Abstract
Among survivor’s patients with cancer, sexual dysfunction (SD) is a common treatment-related disturbance that significantly affects their quality of life. The disorder can be a short-term effect, but frequently it can be permanent and patients often do not receive adequate support to overcome the problem. The characteristics of SD may differ depending on the type of neoplasm and oncological treatment. The most common long-term effects are those related to treatment-induced menopause, ablative oncologic surgery, and altered gonadal function. Breast, prostate, and gynecologic cancers are the tumors most commonly associated with disturbances in sexual health, although there is evidence of SD in patients with other types of neoplasms. It is necessary for the healthcare team caring for oncological patients to be aware of the characteristics of sexual dysfunction in this population. This will make it possible to offer patients a comprehensive and personalized approach to improve their quality of life.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Progress in oncological treatment in recent years has led to a growing number of survivors patients with cancer. This survival improvement has made it possible to identify long- and medium-term quality-of-life issues related to antitumor therapy. Oncological approaches currently include surgery, radiation therapy, and chemotherapy. In addition, new systemic treatment alternatives are available, including immunotherapy with immune checkpoint inhibitors and targeted therapy. All these interventions may cause physical changes and adverse psychosocial effects in patients, either acutely or long after the end of the treatment. Among these conditions, sexual dysfunction (SD) is a common and relevant disorder that can be the result of biological, physiological, and psychological changes associated with cancer itself and its treatment [1,2,3,4].
Despite the high prevalence of sexual dysfunction associated with cancer treatment, sexual problems are often not adequately discussed with patients during medical care, and cultural and relationship factors that also influence sexuality are often ignored in clinical practice. It has been reported that up to 50% of patients claim to be dissatisfied with the information they received from their physician prior to starting cancer treatment, and therefore were not well prepared to overcome the problem. Meanwhile, time pressure, insufficient training, religious convictions, and variability in patient and insurance access are the main limitations described by healthcare professionals in approaching sexual dysfunction [5,6,7].
The purpose of this review is to examine the available information on the epidemiology and characteristics of sexual dysfunction in patients with cancer, including differences by gender and type of malignancy, as well as general recommendations for management and treatment. This knowledge will support physicians and health care professionals involved in the management of oncological patients to provide tailored interventions and overcome disparities in the medical approach of these patients.
The search for information to conduct this manuscript was carried out in Pubmed, selecting publications using the terms: sexual dysfunction, cancer patient, survivors, chemotherapy, surgery, and radiotherapy. Oral and written abstracts presented at international oncology meetings were eligible. Inclusion criteria for articles were: publications since 1995, including abstracts and full text of clinical trials, systematic reviews, meta-analyses, randomized clinical trials, and relevant and current reviews.
Epidemiology and etiology of sexual dysfunction in women and men with cancer
Sexual dysfunction has distinct features in the global population according to gender. In general, men are more likely than women to be sexually active and to report a good-quality sex life. A prevalence of sexual dysfunction of 40% has been reported in women, of which approximately 12% is associated with personal or interpersonal distress, including poor self-perceived health status, low educational level, and depression. In contrast, the incidence of sexual dysfunction in men ranges from 30–60%, increases with age, and is more likely to worsen between the ages of 75 and 85 years [1,2,3, 8, 9].
Globally, the risk factors associated with sexual dysfunction in women and men, include psychological/psychiatric conditions, chronic illness, hormonal changes, aging, chronic medication use, malignancy, and exposure to toxics. Table 1 [7, 8].
Specifically in relation to patients with cancer, there are limited and heterogeneous reports on the incidence of sexual dysfunction related with oncological treatment. A prevalence ranging from 40 to 100% has been informed in some series of patients. This prevalence may differ depending on the route of administration systemic or local as well as the type of malignancy and anticancer treatment. Furthermore, it has been documented that all types of oncological treatment have the potential to affect sexual health [6, 7, 10, 11].
Age has been described as a determining factor in the presence of sexual dysfunction in patients with cancer. According to available reports, women older than 65 years are more likely to permanently discontinue sexual activity after gynecologic malignancy, and men younger than 50 years at diagnosis are more likely to recover sexual function after nerve-sparing radical prostatectomy than men older than 70 years [2, 6, 8, 10].
