Abstract
Background
To evaluate the diagnostic accuracy of 18F-FDG PET/CT in detection of recurrent differentiated thyroid cancer (DTC) in patients with elevated stimulated thyroglobulin (Tg) or anti-Tg antibody (Ab) levels, and negative 131I whole body scan according to the Tg level.
Methods
PET/CT images of well DTC patients who had total thyroidectomy and radioiodine ablation were included. Patients showing elevated Tg titer (≥2.0 ng/ml after TSH stimulation) or elevated anti-Tg titer (≥70.0 IU/ml) while diagnostic radioiodine scan was negative were enrolled. PET/CT was classified as positive or negative on the basis of visual interpretation. The maximum standard uptake values (SUVmax) of the suspected lesions on PET/CT were also recorded. The PET/CT findings were compared with histological or clinical follow-up results based on other imaging modalities and serum Tg/anti-Tg Ab titers. The diagnostic performance of PET/CT was compared among 4 subgroups according to the Tg level [2 ng/ml ≤ stimulated Tg (sTg) < 5 ng/ml, 5 ng/ml ≤ sTg < 10 ng/ml, 10 ng/ml ≤ sTg < 20 ng/ml, and ≥20 ng/ml].
Results
A total of 68 PET/CT images from 60 patients were included, and histological confirmations were available in 32 images. The sensitivity, specificity, accuracy, positive predictive value and negative predictive value of PET/CT were 69.4, 66.7, 69.1, 95.6, and 17.4%, respectively. There were 3 PET/CT cases with high anti-Tg Ab level, and low (<2 ng/ml) Tg level, and all 3 were positive of recurrence. The mean SUVmax of the suspected lesions on PET/CT was 2.9 ± 4.5 (range 1.3–29.7). The sensitivity of PET/CT according to Tg levels was 28.6% when Tg was between 2 and 5, 57.1% between 5 and 10, 60.0% between 10 and 20, and 85.7% when Tg was equal to or greater than 20 ng/ml sub-groups, respectively.
Conclusion
Diagnostic accuracy of FDG PET in radioiodine negative thyroid cancer may vary depending on serum Tg levels at imaging. 18F-FDG PET/CT is useful in detection and localization of recurrent thyroid cancer in patients with negative diagnostic radioiodine scan despite elevated Tg greater than 20 ng/ml or high anti-Tg Ab titers. In contrast, PET/CT provides little additional information when the Tg is less than 5 ng/ml.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
After total thyroidectomy and radioiodine remnant ablation therapy, patients with differentiated thyroid cancers (DTCs) are screened for recurrence by serum thyroglobulin (Tg) titers and 131I whole body scans (WBSs). Because the Tg protein is unique to thyroid tissue, serum thyroglobulin measurement is used as a postoperative tumor marker test in DTC [1]. In successfully treated patients, the scan results are usually negative, and Tg values undetectable or very low even when serum thyrotropin is elevated [2]. Patients with elevated serum thyroglobulin levels undergo 131I scan, which may localize recurrence in 50–60% of patients with papillary carcinoma and 64–67% of patients with follicular carcinoma [3, 4]. However, patients with negative 131I WBS, but elevated serum Tg level are not uncommonly reported [5]. It becomes necessary to investigate with other modalities to identify possible recurrent disease. In recent years, neck ultrasonography has shown high sensitivity and specificity on evaluation of thyroid bed and locoregional lymph nodes [6, 7]. However, in most instances, it is necessary to perform total body assessment of the extent of the disease. It is well established that tumors without functional differentiation for 131I uptake show high glucose metabolism [8]. So 18F-FDG PET has been widely accepted as a method for detecting recurrent DTC with low or no 131I avidity [9–12]. Co-registered PET/CT provides further anatomical detail, enhancing diagnostic accuracy. However, only a few studies have evaluated the usefulness of 18F-FDG PET/CT according to Tg level. Therefore, the aim of this retrospective study was to evaluate the usefulness of integrated 18F-FDG PET/CT for localization of recurrent or metastatic tumor in differentiated thyroid cancer (DTC) patients with negative iodine scan and elevated serum Tg according to the stimulated Tg levels.
