Abstract
Transmissible spongiform encephalopathies (TSEs) are a group of neurodegenerative diseases affecting humans and animals, and scrapie in small ruminants is considered the archetype of TSEs. Derivata di Siria is a native dairy goat of Sicily (south Italy), which is related to Syrian goat breeds. Scrapie disease is considered endemic in Sicily since 1997, following the administration of an infected vaccine. Derivata di Siria goats were involved in six of 66 scrapie-infected flocks in Sicily. Prion protein gene (PRNP) analysis revealed that none of the scrapie cases carried the p.Gln222Lys variant. Sequencing of PRNP in this goat population showed a high frequency (15%) of p.Gln222Lys variant confirming its association with scrapie resistance. PRNP polymorphisms were also analysed in the population of Pantelleria, a small Sicilian Island, where scrapie has never been reported. The native goat breed ‘Pantesca’ was maintained up to almost 80 years and the size of the sheep population on this island has historically been very low. Currently, a crossbreed goat population of 253 heads is present on the island. PRNP genotyping of Pantelleria goats showed genetic variation, with low presence of wild-type goats and the lack of protective alleles. These data reinforce the association between PRNP polymorphisms in small ruminants and scrapie incidence.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Transmissible spongiform encephalopathies (TSEs) or prion diseases are a group of neurodegenerative diseases that can affect humans and animals. Scrapie is considered to be the archetype of the prion diseases and the breeding programmes for genetic resistance are considered the best strategy for the complete eradication of the disease. Polymorphisms of the ovine PRNP gene at codons 136 (Ala or Val), 154 (Arg or His) and 171 (Arg, Gln or His), strongly modulate susceptibility for the disease in sheep (Hunter et al. 1996).
Recent association studies showed that allelic variation of the goat PRNP gene can modulate scrapie susceptibility in goats too. In particular, seven amino acid substitutions have been related to susceptibility or resistance: p.Gly127Ser, p.Ile142Met (Goldmann et al. 2011), p.His143Arg (Billinis et al. 2002), p.Asn146Ser/Asp (Papasavva-Stylianou et al. 2007), p.His154Arg (Billinis et al. 2002; Vaccari et al. 2006; Papasavva-Stylianou et al. 2007; Barillet et al. 2009), p.Gln211Arg (Barillet et al. 2009) and p.Gln222Lys (Acutis et al. 2006; Vaccari et al. 2006; Barillet et al. 2009). Case/control studies performed in Italy (Acutis et al. 2006; Vaccari et al. 2006) and France (Barillet et al. 2009) and experimental challenges with scrapie isolates (Acutis et al. 2012; Corbière et al. 2013; Aguilar-Calvo et al. 2014; Lacroux et al. 2014) reinforced the hypothesis that p.Gln222Lys is a protective PRNP variant also in heterozygosis. In addition, a recent study suggested that Lys222 variant has a protective effect after oral infection with goat bovine spongiform encephalopathy (Aguilar-Calvo et al. 2015).
However, in Greece, p.Gln222Lys variants were detected in scrapie-positive goats in four different, highly endemic geographical areas suggesting either a very high infection pressure or a different scrapie-strain may be responsible for these cases (Fragkiadaki et al. 2011; Boukouvala et al. 2014).
The aim of this study was the characterization of PRNP polymorphisms in Derivata di Siria goats reared in a high-scrapie endemic area of Sicily (southern–central) and in the goat population of Pantelleria island, where scrapie has never been detected. Derivata di Siria animals were involved in six of 29 infected flocks since 1997 (table 1).
Derivata di Siria (Syria-derived) also known as Rossa Mediterranea (Mediterranean Red) is a Sicilian domestic dairy goat which derives from Damascus goat of Syria as other goat breeds in Mediterranean regions. This goat breed has a peculiar reddish-brown coat, it has a milk production of \(\sim 570\) kg per lactation, but it may reach 750 kg. The milk contains 4.11% fat and 3.53% protein, and it is used predominantly for typical cheese production (Noè et al. 2005). The total number of Derivata di Siria in Italy is estimated at about 56,000 and 52% of them are raised mainly in the southern central area of Sicily (Noè et al. 2005).
