Abstract
One of the advantages of postmortem imaging is its ability to obtain diagnostic findings in a non-destructive manner when autopsy is either difficult or may destroy forensic evidence. In recent years, efforts have been made to incorporate computed tomography (CT) based postmortem angiography into forensic pathology; however, it is not currently clear how well the modality can determine sites of bleeding in cases of subarachnoid hemorrhage. Therefore, in this study, we investigated the utility of postmortem cerebral angiography using multi-detector row CT (MDCT) by injecting a contrast medium through a catheter inserted into the internal carotid and vertebral arteries of 10 subarachnoid hemorrhage cases. While postmortem MDCT angiography (PMCTA) was capable of detecting aneurysms in a non-destructive manner, it was sometimes difficult to identify the aneurysm and bleeding sites because of a large amount of contrast medium leaking into the extravascular space. To overcome this problem, we developed the novel contrast imaging method “dynamic cerebral angiography,” which involves scanning the same area multiple times while injecting contrast medium to enable real-time observation of the contrasted vasculature. Using multiphase contrast images acquired by this method, we successfully captured the moment when contrast medium leaked from the hemorrhage site. This method will be useful for identifying exact bleeding sites on PMCTA.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The key aim in the forensic diagnosis of subarachnoid hemorrhage (SAH) is to determine whether the cause was intrinsic or extrinsic because of the legal implications of the different causes of SAH [1]. For example, traumatic SAH, which is secondary to an impact to the face or head and sometimes has the appearance of an intrinsic SAH on CT, could point to a criminal act as the cause of death. To make this determination, the circumstances of death are taken into consideration when an autopsy is conducted to identify the site of the aneurysm, which generally indicates whether there was intrinsic onset or bleeding from a ruptured vertebral artery, which is the most frequent site of bleeding in traumatic SAH. However, identifying these sites, especially on the vertebral artery, can be extremely difficult at times and there is a risk of destroying forensic findings [2]. Therefore, for definitive forensic diagnosis of SAH, a non-destructive postmortem angiographic tool is needed that allows such lesions to be evaluated without destroying any evidence. With the introduction of postmortem diagnostic imaging into the field of forensic medicine, the number of postmortem angiographic studies using multi-detector row computed tomography (MDCT) has been increasing in recent years [3–8]. However, because only a few cases of SAH were investigated in these studies, it is not clear how well postmortem MDCT angiography (PMCTA) can determine the site of bleeding in SAH. Therefore, in this study we developed postmortem selective cerebral CT angiography, which we refer to as dynamic cerebral angiography, to use in cases of SAH. Based on our results, we discuss the utility of, and current issues associated with PMCTA in the forensic diagnosis of SAH.
Materials and methods
This study examined 10 cases of SAH diagnosed by pre-autopsy CT imaging conducted at our institute. As described below, we performed selective cerebral, but not whole body, CT angiography by injecting contrast medium through a catheter inserted into the internal carotid artery to perfuse the anterior brain and the vertebral artery in the posterior brain. Postmortem selective cerebral CT angiography was performed in the first seven cases to acquire post-injection images as preliminary angiograms. In the last three cases, our newly developed imaging modality, dynamic angiography, was used to establish the exact bleeding site.
Preliminary selective cerebral angiography
On autopsy, prior to opening the skull, a 6Fr catheter was inserted into the internal carotid artery. Another was inserted into the vertebral artery on the contralateral side and both were tightly ligated.
Water was then injected into each artery to confirm that the catheters were correctly inserted into the target arteries, and to expel any air in the vasculature before the contralateral end was clamped.
After this preparation in the autopsy room, the body was moved to the CT room. CT scans were performed using a 16-MDCT (ECLOS, Hitachi Medical Systems, Tokyo, Japan). Raw data were acquired at 120 kV, 200 mAs, and collimation 0.625 × 16 mm, and images were reconstructed at 0.625 mm slice thickness in the craniocervical region. Angiography was preceded by non-contrast imaging.