Methods and materials
Patients
From February 2004 to June 2007, a total of 68 PET/CT images from 60 patients (female 41, male 19, mean age 49 ± 12 years, range 26–75 years) who had total thyroidectomy and high dose radioiodine ablation at least once for the treatment of DTC were examined. Patients with elevated Tg titer (>2.0 ng/ml with TSH stimulation) or elevated anti-Tg titer (>70.0 IU/ml) but negative radioiodine scans were included. The patients had previously received high dose 131I 1–6 (mean 1.9 ± 1.1) times for ablation of remnant tissue or treatment of recurrent disease. Total therapeutic dose ranged from 1.11 to 24.42 (mean 7.47 ± 4.29) GBq. The primary tumor histology type was papillary thyroid carcinoma in all cases.
Radioiodine WBS
Radioiodine WBS was performed after T4 withdrawal for at least 4 weeks and strict low iodine diet for at least 2 weeks. Patients stimulated by recombinant human TSH were not included in the study. Anterior and posterior whole body images were obtained 18–48 h after administration of 111 MBq of 123I or 74 MBq of 131I using a dual head gamma camera. 57 WBSs were done with 123I, and 11 with 131I.
Thyroglobulin and anti-thyroglobulin antibody measurement
Thyroid-stimulating hormone (TSH), thyroglobulin (Tg) and anti-Tg antibody levels were measured at the time of the radioiodine WBS. All patients were in TSH stimulation state. 65 patients presented with TSH greater than 30 mU/l, but 3 patients showed incomplete stimulation of TSH (17–23 mU/l). Serum Tg and anti-Tg levels were measured by radioimmunoassay using a commercial kit (THYROGLOBULINE IRMA, CIS bio international, France; RIAZENco TG Ab, ZenTech s.a., Belgium). Elevated Tg level was defined as being greater than 2 ng/ml when TSH was stimulated. 65 of 68 cases showed Tg greater than 2 ng/ml ranging from 2.04 to 1015.65 ng/ml. The 65 patients were divided into 4 groups by the Tg level [2 ng/ml ≤ stimulated Tg (sTg) < 5 ng/ml, 5 ng/ml ≤ sTg < 10 ng/ml, 10 ng/ml ≤ sTg < 20 ng/ml, and ≥20 ng/ml]. Two of the 65 also had elevated anti-Tg Ab titers (94.1 and 97.7 IU/ml). Other 3 patients presented with anti-Tg Ab greater than 70 IU/ml ranging from 429 to 3078 IU/ml, but the Tg was less than 2 ng/ml.
18F-FDG PET/CT imaging
Patients fasted for at least 6 h before the 18F-FDG PET/CT study. 370–555 MBq of 18F-FDG was injected intravenously and scanning began 60 min later. None of the patients had blood glucose levels exceeding 130 mg/dl before the injection. No intravenous contrast agent was used. Two combined PET/CT in-line system (Biograph DUO, Biograph Truepoint; Siemens Medical Solutions, Knoxville, TN, USA) was used to acquire all data. There were 6–8 bed positions, and the acquisition time was 2–3 min per bed position. CT began at the orbitomeatal line and progressed to the upper thigh (30 mA s, 130 kV, 5-mm slice thickness; 80 mA s, 130 kV, 5-mm slice thickness). PET followed immediately over the same body region. The CT data were used for attenuation correction, and images were reconstructed using a standard ordered-subset expectation maximization algorithm. The axial spatial resolution was 6.5 and 4.5 mm at the center of the field of view. PET/CT images were obtained within 1 month from the diagnostic iodine scan. PET/CT scans were interpreted independently by 2 experienced nuclear medicine physicians. PET/CT was considered positive when visually perceptible and discrete FDG uptake against background activity was noted. The maximum standard uptake values (SUVmax) of the suspected lesions on PET/CT were also recorded.
Recurrence evaluation
PET/CT findings were compared with histology, other imaging modalities such as ultrasound, CT and MRI, post-therapeutic 131I WBS, and subsequent Tg and anti-Tg titers. Cases were considered positive for recurrence when: histopathologic results by reoperation or biopsy were positive, other imaging modalities showed abnormal lesions, follow-up Tg and anti-Tg titers were persistently abnormal, and the elevated Tg titer declined after empirical high dose radioiodine treatment. Cases were considered negative for recurrence when: the histopathologic results were negative, various follow-up imaging studies were all consistently negative, and serum Tg titer was normalized without any further treatment.
