Abstract
Introduction
We analyzed the impact of cause, severity, and duration of fever on functional outcome in patients with aneurysmal subarachnoid hemorrhage (aSAH).
Methods
Fever characteristics were analyzed in 584 consecutive patients with aSAH. Fever was defined as core body temperature ≥38.3 °C on ≥2 consecutive days. Subfebrile measurements were those between 37 and 38.2 °C. Febrile and subfebrile loads were the number of hours with fever or subfebrile measurements, respectively. Univariate and multivariate logistic regression models were developed to define predictors of outcome using various categorizations of fever cause, severity, and duration.
Results
Febrile measurements were observed in 281/584 (48.1 %) patients, recurring over a mean duration of 2.1 ± 3.0 days. Early fever within 24 and 72 h was encountered in 69 (11.9 %) and 110 (18.9 %) of patients, respectively. An infectious source was discovered in 126 (44.8 %) febrile patients. On univariate analysis, days of fever, febrile load, and fever onset within 24 and 72 h were associated with poor outcome (all p < 0.001); but subfebrile load was not (p = 0.56). On multivariate model constructed with all variables associated with outcome on univariate analyses, days of fever remained independently associated with poor outcome (OR 1.14 of poor outcome per day of fever, 95 % CI 1.06–1.22; p = 0.0006) displacing all other fever measures from the final model.
Conclusions
Early onset of fever, number of hours with fever, and especially days of fever are associated with poor functional outcome. Conversely, subfebrile load does not influence clinical outcome. These data suggest prolonged fever should be avoided, but subfebrile temperatures may not justify intervention.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Fever is a frequent complication following neurological injury and can exacerbate neuronal damage through increased metabolic demand and the resultant hyperemia that worsens excitotoxicity and inflammation and increases cerebral edema and intracranial pressure [1–3]. Multiple studies have shown fever to be independently associated with worse outcome and longer hospital stays in patients with various types of neurologic illness [4, 5]. Patients with aneurysmal subarachnoid hemorrhage (aSAH) are particularly prone to the development of fever, perhaps due to the development of vasospasm and the presence of blood in the subarachnoid cisterns and within the ventricles [6, 7].
Prior studies have indicated that up to three quarters of patients with aSAH develop abnormal temperature elevations during their hospital stay, with up to 50 % of fevers being attributed to infectious causes [8, 9]. Furthermore, these studies have confirmed that the presence of fever in patients with aSAH is also independently associated with poorer outcomes [6, 9–13]; yet the definitions of fever intensity, duration, and etiology have been heterogeneous, leaving uncertainty in the literature as to the “dose” of fever associated with adverse outcomes. For example, one study found an association between even a single temperature elevation ≥38.5 °C and poor outcome at 3 months in patients with aSAH [10]. Other studies have found correlations with worse outcome in patients with isolated or cumulative degrees of hyperthermia above differing temperature thresholds [9, 13]. Only some studies have investigated the effect of fever etiology (i.e., infectious versus noninfectious) on outcome. The effect of subfebrile temperature elevation (>37 °C) in patients with aSAH is unknown. Recent studies, to our knowledge, have confirmed the deleterious effects of fever in patients with brain injury but have not chosen to focus on the specific characteristics of fever in the aSAH population [14, 15]. The recently published guidelines for the management of aSAH recommend management of fever in this population but have not offered a specific threshold for treatment [16]. As treatment of fever with more aggressive measures can result in deleterious side effects, more specific knowledge of the impact of temperature elevation in the aSAH population is needed to better assess the risk/benefit ratio of treatment and perhaps to aid in prognostication [17]. Therefore, a more detailed analysis of the factors underlying the association of fever with poor outcome in aSAH is necessary.
We pursued this study to evaluate the effect of febrile and subfebrile temperature elevation due to infectious and noninfectious causes on clinical outcome in a large cohort of patients with aSAH.