After non-contrast imaging, a contrast medium was injected at 2 ml/h (20–40 ml each; total 40–80 ml) using an injector for clinical use and was immediately followed by image acquisition. The non-ionic water-soluble contrast medium Iohexol (Omnipaque 300®; Daiichi Sankyo Co., Tokyo, Japan) was prepared by dilution with water and polyethylene glycol (ratio of contrast medium:water:polyethylene glycol = 1:5:10).
Immediately after scanning, planar images and volume rendering images, which were generated by the CT workstation software, were interpreted by the operator. Image findings were then compared with the corresponding autopsy findings by the dissector.
After autopsy, volume renderings were generated again using imaging analysis software (SYNAPSE VINCENT 3D image analysis system; Fujifilm Medical, Tokyo, Japan). We also used the subtraction method to visualize the blood vessels. In this method, only blood vessels are presented, by subtracting the information of bones by performing pre- and post-contrast imaging in the same position.
Dynamic selective cerebral angiography
Autopsy preparation was performed using the same methods as the preliminary selective cerebral angiography.
Raw data acquisition and image reconstruction were performed at 120 kV, 200 mAs, collimation 1.25 × 16 mm, and reconstruction at 1.25 mm slice thickness. Scanning range was set to 275 mm to cover the entry site of the vertebral artery into the vertebral column and the blood vessels at the top of the skull. A total of five scans were performed at 2, 25, 48, 71, and 117 s during the injection of contrast medium. This was the shortest scanning interval that the 16-MDCT used in this study could achieve at the abovementioned settings.
Using two injectors, 25 ml of the same contrast medium as above was injected into the catheter of the internal carotid artery at a rate of 0.2 ml/s, and 12.5 ml of contrast medium was injected into the catheter of the vertebral artery at a rate of 0.1 ml/s. Therefore, the amount of the contrast medium injected into the body at the time of each scan was 0.6, 7.5, 14.4, 21.3, and 35.1 ml. Imaging analysis was performed as described above.
Radiographic interpretation of the preliminary and dynamic angiographic images was performed by a radiology specialist with over 5 years of experience in postmortem CT image interpretation.
Identification of bleeding sites on autopsy
The sites of bleeding were determined on autopsy according to the method of Ikegaya et al. [2]. In brief, the brain was removed while injecting milk through the catheters used in angiography to determine the site of milk leakage as the bleeding site.
Results
Table 1 summarizes the details of all 10 cases. Preliminary angiography in three of the seven cases revealed the site of the aneurysm. Of these three cases, two had findings indicative of aneurysmal bleeding, but it was not possible to identify bleeding in the remaining case. In two of the other four cases, a large amount of contrast medium in the extravascular space made it difficult to identify the site of the aneurysm. One of these two cases was a case of suspected traumatic SAH, where there were no aneurysms or no bleeding sites present in the circle of Willis and contrast medium had accumulated around the vertebral artery. This strongly suggested that the bleeding originated from the vertebral artery; however, we were unable to identify the exact bleeding site on the artery. In the other case of brain death, the bleeding site and almost all of the cerebral blood vessels could not be visualized. In two of the three cases of dynamic cerebral angiography, multiphase CT-angiography allowed us to identify the exact vessel rupture site. In the other case, however, despite determining the site of the enlarged artery, the phase images did not capture the leakage of contrast medium.
The detailed results of preliminary angiography are presented as Cases A–D, and those of dynamic angiography as Cases E and F.
Case A
A 43-year-old man was found dead on the bed of a hotel room.
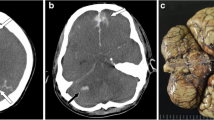
PMCTA findings: Post-contrast axial images revealed a right middle cerebral artery aneurysm and contrast medium leakage from the same site into the intraventricular space (Fig. 1a, arrow). Subtraction 3D CT images generated by the image analysis software confirmed a clear saccular aneurysm at the bifurcation of the right middle cerebral artery (Fig. 1b, arrow) as well as complex patterns of blood vessels inside the skull, which were difficult to visualize even with volume rendering imaging (Fig. 1b, circle).