Statistical analysis
Overall sensitivity, specificity and accuracy of 18F-FDG PET/CT were calculated. The sensitivities of PET/CT were further evaluated according to 4 different Tg levels. The correlation between SUVmax and Tg level was assessed using statistical method (SPSS 13.0).
Results
Patients characteristics are shown in Table 1.
Positive PET/CT
45 of 68 PET/CT images presented positive findings of local recurrence or distant metastasis. 27 of 45 cases were confirmed by biopsy or surgery, and other 18 cases were validated by clinical follow up based on other imaging modalities and subsequent Tg or anti-Tg titers. 43 cases were confirmed as true positive for recurrence or metastasis, and 2 cases were considered as false positive. Table 2 shows the sites of positive lesions identified in PET/CT. All 14 cases showing recurrence or metastasis in only thyroidectomy bed were TP. 16 of 18 PET/CT showing FDG uptake in cervical or mediastinal lymph nodes (LN) were TP, and 2 cases with FDG uptake in cervical LN were FP by pathologic result and clinical follow up. In 2 cases, PET/CT detected metastatic lesions in lung and rib in addition to recurrent tumor in the thyroidectomy bed and LN metastasis. These 2 cases were proven to metastatic papillary carcinoma after surgical resection (Fig. 1). SUVmax ranged widely from 1.3 to 29.7 (mean 4.7 ± 5.7) in lesions suspicious for recurrence on PET/CT. The mean SUVmax by recurrence site was 3.4 ± 3.8 (range 1.5–20.1, n = 25) for thyroid bed, 3.9 ± 2.4 (range 1.3–8.7, n = 26) for cervical LN, 9.3 ± 11.1 (range 1.7–29.7, n = 7) for mediastinal LN and 15.3 ± 16.6 (3.5 and 27.0, n = 2) for distant metastasis. SUVmax of cervical LNs in the 2 FP PET/CT cases was relatively low, 1.6 and 1.7.
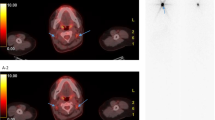
A 58-year-old male with papillary thyroid cancer received total thyroidectomy and then 150 mCi of 131I for remnant ablation. Follow-up radioiodine whole body scan done 6 months later shows no abnormal uptake (a). The patient’s stimulated Tg was 1015.7 ng/ml. In maximum intensity projection PET image, multiple intense focal FDG uptake areas are present (b). Axial PET/CT images show focal FDG uptake in mediastinal lymph nodes (c, d), rib (d) and lung (e). Lung setting CT image shows small nodular opacity in RLL (f). Surgical resection of lung mass and right 6th rib confirmed papillary cancer metastasis
Negative PET/CT
23 of 68 PET/CT images showed no abnormal FDG uptake suggestive of locoregional recurrence or distant metastasis. 19 of 23 cases were confirmed as false negative by pathologic results in 9 cases (Fig. 2) and clinical follow up based on other imaging modalities and follow-up Tg titer in 10 cases. Seven of 10 patients without pathologic confirmation received empirical radioiodine therapy. One case showed a positive post-therapeutic radioiodine scan, while the 6 cases with negative post-therapeutic scans showed gradual decreases in Tg titers. Remaining 3 patients were initially followed up without any further treatment for over 1 year. Though the neck ultrasonography was negative, the Tg titer continued to rise, and 3 patients also received empiric high dose radioiodine therapy. The post-therapeutic scan showed uptake in thyroid bed with or without uptake in the mediastinum, and serum Tg level decreased following therapy. Of four PET/CT true negative cases, 2 cases were confirmed by pathologic results, and 2 cases showed normalized Tg titer without any additional treatment.
