Abstract
Dysfunction of the neuromuscular junction is involved in a wide range of muscular diseases. The development of neuromuscular junction through which skeletal muscle is innervated requires the functional modulation of acetylcholine receptor (AchR) clustering on myofibers. However, studies on AchR clustering in vitro are mostly done on monolayer muscle cell culture, which lacks a three-dimensional (3D) structure, a prominent limitation of the two-dimensional (2D) system. To enable a better understanding on the structure–function correlation underlying skeletal muscle innervation, a muscle system with a well-defined geometry mimicking the in vivo muscular setting is needed. Here, we report a 3D bio-artificial muscle (BAM) bioengineered from green fluorescent protein-transduced C3H murine myoblasts as a novel in vitro tissue-based model for muscle innervation studies. Our cell biological and molecular analysis showed that this BAM is structurally similar to in vivo muscle tissue and can reach the perinatal differentiation stage, higher than does 2D culture. Effective clustering and morphological maturation of AchRs on BAMs induced by agrin and laminin indicate the functional activity and plasticity of this BAM system toward innervation. Taken together, our results show that the BAM provides a favorable 3D environment that at least partially recapitulates real physiological skeletal muscle with regard to innervation. With a convenience of fabrication and manipulation, this 3D in vitro system offers a novel model for studying mechanisms underlying skeletal muscle innervation and testing therapeutic strategies for relevant nervous and muscular diseases.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Native skeletal muscle is innervated and controlled by motor neurons through the neuromuscular junction (NMJ), a synapse where motor neuron axons form presynaptic apparatus releasing acetylcholine, a neurotransmitter, to activate acetylcholine receptors (AchRs) on the postsynaptic side resided on the muscle cell membrane [1–3]. During NMJ development, AchRs are densely clustered on the postsynaptic side of the future neuromuscular junction before the nerve contacts the skeletal muscle at this NMJ [1, 2, 4].
A growing number of studies have suggested that defects in the structure, differentiation, and function of NMJ are a key pathological alteration involved in a wide range of diseases, such as age-related muscle wasting [5–7], congenital myasthenic syndromes [8, 9], and myasthenia gravis (an autoimmune syndrome) [10]. Impaired NMJ can lead to muscle weakness, fatigue, and even muscle loss. Furthermore, diseased skeletal muscle often contains large areas of fibrotic tissue lacking innervation, which could result in further functional deterioration, such as atrophy. Thus, the modulation of innervation through NMJ in muscle tissue can be a critical approach for developing therapeutic strategies for these neuromuscular disorders.
Skeletal muscle innervation requires clustering of mature AchRs on the postsynaptic side, which provides a functional and structural foundation for developing NMJ through which neurons innervate myofibers [4]. Previous studies on in vitro NMJ development and innervation are mostly based on two-dimensional (2D) culture systems [11–13]. Here, we propose that a tissue-engineered bio-artificial muscle (BAM) can be used as a three-dimensional (3D) in vitro system to study NMJ formation and muscle innervation. The advantages of tissue-based in vitro systems reside in that cells organized into 3D structures are often more representative of in vivo tissues than 2D monolayer culture systems [14], allowing investigations of biomedical processes at the functional tissue level [15]. Moreover, like 2D monolayer culture, this 3D system retains the convenience of manipulating the system variables, dissecting the mechanisms or pathways and analyzing results [16].
In this study, skeletal muscle myoblasts were tissue engineered into a miniature 3D bio-artificial muscle [17]. The BAM developed aligned and well-organized myofibers resembling in vivo muscle tissue. The gene expression profiling of myosin heavy chain isoforms showed that the muscle cells within the BAM were differentiated approximately to the perinatal stage. The BAM exhibits the ability to effectively cluster AchRs and improve maturation of AchR clusters in response to agrin, a nerve-derived protein [3, 18], and laminin, a major basal lamina component that plays an important role in NMJ development [19, 20], respectively. Our results suggest that this BAM partially resembles real muscle tissue topologically and molecularly. The BAM maintains physiological capabilities toward innervation. This simplified 3D system can be used to investigate innervation and NMJ development and for developing treatment for muscle-related diseases. The mechanistic insight gained through this 3D BAM system may be a more faithful reflection of in vivo muscle tissue.