Angiography reveals bleeding from an aneurysm in the middle cerebral artery (a, arrow). A subtraction image clearly shows the site of the aneurysm (b, arrow) as well as the cerebral arteries located deep in the skull (b, circle) that are difficult to examine non-destructively. The ruptured aneurysm in the right middle cerebral artery was identified on autopsy (c, circle)
Autopsy findings: Autopsy successfully identified a saccular aneurysm (0.5 cm in diameter) at the bifurcation of the right middle cerebral artery (yellow circle) and this site was the bleeding site (Fig. 1c).
Case B
A 72-year-old woman crossing the street was hit by a motorcycle and immediately went into cardiopulmonary arrest.
PMCTA findings: Post-contrast axial images revealed a large amount of accumulated contrast medium in the basal cistern, with no obvious aneurysm or bleeding sites (Fig. 2a, question-marked arrow). The same leakage was confirmed on 3D volume rendering images, but no aneurysm or bleeding site was detected because the artery was not visualized (Fig. 2b).
A post-contrast axial image (a) and volume rendering image (b) show leakage of a large amount of contrast medium into the cisterns, with no obvious aneurysm or bleeding site (question-marked arrow). Autopsy, however, revealed the presence of an aneurysm at the bifurcation of the internal carotid artery and posterior communicating artery (c)
Autopsy findings: Autopsy identified the site of the ruptured aneurysm as the bifurcation of the left internal carotid-posterior communicating artery (Fig. 2c).
Case C
A 23-year-old man lost consciousness and went into cardiopulmonary arrest after being struck with an iron bar during an argument.
PMCTA findings: Leaked contrast medium was observed in the vicinity of the left vertebral artery on axial images (Fig. 3a). Subtraction images also revealed the accumulation of contrast medium around the vertebral arteries inferior to the junction with the basilar artery, with no obvious aneurysms in other parts of the vasculature including the circle of Willis (Fig. 3b). Although imaging findings suggested bleeding from the left vertebral artery, it was difficult to determine the exact bleeding site or the presence of an aneurysm.
Leakage of contrast medium around the left vertebral artery on axial images (a, arrow). Subtraction images also show the accumulation of contrast medium around the vertebral artery with no obvious ruptured site (b, question-marked arrow). No aneurysm or bleeding sites were observed on other blood vessels including the circle of Willis. Autopsy revealed a ruptured vessel lesion on the posterior wall of the left vertebral artery (c, circle)
Autopsy findings: On autopsy, a tear (0.2 cm in length) running longitudinally on the posterior wall of the left vertebral artery was successfully located (Fig. 3c). No lesions indicative of intrinsic SAH were observed even on histological examination, suggesting that the cause of death was extrinsic SAH.
Case D
A 49-year-old man with a psychiatric disorder stuck a chopstick into his eye. Because cerebral angiography performed in the hospital suggested SAH caused by damage to the bifurcation of the posterior cerebral artery, coil embolization was performed to treat the site. The patient died after being brain-dead for 9 days.
PMCTA findings: Even though post-contrast axial images were suggestive of SAH, brain swelling was substantial, which narrowed the ventricles and cisterns (Fig. 4a). Subtraction images also failed to visualize the cerebral vasculature properly, except for those close to the contrast medium injection sites (Fig. 4b).
Autopsy findings: Autopsy could not locate the site of the bleeding because of severe brain malacia. The cause of death was considered to be cerebral edema caused by brain contusion.
Case E
A 59-year-old woman was discovered deceased by her husband in their bedroom.
PMCTA findings: Among five phase images obtained by dynamic angiography, the second-phase image (25 s after injection of a total of 7.5 ml contrast medium) captured the moment when contrast medium leaked from a saccular aneurysm in the left vertebral artery. At this point, the blood vessels were partially contrasted. Later-phase images clearly showed contrast medium accumulating in the extravascular space. In the fourth phase (71 s, 21.3 ml), it was not possible to identify the exact bleeding site because of contrast medium leakage, even though the majority of the cerebral arteries had contrast at this point (Fig. 5).