Methods
Study Population
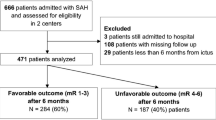
After obtaining Institutional Review Board approval, we performed a retrospective chart review of adult patients with aSAH who were admitted to the Neurologic Intensive Care Unit (ICU) at Saint Marys Hospital/Mayo Medical Center in Rochester, MN between 1/2/2001 and 9/16/2013. The diagnosis of subarachnoid hemorrhage was confirmed radiographically or by the presence of cerebrospinal fluid (CSF) xanthochromia. Inclusion into the study required the presence of a ruptured aneurysm documented on cerebral angiography. Patients with all other causes of aSAH were excluded from the study.
Fever Definitions, Temperature Monitoring, and Fever Treatment
Fever was defined as a body temperature ≥38.3 °C on at least one measurement within a 24-h period for two consecutive days [18, 19]. Days of fever was defined as the number of days the patient had at least one temperature reading ≥38.3 °C during their ICU stay. Fever-free ICU days was the number of days a patient spent without any temperature measurements ≥38.3 °C during their ICU stay. Febrile load was defined as the number of hours a patient spent at or above 38.3 °C during their ICU stay. In contrast, subfebrile temperatures were defined as body temperatures between 37 and 38.2 °C; and subfebrile load was the duration, in hours, a patient spent within this temperature range. Body temperature was measured hourly throughout the ICU stay using a bladder temperature probe, as is routine practice in this patient population at our institution. A fever was considered infectious if either (1) a pathologic organism was isolated from a culture or, (2) the patient received a full course of antibiotics [19]. Noninfectious fever was defined as a temperature reading ≥38.3 °C that did not fulfill the criteria for infectious fever. No cases of associated deep venous thrombosis, pulmonary emboli, or drug fever were identified in the cohort; therefore, we use the terms “noninfectious fever” and “central fever” synonymously. Subfebrile temperatures were not routinely treated. Fever was treated in a progressive and individualized manner, generally beginning with acetaminophen and progressing to the use of ice packs and cooling blankets, with the occasional use of advanced cooling devices for more refractory cases.
Included patients were analyzed with regard to age, sex, smoking status, pre-existing comorbidities, Glasgow Coma Scale, World Federation of Neurosurgical Societies (WFNS), modified Fisher scale, aneurysm location, intraventricular hemorrhage (IVH), intracerebral hemorrhage (ICH), presence and type of aneurysm treatment, neurological and systemic complications, radiographic evidence of ischemic infarction, and need for transfusion. Neurological complications included aneurysmal rebleeding, symptomatic vasospasm, delayed cerebral ischemia (DCI), seizures, and hydrocephalus. Systemic complications included anemia (hemoglobin < 8 g/dL) and acute lung injury/acute respiratory distress syndrome (P/F ratio <200 mmHg with bilateral noncardiogenic infiltrates). Temperature-specific variables collected included the presence of fever and subfebrile measurements, febrile and subfebrile load, days of fever, fever-free ICU days, and fever onset within 24 and 72 h.
Outcome Measures
The WFNS grade was assigned to all cases to assess clinical severity, and we used the WFNS grade after neurological resuscitation for the analysis because we have previously shown that it bears a stronger association with functional outcome than the score upon first evaluation or at nadir [20]. The modified Fisher scale was used to assess the degree of sanguineous extension of hemorrhage on the initial head CT. Aneurysm treatment with clipping or coiling was determined by multidisciplinary consensus based on aneurysmal location and morphology and the neurological and systemic condition of the patient. Routine surveillance for vasospasm was performed using transcranial Doppler ultrasonography, CT angiography and/or perfusion, and catheter angiography. Treatment of symptomatic vasospasm occurred in a graded and individualized manner, beginning with hemodynamic augmentation and progressing to the injection of intra-arterial vasodilators and/or cerebral angioplasty. DCI was defined by clinical deterioration not explained by another cause and/or a new infarction on brain imaging. Details of our multidisciplinary management of aSAH, imaging thresholds, and pertinent definitions have been previously described [20, 21]. Functional outcome was assessed at the last follow-up 6–12 months after the aSAH using the modified Rankin Scale (mRS) [22]. Our primary endpoint was poor functional outcome defined as a score of 3–6 on the mRS. The mRS is a 7-point scale ranging from 0, indicating no symptoms, to 6, indicating death. Functional independence is indicated by a score of 2 or less.