Importantly, chemotherapy-induced fatigue has also been identified as a factor strongly associated with sexual dysfunction and is directly related to loss of libido and sexual performance [2, 6, 8, 11].
There are some specific disorders of sexual function that have been well identified according to gender, as well as some anticancer treatments that affect men and women equally. Regarding the type of neoplasm and gender, frequencies of sexual dysfunction up to 63% had been reported in women with breast cancer, 96% in men with prostate cancer, 91% in women with gynecological cancer, 78% in hematological neoplasms, and 79% in colorectal cancer [2, 6, 10,11,12,13,14,15].
In most cases, overall risk factors for sexual dysfunction in patients with cancer include age, presence of a solid pelvic neoplasm, diagnosis of hematologic malignancy, treatment with stem cell transplantation/alkylating agents, and total body irradiation [1, 8, 9, 11, 16].
Similar characteristics of sexual dysfunction have been described for women and men who have suffered from some tumors such as colon and bladder, the oncological treatment for which includes surgery, radiotherapy, and chemotherapy. All this oncological interventions can affect body image, damage pelvic nerves, and generate erectile dysfunction and ejaculatory disorders in men and dyspareunia and decreased libido and orgasm changes in women [8,9,10,11, 13, 15, 17].
In case of bone marrow transplantation, surviving patients have been reported to have severe sexual dysfunction after years of treatment related to the high doses of chemotherapy received and fear of infection related to chronic immunosuppression [9, 11].
Similarly, male and female survivors of head and neck cancer suffer from disturbances such as poor body image, loss of salivation that limits some sexual practices, and in some cases, feelings of guilt related to a history of human papillomavirus infection that negatively affect sexual performance and have a significant psychological impact [8, 9, 11, 13].
Most data of sexual dysfunction in oncological patients are based on retrospective studies and case series, using heterogeneous tools to assess sexual function. This has made it difficult to interpret the magnitude of the problem due to the lack of consistency in the findings [1, 8, 10, 13].
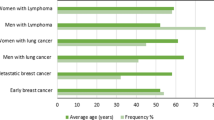
The results of a recent prospective study of 383 patients, 68.1% with breast cancer, 26.3% with lung cancer and 5% with lymphoma, assessed with validated questionnaire for each gender, confirmed important differences according to gender and type of malignancy. This research found that 69% of men versus 31% of women were sexually active and that the prevalence of sexual dissatisfaction was higher in women than in men (76% versus 24%, respectively). In addition, a high incidence of sexual dysfunction was reported in patients with lung cancer and confirmed disturbances in patients with breast cancer reported in previous series [5, 18, 19].
Features of sexual dysfunction in women with cancer
Among women with cancer, sexual dysfunction is one of the most common and distressing side effects of oncological treatment. Low desire, pain, loss of lubrication, and arousal disturbances are the most commonly identified sexual problems in this population [20,21,22].
The predominant literature on the characteristics of sexual dysfunction in women with cancer has been derived from patients with breast, genitourinary, and gynecologic cancers. However, other populations of female cancer survivors also experience high rates of sexual dysfunction related to cancer treatment, such as those with hematologic malignancies treated with hematopoietic cell transplantation and women with head and neck cancer. Table 2 [10, 11, 21,22,23,24,25].
In general, breast cancer patients have been the most studied population and have been described as highly affected because they may suffer the effects of multiple treatments simultaneously, including cancer surgery, radiation therapy, chemotherapy, and hormonal therapy. Most of the information on sexual dysfunction in women with cancer comes from small studies, some retrospective and using heterogeneous instruments for measuring sexual function, which have identified the effect of altered body image in relation to mastectomy scarring and the overall effects of hormone therapy as the main factors associated with sexual dysfunction [1, 6, 16, 25, 26].
Physical changes such as alopecia, surgical ostomy, or mastectomy may result from cancer treatment. The negative impact of such physical changes on the perception of body image is well known. In one study, body image problems, most commonly in the immediate postoperative and treatment periods, were reported by approximately 50 to 75 percent of younger female cancer survivors [27].
On the other hand, depression and its treatment may also have an impact on sexual dysfunction. In one study, an important determinant of decreased sexual desire was the use of antidepressants in up to one-third of breast cancer survivors [28].