A 32-year-old male with papillary thyroid cancer received high dose radioiodine remnant ablation 1 year ago. Follow-up radioiodine scan was negative. Stimulated Tg titer was 4.2 ng/ml. Axial PET (a) and PET/CT (b) images show mild physiologic FDG activity in esophagus, but no discrete abnormal FDG uptake is perceptible. Neck ultrasonogram (c) shows homogeneous hypoechoic nodule in left thyroid bed, and ultrasound-guided biopsy (d) confirmed papillary thyroid cancer
The performance of PET/CT according to the serum Tg level
Figure 3 shows the distribution of the stimulated Tg levels and the results of PET/CT. Overall sensitivity, specificity, accuracy, positive predictive value and negative predictive value of 18F-FDG PET/CT for detection of papillary thyroid cancer local recurrence or metastasis were computed as 69.4, 66.7, 69.1, 95.6, and 17.4% in patients with elevated Tg but negative radioiodine WBS (Table 3). The sensitivity became greater at higher Tg levels (Table 4). In patients with Tg titer greater than 55 ng/ml, sensitivity of PET/CT was 100%. The SUVmax had a very wide range in each Tg level subgroup (Table 4). The SUVmax showed no correlation with the Tg titer (p = 0.073).
All 3 PET/CT cases of patients with anti-Tg Ab over 70 IU/ml but Tg less than 2 ng/ml were true positive for recurrence in thyroidectomy bed and cervical lymph nodes.
Discussion
Both serum thyroglobulin and 131I WBSs have established roles in detecting recurrence or metastasis from DTC after total thyroidectomy and radioiodine ablation [13]. However, elevated Tg levels with negative 131I WBSs represent both diagnostic and therapeutic problems. Studies have shown that in 10–15% of thyroid cancer patients, detectable serum Tg levels are found despite negative WBS [14, 15]. The loss of ability to concentrate iodine when Tg levels are elevated necessitates resorting to different imaging tools other than radioiodine scintigraphy. Feine et al. [8, 16] were the first to formally propose a pattern in which an inverse relationship between 131I and FDG uptake, “flip-flop” existed in metastatic thyroid cancer lesions. Many studies showed wide range of sensitivity of FDG PET or PET/CT for detecting local recurrence or metastasis in Tg-positive and 131I WBS-negative patients, from 40 to 100% [9, 12, 17–22]. In one recent 18F-FDG PET/CT study, the sensitivity was 68.4% on 61 patients with suspected recurrence [17]. We reviewed 68 PET/CT images of 60 patients with elevated stimulated Tg level but negative radioiodine WBS. Our study shows comparable results, with overall sensitivity of 69.4% and specificity 66.7% for detecting and localizing recurrent or metastatic thyroid cancer on 18F-FDG PET/CT.
The serum Tg level obtained after withdrawal of thyroid hormone is strongly related to the tumor volume. Therefore, patients with higher serum Tg levels tend to have more extensive disease [23]. Recently, several studies reported the effectiveness of 18F-FDG PET/CT for detecting recurrent thyroid cancer according to the serum Tg levels [10–12, 17, 24]. Many studies showed that the chance of positive findings in PET increases with increasing Tg level [25]. Schluter et al. [12] presented that true-positive FDG PET finding correlated positively with increasing Tg levels, and they suggested FDG PET is useful at Tg levels of >10 ng/ml. Altenvoerde et al. [10] reported that 18F-FDG PET is a potentially useful method for the detection of metastatic lesions or recurrence, with serum thyroglobulin cut-off level of about 20 ng/ml. Bertagna et al. [26] showed that the highest accuracy was reached when performed for patients with Tg levels higher than 21 ng/ml. However, some studies represented the usefulness of the 18F-FDG PET/CT regardless of the serum Tg level. Shammas et al. [17] reported the sensitivity of FDG PET/CT is 60, 63, and 72% for serum Tg level less than 5, between 5 and 10, and greater than 10 ng/ml. Dahele et al. [27] reviewed sixteen patients with DTC and reported that FDG PET/CT imaging was able to detect recurrent DTC in patients with low Tg levels below 10 ng/ml. Kim et al. [24] show high sensitivity and specificity in 18F-FDG PET/CT for detection of recurrent papillary thyroid cancer in 61 patients, regardless of serum Tg level. Our study reviewed the performance according to stimulated Tg level. The sensitivities of PET/CT jumped greatly from 28.6% in 2–5 ng/ml Tg level subgroup to 57.1% in 5–10 ng/ml subgroup. There was not much difference between the sensitivities of PET/CT between the 5–10 and 10–20 ng/ml subgroups at 57.1 and 60%. The higher Tg level subgroups with levels of 20–40, 40–80 and >80 ng/ml, showed high sensitivity at 78, 75, and 100%, respectively. Our results suggest that PET/CT might also be useful in patients with relatively low stimulated Tg between 5 and 10 ng/ml, as well as having clinical impact in patients with higher Tg level widely known. However, our study suggests that FDG PET/CT is often not helpful when the Tg titer is low below 5 ng/ml, in contrary to several previous studies [17, 24, 27]. As mentioned earlier, Tg level is strongly related to the tumor volume. Considering that the whole body radioiodine scans of enrolled patients are negative and that patients with relatively low stimulated Tg levels tend to show negative results in PET/CT, we may conclude that the small size of the tumor is one of the main causes of negative results in PET/CT.