Pantelleria is the largest volcanic satellite island of Sicily with an area of \(83\,\hbox {km}^{2}\). The island is located in the Mediterranean sea, 100 km southwest of Sicily and just 60 km east of the Tunisian coast. Historically, its small ruminant population has been mainly goats (up to 90%) with a prevalent domestic breeding system for the production of ‘drinking-milk’ for family consumption. The local Mediterranean goat breed ‘Pantesca’ is unfortunately extinct, but some genetic characters might still be present in the local mixed inbreeds. It was probably the first domestic animal to be introduced into the island by prehistoric populations and it has adapted and evolved into an exceptionally robust animal. To increase milk production, local goats were crossed with Saanen in 2008 and Camosciata delle Alpi in 2012. The actual population consists of 253 crossbred animals with both Alpine and Mediterranean phenotypical traits reared in 73 holdings (Italian Anagrafe Zootecnica BDT dataset 2015, http://statistiche.izs.it/portal/page?pageid=73,12918&dad=portal&schem=PORTAL&op=view_rep&p_report=plet_rep_r1_ovi_capr_new&p_liv=R&p_sigla_liv=190). In each farm, <40% kept their own bucks that are often shared between the local farms with a consequent high inbreeding level inside the population (D’Avola, personal communication).
Sicily is considered as a scrapie-endemic region particularly, in the central area of the island after a contaminated vaccine against Mycoplasma agalactiae was used in several flocks (Bertolini et al. 2012). Contaminated vaccine and scrapie outbreaks have never been described in Pantelleria. Our results showed a substantial difference of PRNP polymorphisms between the two analysed populations.
Materials and methods
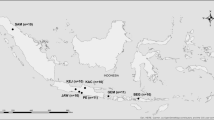
The sample size was calculated with an confidence interval of 95%, considering the officially registered Derivata di Siria goat population in Sicily of 1317 animals (ASSONAPA dataset 2015, http://www.assonapa.com/Consistenze/Cons_razza.asp?Anno=2014&Cod_razza=48) and an expected prevalence of 12% of p.Gln222Lys allele, according to previous preliminary data in goat breeds of southern Italy (Acutis et al. 2008). The minimum sample size was estimated as 149 animals. We have analysed 157 clinically healthy goats (94 males and 63 females) in more than 20 holdings with no history of scrapie-positive animals. All pure-bred farms are located in an area of Sicily particularly endemic for scrapie (figure 1). The animals between 5 and 25% were sampled per herd, including all reproductive males and the best females as indicated by the farmer, based on morphological and reproductive traits. In Pantelleria island, we sampled 56 goats (22.5% of goat population) reared in more than 20 farms, including all males per herd (total 14 bucks).
The area where reproductive males of Derivata di Siria carrying K222 allele were detected. Derivata di Siria goats are mostly reared in the area with the highest incidence of scrapie in Sicily (dotted line). This area is characterized by high frequency of heterozygous p.Gln222Lys (open circles) and homozygous p.Lys222Lys (black circles) males. The total number of males are shown inside each circle.
Peripheral blood sample collection, DNA extraction and genetic analysis were performed as described in Migliore et al. (2015). Statistical analysis was performed by Fisher’s test. Hardy–Weinberg equilibrium (HWE) for the two analysed goat populations was established by Genotype Trasposer V.1.0.
Results
PRNP analysis in Derivata di Siria goats
The PRNP analysis in Derivata di Siria revealed a moderate degree of variability with six polymorphic sites combined in 19 different genotypes (table 2). Polymorphisms, p.Gln37Val, p.His143Arg, p.Arg154His, p.Pro168Gln, p.Gln222Lys and p.Ser240Pro were in HWE as all previously observed in Italian goats (Vaccari et al. 2009). Silent mutations, p.Pro42Pro (ccg/cca) and p.Ser138Ser (agc/agt) were detected, while no novel amino acid substitutions were found.