All of these factors, it may be difficult to rebuild the intimacy of the relationship after cancer treatment. In addition, couples may not feel comfortable discussing changes in sexual function, which can lead to avoidance of this problem [16, 27,28,29].
In a prospective study of 260 breast cancer patients cited previously, there was a significant tendency for patients with both early and metastatic breast cancer to have severe dysfunction of the sexual response phases. This research also found that women with early breast cancer who received chemotherapy progressively improved their sexual function impairment within 5 years of diagnosis. On the other hand, patients who received hormone therapy had moderate impairment, which also improved within 5 years of diagnosis. Statistically significant associations were found in women with metastatic breast cancer and severe impairment of: arousal (p 0.000), lubrication (p 0.002), orgasm (p 0.000), as well as dissatisfaction with sexual performance (p 0.000) and global sexual dissatisfaction (p 0.000) [19].
On the other hand, another recently recognized group of women with cancer who suffer significant deterioration in their sexual health are those with lung cancer. One study documented that 95% of women with this neoplasm had sexual function scores below the 50th percentile [30]. In the CLARIFY study, there was also evidence of a high percentage of sexual dysfunction in a group of women with lung cancer in which 35.2% were receiving chemotherapy, 26.4% immunotherapy, and 20.5% targeted therapy. Statistically significant associations were documented with severe arousal dysfunction (p 0.016) and global sexual dissatisfaction (p 0.044) [19]. There is another recent study of women with lung cancer in which 64% had stage IV disease, almost half of whom were treated with targeted therapy, and 77% reported moderate to severe sexual dysfunction [31].
Criteria for diagnosis of sexual dysfunction in women with cancer
To evaluate and diagnose sexual dysfunction in women, it is important to know that the sexual response cycle is divided into four phases: desire (libido), arousal, orgasm, and resolution. For many patients, these stages may fluctuate in order, may overlap or be absent in some or all of their sexual encounters. It may not be necessary to reach all stages of response, including orgasm, for subjective satisfaction with the sexual experience [32].
Female sexual dysfunction is diagnosed by identification of diagnostic criteria through medical and sexual history. According to the American Psychiatric Association (APA), based on the Fifth Edition of the Diagnostic and Statistical Manual of Mental Disorders (DSM-5), a diagnosis of sexual disorder in women requires that the sexual problem be recurrent or persistent and cause personal distress or interpersonal difficulties. In addition, the symptom(s) must have been present for at least 6 months and cannot be explained by another isolated disorder [32, 33].
The diagnosis is defined in relation to the presence of alteration of one or more phases of the sexual response cycle, being uncommon to observe a disorder limited to only one phase. The main categories of sexual dysfunction that have been determined in women are: [32, 33].
-
1.
Sexual interest disorder (including hypoactive sexual desire disorder and arousal disorder).
-
2.
Female orgasmic disorder.
-
3.
Genitopelvic pain/ penetration disorder (includes dyspareunia and vaginismus).
-
4.
Substance/drug-induced sexual dysfunction.
-
5.
Other specified sexual dysfunction.
-
6.
Unspecified sexual dysfunction.
The evaluation of the female patients with cancer should include a complete medical, sexual, and gynecologic history. All oncologic treatments received, including previous surgery, radiation therapy, and systemic agents, should be considered. Concomitant medications, including antidepressants, analgesics, and neuropathic medications, should also be reviewed [29, 32, 33].
Short screening questionnaires such as the Sexual Symptom Checklist for Female Patients After Cancer proposed by Bober SL, Reese JB, Barbera can be used to identify sexual symptoms related to cancer treatment and for follow-up. It includes questions about sexual desire, arousal or genital sensitivity, orgasm, dyspareunia, and psychosocial factors affecting sexual function such as anxiety or depression, among others [6, 29].
Self-administered outcome questionnaires can also be used. However, integrating these tools into routine clinical practice can be complex and time-consuming. Specific patient reported validated tools are: [34,35,36,37,38].
-
1.
The Female Sexual Function Index (FSFI): 19-item questionnaire that measures pain, arousal, satisfaction, and orgasm.
-
2.