The wide range of SUVmax from 1.7 to 7.1 was represented in low Tg level, 5–10 ng/ml. Bivariate correlation between SUVmax and Tg level shows statistically significance (p = 0.000); however, we get a different result (p = 0.073) in correlation when an outlier case of Tg, 1015.65 ng/ml is excluded. Therefore, intense FDG uptake in PET/CT cannot be expected because the Tg titer is high.
Other various imaging methods are also used to detect recurrent thyroid cancer. CT of the neck is difficult to interpret without administering intravenous contrast material, and MRI, although a sensitive tool, lacks specificity [28]. Ultrasound has an emerging role, with advances in technology such as high-resolution phased-array transducers, color flow Doppler, and power Doppler providing detailed information and improved detection of local recurrences and lymph node metastases [29]. Also, pathologic confirmation such as fine-needle aspiration or biopsy is possible at once when suspicious lesion is noted by ultrasound. From our retrospective study, it is hard to see which modality is more effective for localization of recurrent disease in patients with negative WBS and elevated serum Tg level because the various imaging modalities such as neck CT, ultrasound (US) and MRI were not performed in all patients. US was performed in 41 of 68 patients with in 6 months from PET/CT. On patient base, two patients had distant metastasis, 10 patients showed discordant results between US and PET/CT, and 29 patients had concordant US and PET/CT findings. Among the 10 discordant cases, five cases showed FN in US and 4 different cases showed FN in PET. One case was FP in US. These analyses show that US and PET/CT are complementary even in evaluation of locoregional disease. It is worth noting that PET/CT is not inferior to US for evaluation of locoregional disease although US is widely used in clinical practice. If the comparison of usefulness of PET/CT and US according to the Tg level is evaluated in more patients in the future, we might be able to establish a guideline for the management of thyroid cancer patients with negative radioiodine scan but elevated Tg level.
In our study, 7 (10.3%) of 68 PET/CT images showed mediastinal LN metastasis or distant metastasis. However, all above-mentioned modalities except PET/CT are useful only for evaluation of locoregional disease. Park et al. [30] reported that recurrent or metastatic thyroid cancer, involving mediastinal LN metastasis, could be detected despite low serum Tg level. Taking these findings into consideration, PET/CT may be valuable as a single procedure for whole body evaluation in patients with mildly increased serum Tg levels. However, in our study PET/CT was negative in majority of the mildly increased stimulated Tg cases (Tg < 5 ng/ml).
In our study, treatment plans are changed in 23 patients (33.8%) after PET/CT is performed. US-guided biopsy and following operations for the lesions identified on PET/CT are carried out in 11 patients (Fig. 4). Operations without further imaging study are performed for debulking in five patients. In one case, radioiodine ablation dose was increased. Four patients have high dose radioiodine therapy as scheduled, but because the post-therapeutic scan was negative, resection of positive lesion on PET/CT was done. Radiation therapy to extensive locoregional site was performed in 2 cases. As demonstrated in the examples from this study, PET/CT can contribute to appropriate management of thyroid cancer patients with negative radioiodine scan but elevated Tg level, by evaluation of location and extent of recurrence or metastasis site, guiding site of biopsy, altering the dose of radioiodine therapy, or providing evidence for tumor debulking surgery. A relatively good prognosis is expected if the PET/CT is negative because false negative PET/CT may represent small-sized or well-differentiated tumor. Therefore, if the PET/CT is negative in patients with elevated stimulated Tg levels and negative radioiodine scans, empirical radioiodine therapy or follow up without treatment may be done, considering the Tg level and previous post-therapeutic radioiodine scans. Further studies evaluating the relationship between the PET/CT findings and the prognosis in such patients could establish the additional role of PET/CT in the patients’ management.