Dynamic angiography was performed to obtain five-phase subtraction images (2 (1), 25 (2), 48 (3), 71 (4), and 117 s (5)). The second-phase image shows the presence of an aneurysm in the left vertebral artery with leakage of contrast medium (arrow, 2). Images in the later phases show increasing amounts of leaked contrast medium. Most blood vessels in the anterior brain are not visible at the second phase. Although the fourth-phase image shows almost all of the major vessels, the exact bleeding site cannot be determined because of the large amount of contrast medium leakage
Autopsy findings: Autopsy successfully detected a ruptured saccular aneurysm in the left vertebral artery.
Case F
A 56-year-old man went into cardiopulmonary arrest immediately after being hit by car and was pronounced dead in the hospital 17 h after the accident.
PMCTA findings: Because CT imaging in the hospital revealed SAH, dynamic cerebral angiography was performed. Axial images were remarkable, showing an enlarged left vertebral artery compared with that in the contralateral side, and a fusiform-like aneurysm was clearly observed on 3D volume rendering images (Fig. 6a, b). However, none of the phase images captured the contrast medium leaking into the extravascular space.
Dynamic angiography was performed to obtain five-phase subtraction images of the vertebral artery. The fifth-phase image shows an enlarged left vertebral artery compared with the right artery, and a fusiform-like aneurysm is clearly visible on 3D volume rendering images. However, none of the phase images shows contrast medium leakage (a, b). Post-autopsy histopathological examination using Elastica van Gieson stain revealed fibrin clot formation at the site and a rupture extending to all layers of the vertebral artery with no vascular disease such as medial degeneration or dissection
Autopsy findings: On autopsy, we confirmed an enlarged artery at the same site, and subsequent histopathological examination revealed fibrin clot formation and a rupture extending to all layers of the vertebral artery with no vascular disease such as medial degeneration or dissection (Fig. 6c).
Discussion
Interest in the use of PMCTA is growing in the field of forensic medicine. Whole body angiography, developed by a Swiss group, has greatly advanced the capability of postmortem imaging [3, 5–8, 17], and other groups have started to report studies on PMCTA [4, 13–16]. The main feature of recent postmortem angiographic research is a focus on minimizing the invasiveness of contrast imaging, with the view that postmortem imaging, including angiography, will eventually replace traditional autopsy [4, 17]. These challenges are bold attempts and we also believe that postmortem imaging combined with certain techniques such as angiography or biopsy might reduce the need for many full autopsies in the future. In this study, we presented a new approach for PMCTA that is quick and easy to perform with improved diagnostic quality, especially in SAH cases. We utilized PMCTA, one of the useful postmortem diagnostic tools at present, in combination with autopsy. Regardless of the different postmortem methods available to investigate the vasculature, this study offers new insight into the application of PMCTA in autopsy.
Preliminary cerebral angiography
To evaluate the diagnostic value of PMCTA in SAH, we performed preliminary angiography after filling the blood vessels with contrast medium. The results showed that PMCTA, as illustrated in Case A (Fig. 1), allowed us to non-invasively identify vascular lesions that are difficult to dissect and thus are at risk of being destroyed during autopsy. On the other hand, this study also showed that a large amount of leaked contrast medium may obstruct the view of the bleeding site and the cause, such as an aneurysm, as illustrated in Case B (Fig. 2). In Case C (Fig. 3), CT angiographic findings clearly eliminated the circle of Willis as a possible site for the aneurysm and as a bleeding site, leaving the vertebral artery as the potential bleeding site; thus, the finding was extremely useful for the subsequent autopsy even though leakage of the contrast medium prevented us from locating the exact bleeding site or properly observing the artery.