The National Institute of Health Patient-Reported Outcome Measurement Information System PROMIS®: assesses and monitors the physical, mental, and social health of adults and children. It can be used with the general population as well as with individuals who are living with chronic health conditions.
-
3.
Function and Sexual Satisfaction (PROMIS SexFS): assesses sexual function specifically.
Pelvic examination is recommended in all women with sexual symptoms to identify possible etiologic factors and associated disturbances, being mandatory in the case of sexual pain disorders. Laboratory and imaging are necessary only for associated disorders identified by history/physical examination.[20, 23, 24].
Features of sexual dysfunction in men with cancer
There is a strong correlation between male sexual dysfunction and aging. Generally, in healthy men, alterations in sexual function begin in the early 40 s and increase with each decade. The risk of erectile dysfunction (ED) has been reported to be higher in men with lower levels of education, psychiatric disorders such as depression or anxiety, medication use, obesity, smoking, and chronic medical conditions such as diabetes, chronic renal failure, heart disease, hypertension, and hypogonadism. Table 1 [39, 40].
In one study of cancer population, 49% of men reported the onset of erectile dysfunction (ED) and orgasmic/ejaculatory problems, as well as a decrease in the rate of self-reported sexual activity from 80 to 60% following a diagnosis of cancer. [35, 41].
Most of the data on sexual dysfunction in men with male cancer come from patients with prostate cancer, whose treatment often results in erectile dysfunction. These men are particularly affected because they can suffer from the effects of surgery, radiation therapy, and hormone therapy. However, there is evidence that men with other types of cancer may also experience sexual dysfunction related to oncological treatment Table 3 [11, 16, 42].
Diagnosis of sexual dysfunction in men with cancer
For diagnosis of sexual dysfunction in men with cancer, it should be noted that the types of male sexual dysfunction include the following: [10, 40, 42].
-
Erectile dysfunction (ED): persistent or recurrent inability to achieve or maintain an erection of sufficient firmness and duration for intercourse for at least 3 months. It is the most common form of sexual dysfunction in men. Prevalence of 18% in men aged 50 to 59 years and 37% in men aged 70 to 75 years have been reported.
-
Decreased libido: refers to the loss of sexual interest. A prevalence of 5 to 15% has been reported. It is usually associated with other sexual and urinary dysfunction and increases with age.
-
Ejaculatory disorders: heterogeneous group of disorders including premature, delayed and retrograde ejaculation and anorgasmia. It is generally age related and may be associated with other male sexual dysfunctions, most notably erectile dysfunction.
A complete medical and sexual history should be obtained to confirm disturbances in sexual function. All oncological treatments received, including previous surgery, radiation therapy, and systemic agents, should be taken into account [16, 41].
For screening of sexual dysfunction, self-administered outcome questionnaires can also be used, for example: Patient-Reported Outcomes Measurement Information System (PROMIS) Sexual Function Profile. This tool is a sexual function scale specifically for men with cancer. This eight-question questionnaire assesses erectile function, satisfaction, orgasm/climax, and enjoyment. After registering, it is available online [34, 35].
There are other alternative questionnaires available, including: [35, 42, 43].
-
1.
The International Index of Erectile Function (IIEF): 15-item self-report measure of erectile function, orgasm, desire, satisfaction, and overall satisfaction.
-
2.
The Prostate Cancer Index (PCI) Sexual Function and Sexual Bother: 20-item self-report measure of health-related quality of life that is specific to men with prostate cancer and includes items on the urinary, bowel, and sexual domains.
-
3.
The Sexual Health Inventory for Men (SHIM) or IIEF-5: five-item questionnaire that has been validated for use in the area of erectile function.
There is no data comparing the available questionnaires to define which one is better. The length and type of questions are the main differences between these tools. [16, 34, 42, 43].
Physical examination, looking for absence or loss of normal male hair patterns, gynecomastia, and small testes, as well as evaluation of the cremasteric reflex, could be useful to identify effects of some anticancer treatments such as hormonal therapy, oncological surgery, and radiation therapy. Only associated disorders identified by history/physical examination will require additional laboratory and imaging studies [16, 29, 41].
Approach and treatment of sexual dysfunction in patients with cancer
Sexual health is a multidimensional concept. Therefore, a multidisciplinary approach to the management of sexual dysfunction in patients with cancer is crucial. To provide adequate oncologic care, biological, psychological, couple relationship, and sociocultural factors should always be considered [16, 29, 44].
