Abstract
Immune thrombocytopenia (ITP) is one of the most common acquired immune-mediated bleeding disorders found in children. Prednisone is usually considered a first-line therapeutic agent for ITP in children. Yet, prolonged exposure to prednisone has been associated with certain side effects. This prospective randomized study comparatively assessed the efficacy and safety of short-course high-dose dexamethasone (HDD) and standard prednisone (PDN) as a first-line treatment for children with previously untreated primary ITP. Two hundred eleven children were randomized into the HDD (n = 110) and PDN (n = 101) groups. There was no difference in baseline characteristics between the two groups (p > 0.05). Early response rates were 92.7% and 93% (p = 0.923); initial response rates were 93.6% and 95% (p = 0.658) and durable response rates were 90% and 91% (p = 0.787) in the HDD and PDN groups, respectively. More remission patients in the HDD group compared with the PDN group (86.3% vs. 80.1%) at 12th month after treatment, yet no statistical difference was observed (p = 0.703). Bleeding events were 10.9% and 14.8% (p = 0.105), and bleeding score improvement rates were 78.2% and 76.2% (p = 0.284) in the HDD and PDN groups, respectively. Cushing’s disease, weight gain and infection rates were higher in the PDN group compared to the HDD group (80% vs. 10%, p = 0.001; 74.2% vs. 13.6%, p = 0.001; and 26% vs. 11.8%, p = 0.012) 1 month after treatment. HDD showed non-inferior efficacy and fewer glucocorticoid-related adverse effects compared with PDN. These findings indicated that HDD could be considered as a first-line treatment in children with previously untreated primary ITP, thus replacing standard PDN.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Immune thrombocytopenia (ITP) during childhood is characterized by isolated thrombocytopenia (platelet count < 100 × 109/L) [1], whose annual incidence in children is about 4–5/100,000. ITP in children is a benign self-limiting disease; in 80% of cases, the platelet count returns to normal levels within 12 months. Glucocorticoids, i.e., prednisone, are usually considered as first-line therapeutic agents for ITP in children. Prednisone is usually administered orally at a single dose of 1–2 mg/kg per day for 4 weeks, then tapered off as standard prednisone (PDN) [2]. Adverse effects of standard PDN treatment include weight gain, Cushing’s disease, infection, rash, behavioral changes, sleep disturbances, and hypertension. In children, some of these effects can be well-tolerated, while others can be very harmful. In addition, prolonged drug exposure can cause reduced growth [3, 4]. Thus, the American Society of Hematology (ASH) [5] and international consensus report [6] recommended shortening the duration of glucocorticoid in the treatment of ITP. Moreover, 2019ASH recommends glucocorticoid therapy for less than seven days in children with ITP. In China, standard prednisone (PDN) is commonly prescribed to treat ITP in children.
Compared to PDN, high-dose dexamethasone (HDD) is considered the first-line treatment for previously untreated ITP in adults [7, 8], and a second-line treatment in children ITP [9,10,11,12,13]; HDD has been associated with less adverse effects compared with standard PDN [14]. Moreover, corticosteroids (dexamethasone, methylprednisolone, prednis (ol)one), intravenous immunoglobulin (IVIG), and anti-d are considered an initial treatment option for newly diagnosed ITP [5]. Yet, so far, no randomized controlled study has investigated HDD and prednisone in children with primary ITP. Also, it still remains unclear whether HDD can be used in the first-line treatment of ITP in children. Pediatric trials have found no evidence of more desirable treatment effects conferred by HDD compared with standard PDN [14]. The aim of this prospective, randomized, single-center study was to investigate whether HDD may be used as the first-line treatment in children with previously untreated ITP.
Materials and methods
Study design
This was a prospective, single-center, randomized controlled clinical trial comparing HDD to PDN for first-line management of children with previously untreated ITP. Patients were enrolled in Beijing Children’s Hospital, Capital Medical University, between November 2016 and February 2018. The local ethics committee approved the study. The parents or guardians of all patients provided written informed consent in accordance with the Declaration of Helsinki before enrollment. This study was registered on chictr.org.cn (ChiCTR-INR-16008827).