Statistical Analysis
Descriptive summaries were recorded as frequency (percentage) for categorical variables and as mean ± standard deviation (SD) for continuous variables. Univariate logistic regression analysis was employed to discern associations between the aforementioned variables and mRS score of 0–2 as a binary outcome. No imputation was done for missing data. Any variable associated with functional outcome with a p value ≤0.10 on univariate analysis was considered as a candidate for future multivariable logistic regression analysis. Area under the receiver operating characteristic curve was reported as a measure of model’s prediction ability. An area under the receiver operating characteristic curve estimate of 0.7–0.8 was regarded as acceptable, 0.8–0.9 was regarded as excellent, and more than 0.9 was regarded as outstanding. Since collinearity affected several of the fever variables, several models using each of the variables individually were explored to identify the best one (i.e., the model with the highest area under the curve).
Results
During the study period, we identified 584 patients with aSAH with a mean age of 55.9 years (range 16–89 years). Risk factors for aSAH in the cohort included active smoking in 267 (45.8 %) patients and hypertension in 294 (50.4 %) patients. After neurological resuscitation, the mean WFNS grade was 2.1 ± 1.67 and the mean modified Fisher scale score was 2.9 ± 1.07. Accompanying ICH and IVH was encountered in 129 (22.1 %) and 288 (49.3 %) patients, respectively. Symptomatic hydrocephalus requiring CSF diversion occurred in 313 (53.6 %) patients, and 176 (31.2 %) patients had DCI during their ICU stay. The majority (367/584, 62.8 %) of patients underwent coiling as their method of securing their aneurysm, and 62 (10.6 %) were managed medically. The mean ICU length of stay for the cohort was 11.0 ± 7.22 days. A good outcome occurred in 409 (70.0 %) of all patients; 99 (16.9 %) patients died. Comprehensive demographic information of the respective cohorts and significant associations with outcome on the univariate analysis for non-temperature-related variables are presented in Table 1.
A temperature reading ≥38.3 °C on at least one measurement within a 24-h period for two consecutive days was experienced by approximately half the patients in the cohort (48.1 %) during their hospital stay. However, elevated temperature readings were very common overall in the patient cohort with at least one temperature measurement above 38 °C occurring in 532 (91.1 %) patients. When fever occurred, it was infectious in 126 patients (44.8 % of febrile cases). However, the type of fever was not associated with functional outcome—approximately half the patients with poor outcome had fevers of infectious etiology while the other half had central fevers (p = 0.22). Early fever (onset within 24 h in 69 [11.9 %] patients and within 72 h in 110 [18.9 %]) patients was associated with nearly twice the incidence of poor outcome (28.6 % vs. 14.7 %; p = 0.0002). The average number of days of fever for the cohort was 2.1 ± 3.04 days, though patients with poor outcomes had approximately twice as many days of fever as patients with good outcomes (3.1 ± 3.85 days vs. 1.6 ± 2.50 days; p < 0.0001).
The mean febrile load was more than twice as high in patients with poor outcomes than in patients with good outcomes (33.8 ± 53.6 vs. 14.9 ± 30.1 h; p < 0.0001). Subfebrile measurements were very common, and the subfebrile load was almost six times greater than the febrile load (117.1 ± 115.0 vs. 20.5 ± 39.4 h). However, although the subfebrile load was higher in the poor outcome group, this difference was not significant (p = 0.5594). The results of the univariate analysis for temperature-related variables are displayed in Table 2.