This biopsychosocial model of care allows patients to receive specific therapeutic interventions, medical management for symptoms and education, as well as early approach for multidisciplinary team with expertise in the management of sexual dysfunction in cancer survivors. This team should include a gynecologist; a psychologist and/or a sex therapist; a psychiatrist, urologist, endocrinologist or/and a pelvic floor therapist [6, 29, 44].
Ideally, all patients should be screened for sexual dysfunction prior to initiation of oncological treatment. Any of the available sexual dysfunction screening questionnaires could be used for this purpose. In addition, because sexual dysfunction can be a late effect of cancer treatment, screening should continue during follow-up after the end of treatment [6, 29, 41].
In an effort to address sexual problems in cancer patients, the American Society of Clinical Oncology, in collaboration with Cancer Care Ontario, has recommended that a member of the health care team have a conversation with each patient to explain the sexual dysfunction he or she may be experiencing as a result of the cancer or its treatment. The patient’s partner may be part of this counseling session, at the patient’s request. The explanation should be appropriate to the patient's educational level, cultural/religious beliefs, and sexual orientation. This conversation should be offered at the time of diagnosis and repeated periodically during follow-up [45, 46].
Overall, in relation to the treatment directed at specific sexual symptoms, for male with cancer, the first line of medical treatment for erectile dysfunction is based on the use of 5-phosphodiesterase inhibitors (sildenafil, vardenafil or tadalafil). A second line is the injection of intracavernous vasodilators at the base of the penis or the use of penile implants. For those who are receiving anti-androgen therapy, the treatment of libido dysfunction is more complicated. [47,48,49,50,51].
In contrast, for women, only the pharmacological treatment of sexual problems has not been found to be enough. The use of systemic hormones such as estrogen or testosterone has not shown benefit and has significant adverse effects. There are some local strategies for treating specific symptoms such as dyspareunia and vaginal stenosis, including lubricants and vaginal dilators, as well as the use of vibrators to improve vaginal blood flow. Globally, it has been found that a multidisciplinary approach is required and non-pharmacological strategies as well as psycho-educational interventions are essential to manage sexual dysfunction in female cancer survivors.[44, 52,53,54,55,56,57,58,59,60,61].
Conclusion
Cancer-related sexual dysfunction is a common condition whose severity varies according to gender, the type of malignancy, and oncological treatment. Given the negative impact on survivors’ quality of life, its detection and management should be incorporated into oncology care guidelines and patients follow-up.
To provide tailored support and overcome barriers and disparities in the management of this condition, it is imperative that all members of the oncology team involved in patient care are aware of the high prevalence and characteristics of sexual health problems in patients with cancer.
A multidisciplinary team that includes psychologists, urologists, gynecologists, psychiatrists, endocrinologists, sex therapists and physiotherapists supporting patients during and after the end of oncological treatment is essential to improve quality of the survivors.
Screening and counseling patients for cancer-related sexual dysfunction prior to treatment is recommended. Providing clear information about the follow-up and rehabilitation options available to address the issue is also critical.
Data availability
Not applicable.
References
Krouwel EM, Albers LF, Nicolai MPJ, Putter H, Osanto S, Pelger RCM, et al. Discussing sexual health in the medical oncologist’s practice: exploring current practice and challenges. J Cancer Educ. 2020;35(6):1072–88.
Carter J, Lacchetti C, Andersen BL, Barton DL, Bolte S, Damast S, et al. Journal of clinical oncology. J Clin Oncol. 2017;36:492–511. https://doi.org/10.1200/JCO.2017.
Walker LM, Wiebe E, Turner J, Driga A, Andrews-Lepine E, Ayume A, et al. The oncology and sexuality, intimacy, and survivorship program model: an integrated, multi-disciplinary model of sexual health care within oncology. J Cancer Educ. 2021;36(2):377–85.
Ferlay J, Colombet M, Soerjomataram I, Mathers C, Parkin DM, Piñeros M, et al. Estimating the global cancer incidence and mortality in 2018: GLOBOCAN sources and methods. Int J Cancer. 2019;144(8):1941–53.