Participants
Grouping
We conducted a non-inferiority study. Eligible patients were randomly assigned using a 1:1 ratio: HDD group and PDN group. Grouping and randomization criteria were determined by assigning random serial numbers.
Intervention
The HDD group received 0.6 mg/kg/day dexamethasone (maximum 40 mg/day) by intravenous infusion for 4 consecutive days. Patients who did reach a response after 2 weeks received a second cycle of HDD.
The PDN group received the standard PDN 2 mg/kg dose (maximum 60 mg/day) for 2 consecutive weeks. In responders, the prednisone was gradually tapered over the 4–8 consecutive weeks. If patients were not able to reach a complete response (CR), a low dose was used, maintaining a platelet count above 30×109/L with an absence of bleeding symptoms; taper schedule was determined by physicians.
Primary outcomes
Effectiveness was evaluated by the IWG criteria [1] and the Updated international consensus report [6]: CR, platelet count ≥ 100 × 109/L and no bleeding; response (R), platelet count ≥ 30 × 109/L and at least twofold increase versus baseline count, and no bleeding; no response, platelet count < 30 × 109/L, less than twofold increase versus baseline platelet count, or bleeding; early response, platelet count ≥ 30 × 109/L and at least twofold increase versus baseline value at 1 week; initial response, platelet count ≥ 30 × 109/L and at least twofold increase versus baseline value at 1 month; durable response, platelet count ≥ 30 × 109/L and at least twofold increase versus baseline value at 6 months; remission, platelet count > 100 × 109/L at 12 months.
Secondary outcomes
Bleeding symptoms were classified from grade 0–5, according to the ITP-specific bleeding scale proposed by Buchanan [15]. Grades 0–2, 3, and 4–5 on the adapted Buchanan bleeding scale indicated mild, moderate, and severe bleeding, respectively.
Safety
Drug-related side effects included hypertension, high intraocular pressure, abdominal discomfort, Cushing’s disease, weight gain, infection, and rash during treatment. Patients who experienced severe adverse effects discontinued the allocated intervention and exited the study as determined by the attending physicians. In patients who received the second cycle of HDD, drug-related side effects were re-evaluated. In the HDD group, drug-related side effects were observed for up to 1 month after treatment discontinuation, as determined by the investigators. In order to ensure that the observation time of the side effects in the two groups of children was uniform, the PDN group was also evaluated for 1 month after treatment.
Data collection
Demographic data were collected at enrollment and included age, sex, pretreatment platelet count, and the degree of bleeding. After 1 week of treatment, platelet counts were recorded; at 1 month, platelet count, bleeding score, and side effect recordings were performed. Platelets were then measured once a month for 12 months.
The primary outcomes were an early response, initial response, and durable response and remission rates in both groups. Secondary outcomes were condition improvement (bleeding score reduction) within 1 month, and glucocorticoid-related side effects (e.g., hypertension, hyperglycemia, abdominal discomfort during HDD administration, and Cushing’s disease), weight gain, rash, and infection complications, which were assessed by the senior investigator 1 month after treatment.
Statistical analysis
The sample size calculated based on the initial response rate of PDN treatment was 85%, and the therapeutic effect of the HDD group was not less than 75%. Therefore, 110 evaluable cases per group were required to detect a difference in the incidence of initial response with 80% power of test at 5% significance level (by 2-sided Fisher’s exact test). We also set up a 10% redundancy to balance possible dropout or other bias and finally determined the sample size to be 121 cases in each group. All statistical analyses were performed with the SPSS (version 19) software. Quantitative data with normal distribution were expressed as mean ± standard deviation (SD), and were compared by the t test. Those with skewed distribution were described as median (upper and lower quartiles), and compared by the Wilcoxon rank-sum test. We included patients who received allocated intervention as per-protocol (PP) population in the description of baseline characteristics and the analysis of efficacy and safety. Classification data were described as frequency (percentage), and compared by the Chi square test. At month 12 of treatment, statistical analysis was performed to determine the non-inferiority of HDD versus PDN. Two-tailed p < 0.05 was considered statistically significant.