As in all Italian goat breeds, two main PRNP alleles are present in Derivata di Siria characterized by the presence of serine or proline at codon 240 (frequencies of 48.7 and 51.3%, respectively). The Val37, Arg143 and Gln168 variants were detected at <3% in Derivata di Siria, but these polymorphisms convey little protection against scrapie disease. The histidine substitution at codon 154 (p.Arg154His) showed a frequency of 9%. This variant has a variable degree of protection against classical scrapie (Billinis et al. 2002; Barillet et al. 2009), but increases susceptibility to atypical scrapie in goats (Colussi et al. 2008).
The protective p.Gln222Lys polymorphism with at least one haplotype encoding Lys222 was found in 40 goats (25.5% of the sample) from different herds. There were 34 p.Gln222Lys heterozygous (24 males and 10 females; 21.7%) and six homozygous goats (five males and one female; 3.8%) (table 2). The frequency of p.Gln222Lys in this goat population was 15% and as in other countries, Lys222 was mostly found in haplotype linkage with p.Ser240Pro. These animals were reared in herds located in different Sicilian provinces and inbreeding problems might be less frequent than in Girgentana goat, another native Sicilian diary breed with a higher frequency of p.Gln222Lys (18.7%) (Migliore et al. 2015). The protective polymorphisms, p.Asn146Ser and p.Asn146Asp, which are very common in the Cyprus goat was not found in Derivata di Siria despite the fact that both are derived from a common ancestor, the Syrian goat.
PRNP analysis in Pantelleria goats
In goat population of Pantelleria, we found seven polymorphic sites in PRNP: p.Gly37Val, p.Gly127Ser, p.Met137Ile, p.Ile142Thr, p.Arg154His, p.Arg211Gln and p.Ser240Pro combined in 18 different genotypes (table 3), two silent mutations, p.Pro42Pro (ccg/cca) and p.Ser138Ser (agc/agt) were also present. The population appeared to be in HWE.
All detected mutations have been previously described in Italian goat breeds (Vaccari et al. 2009). Proline at codon 240 is the most frequent allelic variant (47.3%) as in other Italian goats, while polymorphisms at codons 37 (Val), 127 (Ser) and 137 (Ile) showed a low frequency inside the population (\(<\!2\%\)). Histidine at codon 154 and threonine at codon 142 were detected with frequencies of 13.3 and 8.4%, respectively. We also found a significant presence of glutamine at codon 211 (23.1%). This polymorphism has not been detected in Sicilian pure goat breeds yet, but it is common in Saanen and Camosciata delle Alpi raised in north Italy with frequencies of 10.2 and 13.7%, respectively (Acutis et al. 2008). Scrapie resistant-associated polymorphisms such as p.Gln222Lys detected in the main island and p.Asn146Ser/Asp of Cyprus were not detected in Pantelleria.
Discussion
PRNP polymorphisms showed relevant differences between the two goat populations of this study (table 4). Polymorphisms in Derivata di Siria goats were similar to another native goat breed Girgentana, with a high frequency of protective polymorphism p.Gln222Lys (15 and 18%, respectively) (Migliore et al. 2015). These frequencies are among the highest reported of goat breeds of Italy. The p.Gln222Lys is present at lower frequency in other countries, i.e., <1% in goats reared for milk productions in UK (Goldmann et al. 2011), 6.3% in Alpine e Saanen breeds in France (Barillet et al. 2009) and 5.6% in Greek goats (Boukouvala et al. 2014). However, a high frequency (25%) of p.Gln222Lys was recently described in Ducht Toggenburger goats (Windig et al. 2016).
Scrapie in Sicily is endemic since 1997 when clinical signs of the disease was observed in a herd with mixed goat and sheep. To date, 29 infected goat flocks have been recorded in Sicily, with six involving Derivata di Siria breed (table 1). The outbreaks occurred mainly in the southern central area of Sicily, where a scrapie-contaminated vaccine against Mycoplasma agalactiae was accidentally administered in 1992 (Caramelli et al. 2001). A recent epidemiological study revealed that this area of Sicily shows a significant long-term effect caused by the contaminated vaccine circulation and presents the highest incidence of scrapie in Sicily (Bertolini et al. 2012). It may therefore, not be a coincidence that in the same area, Girgentana (Migliore et al. 2015) and Derivata Siria goats carrying high frequency of the p.Gln222Lys are mainly reared (figure 1). High frequencies for ARR resistant allele (30–40%) have already been reported in Sicilian sheep before starting of the regional breeding plan for genetic resistance (Van Kaam et al. 2008). We speculate that our data indicate that positive selection for resistance-associated PRNP polymorphisms is in place when the infection incidence is high.