Materials and Methods
Myoblast Tissue Culture
The green fluorescent protein (GFP)-transduced skeletal myoblasts, as previously described [21, 22], were maintained in a primary mouse myoblast growth medium (PMMGM) [20 % fetal bovine serum (Gibco, Grand Island, New York), 1:1 Dulbecco’s modified Eagle medium (DMEM; Gibco)/fibroblast growth medium (FGM; Lonza, Gaithersburg, Maryland), 1 % ITS + 1 (Sigma, St. Louis, Missouri), and 1 % penicillin–streptomycin] in collagen-coated tissue culture dishes. FGM contained 2 μg/ml insulin and 400 ng/ml human fibroblast growth factor. When near confluent, the cells were trypsinized with 0.5 mg/ml trypsin, pelleted by centrifugation, resuspended in PMMGM, and harvested for bio-artificial muscle tissue engineering.
Preparation of BAMs
BAMs were prepared as previously described [17]. Briefly, 0.2 million GFP-transduced primary mouse myoblast (PMMGFP) cells were trypsinized and suspended in 120 μl fibrin gel made by mixing 0.5 mg/ml fibrinogen and 1 U/ml thrombin (both purchased from Tisseel, Baxter Health Care, Westlake Village, California) and cast in silicone rubber molds [17]. The cell–gel mixture was allowed to sit for 30 min at 37 °C and was then overlaid gently with 150 μl growth medium per well. The cell–gel mixture gelled rapidly within 48 h post-casting. BAMs were formed and kept under tension between two silicone posts. The passive forces in the contracted fibrin gel allow the myoblasts become aligned to the long axis of the BAMs. BAMs were maintained in growth medium for the first 2 days and then switched to differentiation medium (1:1 FGM/DMEM, 10 % horse serum, and 1 % penicillin–streptomycin supplemented with aprotinin) for 14 days. Medium was changed daily. BAMs were treated with 1 μg/ml cytosine arabinoside (Ara C) from the third day for 11 days to eliminate undifferentiated proliferating cells.
Treatment of BAMs with Agrin
On the 14th day after casting BAMs, 6 μl 1 U/ml mouse agrin (generously provided by Dr. Justin Fallon, Brown University) was added into 150 μl differentiation medium plus aprotinin and incubated with BAMs overnight.
Treatment of BAMs with Laminin
The BAMs were fabricated from 0.2 million PMMGFP suspended in 120 μl fibrin gel supplemented with laminin (10, 25, and 50 nM) (BD Bioscience, San Jose, California). BAMs were cast into the molds and incubated as described above, but with Ara C treatment from the third day to the fifth day only.
Quantitative PCR
Total RNA was extracted from BAM samples at day 6, and myoblasts were cultured in Petri dishes, respectively, with a RiboPure kit (Ambion, Austin, Texas). The expression of myosin heavy chain isoforms was investigated using TaqMan gene expression assays (Applied Biosystems, Foster City, California). The following myosin heavy chain transcripts were investigated: embryonic (Myh3, Mm01332463_m1), perinatal (Myh8, Mm01329512_m1), adult fast IIa (Myh2, Mm-00454991_m1), adult fast IIb (Myh4, Mm01332518_m1), and adult fast IId (Myh1, Mm01332489_m1) with mouse GAPDH as an endogenous control. BAM and cell samples were tested in triplicate on a 7500 Fast Real-Time PCR system (Applied Biosystems) using TaqMan one-step RT-PCR master mix (Applied Biosystems). Data were analyzed with Sequence Detection Software v1.3.1 (Applied Biosystems).
Confocal Microscopy
After 14 days of incubation, the BAM samples were fixed in 2 % formaldehyde for 20 min at room temperature and rinsed with phosphate-buffered saline (PBS) for 5 min; then, BAMs were stained with Alexa 594-bungarotoxin (Invitrogen, Grand Island, New York) for 45 min and rinsed with PBS twice. Confocal images were captured with a Leica confocal microscope (TCS SP2 AOBS) (Leica Microsystems, Wetzlar, Germany). Ten random fields were recorded for each BAM sample. The average number of AchR clusters on muscle fibers per 104 μm2 GFP-positive area was quantified using MetaMorph software.
Statistical Analysis
All results are expressed as mean ± SD. A t test was performed using Sigma Stat software (Systat Software, San Jose, California).
Results
BAMs Were Successfully Fabricated In Vitro
We prepared a tissue-engineered three-dimensional bio-artificial muscle as described previously [17]. This BAM was formed and maintained between two parallel flexible silicone posts (Fig. 1a) (see details in “Materials and Methods”). Muscle cells within the BAMs were self-organized into aligned myofibers and stably expressed GFP protein from a construct transduced into the isolated C3H skeletal muscle myoblasts [17] (Fig. 1b, c).