Results
Two hundred thirty patients underwent randomization. Of these, 19 withdrew from the study, which resulted in 211 patients being allocated to the HDD (n = 110) and PDN (n = 101) groups (Fig. 1).
Baseline characteristics did not differ between the two groups (Table 1; p > 0.05). There were more males than females, with no significant difference (p > 0.05). The median age of children with ITP was less than 3 years. Most children had platelet counts below 20 × 109/L before enrollment, with mild to moderate bleeding. The median bleeding score was 2 (range, 0–4) on the Buchanan bleeding score scale (1–2 points, 58.3% or 123/211; 3 points, 27.4% or 58/211). Visceral bleeding was observed in nine patients (4.2%). Four cases had gastrointestinal bleeding, three cases had urinary bleeding, and two cases had pulmonary bleeding.
Primary outcomes
Primary outcomes are shown in Table 2. The proportion of patients achieving CR was higher in the PDN group compared with the HDD group during the first week (67.3% vs. 78.2%; p = 0.075); however, no significant difference was observed (p > 0.05). More than 90% of the children reached an early response in both treatment groups. The HDD group showed reduced initial response rate compared with the PDN group; however, the difference was not significant when evaluated by the PP set (93.6% vs. 95.5%; p = 0.658). Moreover, more than 90% of patients reached an initial response after one month in both groups. In the HDD group, 92.7% of patients responded to the initial course of treatment, with a CR rate of 67.4%. Twelve cases underwent an additional two cycles of HDD.
The durable response rate was slightly higher in the PDN group compared with the HDD group, without statistical significance (p > 0.05). At 12 months, more patients achieved remission in the HDD group compared to the PDN group; yet, again, no statistical significance was observed (p = 0.703).
Non-inferiority statistical analysis was performed 12th month after treatment (Table 3). The CR plus R rate of patients in HDD and PDN group was 97.2% vs. 95.1%, respectively (p < 0001).
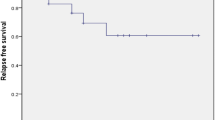
Throughout the follow-up period, the overall duration of response showed no significant difference between the two groups. Moreover, nearly 40% of patients who did not reach CR at 1 month of treatment did not reach CR at 6 (39.3% vs. 50%; p = 0.512) and 12 (35.5% vs. 38.9%; p = 0.828) months, respectively in both groups.
Twelve patients received two cycles of HDD. The median time during the 1st and 2nd HDD cycles was 11 days (range, 7–14). The median platelet count before the 2nd cycle was 16 × 109/L (range, 5–30 × 109/L). After the 2nd cycle, 50% of patients (n = 6) reached CR, 25% (n = 3) achieved R, and 25% (n = 3) reached NR. Three (2.7%) and 4 (4.0%) cases relapsed in the HDD and PDN groups at 3–6 months, respectively (p = 0.712).
Secondary outcomes
After 1 month of treatment, bleeding events (bleeding scores ≥ 1) occurred in 10.9% (12/110) and 14.8% (15/101) of patients in the HDD and PDN groups, respectively. These rates did not significantly differ (p = 0.416). Furthermore, more than 95% of children had a bleeding score of two or less in the HDD and PDN groups. Nearly 80% of all children had bleeding symptoms controlled after treatment, with no significant difference between the two groups (Table 2).
Safety
No severe adverse events occurred, and no patients discontinued glucocorticoid treatment in either group (Table 4). The frequency of adverse events was higher in the PDN group compared with HDD-treated patients. Cushing’s disease and weight gain occurred in nearly 80% of the patients. The infection rate was higher in the PDN group compared with the HDD group (11.8% vs. 24.8%, p = 0.012); these included upper respiratory tract and mild digestive tract infections requiring oral anti-infective treatment for 3–5 days. Several other adverse events occurred in more than 5% of the PDN group, including rashes. Five patients in the HDD group developed elevated intraocular pressure. In patients who received two cycles of HDD, three developed high blood pressure, high intraocular pressure, and abdominal discomfort, all of which occurred during the first treatment cycle. After treatment discontinuation, these symptoms disappeared. No patients in either group exited the study because of adverse effects.
