver magnetic resonance imaging. Radiol Phys Technol 11, 91–99 (2018). https://doi.org/10.1007/s12194-017-0441-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12194-017-0441-2