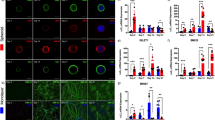
An in vitro prepared BAM. a The BAM (indicated by a yellow arrow) is grown between two flexible silicone posts (indicated by white arrowheads). Scale bar, 5 mm. b The confocal image of the BAM after being treated with agrin. Muscle fibers express GFP (green) and form AchR clusters (red) stained with Alexa 594-bungarotoxin. c The enlarged image of the dash-lined rectangular region in b. The representative parallel myofibers are outlined by white dash lines. Scale bar, 75 μm
Skeletal Muscle Myoblasts Underwent Differentiation Within 3D BAMs
Myosin heavy chain (MHC) isoforms are expressed in a developmentally regulated manner in skeletal muscle [23, 24]. For embryonic and perinatal stages, MHC3 and MHC8 are expressed, respectively. Three MHC1 (adult fast IId), two (adult fast IIa), and four (adult fast IIb) isoforms are expressed at the adult stage. The maturation degree of BAMs can be estimated by examining the types and levels of MHC isoforms expressed. Quantitative PCR was employed to profile relative expression levels of these isoforms in BAMs and myoblasts cultured in a monolayer in Petri dishes. Compared to 2D cultured cells that expressed all MHC isoforms at the relatively low level, the cells from the BAMs harvested at day 6 post-casting highly expressed four of the five isoforms measured (Fig. 2). Among these four types, the expression of the embryonic and perinatal isoforms (MHC3 and MHC8) was significantly higher than that of two other adult isoforms (MHC1 and MHC4), suggesting that muscle cells within BAMs can differentiate to the level comparable to the perinatal skeletal muscles under the culture condition.
Agrin Effectively Induces the Formation of AchR Clusters on BAMs
During synaptogenesis, agrin, a nerve-derived protein, acts to activate muscle-specific kinase, thereby promoting AchR clustering [3, 18]. To test whether BAMs are capable of forming AchR clusters in response to agrin, BAMs were incubated with agrin overnight (see details in “Materials and Methods”). Compared with non-treated BAMs, the number of AchR clusters in the BAMs treated with agrin was increased 7.3-fold from 0.8 to 5.8 AchR clusters per 104 μm2 GFP-positive area (p < 0.005) (Fig. 3a–e).
Agrin increases the number of AchR clusters on BAMs. Myofibers (green) in BAMs express GFP and AchR clusters (red) stained with Alexa 594-bungarotoxin. a AchR clusters are formed on the myofibers in the control BAM. b Compared to the control, more AchR clusters are formed on the muscle cells in the BAM treated with agrin. c, d The enlarged images of the dash-lined rectangular regions in a and b. AchR clusters are indicated by white arrowheads. e The quantification of the density of AchR clusters (mean ± SD) is shown (p < 0.005, n = 4 samples for each group, ten random fields analyzed for each sample). Scale bar, 75 μm
Laminin Promotes the Morphological Maturation of AchR Clusters on BAMs
The mature AchR clusters often aggregate in a pretzel shape (Fig. 4b), offering morphological basis for functional innervation. Although agrin effectively increased the number of AchR clusters in the BAMs, agrin-induced AchR clusters were in an oval plaque shape, an early stage of AchR clustering (Fig. 4a). To examine whether 3D BAMs can generate mature AchR clusters in vitro under the induction of laminin, we incubated BAMs with laminin at three different concentrations: 10, 25, and 50 nM. At all these concentrations, the pretzel-like aggregation of AchR clusters was observed (Fig. 5a), indicating that laminin promotes the maturation of AchR clusters, consistent with its known role [19]. Addition of agrin to laminin-treated BAM did not increase the number of mature (pretzel-shaped) AchR clusters, and pretzel-shaped AchR clusters were not further matured morphologically (Fig. 5a). However, the total number of AchR clusters (immature) was increased when adding agrin to the laminin treatment (Fig. 5b–e), consistent with the agrin’s role in inducing effective formation of AchR clusters. Thus, in the 3D BAMs, laminin but not agrin promotes the appearance of morphologically mature AchR clusters.