The p.Asn146Ser/p.Asn146Asp polymorphisms which are very common and related to resistance in Damascus goats of Cyprus (Papasavva-Stylianou et al. 2007), was not detected in Derivata di Siria although, the two breeds derived from a common ancestor. It might be possible that p.Asn146Ser protective variant in Cyprus was positively selected during the epidemic of scrapie in the mid 1980s (Gravenor et al. 2004). Prion proteins persist in the soil for long period (Georgsson et al. 2006) and environmental factors can contribute to the appearance of different prion strains (Smith et al. 2011). Scrapie in small ruminants and chronic washing disease in cervids can be transmitted through soil (Smith et al. 2011). In a natural cycle, in which scrapie strains from animals go to the environment and vice versa, the older infected animals (e.g. milk-producing animals) have higher possibility to contaminate the environment through fluids, placentas or carcasses and naïve animals acquire the pathologic prions, while grazing (O’Rourke et al. 2011; Smith et al. 2011). It is well established that lateral infection occurs within sheep since scrapie-free animals can get infected when transferred to affected herds (Foster et al. 2006). For this reason, different alleles in native breeds might be positively selected in relation to the local circulating scrapie strains originated in specific environmental conditions, establishing a kind of natural equilibrium between pathogenic prions and PRNP polymorphisms in specific geographic areas. The use of contaminated vaccine in Italy altered this equilibrium with a consequent increase in scrapie incidence followed by an increase in the frequency of the local protective alleles. It is well known that the prevalence of specific alleles in local sheep breeds differs among different countries, i.e. VRQ allele carriers in Italian breeds (Sarda, Valle del Belice and Comisana sheep) (Van Kaam et al. 2008) are fewer than in the so-called ‘valine breeds’ (Cheviot, Swaledales and Shetland sheep) in the UK (Hunter 1997). A large variety of alleles are observed in scrapie-free (Goldmann et al. 2011) as in Pantelleria goats population, which showed several marked differences from the goats of Sicily. A high frequency was found for the p.Arg211Gln polymorphism (23.1%) in Pantelleria goats, while it was absent from Derivata di Siria; because this polymorphism is very common in Alpine goat breeds reared in north Italy (Acutis et al. 2008), its presence might be explained by the recent crossbreeding on the island. Interestingly, threonine at codon 142 was detected at higher frequency (8.4%) compared to other southern Italian goats (0.6%) (Acutis et al. 2008). This polymorphism was not present in Sicilian native breeds Derivata di Siria, Girgentana and in the Alpine breeds, suggesting that it might be derived from the original native ‘Pantesca’ breed. The association of polymorphism p.Ile142Thr with scrapie susceptibility is unknown. A statistically significant difference (\(P{<}0.001\)) based on allele frequencies among the two populations was found in the ratio of wild type and polymorphic variants. Surprisingly, while Derivata di Siria showed 70% of wild-type genotypes, Pantelleria goats showed only 52.7%. Statistically significant difference (\(P{<}\,0.001\)) was also found, while comparing wild-type genotypes to any other carrier of a polymorphic variant. We found 52 and 25% of wild-type homozygotes in Derivata di Siria and in Pantelleria goats, respectively. Whether absence of scrapie in Pantelleria is associated with low presence of wild-type PRNP alleles in the island remains to be studied in future.
This Sicilian genetic study suggests that polymorphisms in the prion protein gene might be related to the relative incidence and selective pressure of scrapie strains. Natural selection might occur in areas highly endemic for scrapie. The presence of protective variants in some breeds in certain geographical areas could be an important resource to facilitate the breeding programmes for TSE resistance in native breeds with a good impact on the preservation of livestock biodiversity and valourization of typical animal productions.
References
Acutis P. L., Bossers A., Priem J., Riina M. V., Peletto S., Mazza M. et al. 2006 Identification of prion protein gene polymorphisms in goats from Italian scrapie outbreaks. J. Gen. Virol. 87, 1029–1033.