In the field of forensic medicine, care must be taken during autopsy because extrinsic SAH may occasionally be encountered (i.e., traumatic rupture of the vertebral artery) in cases of suspected intrinsic SAH, despite no obvious external injuries on non-contrast CT imaging or external examination [10, 11]. Consequently, forensic pathologists need to establish the mechanism of the SAH based on information obtained from the investigation of the scene, external findings, and the presence of an aneurysm and bleeding site. If identification of the exact location of the bleeding site is difficult because of a large amount of contrast medium leaking into the extravascular space, this is a huge setback in the forensic diagnosis of SAH.
In antemortem CT angiographic imaging of SAH, the leakage of contrast agents from a ruptured blood vessel is reportedly extremely rare [12]. In Case F, because the individual was alive for 17 h after the onset of SAH, clot formation at the rupture site completely prevented leakage of contrast medium in postmortem angiography. When the patient is alive, active bleeding from a blood vessel tear is stopped by hemostasis, which involves vascular contraction and clot formation. A major cause of the large amount of leakage of contrast medium after death is the lack of hemostasis, and this is particularly true in cases of sudden death where death occurred almost instantaneously after the onset of an acute event. Although we did not strictly define the injection pressure of contrast medium in this study, a previous X-ray angiographic study of the vertebral artery conducted using vital pressure also reported similar leakage of contrast medium [18]. Therefore, regardless of the method used, the use of contrast medium can result in problematic leakage during postmortem angiography.
Dynamic cerebral angiography
After being inspired by reading about “dynamic CT angiography” used in clinical practice [9], we developed the novel method of “dynamic cerebral angiography” to overcome the abovementioned problems. In this method, we inject contrast medium at a low rate and scan the area of interest multiple times, starting immediately after the injection, to capture the moment when the contrast medium leaks into the extravascular space, as illustrated in the second phase in Case E (Fig. 5). The subsequent phases confirm the accumulation of contrast medium over time, suggesting that this method not only overcomes the problem of contrast media leakage but can visualize the exact site of vessel rupture. Therefore, this may be an effective tool for forensic pathologists in autopsies for SAH. Because our dynamic cerebral angiography method requires multiple scans over a short period, it might be difficult to apply this method in whole body angiography. However, such application may be possible if 64-MDCT or 320-MDCT scanners, which are commonly used in clinical practice, become more readily available in the field of forensic medicine.
In the dynamic angiography cases, we noticed a characteristic finding from the pattern of spread of extravasated contrast medium. In Case 8 (Case E; Fig. 5) and Case 10, accumulation of contrast medium spread to a low level not to high level from the bleeding point. Specifically, contrast medium was seen at the subtentorial level around the vertebral artery in Case 8 and at the level of the circle of Willis in Case 10. A similar phenomenon was seen in Case B and C where a large amount of leaked contrast medium was seen on preliminary angiography. In standard PMCTA for SAH cases, this finding may be used to identify the bleeding site.
In Case F (Fig. 6), even though we observed an enlarged left vertebral artery resembling a fusiform aneurysm, none of the phase images showed contrast medium leakage from the blood vessel. Subsequent autopsy and detailed histological examination finally revealed a transmural rupture of the vertebral artery with no vascular disease such as medial degeneration or dissection, suggesting traumatic rupture of the vertebral artery. We believe the failure to observe this leakage was because the hemostatic and repair mechanisms were activated as part of physiological reactions and stopped the bleeding in the antemortem period. In antemortem imaging diagnosis, artery diameter enlargement is considered to be an indicator of a fusiform aneurysm or dissecting aneurysm. However, great care must be taken because of the potential for false-positive findings [19, 20]. In fact, this case was not diagnosed as intrinsic SAH due to ruptured aneurysm, but as traumatic rupture of the vertebral artery. We believe it is necessary to assess ruptured blood vessels using both imaging and histological findings, even in future studies, to correctly distinguish intrinsic from extrinsic cases. At present, postmortem angiography can be used as an effective tool to aid determination of the cause of death.