Sánchez F, Pérez Conchillo M, Borrás Valls JJ, Gómez Llorens O, Aznar Vicentee J. Caballero Martín de las Mulas A. Diseño y validación del cuestionario de función sexual de la Mujer (FSM). Aten Primaria. 2004;34(6):286–94.
Bober SL, Varela VS. Sexuality in adult cancer survivors: challenges and intervention. J Clin Oncol. 2012;30(30):3712–9.
Figueroa-Martín L, Duarte-Clíments G, Sánchez-Gómez MB, Ruyman B-B. Abordaje de la sexualidad en atención primaria: ¿qué valorar? Ene. 2015. https://doi.org/10.4321/S1988-348X2015000200006.
Lewis RW, Fugl-Meyer KS, Bosch R, Fugl-Meyer AR, Laumann EO, Lizza E, et al. Epidemiology/risk factors of sexual dysfunction. J Sex Med. 2004;1(1):35–9.
Cakar B, Karaca B, Uslu R. Sexual dysfunction in cancer patients: a review. J Buon. 2013;18(4):818–23.
McCabe MP, Sharlip ID, Lewis R, Atalla E, Balon R, Fisher AD, et al. Incidence and prevalence of sexual dysfunction in women and men: a consensus statement from the fourth international consultation on sexual medicine 2015. J Sex Med. 2016;13(2):144–52.
Salter CA, Mulhall JP. Oncosexology. Urol Clin North Am. 2021;48(4):591–602.
Lovelace DL, McDaniel LR, Golden D. Long-term effects of breast cancer surgery, treatment, and survivor care. J Midwifery Womens Health. 2019;64(6):713–24.
Stulz A, Lamore K, Montalescot L, Favez N, Flahault C. Sexual health in colon cancer patients: a systematic review. Psychooncology. 2020;29(7):1095–104.
Tramacere F, Lancellotta V, Casà C, Fionda B, Cornacchione P, Mazzarella C, et al. Assessment of sexual dysfunction in cervical cancer patients after different treatment modality: a systematic review. Medicina. 2022;58(9):1223.
Celentano V, Cohen R, Warusavitarne J, Faiz O, Chand M. Sexual dysfunction following rectal cancer surgery. Int J Colorectal Dis. 2017;32(11):1523–30.
Jackson SE, Wardle J, Steptoe A, Fisher A. Sexuality after a cancer diagnosis: a population-based study. Cancer. 2016;122(24):3883–91.
Wallington DG, Holliday EB. Preparing patients for sexual dysfunction after radiation for anorectal cancers: a systematic review. Pract Radiat Oncol. 2021;11(3):193–201.
Sánchez-Sánchez F, Ferrer-Casanova C, Ponce-Buj B, Sipán-Sarrión Y, Jurado-López AR, San Martin-Blanco C, et al. Design and validation of a male sexual function questionnaire. SEMERGEN. 2020;46(7):441–7.
Ospina Serrano AV, Maximiano C, de Sanchez CBI, Torrente M, Mendez M, Sanchez JC, et al. Sexual dysfunction in patients with cancer: Results from the CLARIFY project. J Clin Oncol. 2023;41(16):12094–12094.
Shifren JL, Monz BU, Russo PA, Segreti A, Johannes CB. Sexual problems and distress in United States women. Obstet Gynecol. 2008;112(5):970–8.
Basson R. Sexual desire and arousal disorders in women. N Engl J Med. 2006;354(14):1497–506.
Sousa Rodrigues Guedes T, Barbosa Otoni Gonçalves Guedes M, de Castro Santana R, Costa da Silva JF, Almeida Gomes Dantas A, Ochandorena-Acha M, et al. Sexual Dysfunction in Women with Cancer: A Systematic Review of Longitudinal Studies. Int J Environ Res Public Health. 2022; 19(19).
Rodrigues-Machado N, Quintana MJ, Gómez-Gómez R, Bonfill-Cosp X. Sexual function in women with breast cancer: an evidence map of observational studies. Int J Environ Res Public Health. 2022;19(21):13976.
Jing L, Zhang C, Li W, Jin F, Wang A. Incidence and severity of sexual dysfunction among women with breast cancer: a meta-analysis based on female sexual function index. Support Care Cancer. 2019;27(4):1171–80.