Acutis P. L., Colussi S., Santagada G., Laurenza C., Maniaci M. G., Riina M. V. et al. 2008 Genetic variability of the PRNP gene in goat breeds from northern and southern Italy. J. Appl. Microbiol. 104, 1782–1789.
Acutis P. L., Martucci F., D’Angelo A., Peletto S., Colussi S., Maurella C. et al. 2012 Resistance to classical scrapie in experimentally challenged goats carrying mutation K222 of the prion protein gene. Vet. Res. 43, 8–17.
Aguilar-Calvo P., Espinosa J. C., Pintado B., Gutiérrez-Adán A., Alamillo E., Miranda A. et al. 2014 Role of the goat K222-PrP(C) polymorphic variant in prion infection resistance. J. Virol. 88, 2670–2676.
Aguilar-Calvo P., Fast C., Tauscher K., Espinosa J. C., Groschup M. H., Nadeem M. et al. 2015 Effect of Q211 and K222 PRNP polymorphic variants in the susceptibility of goats to oral infection with goat bovine spongiform encephalopathy. J. Infect. Dis. 212, 664–672.
Barillet F., Mariat D., Amigues Y., Faugeras R., Caillat H., Moazami-Goudarzi K. et al. 2009 Identification of seven haplotypes of the caprine PrP gene at codons 127, 142, 154, 211, 222 and 240 in French Alpine and Saanen breeds and their association with classical scrapie. J. Gen. Virol. 90, 769–776.
Bertolini S., Maurella C., Bona C., Ingravalle F., Desiato R., Baioni E. et al. 2012 A relevant long-term impact of the circulation of a potentially contaminated vaccine on the distribution of scrapie in Italy. Results from a retrospective cohort study. Vet. Res. 43, 63.
Billinis C., Panagiotidis C. H., Psychas V., Argyroudis S., Nicolaou A., Leontides S. et al. 2002 Prion protein gene polymorphisms in natural goat scrapie. J. Gen. Virol. 83, 713–721.
Boukouvala E., Katharopoulos E., Stewart P., Palaska V., Giadinis N., Arsenos G. et al. 2014 PRNP polymorphisms in Greek goats affected with natural scrapie. Prion 8, suppl. 87, 65.
Caramelli M., Ru G., Casalone C., Bozzetta E., Acutis P. L., Calella A. et al. 2001 Evidence for the transmission of scrapie to sheep and goats from a vaccine against Mycoplasma agalactiae. Vet. Res. 148, 531–536.
Colussi S., Vaccari G., Maurella C., Bona C., Lorenzetti R., Troiano P. et al. 2008 Histidine at codon 154 of the prion protein gene is a risk factor for Nor98 scrapie in goats. J. Gen. Virol. 89, 3173–3176.
Corbière F., Perrin-Chauvineau C., Lacroux C., Costes P., Thomas M., Brémaud I. et al. 2013 PrP-associated resistance to scrapie in five highly infected goat herds. J. Gen. Virol. 94, 241–245.
Foster J., McKenzie C., Parnham D., Drummond D., Chong A., Goldmann W. et al. 2006 Lateral transmission of natural scrapie to scrapie-free New Zealand sheep placed in an endemically infected UK flock. Vet. Rec. 159, 633–634.
Fragkiadaki E. G., Vaccari G., Ekateriniadou L. V., Agrimi U., Giadinis N. D., Chiappini B. et al. 2011 PRNP genetic variability and molecular typing of natural goat scrapie isolates in a high number of infected flocks. Vet. Res. 42, 104.
Georgsson G., Sigurdarson S. and Brown P. 2006 Infectious agent of sheep scrapie may persist in the environment for at least 16 years. J. Gen. Virol. 87, 3737–3740.
Goldmann W., Ryan K., Stewart P., Parnham D., Xicohtencatl R., Fernandez N. et al. 2011 Caprine prion gene polymorphisms are associated with decreased incidence of classical scrapie in goat herds in the United Kingdom. Vet. Res. 42, 110–117.
Gravenor M. B., Papasozomenos P., McLean A. R. and Neophytou G. 2004 A scrapie epidemic in Cyprus. Epidemiol. Infect. 132, 751–760.