In this study, we also found that the contrast medium hardly penetrated into the brain arteries when intracranial pressure was significantly increased. A similar phenomenon has been reported in a PMCTA study in a case of SAH with increased intracranial pressure and on a heart–lung machine [21]. In the present study, the cerebral arteries could be visualized in most cases because we injected contrast medium directly into the cerebral arteries at a constant flow rate without setting the infusion pressure, except in Case D (brain death with increased intracranial pressure) (Fig. 4) in which no intracranial blood vessels were contrasted. An increase in flow rate and infusion pressure may overcome this problem. However, we were concerned about unexpected extravasation, leading to an artifact, with the increase in the intravascular pressure rather than a filling defect. We believe this phenomenon may be a limitation of PMCTA.
Limitations
There are some limitations of the present technique. The first limitation is that this technique is selective angiography for the cerebral arteries, not for the whole vascular system. For this reason, we cannot assess other vascular pathology that might be detected by whole body angiography. This would be important if the decedent had an underlying disease, such as one that involved the systemic vasculature other than the cerebral vessels (e.g., systemic disease such as Marfan syndrome, Ehlers-Danlos syndrome or fibromuscular dysplasia, and arteritis), or conditions that spread to involve the cerebral vessels from other vascular lesions (e.g., aortic dissection secondary to both intrinsic and traumatic causes). The second limitation is a failure to assess lesions at the proximal level of the internal carotid artery and vertebral artery due to the placement of the catheter. Future research should also evaluate angiography of the venous system to assess venous lesions that could be sources of hemorrhage.
Conclusion
In this study, we showed that PMCTA is useful for the non-destructive observation of ruptured blood vessels and aneurysms. This new imaging modality, especially the dynamic protocol, is easy to perform and is an effective tool to aid determination of the cause of death, and it has the potential to distinguish between intrinsic and extrinsic causes by correctly identifying the bleeding site or the presence of aneurysm. The number of examined cases was quite small and we encountered a case that required both an autopsy and histological examination to achieve a diagnosis, therefore further studies on the diagnostic value of PMCTA are needed.
Key points
-
1.
PMCTA enables observation of aneurysms in a non-destructive manner and provides the forensic pathologist with useful diagnostic information about the cause of death.
-
2.
If a large amount of contrast medium leaks, PMCTA may not be effective for visualizing the vasculature, thus complicating identification of the aneurysm and bleeding site. Dynamic cerebral angiography has the potential to overcome these problems.
-
3.
The findings of the present, albeit small, study indicate that even if aneurysms are identified on PMCTA, the bleeding site (leakage of contrast medium) may not be observed and thus the cause cannot be correctly diagnosed as intrinsic or extrinsic without detailed histopathological examination.
-
4.
Blood vessels may be poorly contrasted on dynamic cerebral angiography in cases of markedly increased intracranial pressure.
References
Saukko P, Knight B. Knight’s forensic pathology. 3rd ed. London: Arnold; 2004.
Ikegaya H, Yajima D, Iwase H. Vertebral artery rupture in a sudden death case after mild trauma. Am J Forensic Med Pathol. 2008;29(3):276–8.
Grabherr S, Djonov V, Yen K, Thali MJ, Dirnhofer R. Postmortem angiography: review of former and current methods. AJR Am J Roentgenol. 2007;188(3):832–8.
Saunders SL, Morgan B, Raj V, Rutty GN. Post-mortem computed tomography angiography: past, present and future. Forensic Sci Med Pathol. 2011;7(3):271–7.
Grabherr S, Gygax E, Sollberger B, Ross S, Oesterhelweg L, Bolliger S, Christe A, Djonov V, Thali MJ, Dirnhofer R. Two-step postmortem angiography with a modified heart-lung machine: preliminary results. AJR Am J Roentgenol. 2008;190(2):345–51.
Grabherr S, Doenz F, Steger B, Dirnhofer R, Dominguez A, Sollberger B, Gygax E, Rizzo E, Chevallier C, Meuli R, Mangin P. Multi-phase post-mortem CT angiography: development of a standardized protocol. Int J Legal Med. 2011;125(6):791–802.