Clayton AH, Valladares Juarez EM. Female sexual dysfunction. Med Clin North Am. 2019;103(4):681–98.
Gozzi E, La Manna AR, Rossi L, Colonna M, Ulgiati MA, Romagnoli L, et al. What hides beneath the scar: sexuality and breast cancer what women don’t say: a single-center study. Clin Ter. 2022;173(4):342–6.
Fingeret MC, Teo I, Epner DE. Managing body image difficulties of adult cancer patients: Lessons from available research. Cancer. 2014;120(5):633–41.
Dusetzina SB, Alexander GC, Freedman RA, Huskamp HA, Keating NL. Trends in co-prescribing of antidepressants and tamoxifen among women with breast cancer, 2004–2010. Breast Cancer Res Treat. 2013;137(1):285–96.
Bober SL, Reese JB, Barbera L, Bradford A, Carpenter KM, Goldfarb S, et al. How to ask and what to do. Curr Opin Support Palliat Care. 2016;10(1):44–54.
Sarna L. Women with lung cancer: impact on quality of life. Qual Life Res. 1993;2(1):13–22.
Duma N, Acharya R, Wei Z, Seaborne L, Heisler C, Fidler MJ, et al. MA14.04 sexual health assessment in women with lung cancer (SHAWL) study. J Thorac Oncol. 2022;17(9):93–4.
Basson R. Women’s sexual dysfunction: revised and expanded definitions. Can Med Assoc J. 2005;172(10):1327–33.
Tiefer L. A new view of women’s sexual problems: why new? Why now? J Sex Res. 2001;38(2):89–96.
Cella D, Yount S, Rothrock N, Gershon R, Cook K, Reeve B, et al. The patient-reported outcomes measurement information system (PROMIS). Med Care. 2007;45(5):S3-11.
Huyghe E, Sui D, Odensky E, Schover LR. Needs assessment survey to justify establishing a reproductive health clinic at a comprehensive cancer center. J Sex Med. 2009;6(1):149–63.
Rosen CBJHSLR. The female sexual function index (FSFI): a multidimensional self-report instrument for the assessment of female sexual function. J Sex Marital Ther. 2000;26(2):191–208.
Flynn KE, Lin L, Cyranowski JM, Reeve BB, Reese JB, Jeffery DD, et al. Development of the NIH PROMIS® sexual function and satisfaction measures in patients with cancer. J Sex Med. 2013;10(Supplement_1):43–52.
Maiorino MI, Chiodini P, Bellastella G, Giugliano D, Esposito K. Sexual dysfunction in women with cancer: a systematic review with meta-analysis of studies using the female sexual function index. Endocrine. 2016;54(2):329–41.
Johannes CB, Araujo AB, Feldman HA, Derby CA, Kleinman KP, McKinlay JB. Incidence of erectile dysfunction in men 40 to 69 years old: longitudinal results from the Massachusetts male aging study. J Urol. 2000;163(2):460–3.
Rosen RC, Fisher WA, Eardley I, Niederberger C, Nadel A, Sand M. The multinational men’s attitudes to life events and sexuality (MALES) study: I. Prevalence oSf erectile dysfunction and related health concerns in the general population. Curr Med Res Opin. 2004;20(5):607–17.
Schover LR. Sexual quality of life in men and women after cancer. Climacteric. 2019;22(6):553–7.
Loi M, Wortel RC, Francolini G, Incrocci L. Sexual function in patients treated with stereotactic radiotherapy for prostate cancer: a systematic review of the current evidence. J Sex Med. 2019;16(9):1409–20.
Rosen RC, Riley A, Wagner G, Osterloh IH, Kirkpatrick J, Mishra A. The international index of erectile function (IIEF): a multidimensional scale for assessment of erectile dysfunction. Urology. 1997;49(6):822–30.
Thomas HN, Thurston RC. A biopsychosocial approach to women’s sexual function and dysfunction at midlife: a narrative review. Maturitas. 2016;87:49–60.
Barbera L, Zwaal C, Elterman D, McPherson K, Wolfman W, Katz A, et al. Interventions to address sexual problems in people with cancer. Curr Oncol. 2017;24(3):192–200.