Discussion
Dexamethasone with a long half-life is used to treat ITP as a short course. The American Society of Hematology (ASH) 2019 guideline panel suggested standard PDN or HDD in adults with previously untreated ITP [5]. Moreover, Chinese and Korean adult ITP consensuses recommend HDD as the first choice for first-line regimens for the same group of patients [8, 16]. Yet, no clinical evidence confirmed a desirable effect of treatment with dexamethasone compared with prednisone for untreated primary immune thrombocytopenia (ITP) in children.
In this study, we demonstrated an improvement in the initial platelet count response with HDD in children with untreated primary ITP. We found a 92.7% early response rate after treatment with HDD. The short-term treatment response in the HDD group was similar to that of the PDN group, and there was no difference in CR between the PDN group (78.2%) and HDD group (67.3%) at 1 week after treatment, which was consistent with previous findings [7]. Similar response rates in both groups were found a month after treatment. Albayrak et al. [17] and Fujisawa et al. [18] found no difference in initial response among different high-dose corticosteroid regimens in children, which is consistent with our findings. In another study, adult patients achieving initial overall R (79% vs. 59%) or CR (64% vs. 36%) for 14 days were more numerous in the HDD group, which indicated that response occurs more rapidly with HDD [14]. In this study, however, no such evidence was obtained. Furthermore, Youssef et al. [13] reported that HDD as a rescue therapy seems to be an effective alternative emergency treatment for uncontrolled bleeding in chronic ITP children. In the present study, approx. 80% of bleeding symptoms were controlled in the HDD group.
HDD is associated with a high rate of remission. The above results showed no statistically significant difference in the durable response rate between the two groups. More patients in the HDD group achieved remission. When HDD was used as the first-line treatment in pediatric ITP, 86.3% of the children experienced remission. In a study by Thomas et al. [19], 78% of patients with chronic ITP achieved remission in 3 days, while long-term remission was found in less than half of children with chronic ITP. Moreover, Nugent et al. [20] reported a remission rate in 18 children with chronic ITP after one year of 44%, which suggested treatment effectiveness. Currently, there are no data on the application of HDD in newly diagnosed children with ITP. The above data confirm the stable long-term efficacy of HDD. In an adult study, the HDD group had significantly better 3-month CR (80% vs. 23.3%) and 6-month CR (73.3% vs. 16.7%) rates [21]. Chen et al. [22] assessed 125 adults with previously untreated ITP, and a single course of oral HDD resulted in a sustained response at 6 months in 53 patients (42%), and initial response in 106 (85%) patients. Moreover, Wei et al. [7] conducted a randomized trial and found no difference in platelet count response at 6 months between the HDD and PDN groups. The total effective rate (82.1% vs. 67.4%) and CR rate (50.5% vs. 26.8%) were higher in the HDD group compared with the PDN group. A meta-analysis reported an initial overall response rate of 79% for dexamethasone, which was similar to existing observational studies [14].
A previous report showed that the choice of initial corticosteroids does not affect immune thrombocytopenia in adults [14]. The present study focused on patients in both groups who did not achieve CR after 1 month. The differences in response rates at 6 and 12 months were not statistically significant, which suggested that different steroid regimens did not change long-term outcomes in children with ITP.
Adverse reactions were significantly reduced in the HDD group compared with the PDN group. The HDD group had lower incidence rates of Cushing’s disease, weight gain and infection compared with the PDN group. Cushing’s disease was resolved, and infections were minor in the HDD group. Mithoowani et al. [14] found that adverse events were less frequent after HDD administration and that weight gain and Cushing’s disease were less common. Wei et al. [7] found that the incidence of adverse reactions was significantly lower in the HDD group compared with the PDN group, possibly because steroid drugs were administered for a significantly shorter duration in the HDD group compared with the PDN group. Regarding hormone-induced psychiatric symptoms, we did not conduct this assessment. However, study has found that dexamethasone was not associated with more neurobehavioral side effects than prednisone [23].