Ross S, Spendlove D, Bolliger S, Christe A, Oesterhelweg L, Grabherr S, Thali MJ, Gygax E. Postmortem whole-body CT angiography: evaluation of two contrast media solutions. AJR Am J Roentgenol. 2008;190(5):1380–9.
Flach PM, Ross SG, Bolliger SA, Preiss US, Thali MJ, Spendlove D. Postmortem whole-body computed tomography angiography visualizing vascular rupture in a case of fatal car crash. Arch Pathol Lab Med. 2010;134(1):115–9.
Matsumoto M, Kodama N, Endo Y, Sakuma J, Suzuki K, Sasaki T, Murakami K, Suzuki K, Katakura T, Shishido F. Dynamic 3D-CT angiography. AJNR Am J Neuroradiol. 2007;28(2):299–304.
Iwase H, Yamamoto S, Yajima D, Hayakawa M, Kobayashi K, Otsuka K, Sato K, Motani H, Kasahara S, Ito H. Can cervical spine injury be correctly diagnosed by post mortem computed tomography? Leg Med. 2009;11(4):168–74.
Opeskin K, Burke MP. Vertebral artery trauma. Am J Forensic Med Path. 1998;19(3):206–17.
Kathuria S, Deveikis JP, Westesson PL, Gandhi D. Improved diagnosis of actively bleeding aneurysm on CT angiography using delayed CT images. Eur J Radiol. 2011;79(2):328–31.
Roberts IS, Benamore RE, Peebles C, Roobottom C, Traill ZC. Technical report: diagnosis of coronary artery disease using minimally invasive autopsy: evaluation of a novel method of post-mortem coronary CT angiography. Clin Radiol. 2011;66(7):645–50.
Saunders SL, Morgan B, Raj V, Robinson CE, Rutty GN. Targeted post-mortem computed tomography cardiac angiography: proof of concept. Int J Legal Med. 2011;125(4):609–16.
Palmiere C, Binaghi S, Doenz F, Bize P, Chevallier C, Mangin P, Grabherr S. Detection of hemorrhage source: the diagnostic value of post-mortem CT-angiography. Forensic Sci Int. 2012;222(1–3):33–9.
Burke M, Parsons S, Bassed R. Management of medicolegal natural deaths from hemopericardium or hemothorax using postmortem CT scanning. Forensic Sci Med Pathol. 2012;8(4):367–72.
Bolliger SA, Filograna L, Spendlove D, Thali MJ, Dirnhofer S, Ross S. Postmortem imaging-guided biopsy as an adjuvant to minimally invasive autopsy with CT and postmortem angiography: a feasibility study. AJR Am J Roentgenol. 2010;195(5):1051–6.
Karhunen PJ, Kauppila R, Penttilä A, Erkinjuntti T. Vertebral artery rupture in traumatic subarachnoid haemorrhage detected by postmortem angiography. Forensic Sci Int. 1990;44(2–3):107–15.
Chen CJ, Tseng YC, Lee TH, Hsu HL, See LC. Multisection CT angiography compared with catheter angiography in diagnosing vertebral artery dissection. AJNR Am J Neuroradiol. 2004;25(5):769–74.
Kaiser Ch, Schnabel A, Berkefeld J, Bratzke H. Traumatic rupture of the intracranial vertebral artery due to rotational acceleration. Forensic Sci Int. 2008;182(1–3):e15–7.
Chew KL, Baber Y, Iles L, O’Donnell C. Duret hemorrhage: demonstration of ruptured paramedian pontine branches of the basilar artery on minimally invasive, whole body postmortem CT angiography. Forensic Sci Med Pathol. 2012;8(4):436–40.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Inokuchi, G., Yajima, D., Hayakawa, M. et al. Postmortem dynamic cerebral angiography for detecting aneurysm and bleeding sites in cases of subarachnoid hemorrhage. Forensic Sci Med Pathol 10, 487–495 (2014). https://doi.org/10.1007/s12024-014-9572-5
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12024-014-9572-5