On multivariate analysis, higher WFNS grade after neurological resuscitation, age, blood transfusion during hospitalization, higher initial modified Fisher scale score, and days of fever were associated with a poor outcome (Table 3). The model constituted by these five variables predicted poor functional outcome with an area under the curve of 0.897. Other fever variables (such as fever load and onset of fever within 72 h) were also significant when used instead of days of fever, but inclusion of days of fever generated the strongest model.
Discussion
The results of our study confirm prior reports that a substantial proportion of patients with aSAH will become febrile during their hospitalization [6, 7, 9, 10, 13]. It also reaffirms the evidence that fever is associated with poor functional outcome. However, our analysis of the relationship of fever with outcome in aSAH has novel aspects that offer new insights into this relationship. Prolonged fever (as characterized by number of days with fever) was a strong and independent predictor of poor outcome. Early onset of fever also portended worse prognosis. Meanwhile, subfebrile temperature elevations were very prevalent but did not impact the chances of achieving good functional recovery.
Previous studies have varied in their definition of fever, and this may explain the differences in prevalence across studies (Table 4). These preceding studies have predominantly focused on reporting the association of their collective heterogeneous definitions of fever with poor functional outcome [6, 9, 10, 14, 15] and, at most, discriminated the strength of the association relative to fever burden [13]. Information regarding the association of other temperature-related variables including fever duration and time of onset after hospitalization with outcome in these studies is, generally, lacking. Unlike previous studies, we differentiated between infectious and central fever. Fever was infectious in less than half of febrile patients in our cohort, and the nature of the fever (infectious vs. central) did not influence functional outcome.
By categorizing the time of onset of fever, we found that patients with poor outcome had fever earlier in their hospital course than patients with good outcome. Early fevers in patients with neurological injury, especially in the aSAH population, have been demonstrated to be predictive of central fever [18, 19]. Central fever is postulated to result from cerebral inflammation induced by cytokine release and the direct effects of cisternal and intraventricular blood [23, 24]. It is possible that early fever is associated with worse outcome because it is a marker of the severity of the hemorrhage, which would explain why the association was no longer significant on the multivariable analysis.
Our study demonstrates that the duration of fever is most strongly associated with poor outcome. Both the febrile load and days of fever were nearly double in aSAH patients with poor outcome relative to patients with good outcome. Days of fever was the fever-related variable independently associated with poor outcome on the multivariate analysis, and adding this variable contributed to the refinement of the prognostic model.
Conversely, the difference in subfebrile load between the outcome groups was not significant. This constitutes a novel and clinically useful observation. Based on this result, subfebrile temperature elevations may not have a deleterious impact on prognosis after aSAH and, therefore, may not require aggressive treatment. It is also reasonable to surmise that treating fever to a target of subfebrile temperatures rather than to strict normothermia may be sufficient. A prospective study comparing different temperature triggers and temperature targets for fever treatment would be needed to confirm these hypotheses. In fact, further study is warranted to determine if aggressive temperature control in febrile patients with aSAH can improve functional outcomes. One small case–control study suggested that induction of normothermia might be associated with improved outcomes at 1 year (though not at earlier time points) but at the expense of increased complications related to the treatment [25].