A 61-year-old female with papillary thyroid cancer received total thyroidectomy and high dose ablation. Radioiodine scan was negative while stimulated Tg was elevated at 6.7 ng/ml 1 year later. FDG PET/CT image shows focal abnormal uptake areas in left thyroid bed (a) and along left internal jugular chain (b–d). The SUVmax measured 3.8 at left mid-internal jugular chain. Neck ultrasonogram showed a 0.5 cm sized, oval, hypoechoic lesion in left thyroid bed (e), and small lymph nodes less than 1 cm in long diameter with preserved central echogenic hilum in the left internal jugular chain (f). Positive finding of PET/CT guided the appropriate biopsy site. And left neck dissection was done and confirmed recurrent papillary thyroid cancer and lymph node metastases
In 3 cases, the anti-Tg Ab was very high while the Tg is normal. Tg value in patients with very high titer of anti-Tg autoantibodies can be unreliable, as these antibodies can interfere with immunometric assays despite improved methodology using mouse monoclonal antibodies with least cross-reactivity to human autoantibodies. The presence of anti-Tg Ab has been reported as a risk factor for recurrence [31, 32]. The three patients with elevated anti-Tg Ab but Tg lower than 2 ng/ml showed positive findings in PET/CT and confirmed with metastatic cervical LNs by pathologic results. Likewise, FDG PET/CT can be useful for evaluation of recurrent or metastatic tumor in patients with elevated anti-Tg Ab titer but low Tg titer and negative WBS.
One limitation of our study is that in 25% of the cases, pathologic confirmation was not performed and clinical follow-up were the only evidences available for recurrence. We assumed the recurrence when follow-up images showed abnormal findings or Tg titer persistently increased regardless of therapy. However, Tg cannot serve as a gold standard for as it can be increased from other causes [33, 34]. Another limitation is that the patient number of each Tg level subgroup may be insufficient to make conclusive statements according to the Tg level.
Conclusions
Diagnostic accuracy of FDG PET in radioiodine negative thyroid cancer may vary depending on serum Tg levels at imaging. 18F-FDG PET/CT is useful for detection and localization of recurrence or metastasis in DTC patients with negative radioiodine scan but elevated stimulated Tg greater than 20 ng/ml or high anti-Tg Ab titers. And PET/CT can also be helpful in patients with relatively low Tg level between 5 and 10 ng/ml. However, FDG PET/CT was not very beneficial in patients with Tg titer below 5 ng/ml.
References
Spencer CA, Lopresti JS. Measuring thyroglobulin and thyroglobulin autoantibody in patients with differentiated thyroid cancer. Nat Clin Pract Endocrinol Metab. 2008;4:223–33.
Mazzaferri EL, Robbins RJ, Spencer CA, Braverman LE, Pacini F, Wartofsky L, et al. A consensus report of the role of serum thyroglobulin as a monitoring method for low-risk patients with papillary thyroid carcinoma. J Clin Endocrinol Metab. 2003;88:1433–41.
Galloway RJ, Smallridge RC. Imaging in thyroid cancer. Endocrinol Metab Clin North Am. 1996;25:93–113.
Lubin E, Mechlis-Frish S, Zatz S, Shimoni A, Segal K, Avraham A, et al. Serum thyroglobulin and iodine-131 whole-body scan in the diagnosis and assessment of treatment for metastatic differentiated thyroid carcinoma. J Nucl Med. 1994;35:257–62.
Pacini F, Agate L, Elisei R, Capezzone M, Ceccarelli C, Lippi F, et al. Outcome of differentiated thyroid cancer with detectable serum Tg and negative diagnostic (131)I whole body scan: comparison of patients treated with high (131)I activities versus untreated patients. J Clin Endocrinol Metab. 2001;86:4092–7.