The present study had several limitations. (1) This was a single-center study, with only 1 year of follow up. (2) Patients with platelet counts of 30 × 109/L were enrolled regardless of bleeding manifestations. Given China's specific national conditions, for children with platelets between 20–30 × 109/L, parents were worried about the risk of bleeding and strongly demanded treatment when living far from the hospital. (3) HDD was intravenously infused, which differed from previous reports. (4) The time for assessing side effects was insufficient, and symptoms, such as insomnia and fatigue, were not assessed in the children. (5) Follow up treatment was inconsistent in some patients after the first platelet elevation treatment. (6) We used the per-protocol (PP) population analysis for statistical analysis.
In conclusion, this study suggested that HDD provides a comparable effective response as a first-line treatment for ITP, with better tolerance than conventional PDN. Therefore, HDD could become a preferred corticosteroid approach for first-line management of children with previously untreated primary ITP.
Authors contributors
LF, Jie Ma: Writing—original draft, RW: Writing–review & editing, LF, JM: Data curation, ZC, HG: Formal analysis, RW: Project administration. All authors have read and approved the final manuscript.
Abbreviations
- HDD:
-
High-dose dexamethasone
- ITP:
-
Immune thrombocytopenia
- PDN:
-
Prednisone
- CR:
-
Complete response
- R:
-
Response
- NR:
-
No response
- IWG:
-
International Working Group
- R:
-
Response
- NR:
-
No response
- ASH:
-
The American Society of Hematology
References
Rodeghiero F, Stasi R, Gernsheimer T, Michel M, Provan D, Arnold DM, et al. Standardization of terminology, definitions and outcome criteria in immune thrombocytopenic purpura of adults and children: report from an international working group. Blood. 2009;113(11):2386–93.
Subspecialty Group of Hematolog. The Society of Pediatrics, Chinese Medical Association Recommendations for diagnosis and treatment of primary immune thrombocytopenia in children. Zhonghua Er Ke Za Zhi. 2013;51(5):382–4.
Allen DB, Julius JR, Breen TJ, Attie KM. Treatment of glucocorticoid-induced growth suppression with growth hormone national cooperative growth study. J Clin Endocrinol Metab. 1998;83(8):2824–9.
Huscher D, Thiele K, Gromnica-Ihle E, Hein G, Demary W, Dreher R, et al. Dose-related patterns of glucocorticoid-induced side effects. Ann Rheum Dis. 2009;68(7):1119–24.
Neunert C, Terrell DR, Arnold DM, Buchanan G, Cines DB, Cooper N, et al. American Society of Hematology 2019 guidelines for immune thrombocytopenia. Blood Adv. 2019;3(23):3829–66.
Provan D, Arnold DM, Bussel JB, Chong BH, Cooper N, Gernsheimer T, et al. Updated international consensus report on the investigation and management of primary immune thrombocytopenia. Blood Adv. 2019;3(22):3780–817.
Wei Y, Ji XB, Wang YW, Wang JX, Yang EQ, Wang ZC, et al. High-dose dexamethasone vs. prednisone for treatment of adult immune thrombocytopenia: a prospective multicenter randomized trial. Blood. 2016;127(3):296–302.
Jang JH, Kim JY, Mun YC, Bang SM, Lim YJ, Shin DY, et al. Management of immune thrombocytopenia: Korean experts recommendation in 2017. Blood Res. 2017;52(4):254–63.
Dubbeld P, van der Heul C, Hillen HF. Effect of high-dose dexamethasone in prednisone-resistant autoimmune thrombocytopenic purpura (ITP). Neth J Med. 1991;39(1–2):6–10.
Andersen JC. Response of resistant idiopathic thrombocytopenic purpura to pulsed high-dose dexamethasone therapy. N Engl J Med. 1994;330(22):1560–4.
Borgna-Pignatti C, Rugolotto S, Nobili B, Amendola G, Stefano PD, Maccario R, et al. A trial of high-dose dexamethasone therapy for chronic idiopathic thrombocytopenic purpura in childhood. J Pediatr. 1997;130(1):13–6.