Morphology of induced AchR clusters. Myofibers are green, and AchR clusters are stained in red. a Agrin induces the formation of an AchR cluster in an oval plaque shape (outlined by a dash line) lacking branches. b Laminin at the concentration of 50 nM induces the aggregation of AchR clusters in a pretzel shape (outlined by a dash line) with multiple branches (indicated by white arrowheads), a morphological indicator for the maturation of AchR clusters
Laminin promotes the morphological maturation of AchR clusters. Myofibers are green, and AchR clusters are stained in red. a All three different concentrations (10, 25, and 50 nM) of laminin induced a mature and pretzel-shaped aggregation of AchR clusters. Addition of agrin did not further increase the maturity of AchR clusters or the number of mature AchR clusters. Scale bar, 5 μm. b AchR clusters are formed on the myofibers of the BAM treated with 25 nM laminin. c More AchR clusters are formed on muscle cells of the BAM treated with 25 nM laminin and agrin. Scale bar for b and c, 75 μm. d, e The enlarged images of the dash-lined rectangular regions in a and b. AchR clusters are indicated by white arrowheads
Discussion
The proper clustering of AchR, an essential step leading to the neuromuscular junction formation, provides the morphological basis for skeletal muscle innervation [1, 3]. Much of our understanding about AchR, NMJ, and muscle innervation comes from studies on conventional 2D culture lacking well-defined spatial structure and a mechanical setting that can limit the reliability and significance of studies. Thus, 3D in vitro models have been proposed to overcome the limitations of 2D culture for various fields, for example, stem cells [25] and cancer research [26]. To determine whether a 3D skeletal muscle system in vitro can be used for skeletal muscle innervation, we fabricated a BAM where myoblasts were tissue engineered into 3D muscle tissue in the hope of mimicking the real physiological skeletal muscle.
We characterized BAMs in terms of MHC gene expression in that myosin is a major component of the contractile apparatus of muscle, and its isoforms are expressed in a tissue-specific and developmentally regulated manner in skeletal muscle [23, 24]. The BAMs prepared under our experimental condition can differentiate to the level similar to the muscle at the perinatal stage. This result indicates that the BAMs more closely resemble in vivo muscle tissue than do monolayer-cultured muscle cells that exhibit almost no differentiation. The higher differentiation state may be associated with greater functional capabilities. These BAMs have been shown to contract and generate force when stimulated [17], suggesting that the BAM system is functionally close to in vivo muscles. The muscle cells/myofibers in BAMs may have the potential to reach a more advanced development stage if BAM culture conditions can be optimized to allow further differentiation.
Functional NMJ requires the topological maturation of the postsynaptic apparatus from an oval plaque of AchR clusters into a complex postnatal topology characterized by a pretzel-shaped and branched morphology [27]. Although the artificial muscle lacks mature AchR clusters that are essential for innervation, the BAM is capable of responding to agrin, a nerve-derived protein, markedly and effectively forming a large number of AchR clusters. However, the agrin-stimulated receptor clusters are morphologically immature, showing an oval plaque shape. Laminin has been reported to induce maturation of AchR clusters in monolayer cell culture [19, 20]. We found that the addition of laminin to BAM fabrication significantly induces the pretzel-shaped aggregation of AchR clusters, an advanced structure. Thus, laminin-mediated maturation of AchR clusters confers a topological advantage on the BAMs for NMJ development.
Together with proper protein induction, the formation and maturation of AchR clusters, a key step toward innervation, can be readily modulated in a nerve-independent defined 3D bio-artificial muscle model. Given this morphological plasticity and functional activity, the BAMs would be a suitable tissue-based in vitro model for studies on skeletal muscle innervation. Our study here serves as an example where tissue engineering and neurobiology are integrated into one single system for the study of the molecules affecting skeletal muscle innervations. This system can also be widely applied for identifying other proteins/factors that might improve AchR formation and maturation and studying single or cumulative effects of different molecules on muscles at the tissue level. The understanding on cross talk between proteins/growth factors and the neuromuscular systems may provide potential targets to advance skeletal muscle innervations and contribute to the development of new potential therapeutic approaches for the treatment of neuromuscular dysfunctions, especially with the recent ability to form functional NMJs in vitro between human skeletal muscle fibers and human stem cell-derived motoneurons [28].