Frilling A, Gorges R, Tecklenborg K, Gassmann P, Bockhorn M, Clausen M, et al. Value of preoperative diagnostic modalities in patients with recurrent thyroid carcinoma. Surgery. 2000;128:1067–74.
Pacini F, Molinaro E, Castagna MG, Agate L, Elisei R, Ceccarelli C, et al. Recombinant human thyrotropin-stimulated serum thyroglobulin combined with neck ultrasonography has the highest sensitivity in monitoring differentiated thyroid carcinoma. J Clin Endocrinol Metab. 2003;88:3668–73.
Feine U, Lietzenmayer R, Hanke JP, Held J, Wohrle H, Muller-Schauenburg W. Fluorine-18-FDG and iodine-131-iodide uptake in thyroid cancer. J Nucl Med. 1996;37:1468–72.
Alnafisi NS, Driedger AA, Coates G, Moote DJ, Raphael SJ. FDG PET of recurrent or metastatic 131I-negative papillary thyroid carcinoma. J Nucl Med. 2000;41:1010–5.
Altenvoerde G, Lerch H, Kuwert T, Matheja P, Schafers M, Schober O. Positron emission tomography with F-18-deoxyglucose in patients with differentiated thyroid carcinoma, elevated thyroglobulin levels, and negative iodine scans. Langenbecks Arch Surg. 1998;383:160–3.
Wang W, Macapinlac H, Larson SM, Yeh SD, Akhurst T, Finn RD, et al. [18F]-2-fluoro-2-deoxy-d-glucose positron emission tomography localizes residual thyroid cancer in patients with negative diagnostic (131)I whole body scans and elevated serum thyroglobulin levels. J Clin Endocrinol Metab. 1999;84:2291–302.
Schluter B, Bohuslavizki KH, Beyer W, Plotkin M, Buchert R, Clausen M. Impact of FDG PET on patients with differentiated thyroid cancer who present with elevated thyroglobulin and negative 131I scan. J Nucl Med. 2001;42:71–6.
Pacini F, Pinchera A, Giani C, Grasso L, Baschieri L. Serum thyroglobulin concentrations and 131I whole body scans in the diagnosis of metastases from differentiated thyroid carcinoma (after thyroidectomy). Clin Endocrinol (Oxf). 1980;13:107–10.
Pacini F, Mariotti S, Formica N, Elisei R, Anelli S, Capotorti E, et al. Thyroid autoantibodies in thyroid cancer: incidence and relationship with tumour outcome. Acta Endocrinol (Copenh). 1988;119:373–80.
Schlumberger M, Arcangioli O, Piekarski JD, Tubiana M, Parmentier C. Detection and treatment of lung metastases of differentiated thyroid carcinoma in patients with normal chest X-rays. J Nucl Med. 1988;29:1790–4.
Feine U. Fluor-18-deoxyglucose positron emission tomography in differentiated thyroid cancer. Eur J Endocrinol. 1998;138:492–6.
Shammas A, Degirmenci B, Mountz JM, McCook BM, Branstetter B, Bencherif B, et al. 18F-FDG PET/CT in patients with suspected recurrent or metastatic well-differentiated thyroid cancer. J Nucl Med. 2007;48:221–6.
Conti PS, Durski JM, Bacqai F, Grafton ST, Singer PA. Imaging of locally recurrent and metastatic thyroid cancer with positron emission tomography. Thyroid. 1999;9:797–804.
Plotkin M, Hautzel H, Krause BJ, Schmidt D, Larisch R, Mottaghy FM, et al. Implication of 2–18fluoro-2-deoxyglucose positron emission tomography in the follow-up of Hurthle cell thyroid cancer. Thyroid. 2002;12:155–61.
Nahas Z, Goldenberg D, Fakhry C, Ewertz M, Zeiger M, Ladenson PW, et al. The role of positron emission tomography/computed tomography in the management of recurrent papillary thyroid carcinoma. Laryngoscope. 2005;115:237–43.