Carter J, Lacchetti C, Andersen BL, Barton DL, Bolte S, Damast S, et al. Interventions to address sexual problems in people with cancer: American society of clinical oncology clinical practice guideline adaptation of cancer care ontario guideline. J Clin Oncol. 2018;36(5):492–511.
Stephenson RA, Mori M, Hsieh YC, Beer TM, Stanford JL, Gilliland FD, et al. Treatment of erectile dysfunction following therapy for clinically localized prostate cancer: patient reported use and outcomes from the surveillance, epidemiology, and end results prostate cancer outcomes study. J Urol. 2005;174(2):646–50.
Pang K, Pan D, Xu H, Ma Y, Wang J, Xu P, et al. Advances in physical diagnosis and treatment of male erectile dysfunction. Front Physiol. 2023;9:13.
Montorsi F, Padma-Nathan H, Glina S. Erectile function and assessments of erection hardness correlate positively with measures of emotional well-being, sexual satisfaction, and treatment satisfaction in men with erectile dysfunction treated with sildenafil citrate (Viagra®). Urology. 2006;68(3):26–37.
Schover LR, Fouladi RT, Warneke CL, Neese L, Klein EA, Zippe C, et al. The use of treatments for erectile dysfunction among survivors of prostate carcinoma. Cancer. 2002;95(11):2397–407.
Miller DC, Wei JT, Dunn RL, Montie JE, Pimentel H, Sandler HM, et al. Use of medications or devices for erectile dysfunction among long-term prostate cancer treatment survivors: potential influence of sexual motivation and/or indifference. Urology. 2006;68(1):166–71.
Brotto LA, Yule M, Breckon E. Psychological interventions for the sexual sequelae of cancer: a review of the literature. J Cancer Surviv. 2010;4(4):346–60.
Carter J, Goldfrank D, Schover LR. Simple strategies for vaginal health promotion in cancer survivors. J Sex Med. 2011;8(2):549–59.
Denlinger CS, Carlson RW, Are M, Baker KS, Davis E, Edge SB, et al. Survivorship: sexual dysfunction (Female), version 1.2013. J Natl Compr Cancer Netw. 2014;12(2):184–92.
Caruso S, Intelisano G, Farina M, Di Mari L, Agnello C. The function of sildenafil on female sexual pathways: a double-blind, cross-over, placebo-controlled study. Eur J Obstet Gynecol Reprod Biol. 2003;110(2):201–6.
Caruso S, Intelisano G, Lupo L, Agnello C. Premenopausal women affected by sexual arousal disorder treated with sildenafil: a double-blind, cross-over, placebo-controlled study. BJOG. 2001;108(6):623–8.
Lindau ST, Gavrilova N. Sex, health, and years of sexually active life gained due to good health: evidence from two US population based cross sectional surveys of ageing. BMJ. 2010;340(mar09 2):c810–c810.
Ratner ES, Foran KA, Schwartz PE, Minkin MJ. Sexuality and intimacy after gynecological cancer. Maturitas. 2010;66(1):23–6.
Bernard S, Tandon P, Waters A, Selmani S, Wiebe E, Turner J, et al. Preferences, barriers and facilitators regarding virtual pelvic healthcare in individuals with gynaecological cancers: protocol for a patient-oriented, mixed-methods study. BMJ Open. 2023;13(1): e067606.
Venkataramu V, Ghotra H, Chaturvedi S. Management of psychiatric disorders in patients with cancer. Indian J Psychiatry. 2022;64(8):458.
Arthur EK, Menon U, Reese JB, Browning K, Overcash J, Rose K, et al. Profiles of women’s adjustment after cancer based on sexual and psychosocial wellbeing: results of a cluster analysis. BMC Cancer. 2022;22(1):1003.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
AVO reports consultant fees from AstraZeneca, Bristol Myers Squibb Company, Pfizer, Merck, Takeda Oncology and Roche; and support for attending meetings and/or travel from Pfizer, Roche, MSD, and Janssen.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ospina Serrano, A.V. Overview of sexual dysfunction in patients with cancer. Clin Transl Oncol 25, 3369–3377 (2023). https://doi.org/10.1007/s12094-023-03311-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12094-023-03311-5