References
Sanes JR, Lichtman JW (2001) Induction, assembly, maturation and maintenance of a postsynaptic apparatus. Nat Rev Neurosci 11:791–805
Hall ZW, Sanes JR (1993) Synaptic structure and development: the neuromuscular junction. Cell 72:99–121
Sanes JR, Lichtman JW (1999) Development of the vertebrate neuromuscular junction. Annu Rev Neurosci 22:389–442
Ferraro E, Molinari BL (2012) Molecular control of neuromuscular junction development. J Cachexia Sarcopenia Muscle 1:13–23
Shigemoto K, Kubo S, Mori S, Yamada S, Akiyoshi T, Miyazaki T (2010) Muscle weakness and neuromuscular junctions in aging and disease. Geriatr Gerontol Int 10:S137–147
Jang YC, Van Remmen H (2010) Age-associated alterations of the neuromuscular junction. Exp Gerontol 46(2–3):193–198
Balice-Gordon RJ (1997) Age-related changes in neuromuscular innervation. Muscle Nerve Suppl 5:S83–87
Engel AG, Sine SM (2005) Current understanding of congenital myasthenic syndromes. Curr Opin Pharmacol 3:308–321
Nogajski JH, Kiernan MC, Ouvrier RA, Andrews PI (2009) Congenital myasthenic syndromes. J Clin Neurosci 1:1–11
Conti-Fine BM, Milani M, Kaminski HJ (2006) Myasthenia gravis: past, present, and future. J Clin Invest 11:2843–2854
Arnold AS, Christe M, Handschin C, Handschin C (2012) A functional motor unit in the culture dish: co-culture of spinal cord explants and muscle cells. J Vis Exp 62. doi:10.3791/3616
Anderson MJ, Cohen MW (1977) Nerve-induced and spontaneous redistribution of acetylcholine receptors on cultured muscle cells. J Physiol 3:757–773
Bernareggi A, Luin E, Formaggio E, Fumagalli G, Lorenzon P (2012) Novel role for prepatterned nicotinic acetylcholine receptors during myogenesis. Muscle Nerve 1:112–121
Nelson CM, Bissell MJ (2006) Of extracellular matrix, scaffolds, and signaling: tissue architecture regulates development, homeostasis, and cancer. Annu Rev Cell Dev Biol 22:287–309
Vandenburgh H, Shansky J, Benesch-Lee F, Skelly K, Spinazzola JM, Saponjian Y, Tseng BS (2009) Automated drug screening with contractile muscle tissue engineered from dystrophic myoblasts. FASEB J 10:3325–3334
Mueller-Klieser W (1997) Three-dimensional cell cultures: from molecular mechanisms to clinical applications. Am J Physiol 4(Pt 1):C1109–1123
Vandenburgh H, Shansky J, Benesch-Lee F, Barbata V, Reid J, Thorrez L, Valentini R, Crawford G (2008) Drug-screening platform based on the contractility of tissue-engineered muscle. Muscle Nerve 4:438–447
Bowe MA, Fallon JR (1995) The role of agrin in synapse formation. Annu Rev Neurosci 18:443–462
Kummer TT, Misgeld T, Lichtman JW, Sanes JR (2004) Nerve-independent formation of a topologically complex postsynaptic apparatus. J Cell Biol 7:1077–1087
Nishimune H, Valdez G, Jarad G, Moulson CL, Muller U, Miner JH, Sanes JR (2008) Laminins promote postsynaptic maturation by an autocrine mechanism at the neuromuscular junction. J Cell Biol 6:1201–1215
Thorrez L, Vandenburgh H, Callewaert N, Mertens N, Shansky J, Wang L, Arnout J, Collen D, Chuah M, Vandendriessche T (2006) Angiogenesis enhances factor IX delivery and persistence from retrievable human bioengineered muscle implants. Mol Ther 3:442–451
Vandenburgh H, Shansky J, Del Tatto M, Chromiak J (1999) Organogenesis of skeletal muscle in tissue culture. Methods Mol Med 18:217–225
Mahdavi V, Izumo S, Nadal-Ginard B (1987) Developmental and hormonal regulation of sarcomeric myosin heavy chain gene family. Circ Res 6:804–814
Wydro RM, Nguyen HT, Gubits RM, Nadal-Ginard B (1983) Characterization of sarcomeric myosin heavy chain genes. J Biol Chem 1:670–678
Lund AW, Yener B, Stegemann JP, Plopper GE (2009) The natural and engineered 3D microenvironment as a regulatory cue during stem cell fate determination. Tissue Eng Part B Rev 3:371–380
Nyga A, Cheema U, Loizidou M (2011) 3D tumour models: novel in vitro approaches to cancer studies. J Cell Commun Signal 3:239–248
Mazhar S, Herbst R (2012) The formation of complex acetylcholine receptor clusters requires MuSK kinase activity and structural information from the MuSK extracellular domain. Mol Cell Neurosci 4:475–486
Guo X, Gonzalez M, Stancescu M, Vandenburgh HH, Hickman JJ (2011) Neuromuscular junction formation between human stem cell-derived motoneurons and human skeletal muscle in a defined system. Biomaterials 36:9602–9611
Acknowledgments
We are grateful to Dr. Justin Fallon for providing mouse agrin. This research was supported by NIH grants R41 AR053386 and R43 AG029705. The authors confirm that no competing financial interests exist, and there has been no financial support for this research could have influenced its outcome.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wang, L., Shansky, J. & Vandenburgh, H. Induced Formation and Maturation of Acetylcholine Receptor Clusters in a Defined 3D Bio-Artificial Muscle. Mol Neurobiol 48, 397–403 (2013). https://doi.org/10.1007/s12035-013-8412-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-013-8412-z