Our study has several limitations. First, our definitions of fever and subfebrile temperature and infectious fever were operational; and we acknowledge that changes in these definitions could affect the associations we encountered. We have opted to define fever as temperature ≥38.3 °C on at least one measurement for two consecutive days in this and previous studies [18, 19] in an effort to minimize the impact of potentially spurious measurements. We chose the cutoff of 37 °C to define subfebrile temperature because this target is often used in protocols designed to maintain strict normothermia, and it was the maximal normal temperature on the Intraoperative Hypothermia for Aneurysm Surgery trial [10]. Our definition of infectious fever comprised culture-proven infection and fever treated with a full course of antibiotics as in a previous study [19]. This was decided to acknowledge that it is not infrequent to find cases of aSAH with a high clinical suspicion of infection despite persistently negative cultures. Second, all patients with fever were treated with temperature-reducing therapies; and therefore, some patients’ fevers may have been “masked” by therapeutic interventions, reducing the apparent febrile load. As such, the results of our study should be viewed as reflecting the effect of refractory fever upon outcome in aSAH. Third, the degree of temperature elevation was recorded semiquantitatively (patients were either febrile or subfebrile) and so the incremental effect of fever >38.3 °C could not be assessed. Fourth, infectious and central fever may coexist in aSAH; and therefore, the distinction between the two types needs to be interpreted with caution. Finally, the nature of this study as a retrospective chart review predisposes it to potential bias as, among other possible confounding scenarios, not all fevers were worked up and treated in a rigorously uniform fashion. Additionally, the retrospective nature of the study did not allow for a specific pre-defined follow-up period for assessment of outcome after hospital discharge; and therefore, the range of associated follow-up visits for our study patients was used (6–12 months). Clinical outcomes can improve for individual patients during this range, and it is possible that our study may have a bias toward poorer outcomes given the mix of patients where outcomes were assessed in the earlier range of follow-up (i.e., around 6 months from hospital discharge).
In conclusion, fever is very prevalent during the acute hospital course of aSAH. Poor outcome is associated with early fever onset, greater febrile load, and especially more days of fever. Conversely, the number of hours with elevated but subfebrile temperatures does not seem to influence clinical outcome. Therefore, fever should be anticipated in patients with aSAH. While early and, especially, prolonged fever should be treated, subfebrile temperatures do not appear to justify intervention.
References
Polderman KH. Induced hypothermia and fever control for prevention and treatment of neurological injuries. Lancet. 2008;371:1955–69.
Greer DM, Funk SE, Reaven NL, Ouzounelli M, Uman GC. Impact of fever on outcome in patients with stroke and neurologic injury: a comprehensive meta-analysis. Stroke. 2008;39:3029–35.
Rossi S, Zanier ER, Mauri I, Columbo A, Stocchetti N. Brain temperature, body core temperature, and intracranial pressure in acute cerebral damage. J Neurol Neurosurg Psychiatry. 2001;71:448–54.
Diringer MN, Reaven NL, Funk SE, Uman GC. Elevated body temperature independently contributes to increased length of stay in the neurologic intensive care unit patients. Crit Care Med. 2004;32:1489–95.
Naidech AM, Bendok BR, Tamul P, Bassin SL, Watts CM, Batjer HH, et al. Medical complications drive length of stay after brain hemorrhage: a cohort study. Neurocrit Care. 2009;10:11–9.
Oliveira-Filho J, Ezzeddine MA, Segal AZ, Buonanno FS, Chang Y, Ogilvy CS, et al. Fever in subarachnoid hemorrhage: relationship to vasospasm and outcome. Neurology. 2001;56:1299–304.
Mees SMD, Luitse MJ, van den Bergh WM, Rinkel GJ. Fever after aneurysmal subarachnoid hemorrhage: relation with extent of hydrocephalus and amount of extravasated blood. Stroke. 2008;39:2141–3.
Commichau C, Scarmeas N, Mayer SA. Risk factors for fever in the neurologic intensive care unit. Neurology. 2003;60:837–41.
Fernandez A, Schmidt JM, Claassen J, Pavlicova M, Huddleston D, Kreiter KT, et al. Fever after subarachnoid hemorrhage: risk factors and impact on outcome. Neurology. 2007;68:1013–9.
Todd MM, Hindman BJ, Clarke WR, Torner JC, Weeks JB, Bayman EO, et al. IHAST Investigators. Perioperative fever and outcome in surgical patients with aneurysmal subarachnoid hemorrhage. Neurosurgery. 2009;64:897–908.