Hunter N. 1997 PrP genetics in sheep and the applications for scrapie and BSE. Trends Microbiol. 5, 331–334.
Hunter N., Foster J. D., Goldmann W., Stear J. M., Hope J. and Bostock C. 1996 Natural scrapie in closed flock of Cheviot sheep occurs only in specific PrP genotypes. Arch. Virol. 141, 809–824.
Lacroux C., Perrin-Chauvineau C., Corbière F., Aron N., Aguilar-Calvo P., Torres J. M. et al. 2014 Genetic resistance to scrapie infection in experimentally challenged goats. J. Virol. 88, 2406–2413.
Migliore S., Agnello S., Chiappini B., Vaccari G., Mignacca S. A., Di Marco Lo Presti V. et al. 2015 Biodiversity and selection for scrapie resistance in goats: genetic polymorphism in “Girgentana” breed in Sicily, Italy. Small Rum. Res. 125, 137–141.
Noè L., Gaviraghi A., D’Angelo A., Bonanno A., Di Trana A., Sepe L. et al. 2005 Le razze caprine d’Italia. In L’ alimentazione della capra da latte (ed. G. Pulinaa), , Bologna, Avenue Media, p. 381–435.
O’Rourke K. I., Zhuang D., Truscott T. C., Yan H. and Schneider D. A. 2011 Sparse PrPSc accumulation in the placentas of goats with naturally acquired scrapie. BMC Vet. Res. 7, 7.
Papasavva-Stylianou P., Kleanthous M., Toumazos P., Mavrikiou P. and Loucaides P. 2007 Novel polymorphisms at codons 146 and 151 in the prion protein gene of Cyprus goats, and their association with natural scrapie. Vet. J. 173, 459–462.
Smith C. B., Booth C. J. and Pedersen J. A. 2011 Fate of prions in soil: a review. J. Environ. Qual. 40, 449–461.
Vaccari G., Di Bari M. A., Morelli L., Nonno R., Chiappini B., Antonucci G. et al. 2006 Identification of an allelic variant of the goat PrP gene associated with resistance to scrapie. J. Gen. Virol. 87, 1395–1402.
Vaccari G., Panagiotidis C. H., Acìn C., Peletto S., Barillet F., Acutis P. et al. 2009 State-of-the-art review of goat TSE in the European Union, with special emphasis on PRNP genetics and epidemiology. Vet. Res. 40, 48.
Van Kaam J., Finocchiaro R., Vitale M., Pinelli F., Scimonelli M. Vitale F. et al. 2008 Prion protein gene frequencies in three Sicilian dairy sheep populations. Ital. J. Anim. Sci. 7, 87–94.
Windig J. J., Hoving R. A., Priem J., Bossers A., van Keulen L. J. and Langeveld J. P. 2016 Variation in the prion protein sequence in Dutch goat breeds. J. Anim. Breed Genet. 133, 366–374.
Acknowledgements
The authors are grateful to Sicilian farmers for their collaboration. This study was supported by EMIDA ERA-NET project: ‘GOAT-TSE-FREE’, by Ricerca Finalizzata 2010 Project Code: RF-2010-2318525 to MV and RC IZSSi 16/11 to SA from Italian Ministry of Health. WG supported by the Biotechnology and Biological Sciences Research Council (Strategic Programme Grant BB/J004332/1 to the Roslin Institute).
Author information
Authors and Affiliations
Corresponding author
Additional information
Corresponding editor: Silvia Garagna
[Migliore S., Agnello S., D’Avola S., Goldmann W., Di Marco Lo Presti V. and Vitale M. 2017 A cross-sectional study of PRNP gene in two native Sicilian goat populations in Italy: a relation between prion gene polymorphisms and scrapie incidence. J. Genet. 96, xx-xx]
Rights and permissions
About this article
Cite this article
Migliore, S., Agnello, S., D’Avola, S. et al. A cross-sectional study of PRNP gene in two native Sicilian goat populations in Italy: a relation between prion gene polymorphisms and scrapie incidence. J Genet 96, 319–325 (2017). https://doi.org/10.1007/s12041-017-0776-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12041-017-0776-9