Alzahrani AS, Mohamed GE, Al Rifai A, Al-Sugair A, Abdel Salam SA, Sulaiman OM, et al. Role of [18F]fluorodeoxyglucose positron emission tomography in follow-up of differentiated thyroid cancer. Endocr Pract. 2006;12:152–8.
Finkelstein SE, Grigsby PW, Siegel BA, Dehdashti F, Moley JF, Hall BL. Combined [18F]Fluorodeoxyglucose positron emission tomography and computed tomography (FDG-PET/CT) for detection of recurrent, 131I-negative thyroid cancer. Ann Surg Oncol. 2008;15:286–92.
Bachelot A, Cailleux AF, Klain M, Baudin E, Ricard M, Bellon N, et al. Relationship between tumor burden and serum thyroglobulin level in patients with papillary and follicular thyroid carcinoma. Thyroid. 2002;12:707–11.
Kim Kun-Ho, Shong Min-Ho, Seo Young-Duk, Kim Seong-Min. Usefulness of 18F-FDG PET/CT in locoregional recurrence of differentiated thyroid cancer: comparison PET/CT to PET and neck ultrasonography for biopsy-proven lesions. Nucl Med Mol Imaging. 2009;43:411–20.
Stokkel MP, Duchateau CS, Dragoiescu C. The value of FDG-PET in the follow-up of differentiated thyroid cancer: a review of the literature. Q J Nucl Med Mol Imaging. 2006;50:78–87.
Bertagna F, Bosio G, Biasiotto G, Rodella C, Puta E, Gabanelli S, et al. F-18 FDG-PET/CT evaluation of patients with differentiated thyroid cancer with negative I-131 total body scan and high thyroglobulin level. Clin Nucl Med. 2009;34:756–61.
Dahele M, Ung YC, Ehrlich L, Silverberg J, Balogh J, Wong CS. 18F-fluorodeoxyglucose positron emission tomography-computed tomography for suspected recurrent papillary thyroid cancer: early experience at Sunnybrook Health Sciences Centre. J Otolaryngol Head Neck Surg. 2008;37:712–7.
Gross ND, Weissman JL, Talbot JM, Andersen PE, Wax MK, Cohen JI. MRI detection of cervical metastasis from differentiated thyroid carcinoma. Laryngoscope. 2001;111:1905–9.
Torlontano M, Attard M, Crocetti U, Tumino S, Bruno R, Costante G, et al. Follow-up of low risk patients with papillary thyroid cancer: role of neck ultrasonography in detecting lymph node metastases. J Clin Endocrinol Metab. 2004;89:3402–7.
Park EK, Chung JK, Lim IH, Park do J, Lee DS, Lee MC, et al. Recurrent/metastatic thyroid carcinomas false negative for serum thyroglobulin but positive by posttherapy I-131 whole body scans. Eur J Nucl Med Mol Imaging. 2009;36:172–9.
Chung JK, Park YJ, Kim TY, So Y, Kim SK, Park DJ, et al. Clinical significance of elevated level of serum antithyroglobulin antibody in patients with differentiated thyroid cancer after thyroid ablation. Clin Endocrinol (Oxf). 2002;57:215–21.
Tumino S, Belfiore A. Appearance of antithyroglobulin antibodies as the sole sign of metastatic lymph nodes in a patient operated on for papillary thyroid cancer: a case report. Thyroid. 2000;10:431–3.
Ma C, Kuang A, Xie J, Ma T. Possible explanations for patients with discordant findings of serum thyroglobulin and 131I whole-body scanning. J Nucl Med. 2005;46:1473–80.
Giovanella L, Keller F, Ceriani L, Tozzoli R. Heterophile antibodies may falsely increase or decrease thyroglobulin measurement in patients with differentiated thyroid carcinoma. Clin Chem Lab Med. 2009;47:952–4.
Conflict of interest
No potential conflicts of interest were disclosed.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Na, S.J., Yoo, I.R., O, J.H. et al. Diagnostic accuracy of 18F-fluorodeoxyglucose positron emission tomography/computed tomography in differentiated thyroid cancer patients with elevated thyroglobulin and negative 131I whole body scan: evaluation by thyroglobulin level. Ann Nucl Med 26, 26–34 (2012). https://doi.org/10.1007/s12149-011-0536-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12149-011-0536-5