Springer MV, Schmidt JM, Wartenberg KE, Frontera JA, Badjatia N, Mayer SA. Predictors of global cognitive impairment 1 year after subarachnoid hemorrhage. Neurosurgery. 2009;65:1043–50.
Wartenberg KE, Schmidt JM, Claassen J, Temes RE, Frontera JA, et al. Impact of medical complications on outcome after subarachnoid hemorrhage. Crit Care Med. 2006;34:617–23.
Naidech AM, Bendok BR, Bernstein RA, Alberts MJ, Batjer HH, Watts CM, et al. Fever burden and functional recovery after subarachnoid hemorrhage. Neurosurgery. 2008;63:212–7.
Rincon F, Hunter K, Schorr C, Dellinger RP, Zanotti-Cavazzoni S. The epidemiology of spontaneous fever and hypothermia on admission of brain injury patients to intensive care units: a multicenter cohort study. J Neurosurg. 2014;121:950–60.
Rincon F, et al. Brain injury as a risk factor for fever upon admission to the intensive care unit and association with in-hospital case fatality: a matched cohort study. J Intensive Care Med. 2015;30:107–14.
Diringer MN, et al. Critical care management of patients following aneurismal subarachnoid hemorrhage: recommendations from the neurocritical care society’s multidisciplinary consensus conference. Neurocrit Care. 2011;15:211–40.
Scaravilli V, Tenchero G, Citerio G. Fever management in SAH. Neurocrit Care. 2011;15:287–94.
Giraldo EA, Mandrekar JN, Rubin MN, Dupont SA, Zhang Giraldo EA, Mandrekar JN, et al. Timing of clinical grade assessment and poor outcome in patients with aneurysmal subarachnoid hemorrhage. J Neurosurg. 2012;117:15–9.
Rabinstein AA, Lanzino G, Wijdicks EF. Multidisciplinary management and emerging therapeutic strategies in aneurysmal subarachnoid haemorrhage. Lancet Neurol. 2010;9:504–19.
Van Swieten JC, Koudstaal PJ, Visser MC, Schouten HJ, van Gijn J. Interobserver agreement for the assessment of handicap in stroke patients. Stroke. 1988;19:604–7.
Rabinstein AA, Sandhu K. Non-infectious fever in the neurological intensive care unit: incidence, causes and predictors. J Neurol Neurosurg Psychiatry. 2007;78:1278–80.
Hocker SE, Tian L, Li G, Steckelberg JM, Mandrekar JN, Rabinstein AA. Indicators of central fever in the neurologic intensive care unit. JAMA Neurol. 2013;70:1499–504.
Yoshimoto Y, Tanaka Y, Hoya K. Acute systemic inflammatory response syndrome in subarachnoid hemorrhage. Stroke. 2001;32:1989–93.
Hanafy KA, Morgan Stuart R, Fernandez L, Schmidt JM, Claassen J, Lee K, et al. Cerebral inflammatory response and predictors of admission clinical grade after aneurysmal subarachnoid hemorrhage. J Clin Neurosci. 2010;17:22–5.
Badjatia N, Fernandez L, Schmidt JM, Lee K, Claassen J, Connolly ES, et al. Impact of induced normothermia on outcome after subarachnoid hemorrhage: a case-control study. Neurosurgery. 2010;66:696–700.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Drs. Kramer, Rabinstein, Pegoli, and Mandrekar report no disclosures. Dr. Lanzino reports a consultant relationship with eV3/Covidien and Codman.
Rights and permissions
About this article
Cite this article
Kramer, C.L., Pegoli, M., Mandrekar, J. et al. Refining the Association of Fever with Functional Outcome in Aneurysmal Subarachnoid Hemorrhage. Neurocrit Care 26, 41–47 (2017). https://doi.org/10.1007/s12028-016-0281-7
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12028-016-0281-7