Ma J, Fu LL, Chen ZP, Ma JY, Zhang R, Su Y, et al. Clinical study of pulsed high- dose dexamethasone treatment in 38 children with primary immune thrombocytopenic purpura. Zhonghua Xue Ye Xue Za Zhi. 2016;37(10):912–5.
Youssef M, Salah EE, Elsayh KI, Taha SF, Gamil M, Abo-Elela M. High dose dexamethasone as an alternative rescue therapy for active bleeding in children with chronic ITP: clinical and immunological effects. Platelets. 2019;30(7):886–92.
Mithoowani S, Gregory-Miller K, Goy J, Miller MC, Wang G, Noroozi N, et al. High-dose dexamethasone compared with prednisone for previously untreated primary immune thrombocytopenia: a systematic review and meta-analysis. Lancet Haematol. 2016;3(10):e489–e496496.
Buchanan GR, Adix L. Grading of hemorrhage in children with idiopathic thrombocytopenic purpura. J Pediatr. 2002;141(5):683–8.
Thrombosis And Hemostasis Group Hematology Society C. Association M. Consensus of Chinese experts on diagnosis and treatment of adult primary immune thrombocytopenia (version 2016). Zhonghua Xue Ye Xue Za Zhi. 2016;37(2):89–93.
Albayrak D, Islek I, Kalayci AG, Gürses N. Acute immune thrombocytopenic purpura: a comparative study of very high oral doses of methylprednisolone and intravenously administered immune globulin. J Pediatr. 1994;125(6):1004–7.
Fujisawa K, Iyori H, Ohkawa H, Konishi S, Bessho F, Shirahata A, et al. A prospective, randomized trial of conventional, dose-accelerated corticosteroids and intravenous immunoglobulin in children with newly diagnosed idiopathic thrombocytopenic purpura. Int J Hematol. 2000;72(3):376–83.
Kuhne T, Freedman J, Semple JW, Doyle J, Butchart S, Blanchette VS. Platelet and immune responses to oral cyclic dexamethasone therapy in childhood chronic immune thrombocytopenic purpura. J Pediatr. 1997;130(1):17–24.
Nugent D, Pendergrass HD. Long-term follow-up of refractory immune-mediated thrombocytopenic purpura (ITP) patients treated with high dose dexamethasone therapy. Blood. 1995;5:10–86.
Mashhadi MA, Kaykhaei MA, Sepehri Z, Miri-Moghaddam E. Single course of high dose dexamethasone is more effective than conventional prednisolone therapy in the treatment of primary newly diagnosed immune thrombocytopenia. Daru. 2012;20(1):7–12.
Cheng Y, Wong RS, Soo YO, Chui CH, Lau FY, Chan NP, et al. Initial treatment of immune thrombocytopenic purpura with high-dose dexamethasone. N Engl J Med. 2003;349(9):831–6.
Waber DP, McCabe M, Sebree M, Forbes PW, Adams H, Alyman C, et al. Neuropsychological outcomes of a randomized trial of prednisone versus dexamethasone in acute lymphoblastic leukemia: findings from Dana-Farber Cancer Institute All Consortium Protocol 00–01[J]. Pediatr Blood Cancer. 2013;60(11):1785–91.
Acknowledgements
This work was supported by the National Natural Science Foundation of China (No. 81970111), the Beijing Natural Science Foundation of China (No. 7192064), the Special Fund of the Pediatric Medical Coordinated Development Center of Beijing Hospitals Authority (No. XTZD20180205), and the National Science and Technology Key Projects (No. 2017ZX0930402900).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing financial interests.
Ethical approval
This study was conducted in accordance with the Declaration of Helsinki and approved by the Institutional Review Board (IRB) of Beijing Children’s Hospital, Capital Medical University. All parents signed informed consent forms.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
About this article
Cite this article
Ma, J., Fu, L., Chen, Z. et al. High-dose dexamethasone as a replacement for traditional prednisone as the first-line treatment in children with previously untreated primary immune thrombocytopenia: a prospective, randomized single-center study. Int J Hematol 112, 773–779 (2020). https://doi.org/10.1007/s12185-020-02977-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12185-020-02977-9